Demand for endoscope reprocessing device in Japan is valued at USD 131.3 million in 2025 and is projected to reach USD 223.7 million by 2035 at a CAGR of 5.5%. Automated endoscope reprocessors dominate deployment across large medical centers, supported by washer disinfectors, sterilizers, ultrasonic washers, and drying and storage cabinets across tiered facilities. Hospitals and central sterile services departments form the core demand base, while specialty clinics and academic institutions contribute steady procurement cycles. Kanto, Kinki, and Chubu remain the operational centers due to dense hospital networks and advanced endoscopy services. Global suppliers such as Olympus, Getinge, STERIS, Steelco, and ASP anchor system installations across public and private institutions.
Utilization patterns across Japan reflect rising contamination control audits, higher endoscope fleet density per hospital, and procedural expansion in gastroenterology, pulmonology, and urology units. Semi-automatic systems continue to serve mid volume facilities, while fully automatic platforms gain placement in urban tertiary centers with high procedural turnover. Floor standing and bench top systems dominate due to space constraints and workflow integration preferences. Regional demand across Tohoku, Kyushu, and Hokkaido follows renovation-driven upgrades rather than greenfield installations. Procurement decisions align closely with chemical compatibility, cycle validation accuracy, and staff workload reduction metrics. Supplier competition centers on system throughput reliability, digital tracking integration, footprint optimization, and long-term service agreements aligned with hospital sterilization compliance programs under national infection control oversight.
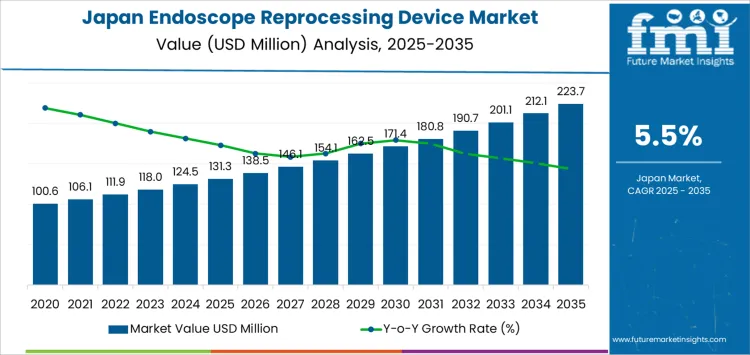
Endoscope reprocessing device demand in Japan stands at USD 131.3 million in 2025 and advances to USD 162.5 million by 2030, representing a USD 31.2 million increase over the five-year period. The climb from USD 100.6 million in 2020 reflects rising endoscopy procedure density across gastroenterology, pulmonology, and urology departments. Hospital infection control frameworks place strong operational pressure on rapid, standardized, and traceable reprocessing cycles. Large hospital networks continue to shift away from manual disinfection toward automated systems that deliver cycle validation, accurate chemical dosing, and audit-ready documentation. Demand during this stage remains closely linked to growth in procedure turnover and tightening national compliance benchmarks for device hygiene.
From 2030 to 2035, demand expands from USD 162.5 million to USD 223.7 million, adding USD 61.2 million in fresh value during the later phase. Annual value gains widen as secondary hospitals and outpatient endoscopy centers scale their own automated reprocessing capacity. Higher screening volumes for colorectal cancer, respiratory disease, and biliary conditions sustain continuous equipment utilization. Replacement-driven demand also strengthens as early-generation units reach end-of-life thresholds. Integration of digital tracking, consumable optimization, and chemical-free disinfection modules increases the average system value per installation.
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 131.3 million |
| Forecast Value (2035) | USD 223.7 million |
| Forecast CAGR (2025-2035) | 5.5% |
Demand for endoscope reprocessing device in Japan is shaped by the very high volume of gastrointestinal screening, strong infection control discipline, and strict hospital accreditation standards. Japan maintains one of the highest rates of routine endoscopy for gastric and colorectal disease due to early cancer detection programs and dense outpatient access. This sustained procedural load places constant pressure on hospitals to maintain fast, repeatable, and verifiably clean reprocessing cycles. Manual cleaning alone proved insufficient as case volumes grew and traceability requirements tightened. Automated reprocessing devices gained early traction in tertiary hospitals where turnaround speed, documentation, and staff exposure reduction became operational priorities. Reprocessing systems are now treated as core infrastructure rather than optional equipment in endoscopy units.
