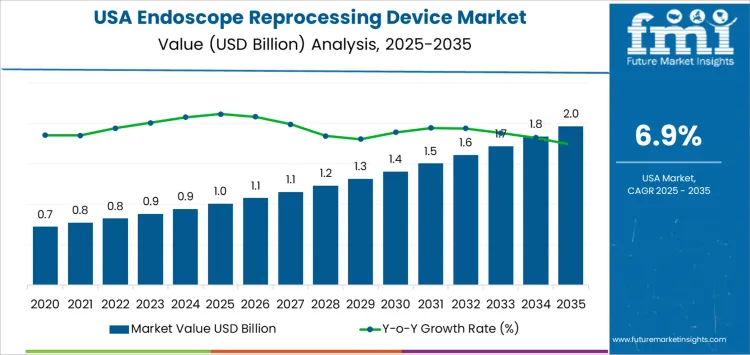
The demand for endoscope reprocessing devices in the USA is expected to grow from USD 1.0 billion in 2025 to USD 2.0 billion by 2035, reflecting a compound annual growth rate (CAGR) of 6.9%. This growth is driven by the increasing number of endoscopic procedures, the rising demand for high-quality, sterile medical equipment, and the growing awareness of the importance of proper disinfection and sterilization in healthcare settings.
Endoscope reprocessing devices play a critical role in ensuring that endoscopes are safely cleaned and disinfected between uses, preventing cross-contamination and ensuring patient safety in diagnostic and therapeutic procedures.
The market is also supported by the increasing focus on infection control and patient safety, particularly as healthcare institutions prioritize the reduction of hospital-acquired infections (HAIs). As healthcare facilities expand their endoscopic procedures and as regulations around disinfection practices tighten, the demand for advanced endoscope reprocessing systems is expected to grow. Technological advancements in automated reprocessing systems, which reduce human error and improve efficiency, further contribute to the growth of this market.
The contribution of volume vs price growth in the endoscope reprocessing device market reveals that volume growth plays a dominant role in the market's expansion, although price growth contributes in later stages as the market matures. Starting at USD 1.0 billion in 2025, the market grows gradually, reaching USD 1.1 billion in 2026 and USD 1.1 billion in 2027. These early years reflect steady volume-driven growth as the demand for endoscopic procedures continues to rise, supported by a focus on improving sterilization processes in healthcare settings.
From 2028 onward, volume growth remains a significant factor, with the market reaching USD 1.2 billion in 2028, USD 1.3 billion in 2029, and USD 1.4 billion in 2030. This steady increase reflects the ongoing expansion of endoscopic procedures and the widespread adoption of reprocessing devices. However, by 2031, the market begins to see a more significant influence from price growth, with higher-priced, more advanced automated reprocessing systems entering the market. The value of the market increases to USD 1.5 billion in 2031, USD 1.6 billion in 2032, and USD 1.7 billion in 2033.
By 2035, the market reaches USD 2.0 billion, with both volume and price contributing to the overall growth. This reflects the increased demand for more advanced, automated systems and the adoption of premium solutions in hospitals and healthcare institutions, where there is a stronger emphasis on quality, efficiency, and patient safety. The contribution of volume and price growth in this stage shows that while volume remains the primary driver in the early years, price growth becomes more important as the market matures and new, more advanced products are introduced.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 1 billion |
| Industry Forecast Value (2035) | USD 2 billion |
| Industry Forecast CAGR (2025-2035) | 6.9% |
Demand for endoscope reprocessing devices in the USA is rising because the number of endoscopic procedures is increasing - for diagnostics and minimally invasive surgeries in gastroenterology, pulmonology, urology, and other specialties. Each reusable endoscope must be thoroughly cleaned, disinfected or sterilized and dried after every use in order to prevent cross-contamination and transmission of infections between patients. This creates a steady need for reliable, efficient reprocessing devices to meet high procedure volumes while ensuring patient safety.
At the same time, stronger regulatory requirements and heightened awareness of infection control are driving adoption of automated reprocessing systems. Health agencies and professional guidelines emphasize strict compliance with validated cleaning and disinfection protocols for endoscopes. Automated endoscope reprocessors (AERs) and related devices help healthcare facilities ensure consistency, traceability, and compliance with safety standards - reducing the risk of healthcare-associated infections (HAIs) linked to improperly reprocessed scopes. Because of these factors - rising procedure volumes, infection control imperatives, and regulatory pressure - the market for endoscope reprocessing devices in the USA is expected to grow substantially in the coming years.
