The medical sharps market is expected to expand from USD 10.9 billion in 2026 to USD 45.3 billion by 2036, registering a CAGR of 15.3% over the forecast period. Growth momentum is underpinned by rising injection volumes, expanding vaccination coverage, and tighter enforcement of occupational safety regulations across healthcare systems. Safety-engineered sharps are increasingly treated as a compliance requirement rather than a discretionary upgrade, strengthening baseline demand across hospitals, clinics, and home-care settings. However, execution risk remains uneven.
Adoption friction persists in cost-sensitive markets where conventional sharps still dominate procurement decisions, slowing full conversion to higher-priced safety devices. Supply reliability, training consistency, and disposal infrastructure also influence outcomes, particularly in decentralized care environments. From an economic standpoint, demand demonstrates low elasticity due to the non-substitutable nature of sharps in clinical care, yet margin pressure is shaped by public tendering, reimbursement limits, and rising waste-management costs. Between 2026 and 2036, performance will favor manufacturers that balance regulatory compliance, scalable production, and disciplined pricing strategies while navigating regional differences in safety enforcement and healthcare spending sensitivity.
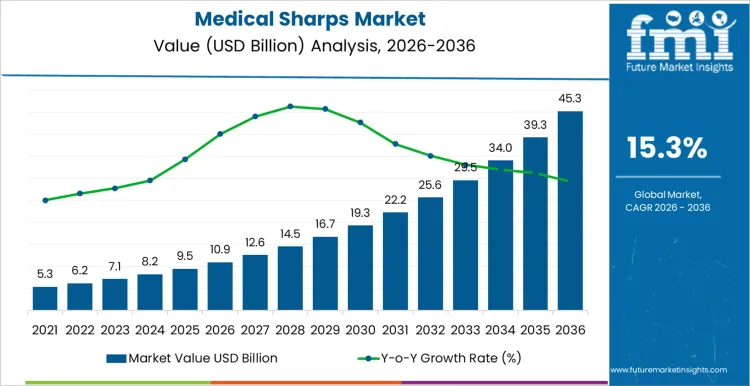
| Metric | Value |
|---|---|
| Medical Sharps Market Value (2026) | USD 10.9 Billion |
| Medical Sharps Market Forecast Value (2036) | USD 45.3 Billion |
| Medical Sharps Market Forecast CAGR 2026 to 2036 | 15.3% |
The medical sharps market is undergoing rapid expansion as healthcare systems intensify focus on needlestick injury prevention and occupational safety. Regulatory mandates and hospital accreditation standards are driving widespread adoption of safety-engineered sharps, including retractable needles, shielded lancets, and passive safety mechanisms. For manufacturers, demand is shifting from basic consumables toward devices that integrate safety features without adding procedural complexity or compromising clinical performance.
Procurement decisions are increasingly driven by total cost of ownership rather than unit pricing. Hospitals and outpatient facilities are evaluating sharps based on injury reduction outcomes, training requirements, and compatibility with existing clinical workflows. Products that reduce disposal volume, support standardized use across departments, and minimize user-dependent activation errors are gaining preference. Suppliers offering bundled portfolios combining safety sharps with compliant containers and training support are strengthening their position with group purchasing organizations and integrated delivery networks.
Disposal infrastructure and waste-management economics are also influencing market dynamics. Rising volumes of sharps waste are increasing pressure on healthcare providers to adopt optimized containment, tracking, and disposal solutions. This is creating opportunities for sharps manufacturers and waste partners that can demonstrate reduced handling risk, improved traceability, and compliance with evolving environmental and reporting standards. For companies across the value chain, competitive advantage in the medical sharps market increasingly depends on regulatory alignment, device reliability, and the ability to support scalable, system-wide safety programs positioning medical sharps as a high-growth, safety-critical segment of global healthcare supplies.
The medical sharps market is segmented by product and application, reflecting how clinical usage frequency and care settings influence demand patterns. By product, hypodermic needles represent the leading category, supported by their essential role across routine and specialized medical procedures. Other products include syringes with needles, lancets, blood collection needles, surgical blades and scalpels, pen needles, and other sharps used across inpatient and outpatient care. By application, drug delivery and injections form the largest demand area, followed by blood collection and diagnostics, diabetes management, vaccination programs, and surgical procedures, highlighting the broad clinical reliance on sharps-based devices.
