The global pancreatic stone protein testing sector is on track to achieve a valuation of USD 2.89 billion by 2036, accelerating from USD 2.30 billion in 2026 at a CAGR of 2.3%. As per Future Market Insights, expansion is structurally underpinned by the growing clinical validation of PSP as an early sepsis biomarker that can detect infection-driven organ dysfunction 24 to 48 hours before conventional markers.
The World Health Organization recognized sepsis as a global health priority in its 2017 resolution WHA70.7, and subsequent WHO reports have continued to emphasize that early detection remains the single most effective intervention for reducing sepsis mortality. This institutional recognition compels hospital systems and emergency departments to evaluate novel biomarkers that can accelerate time-to-detection. Simultaneously, the regulatory landscape is shifting from research-use-only PSP assays to cleared in-vitro diagnostic (IVD) platforms, which forces a transition in how clinicians integrate PSP results into existing sepsis screening protocols.
Patrick Pestalozzi, CEO of Abionic, stated: 'This clearance will allow us to deploy our solutions across the United States and provide clinicians in acute care settings with a proven solution to accelerate the Time-To-Detection of sepsis.' This statement followed Abionic's October 2024 receipt of 510(k) clearance from the USA FDA for its IVD Capsule PSP test, which runs on the nanofluidic abioSCOPE platform and delivers results from a single drop of blood in five minutes. FMI opines that the USA market entry of PSP-based diagnostics is the most significant inflection point for this biomarker since its initial clinical adoption in European ICUs. The 510(k) pathway, rather than a de novo classification, indicates that the FDA recognized sufficient predicate equivalence, which will streamline future PSP assay submissions from competing manufacturers.
The operational reality for PSP testing suppliers is defined by the need to expand clinical adoption beyond adult ICU settings into pediatric and emergency department workflows. In January 2026, Abionic announced that its 7-minute PSP test has been approved for pediatric use, extending its IVDR-compliant coverage from adults to children.
As per FMI, this pediatric extension is clinically significant because sepsis is a leading cause of death among pediatric patients globally, and existing biomarkers have well-documented limitations in sensitivity for neonatal and pediatric populations. The expansion of PSP testing into pediatric applications creates a new addressable segment that had been previously underserved by rapid biomarker platforms. Abionic's nanofluidic platform architecture, which requires only a capillary blood sample, is particularly well-suited to pediatric settings where venous access is often difficult and sample volumes are limited.
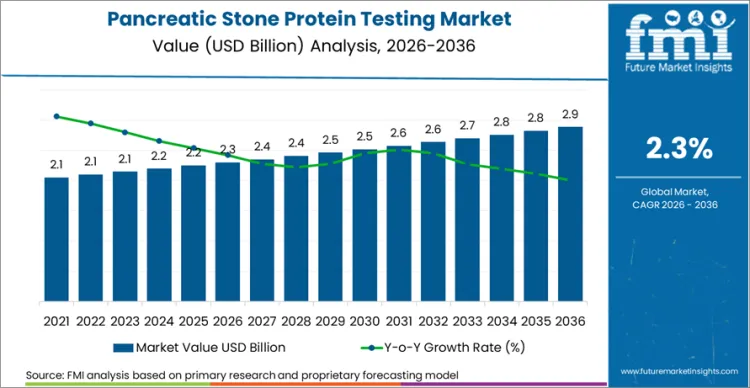
Future Market Insights projects the pancreatic stone protein testing market to expand at a CAGR of 2.3% from 2026 to 2036, increasing from USD 2.30 Billion in 2026 to USD 2.89 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on sepsis diagnostic test volumes and ICU bed utilization rates.
FMI analysts perceive the market evolving from a niche European ICU biomarker toward a globally adopted rapid sepsis screening tool, with USA FDA clearance being the inflection point for volume growth.
FMI Research Approach: Abionic 510(k) FDA clearance (October 2024) and IVDR pediatric extension (January 2026).
The United States holds a significant share of the global pancreatic stone protein testing market by value, which is supported by the 2024 FDA 510(k) clearance and the largest installed base of acute care hospital beds globally.
FMI Research Approach: FMI country-level revenue modeling by ICU bed counts and sepsis diagnostic test procurement data.
The global pancreatic stone protein testing market is projected to reach USD 2.89 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from WHO sepsis prevalence estimates and point-of-care diagnostic adoption curves.
The pancreatic stone protein testing market includes revenue from IVD assays and point-of-care testing platforms that measure PSP levels in blood for the early detection and monitoring of sepsis and infection-related organ dysfunction.