Overview The endoscope reprocessing device market in the USA is segmented by product type and operation mode. Automated endoscope reprocessors lead the product segment, accounting for 51% of the market share. In terms of operation mode, semi-automatic systems dominate, holding 57% of the market. The increasing demand for hygienic, efficient, and reliable reprocessing solutions for medical endoscopes is driving the adoption of advanced reprocessing devices across healthcare facilities in the USA.
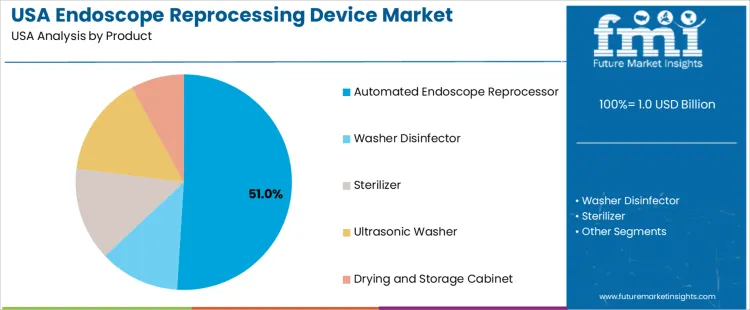
Automated endoscope reprocessors dominate the endoscope reprocessing device market, holding 51% of the total demand in the USA. These devices are preferred for their efficiency, consistency, and ability to ensure proper disinfection and sterilization of endoscopes. Automated reprocessing systems help healthcare facilities maintain high standards of infection control by automating the cleaning, disinfection, and drying processes. They are especially valuable in high-volume settings, such as hospitals and outpatient surgical centers, where large numbers of endoscopes must be processed quickly and thoroughly.
The growing emphasis on patient safety and reducing the risk of healthcare-associated infections is driving the widespread adoption of automated endoscope reprocessors. While other reprocessing devices, such as washer disinfectors, sterilizers, ultrasonic washers, and drying and storage cabinets, are also used, automated endoscope reprocessors remain the most popular due to their comprehensive features and enhanced efficiency in reprocessing medical instruments.
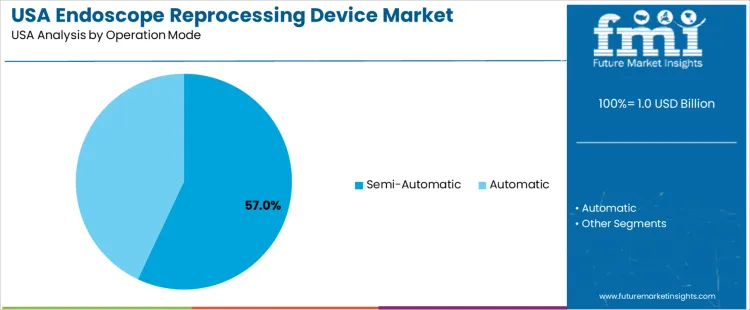
Semi-automatic systems are the most widely used operation mode for endoscope reprocessing devices, accounting for 57% of the market share. Semi-automatic systems offer a balance between manual oversight and automation, allowing healthcare providers to maintain some level of control over the reprocessing cycle while benefiting from automated features such as washing and disinfection. These systems are particularly appealing in settings where flexibility is important, and the need for consistent results is high.
Automatic systems, while gaining in popularity for their ease of use and complete automation, account for a smaller portion of the market compared to semi-automatic devices. The demand for semi-automatic systems continues to rise due to their versatility, cost-effectiveness, and ability to meet the reprocessing needs of healthcare facilities while maintaining high standards of infection control.
The demand for endoscope reprocessing devices in the USA is growing due to increasing awareness of the importance of infection control in medical settings. Endoscopes are critical tools in diagnostic and surgical procedures, but they can pose a risk for cross-contamination if not properly cleaned and disinfected. As healthcare facilities place greater emphasis on patient safety and infection prevention, the need for reliable and efficient endoscope reprocessing devices has risen. This market is expected to expand with advances in cleaning and sterilization technologies, as well as increasing regulations regarding reprocessing standards.