Future demand for endoscope reprocessing devices in Japan will be shaped by aging-driven procedure growth, rising antimicrobial resistance awareness, and expanding outpatient endoscopy capacity. Shorter hospital stays and higher same-day procedure turnover increase reliance on automated, validated disinfection workflows. Remote audit capability and digital cycle tracking will become more relevant as hospitals face increased regulatory oversight. Barriers include high capital cost for advanced systems, space limitations in older hospital buildings, and slower adoption in small regional clinics. Staff training and maintenance workload also constrain wider deployment. Long-term demand will depend on how effectively Japanese healthcare systems balance infection control rigor, procedural throughput, and cost containment across both urban and regional endoscopy centers.
Demand for endoscope reprocessing devices in Japan is shaped by high endoscopy procedure volume, strict infection control standards, and centralized hospital-based reprocessing workflows. Automated endoscope reprocessors lead by product due to the standardization of cleaning and disinfection cycles. Semi-automatic operation mode leads the operation mode because it balances process control with workflow flexibility. Acute care hospitals, university medical centers, and specialty endoscopy clinics drive procurement. Import reliance remains present for advanced control systems and chemical dosing components. Substitution pressure across product categories remains limited by regulatory audit requirements. Demand stability is supported by rising diagnostic endoscopy volumes and national patient safety enforcement.
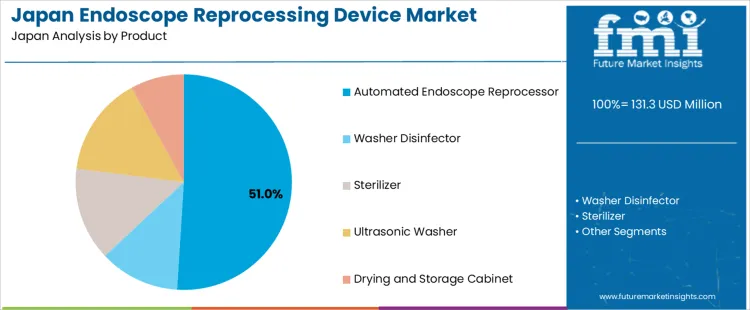
Automated endoscope reprocessors account for 51% of the demand for endoscope reprocessing device in Japan by product, reflecting preference for standardized cleaning validation and reduced manual handling. Consumption intensity is driven by high throughput gastroenterology units and surgical endoscopy suites that require consistent cycle performance. Usage remains stable because automated systems support repeatable disinfection protocols aligned with national infection prevention guidance. Procurement is led by large hospitals operating multi room endoscopy departments. Price sensitivity remains moderate because device reliability reduces reprocessing errors and contamination risk. Specification control emphasizes channel flush pressure stability, disinfectant concentration accuracy, and automated leak testing capability.
Automated endoscope reprocessors also generate steady replacement demand linked to sensor wear, pump degradation, and control board life cycles. Repeat utilization remains predictable under continuous daily operation schedules. Buyers favor standardized platform models to reduce staff retraining and spare part diversity. Margin structure remains controlled under tender based hospital purchasing. Regulatory exposure remains elevated due to audit traceability and outbreak prevention oversight. Import reliance persists for dosing pumps and control electronics. Substitution pressure from washer disinfectors remains limited for flexible endoscopes that require validated channel specific reprocessing.
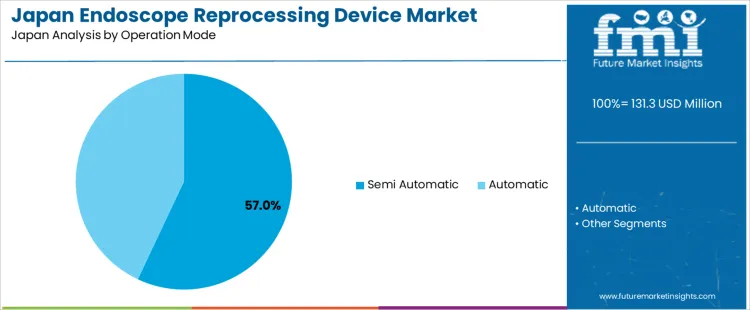
Semi-automatic operation represents 57.0% of the demand for endoscope reprocessing device in Japan by operation mode, reflecting its balance between automation and operator control. Consumption intensity is driven by mid-sized hospitals and outpatient endoscopy centers that require efficiency without full automation cost exposure. Usage remains stable because semi-automatic systems allow adjustment of cycle steps under varied scope types. Procurement is managed by hospital departments with constrained capital budgets and workflow customization needs. Price sensitivity remains moderate because these systems reduce labor burden while retaining manual supervision. Specification control emphasizes timing accuracy, rinse water quality control, and disinfectant exposure consistency across cycle steps.