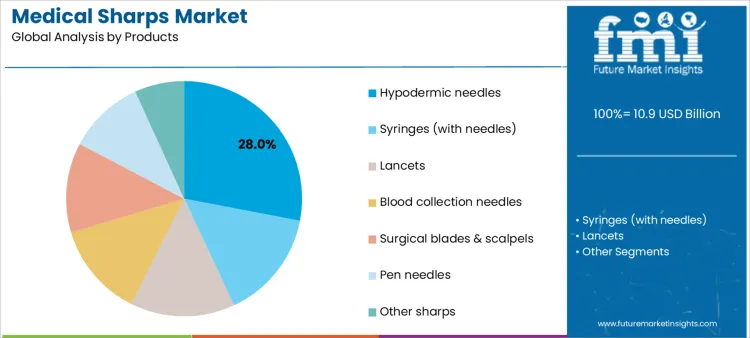
Hypodermic needles account for 28% share of the medical sharps market because they are universally required for injections, fluid delivery, and medication administration. Hospitals, clinics, and home-care settings use hypodermic needles across therapeutic areas such as antibiotics, analgesics, insulin, and biologics. Their standardized design enables compatibility with multiple syringe types and delivery systems. High procedure frequency drives continuous replacement demand. Safety-engineered variants further expand usage in regulated healthcare environments. For manufacturers, hypodermic needles offer high-volume production, predictable procurement cycles, and broad geographic demand. These clinical necessity and scale advantages explain their leading position among medical sharps products.
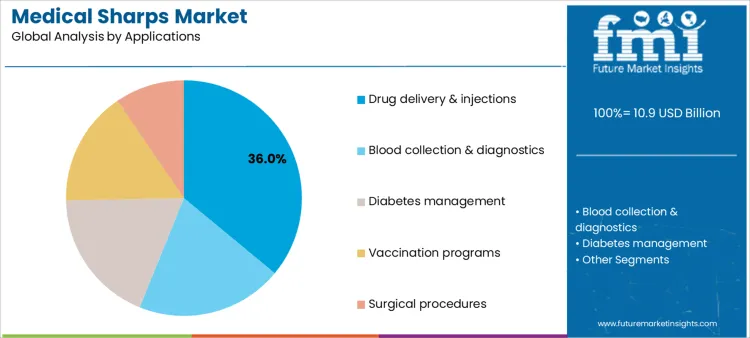
Drug delivery and injections hold 36% share of the medical sharps market because injectable therapies remain central to acute and chronic disease management. Many medications require parenteral administration to ensure rapid onset or accurate dosing. Hospitals and outpatient facilities perform high volumes of injections for treatment, prevention, and supportive care. Growth in chronic disease prevalence increases reliance on injectable therapies administered in both clinical and home settings. Vaccines, biologics, and emergency medications further contribute to injection frequency. For sharps suppliers, this application segment ensures consistent demand tied to patient volume and treatment intensity, explaining its dominance.
The medical sharps market is driven by the widespread need for devices that enable safe and effective collection, containment, and disposal of needles, scalpels, lancets, and other sharp instruments used in healthcare, home care, and clinical settings. Rising incidence of chronic diseases such as diabetes increases use of self-administered injections, expanding demand for sharps with safety features. Healthcare facilities and clinics require compliant containers and sharps systems to minimize occupational injuries and infection risks. For manufacturers and suppliers, product safety design, regulatory compliance, ease of use, and compatibility with waste management infrastructure are critical factors influencing procurement decisions by hospitals, long-term care facilities, and home health providers.
Safety and design trends are shaping the medical sharps market by emphasizing engineered protection mechanisms, ergonomic features, and disposal convenience. Sharps with built-in safety guards, automatic retraction, and tamper-resistant closures reduce needlestick injury risk for clinicians and patients. Standardized container designs with secure locking lids and fill-level indicators improve handling and reduce exposure risks during transport and disposal. Innovations in compact, lightweight, and wall-mountable designs support space-constrained environments and home care adoption. For suppliers, offering solutions that align with occupational safety guidelines and ease training for end users strengthens product acceptance and repeat purchasing.
Regulatory, disposal, and cost challenges restrain growth in the medical sharps market by adding complexity and operating expense for healthcare providers and waste handlers. Strict regulations govern sharps collection, labeling, transport, and disposal to prevent injury and contamination, requiring facilities to maintain compliant waste streams and documentation. Investment in approved sharps containers, service contracts with waste disposal vendors, and staff training increases operational costs. Home care sharps disposal presents unique challenges as patients must comply with local ordinances for safe disposal, sometimes limiting utilization of advanced safety devices. Price sensitivity in outpatient and home settings can constrain rapid adoption of premium sharps products.