Several factors are driving the demand for endoscope reprocessing devices in the USA. First, the increasing number of minimally invasive procedures, such as gastrointestinal, respiratory, and urological endoscopies, is boosting the use of endoscopes and creating a need for efficient reprocessing systems. Second, the growing focus on infection control and the prevention of healthcare-associated infections (HAIs) is encouraging healthcare facilities to adopt advanced reprocessing devices that ensure thorough cleaning and sterilization of endoscopes.
Third, stringent regulatory standards and guidelines, such as those set by the FDA and CDC, are driving healthcare providers to invest in endoscope reprocessing devices to meet compliance requirements. Additionally, technological advancements in reprocessing systems, such as automated cleaning and disinfection technologies, are making these devices more efficient, reducing human error and improving overall patient safety.
Despite the growing demand, several factors may limit the adoption of endoscope reprocessing devices in the USA. One of the primary constraints is the high cost of advanced reprocessing devices, which can be a significant financial burden for smaller healthcare facilities or those with limited budgets. Another challenge is the complexity of some reprocessing systems, which may require specialized training for staff to operate effectively, creating barriers to implementation in certain settings.
Additionally, concerns about the effectiveness of reprocessing in eliminating pathogens, particularly with older equipment, can lead to hesitancy in adopting new devices. Lastly, variations in local or institutional policies regarding reprocessing methods may hinder the standardization and widespread use of endoscope reprocessing devices in some healthcare environments.
Several key trends are influencing the demand for endoscope reprocessing devices in the USA. One significant trend is the shift toward automation in reprocessing systems, which helps improve the efficiency, consistency, and safety of the cleaning process while reducing human error. Another trend is the increasing focus on sustainable and environmentally friendly reprocessing solutions, as healthcare facilities seek methods that minimize waste and environmental impact.
Additionally, the rise of digital technologies, such as monitoring systems that track and log cleaning processes, is enhancing the overall effectiveness of reprocessing devices by providing real-time feedback and documentation. The growing emphasis on infection prevention and patient safety in healthcare facilities is also driving the adoption of advanced endoscope reprocessing devices to meet regulatory standards and reduce the risk of cross-contamination.
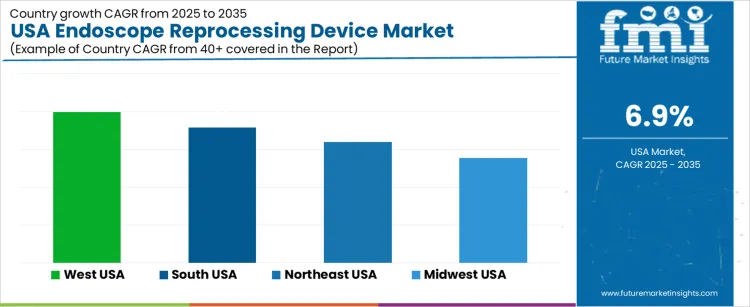
The Endoscope Reprocessing Device market in the USA is expected to grow steadily, with the West USA leading the way at a projected CAGR of 8.0%. South USA follows closely with a growth rate of 7.1%, while the Northeast USA is expected to grow at 6.4%. The Midwest USA shows moderate growth with a CAGR of 5.5%. Endoscope reprocessing devices, which are crucial in ensuring the safe use of endoscopes through proper cleaning and sterilization, are in high demand due to increasing healthcare regulations and the growing number of endoscopic procedures. As healthcare facilities continue to prioritize patient safety and infection control, the market for endoscope reprocessing devices is expected to rise across the United States.