Semi-automatic systems also generate consistent repeat demand through gradual fleet upgrades and endoscopy capacity expansion. Repeat utilization remains predictable as reprocessing loads rise with procedure growth. Buyers favor modular designs that allow future automation upgrade without full system replacement. Margin structure remains controlled under equipment competition and distributor contracting. Regulatory exposure remains elevated due to compliance with reprocessing documentation standards. Import reliance persists for valves, solenoid controls, and monitoring sensors. Substitution pressure from fully automatic systems rises in large tertiary centers, though semi-automatic operation retains preference in cost sensitive procedural settings.
Demand for endoscope reprocessing devices in Japan is shaped by strict infection control rules, dense hospital utilization, and rising procedural complexity across gastroenterology and pulmonology. High daily case turnover in urban hospitals places heavy pressure on cleaning accuracy, cycle speed, and traceability. Elderly patient volume increases exposure risk from cross contamination, raising tolerance for manual processing errors. Automated reprocessing systems support standardized disinfection, drying, and documentation under safety audits. Hospitals treat reprocessing as a core clinical infrastructure function rather than a support task. Demand grows through equipment replacement cycles, workflow modernization, and compliance readiness instead of episodic outbreak response activity.
Urban medical centers in Japan perform exceptionally high volumes of endoscopic examinations for cancer screening, bleeding control, and respiratory diagnostics. Turnover speed between cases determines room utilization and staff efficiency. Automated reprocessing devices shorten downtime through verified wash, high level disinfection, and controlled drying cycles. Manual reprocessing struggles to match this throughput without increasing human error risk. Nurses and technicians rely on automation to sustain late evening procedure schedules. Demand rises in parallel with colonoscopy screening participation and pulmonary diagnostics. Hospitals select systems that balance cycle time with documented microbial reduction under inspection standards for national accreditation compliance requirements today.
Hospital accreditation in Japan places strong emphasis on traceable disinfection records and documented exposure control. Reprocessing devices now integrate barcode tracking, cycle verification, and digital archiving of every scope processed. These features support audit preparation and post incident review without reliance on handwritten logs. Central sterile departments use this data to match device performance against inspection thresholds. Procurement teams increasingly judge suppliers on validation reporting depth rather than unit price alone. Demand follows inspection rigor and public reporting sensitivity tied to healthcare associated infection metrics across prefectural hospital networks where accountability pressures influence capital equipment approval pathways each fiscal cycle.
Japan hospital staffing patterns show continued tightening across nursing and technical roles. Endoscope reprocessing demands repetitive precision tasks that elevate fatigue risk during long shifts. Automated devices reduce manual handling while stabilizing output quality across staffing fluctuations. Many facilities operate under strict space limits that favor compact closed systems over open bath methods. Centralized reprocessing rooms serve multiple departments through scheduled batch workflows. Device placement must align with patient transport corridors and contamination zoning. Demand is shaped by labor substitution value, footprint efficiency, and safe workflow separation inside compact acute care facilities that support daily procedural throughput without bottleneck formation.
| Region | CAGR (%) |
|---|---|
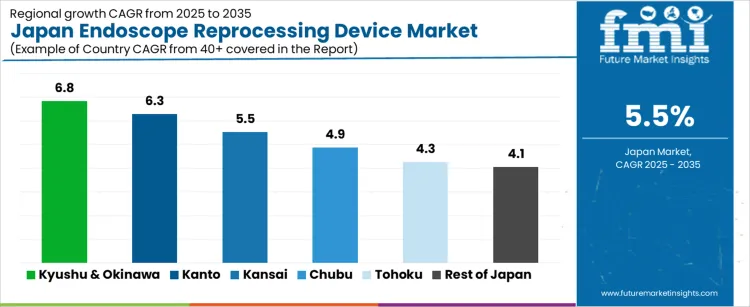
| Kyushu & Okinawa | 6.8% |
| Kanto | 6.3% |
| Kansai | 5.5% |
| Chubu | 4.9% |
| Tohoku | 4.3% |
| Rest of Japan | 4.1% |
The demand for endoscope reprocessing devices in Japan is rising steadily across hospital networks, led by Kyushu and Okinawa at a 6.8% CAGR. Growth in this region is supported by increasing endoscopic procedure volumes, stricter infection control protocols, and modernization of sterilization infrastructure in regional hospitals. Kanto follows at 6.3%, driven by dense tertiary care hospitals, high diagnostic endoscopy throughput, and strong compliance with automated reprocessing standards. Kansai records 5.5% growth, reflecting stable replacement demand and upgrades in university hospitals and private gastroenterology centers. Chubu at 4.9% shows moderate uptake linked to regional hospital expansions and outpatient endoscopy units. Tohoku and the Rest of Japan, at 4.3% and 4.1%, reflect slower growth shaped by lower procedure density, longer equipment replacement cycles, and budget constraints in smaller healthcare facilities.