FMI Research Approach: FMI market taxonomy aligned with WHO ICD sepsis classifications and IVD regulatory frameworks.
Globally unique trends include the FDA 510(k) clearance of the first PSP rapid test, the extension of PSP diagnostics into pediatric populations, and the integration of nanofluidic platforms that require only capillary blood samples.
FMI Research Approach: Abionic FDA clearance (October 2024) and pediatric PSP test approval (January 2026).
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.30 Billion |
| Industry Value (2036) | USD 2.89 Billion |
| CAGR (2026 to 2036) | 2.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Sepsis, which is a life-threatening ailment, caused due to the body’s extreme response to infection is a major factor behind mortality in hospitals. Its occurrence is surging across the globe, specifically in aging demographics.
As per ENDSEPSIS.ORG, the USA reports over 1.5 million cases of sepsis yearly. In UK also, the number of cases of sepsis reached 245,000 annually, as per the UK Sepsis Trust. Its early detection using biomarkers, including pancreatic stone protein, has shown significant improvements in patient outcomes, boosting the demand for such testing.
Healthcare institutions and governments across the world are offering support for funding the development of diagnostic technologies, such as PSP testing to decrease mortalities related to acute inflammatory conditions and sepsis. As per an ENDSEPSIS report, CDC was awarded USD 3 million in the FY24 Congressional Budget to address sepsis. Citing another instance, in December 2024, the UK government allocated £100 million for public-private investments to construct 20 research facilities.
The critical care segment is witnessing advancements in PSP testing, specifically in point-of-care diagnostics. Additionally, accurate and quick outcomes are offered by ELISA-based and immunoassay-based PSP tests, which decrease the diagnostic turnaround time. Owing to their efficiency in critical care situations, multiplex assays which can identify multiple biomarkers simultaneously have gained popularity.
PSP testing is being widely embraced by healthcare providers as a part of their daily critical care monitoring to identify early signs of infection. This is owing to PSP’s excellence as compared to other conventional biomarkers such as procalcitonin (PCT) in the diagnosis of sepsis.
Deployment of AI in Diagnostics
AI algorithms are now being used with pancreatic stone protein testing platforms to enhance the accuracy of diagnostic interpretations and offer predictive insights for the management of patients. AI-powered platforms can determine PSP levels with other biomarkers to identify early warning signs of deadly diseases including sepsis.
Moreover, AI minimizes manual errors in diagnosis and provides improved patient outcomes. Additionally, predictive analytics from AI tools help in proactive interventions, which streamlines resource allocation.
Enhancement in Multiplex Diagnostic
Multiplex assays that detect multiple biomarkers, including PSP, in a single test are gaining popularity. They boost efficiency and lower costs by offering comprehensive diagnostics with minimal sample volume. Moreover, they also enhance workflows in laboratories and ICUs, minimizing the time and labor required for diagnostics. By merging PSP with other biomarkers such as C- C-reactive protein and Procalcitonin, these assays increase the clinical utility of PSP testing.
Growing Use of Telemedicine and Remote Diagnostics
PSP testing and telemedicine platforms are being jointly used, which allows for remote diagnostics and consultation. Remote diagnostic capabilities are particularly useful in managing critical care in regions with finite healthcare access. Additionally, early detection of diseases such as sepsis has now been made possible through telemedicine, which helps reduce the incidence of delayed treatment.
Moreover, many healthcare companies are also investing heavily in telemedicine to boost their diagnostic capabilities. In March 2021, Abbott launched NeuroSphere™ Virtual Clinic in the USA, the first technology that enables communication between physicians and patients, which helps patients get remote treatment. This clinic has also got approval from the USA FDA and can help patients suffering from movement disorders or chronic pain.
Prohibitive Costs and Restricted Accessibility Can Hamper Sales
The costs related to PSP testing devices and kits are more than those of conventional diagnostic devices, including Procalcitonin (PCT) or c-reactive protein (CRP) tests. This relatively higher cost makes these tests out of reach for healthcare providers. Additionally, clinics and hospitals in rural areas often do not have the infrastructure or financial resources to spend on PSP testing equipment. For instance, in areas including sub-Saharan Africa and parts of Southeast Asia, the steep cost prevents the integration of PSP testing into daily diagnostic workflows.

By Product Type
| Attributes | Details |
|---|---|
| Top Product Type | PSP Testing Kits |
| Market Share in 2026 | 47.9% |
By product type, the market is divided into PSP testing kits, laboratory-based PSP assays, and point-of-care (POC) PSP devices. The PSP testing kits segment leads accounting for 47.9% of the market share in 2026. PSP testing kits are becoming popular owing to innovations in PSP testing, specifically in point-of-care (POC) diagnostics, which are transforming the critical care segment.