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 8% |
| South USA | 7.1% |
| Northeast USA | 6.4% |
| Midwest USA | 5.5% |
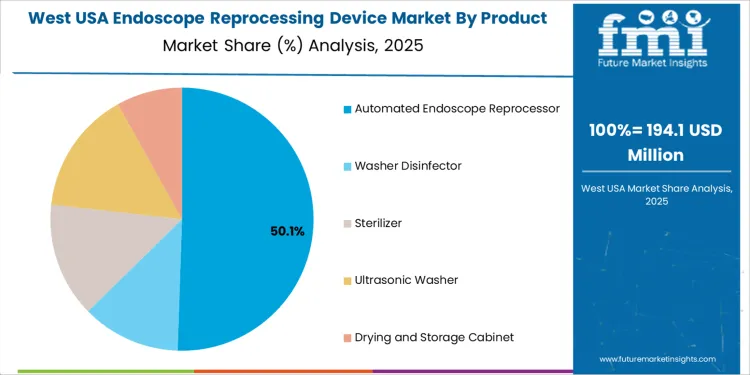
The West USA is projected to experience the highest growth in the Endoscope Reprocessing Device market, with a projected CAGR of 8.0%. The region’s healthcare systems, particularly in states like California, Washington, and Oregon, are increasingly adopting advanced infection control and sterilization practices. The West’s strong emphasis on patient safety, coupled with a large number of hospitals and medical centers performing endoscopic procedures, is a key driver of this growth.
With healthcare facilities under increasing regulatory pressure to maintain high standards of infection control, endoscope reprocessing devices have become essential. Additionally, the region’s focus on technological innovation and improving operational efficiency in healthcare is contributing to the demand for automated and effective endoscope reprocessing solutions. As the population in the West continues to grow and the number of endoscopic procedures increases, the market for these devices is expected to continue expanding.
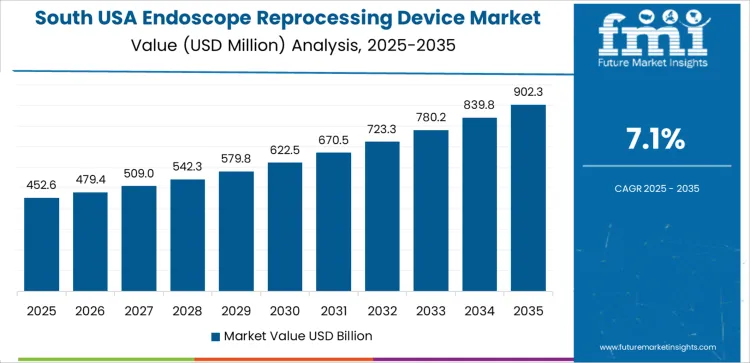
In South USA, the demand for endoscope reprocessing devices is projected to grow at a CAGR of 7.1%. The region’s expanding healthcare infrastructure and rising healthcare spending are significant factors in the adoption of these devices. States like Texas, Florida, and Georgia are seeing an increase in medical procedures involving endoscopy, driven by both an aging population and advancements in diagnostic technologies. With the rising number of endoscopic procedures, there is a growing need for proper sterilization and infection control, which drives the demand for endoscope reprocessing devices.
The South is also witnessing increased awareness of the importance of infection prevention in healthcare settings, further boosting the market. As healthcare providers in the South invest in modernizing their infection control processes, endoscope reprocessing devices are becoming a critical part of ensuring patient safety and improving the efficiency of medical operations.
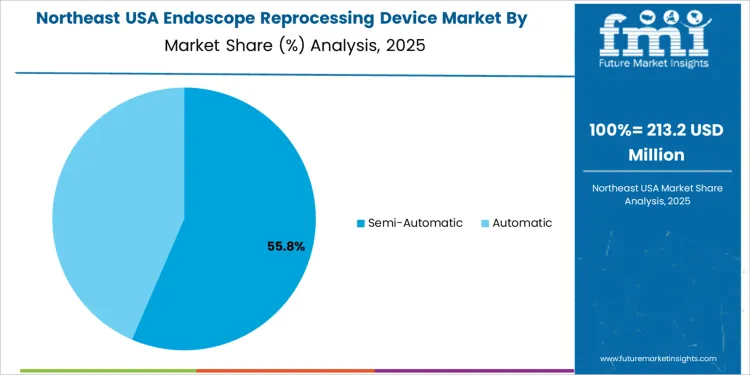
In Northeast USA, the Endoscope Reprocessing Device market is projected to grow at a CAGR of 6.4%. The region's well-established healthcare systems, particularly in cities like New York, Boston, and Philadelphia, are driving the demand for endoscopic procedures, which in turn increases the need for effective reprocessing solutions. The Northeast’s focus on providing high-quality care and adhering to stringent healthcare regulations is contributing to the adoption of endoscope reprocessing devices.