Demand for endoscope reprocessing device in Kyushu and Okinawa is advancing at a CAGR of 6.8% through 2035, supported by rising gastrointestinal procedure volumes, steady hospital equipment upgrades, and strict infection control protocols in regional medical centers. Public hospitals in Fukuoka and Okinawa drive most automated reprocessing system installations. This region reflects strong dependence on centralized sterilization departments rather than unit level reprocessing. Growth is linked to expanding colonoscopy screening programs, higher surgical endoscopy use, and stronger regulatory focus on patient safety and device hygiene standards.
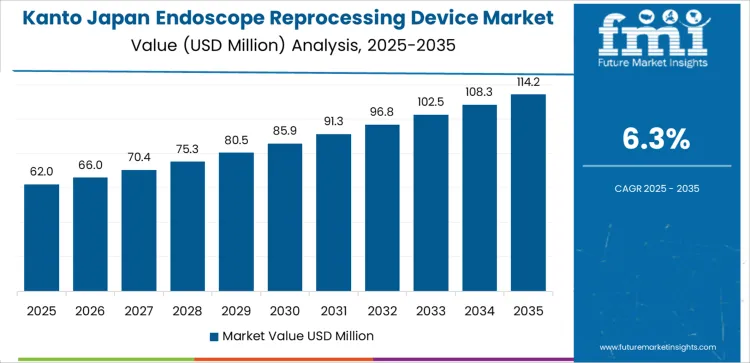
Demand for endoscope reprocessing device in Kanto is rising at a CAGR of 6.3% through 2035, driven by high endoscopy procedure density, advanced hospital infrastructure, and widespread adoption of automated disinfection systems across Tokyo and surrounding areas. Kanto leads in deployment of high throughput reprocessing units within tertiary hospitals and private specialty clinics. This region records higher unit turnover due to intense daily procedure volumes. Growth reflects continuous hospital modernization, outpatient endoscopy expansion, and strong regulatory oversight of device hygiene and traceability systems.
Demand for endoscope reprocessing device in Kansai is progressing at a CAGR of 5.5% through 2035, supported by steady endoscopy volumes, stable hospital investment cycles, and consistent infection prevention standards across Osaka and Kyoto. Kansai shows balanced deployment across public teaching hospitals and mid sized private facilities. This region reflects controlled capital investment rather than rapid expansion. Growth is shaped by periodic equipment refresh programs, stable gastrointestinal disease diagnosis rates, and steady transition from manual to automated reprocessing workflows.
Demand for endoscope reprocessing device in Chubu is advancing at a CAGR of 4.9% through 2035, supported by general hospital coverage, steady diagnostic endoscopy use, and moderate capital equipment spending in Aichi. Chubu shows stronger reliance on shared reprocessing facilities across hospital departments rather than dedicated endoscopy units. Demand remains focused on mid capacity automated systems. Growth reflects workforce linked hospital utilization, routine gastrointestinal diagnostics, and gradual compliance upgrades for infection prevention standards.
Demand for endoscope reprocessing device in Tohoku is rising at a CAGR of 4.3% through 2035, supported by hospital consolidation, aging patient demographics, and stable gastrointestinal disease treatment volumes. Miyagi anchors most regional equipment installations through large referral hospitals. Tohoku reflects lower private clinic penetration and slower capital equipment renewal. Demand remains centered on essential automated reprocessing units used within hospital based endoscopy departments serving broad rural catchment areas.
Demand for endoscope reprocessing device in Rest of Japan is advancing at a CAGR of 4.1% through 2035, supported by small city hospital networks, routine diagnostic endoscopy services, and basic infection control compliance upgrades. These areas show limited adoption of high throughput systems due to lower procedure volumes. Demand remains focused on compact automated reprocessors serving outpatient and inpatient departments. Growth is steady and shaped by regulatory hygiene requirements and gradual healthcare facility modernization.