The medical sharps market is expanding steadily as healthcare systems continue to rely on needles, syringes, lancets, and other sharp instruments for diagnostics, drug delivery, vaccination, and chronic disease management. Demand is driven by rising procedure volumes across hospitals, clinics, laboratories, and home care settings. Growth varies by country based on healthcare access, injection frequency, adoption of safety-engineered sharps, and regulatory requirements for injury prevention. Mature markets emphasize safety mechanisms, compliance with occupational health standards, and replacement of conventional devices, while higher-growth regions are supported by expanding healthcare infrastructure, vaccination programs, and chronic disease prevalence. Market value is shaped by recurring consumption, regulatory-driven product upgrades, and distribution reach across institutional and retail healthcare channels.
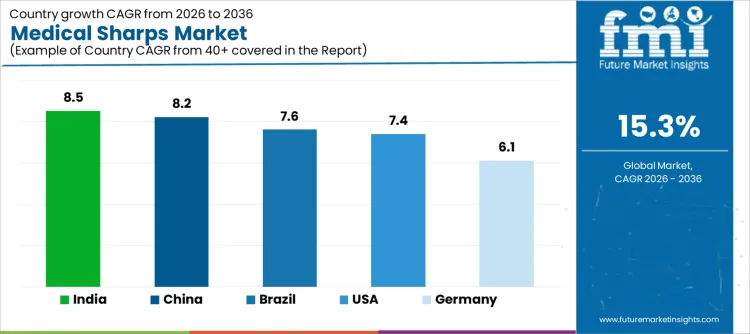
| Country | CAGR (%) |
|---|---|
| India | 8.5 |
| China | 8.2 |
| Brazil | 7.6 |
| United States | 7.4 |
| Germany | 6.1 |
The medical sharps market in the United States is expanding at a CAGR of 7.4% during 2026-2036, driven by consistent demand across hospitals, clinics, laboratories, and home healthcare settings. High injection volumes associated with vaccination programs, diabetes management, oncology care, and diagnostic testing sustain market demand. Adoption of safety-engineered sharps is widespread, supported by strict occupational safety regulations and strong enforcement. Buyers prioritize needle-stick injury prevention, product reliability, and compatibility with established clinical workflows. Procurement decisions are influenced by hospital safety committees, group purchasing organizations, and long-term supplier contracts. Market value is supported by recurring consumption, replacement of conventional sharps with safety variants, and expansion of home-based care. The United States represents a mature market where growth is steady and driven by safety compliance, procedure volume stability, and incremental product upgrades rather than rapid expansion.
Brazil’s medical sharps market is growing at a CAGR of 7.6% during 2026-2036, supported by expanding healthcare access and rising procedure volumes. Demand is driven by vaccination programs, chronic disease treatment, and diagnostic testing in public and private healthcare facilities. Adoption of safety-engineered sharps is increasing, although conventional devices remain in use due to cost considerations. Buyers emphasize affordability, availability, and durability of products. Procurement decisions are influenced by public healthcare budgets, distributor reach, and clinician familiarity. Market growth is reinforced by expansion of healthcare infrastructure in urban centers and improving access in regional hospitals. Brazil represents a developing market where growth is driven by increasing procedure volumes, gradual adoption of safer devices, and improved distribution coverage across healthcare systems.
China’s medical sharps market is expanding at a CAGR of 8.2% during 2026-2036, driven by rising healthcare utilization and large-scale vaccination and diagnostic programs. Demand is supported by growing hospital networks and increased access to injectable therapies. Safety-engineered sharps are gaining traction, particularly in major hospitals, while conventional sharps remain common in smaller facilities. Buyers prioritize supply reliability, cost control, and compliance with national standards. Procurement decisions favor suppliers with local manufacturing, regulatory approvals, and high production capacity. Market value is driven by population scale, increasing procedure volumes, and standardization of medical practices. China represents a volume-driven market where competitiveness depends on manufacturing scale, distribution strength, and ability to meet high demand consistently.