As healthcare facilities increasingly prioritize infection control and safe medical practices, these devices are essential in maintaining the cleanliness and safety of endoscopes used in diagnostic and therapeutic procedures. The growing awareness of the risks associated with improper reprocessing of medical equipment and the need for compliance with infection prevention protocols are also driving the market in the Northeast USA.
In Midwest USA, the Endoscope Reprocessing Device market is expected to grow at a more moderate rate of 5.5%. The Midwest’s healthcare systems, particularly in large cities like Chicago, Detroit, and Minneapolis, are focusing on improving operational efficiency and patient safety. As the number of endoscopic procedures rises, there is a growing need for devices that ensure proper cleaning and sterilization of medical equipment.
While the Midwest’s growth rate is slower compared to other regions, the demand for endoscope reprocessing devices is increasing as healthcare providers seek to meet infection control regulations and improve patient outcomes. The Midwest’s focus on improving healthcare infrastructure and expanding access to advanced medical technologies, including reprocessing solutions, is contributing to the steady growth of this market in the region.
Demand for endoscope reprocessing devices in the USA has surged in recent years as the volume of endoscopic procedures increases. Rising incidence of gastrointestinal disorders, cancers, and older-age related conditions leads to a growing number of diagnostic and therapeutic endoscopies. This growth makes reliable cleaning, disinfection, and re-processing essential to prevent healthcare-associated infections and ensure patient safety. Increasing regulatory scrutiny, stricter hygiene protocols, and growing awareness of risks associated with improperly cleaned endoscopes - especially reusable flexible endoscopes - further fuel demand for automated reprocessing solutions.
On the supply side, a handful of global medical-device and sterilization companies lead the market. According to your list, primary suppliers include Getinge, Olympus Corporation, STERIS plc, ASP India Private Ltd., and Steelco S.p.A. These firms provide a wide range of reprocessing solutions including automated endoscope reprocessors (AERs), high-level disinfectants, drying and storage systems, and traceability tools.
Competition among suppliers centers on disinfection reliability, throughput capacity, ability to meet regulatory requirements, ease of use in hospital sterile-processing departments, and total cost of ownership. Vendors that offer validated, efficient, and user-friendly reprocessing systems combined with strong service and training support are best positioned to capture growth as USA hospitals and ambulatory surgery centers expand endoscopy services and tighten infection-control standards.
| Items | Details |
|---|---|
| Quantitative Units | USD Billion |
| Regions Covered | USA |
| Product | Automated Endoscope Reprocessor; Washer Disinfector; Sterilizer; Ultrasonic Washer; Drying & Storage Cabinet |
| Operation Mode | Semi-Automatic; Automatic |
| Modality | Floor-standing / Bench-top; Mobile units |
| End User | Hospitals; Specialty Clinics; Ambulatory Surgical Centers (ASCs); Academic & Research Institutes; Central Sterile Services Departments (CSSD) |
| Key Companies Profiled | Getinge; Olympus Corporation; STERIS plc; ASP India Private Ltd.; Steelco S.p.A. |
| Additional Attributes | Dollar sales by device type and end-user reflect increasing adoption of automated and high-efficiency reprocessing systems. Growth is driven by rising endoscopic procedure volume, stricter infection-control regulations, and demand for standardized disinfection and sterilization workflows in hospitals, ASCs, and clinics. |
How big is the demand for endoscope reprocessing device in USA in 2025?
The demand for endoscope reprocessing device in USA is estimated to be valued at USD 1.0 billion in 2025.
What will be the size of endoscope reprocessing device in USA in 2035?
The market size for the endoscope reprocessing device in USA is projected to reach USD 2.0 billion by 2035.
How much will be the demand for endoscope reprocessing device in USA growth between 2025 and 2035?
The demand for endoscope reprocessing device in USA is expected to grow at a 6.9% CAGR between 2025 and 2035.
What are the key product types in the endoscope reprocessing device in USA?
The key product types in endoscope reprocessing device in USA are automated endoscope reprocessor, washer disinfector, sterilizer, ultrasonic washer and drying and storage cabinet.
Which operation mode segment is expected to contribute significant share in the endoscope reprocessing device in USA in 2025?
In terms of operation mode, semi-automatic segment is expected to command 57.0% share in the endoscope reprocessing device in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.