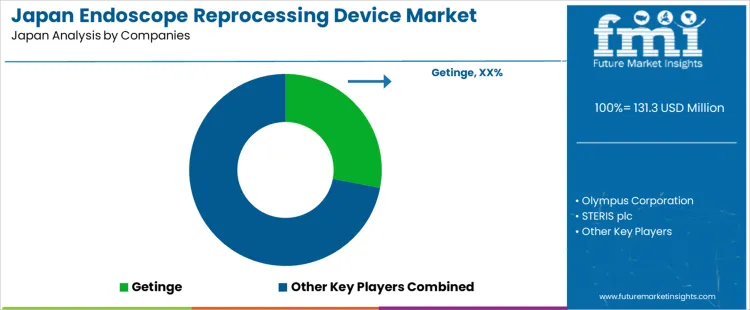
The demand for endoscope reprocessing devices in Japan is shaped by high procedural volumes in gastroenterology, strict infection control standards, and continuous regulatory oversight of hospital hygiene systems. Olympus Corporation holds a central domestic position through deep integration of endoscope platforms with dedicated washer disinfectors used across public and private hospitals. The company benefits from long standing service networks, user training programs, and compatibility between imaging systems and reprocessing workflows. Getinge supports large hospital deployment through automated washer disinfectors and drying cabinets used in high throughput endoscopy units. STERIS contributes through low temperature sterilization and reprocessing systems adopted by tertiary care hospitals and academic medical centers focused on multi-specialty infection control.
Steelco S.p.A. participates through imported automated cleaning and disinfection systems installed in selected surgical centers and teaching hospitals. ASP India Private Ltd. supports limited demand through chemical sterilants and low temperature processing solutions supplied through distributor led channels. System selection in Japan is governed by PMDA approval, microbial reduction performance, cycle time consistency, and compatibility with domestic endoscope inventories. Procurement decisions reflect central sterile department capacity, capital budgeting under prefectural planning, and long term maintenance coverage. Buyer preference favors suppliers with domestic engineering support, Japanese language software interfaces, and validated performance data under national infection prevention guidelines. Demand visibility tracks growth in colonoscopy screening programs, aging population health checks, and continued tightening of hospital hygiene audit requirements across regional medical networks.
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Product | Automated Endoscope Reprocessor, Washer Disinfector, Sterilizer, Ultrasonic Washer, Drying and Storage Cabinet |
| Operation Mode | Semi-Automatic, Automatic |
| Modality | Floor Standing/Bench Top, Mobile |
| End User | Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Academic & Research Institutes, Central Sterile Services Department (CSSD) |
| Region | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Getinge, Olympus Corporation, STERIS plc, ASP India Private Ltd., Steelco S.p.A. |
| Additional Attributes | Dollar by sales by product, operation mode, and modality; Regional CAGR and hospital adoption rates; Gastroenterology, pulmonology, and urology procedural throughput; Automated vs semi-automatic system utilization; Replacement demand and fleet modernization; Workflow integration and space optimization; Digital cycle tracking and audit readiness; Staff workload reduction metrics; Chemical compatibility and disinfectant dosing control; PMDA approval and infection control compliance; Import reliance for advanced control systems; Substitution pressure across washer/disinfector categories |
How big is the demand for endoscope reprocessing device in Japan in 2025?
The demand for endoscope reprocessing device in Japan is estimated to be valued at USD 131.3 million in 2025.
What will be the size of endoscope reprocessing device in Japan in 2035?
The market size for the endoscope reprocessing device in Japan is projected to reach USD 223.7 million by 2035.
How much will be the demand for endoscope reprocessing device in Japan growth between 2025 and 2035?
The demand for endoscope reprocessing device in Japan is expected to grow at a 5.5% CAGR between 2025 and 2035.
What are the key product types in the endoscope reprocessing device in Japan?
The key product types in endoscope reprocessing device in Japan are automated endoscope reprocessor, washer disinfector, sterilizer, ultrasonic washer and drying and storage cabinet.
Which operation mode segment is expected to contribute significant share in the endoscope reprocessing device in Japan in 2025?
In terms of operation mode, semi automatic segment is expected to command 57.0% share in the endoscope reprocessing device in Japan in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.