India’s medical sharps market is projected to grow at a CAGR of 8.5% during 2026-2036, driven by expanding healthcare access and increasing reliance on injectable therapies across public and private care settings. Large-scale vaccination programs, rising diabetes prevalence, oncology treatment, and infectious disease management continue to generate high sharps consumption. Adoption of safety-engineered sharps is gaining momentum, particularly in tier-one urban hospitals and accredited facilities, while conventional low-cost sharps remain widely used in cost-sensitive environments. Buyers prioritize affordability, consistent availability, and ease of use due to high patient throughput and operational constraints. Procurement cycles are relatively short, allowing hospitals and clinics to switch suppliers quickly when pricing or supply conditions change. Market value is supported by frequent injection use, growing diagnostic activity, and continued expansion of hospital and clinic infrastructure. India represents a high-growth, volume-driven market where competitive success depends on pricing flexibility, reliable distribution coverage, and the ability to serve diverse care settings at scale.
Germany’s medical sharps market is growing at a CAGR of 6.1% during the forecast period, reflecting a highly regulated and protocol-driven healthcare environment. Healthcare providers emphasize strict compliance with occupational safety standards and routinely prioritize safety-engineered sharps to reduce needle-stick injuries. Demand is stable, supported by routine clinical procedures, chronic disease management, and laboratory diagnostics rather than rapid expansion of injection volumes. Procurement decisions are typically managed through hospital committees and insurance-aligned purchasing frameworks, resulting in extended evaluation and approval timelines. Product selection is guided by documented safety performance, regulatory certification, and long-term supplier reliability. Replacement demand, rather than first-time adoption, drives market value as facilities update existing inventories to meet evolving safety requirements. Germany represents a mature market where growth is steady and predictable, shaped by compliance obligations, clinical governance, and disciplined procurement practices rather than aggressive volume growth.
Competition in the medical sharps market is driven by product safety features, ergonomic design, and documented performance that matter to clinicians, infection control teams, and procurement decision-makers. BD (Becton Dickinson) positions its sharps portfolio around engineered needle and lancet systems with official materials emphasizing safety-engineered protective mechanisms, sterile integrity, and compliance with global needlestick injury prevention standards. Brochures highlight passive safety features and clear activation feedback to support healthcare worker safety and reduce exposure risk.
Terumo Corporation competes through precision manufacturing and clinically validated sharp instruments designed for consistent penetration force and minimal tissue trauma. Product literature underscores ergonomic housings, dimensional control, and validated lot traceability, appealing to high-volume infusion, injection, and blood collection settings. B. Braun Melsungen differentiates by aligning sharps with broader infusion and anesthesia platforms, with documentation that stresses integrated safety features and compatibility with complementary devices such as IV sets and catheters.
Regional manufacturers and specialty sharps expand competitive breadth. Nipro Corporation offers precision needles and syringes with materials highlighting bidirectional safety protection and manufacturing control, supported by sterile packaging designed for busy clinical environments. Smiths Medical positions its sharps with official brochures focused on reliable performance in critical care and perioperative settings, underscoring secure handling and reduced risk of accidental sticks.
Others in the market include niche sharps producers and regional suppliers that offer custom gauges, specialty lancets, and economically positioned disposable systems. These products are supported by technical specifications on gauge consistency, material composition, and sterilization standards. Across all suppliers, competitive differentiation is built on brochure-verified safety mechanisms, ergonomic design, and documented clinical acceptance rather than price alone.
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Billion |
| Products | Hypodermic Needles, Syringes (with Needles), Lancets, Blood Collection Needles, Surgical Blades & Scalpels, Pen Needles, Other Sharps |
| Applications | Drug Delivery & Injections, Blood Collection & Diagnostics, Diabetes Management, Vaccination Programs, Surgical Procedures |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | BD (Becton Dickinson), Terumo Corporation, B. Braun Melsungen AG, Nipro Corporation, Smiths Medical, Others |
| Additional Attributes | Dollar sales by product type and application are evaluated across hospitals, clinics, diagnostics labs, and home-care settings. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of safety-engineered sharps adoption, evaluation of infection-control compliance, competitive benchmarking of manufacturers, and assessment of vaccination expansion and chronic disease management trends globally. |
How big is the medical sharps market in 2026?
The global medical sharps market is estimated to be valued at USD 10.9 billion in 2026.
What will be the size of medical sharps market in 2036?
The market size for the medical sharps market is projected to reach USD 45.3 billion by 2036.
How much will be the medical sharps market growth between 2026 and 2036?
The medical sharps market is expected to grow at a 15.3% CAGR between 2026 and 2036.
What are the key product types in the medical sharps market?
The key product types in medical sharps market are hypodermic needles, syringes (with needles), lancets, blood collection needles, surgical blades & scalpels, pen needles and other sharps.
Which applications segment to contribute significant share in the medical sharps market in 2026?
In terms of applications, drug delivery & injections segment to command 36.0% share in the medical sharps market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.