Moreover, immunoassay-based and ELISA-based PSP tests offer accurate and quick outcomes, which decreases the diagnostic turnaround time. Additionally, multiplex assays, which can detect multiple biomarkers simultaneously, are witnessing momentum due to their efficiency in critical care scenarios.
By application, the market is segmented into acute pancreatitis monitoring, critical care monitoring, systemic inflammatory response syndrome (SIRS) diagnosis, and sepsis diagnosis. PSP testing is being widely used for acute pancreatitis monitoring.
The rising cases of acute pancreatitis are boosting the demand for such testing solutions. According to Pancreas.org, around 50,000 to 80,000 cases of acute pancreatitis occur in the USA every year.
By technology, the market is divided into immunoassay-based testing, point-of-care diagnostic devices, ELISA-based testing, and multiplex assays. The recent innovations in point-of-care biosensors for pancreatic disorders are increasing the demand for point-of-care diagnostic devices. Additionally, the high accuracy, sensitivity, and predictive value of point-of-care devices is further augmenting segment expansion.

By End-user
| Attributes | Details |
|---|---|
| Top End-user Type | Hospitals |
| Market Share in 2026 | 38.9% |
By end-user, the market is divided into specialty clinics, academic and research institutions, diagnostic laboratories, and hospitals. The hospitals segment holds 38.9% of the market share in 2026. Hospitals are growingly adopting PSP testing as part of their daily critical care monitoring to determine early signs of systemic inflammation or infection. This can be credited to PSP’s superiority over traditional biomarkers such as procalcitonin (PCT) in diagnosing sepsis.
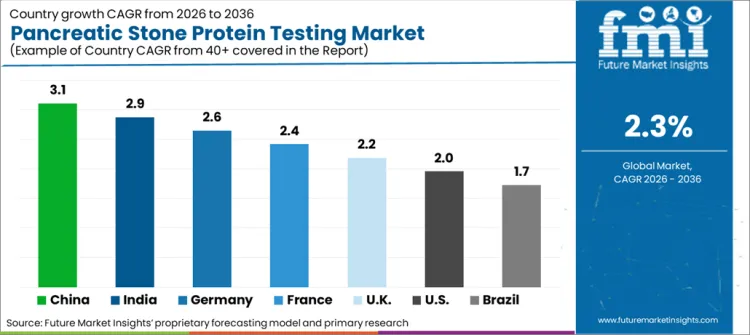
India is expected to grow at a CAGR of 3.2% during the forecast period. The quick embracement of PSP testing in emerging nations, such as China, and Southeast Asia, owing to growing investments in healthcare and rising cases of critical conditions is leading to regional expansion.
Additionally, the emphasis of governments in these regions is on improving healthcare infrastructure and providing subsidies for diagnostic devices for underserved populations.
Furthermore, the availability of a well-established healthcare system in Japan is contributing to market growth. The aging demographic in the nation is resulting in a greater prevalence of sepsis, which is contributing to higher uptake of advanced diagnostic devices.
Additionally, PSP testing kits with enhanced specificity and sensitivity are being launched by AMS Biotechnology and Creative Diagnostics, which is further adding to the market growth. The market expansion in the country can further be credited to the growing incidence of systemic inflammatory diseases.
In North America, the USA is slated to expand at a CAGR of 0.9% during the forecast period. It dominates the market owing to the rising cases of critical diseases such as sepsis and acute pancreatitis. The market growth is also driven by the increasing uptake of POC diagnostics in hospitals and ICUs to decrease deaths associated with sepsis. Moreover, significant investment in research & development by major companies including Ray Biotech and Thermo Fisher Scientific is being made in the region.
In Europe, Germany is the largest market for PSP testing, driven by government-supported healthcare policies and a robust emphasis on early diagnosis of sepsis and inflammatory diseases.
High uptake of multiplex diagnostic assays in hospitals and diagnostic laboratories improves PSP testing perception. The presence of a strong healthcare infrastructure ensures the affordability and accessibility of advanced diagnostic devices. Besides this, growing awareness among healthcare professionals about PSP as a sepsis biomarker is further contributing to market growth.
In the UK, the uptake of PSP testing for sepsis and other critical diseases is being encouraged by the UK’s National Health Service (NHS) owing to its emphasis on enhancing healthcare efficiency and decreasing mortalities.
The rising occurrences of sepsis and awareness campaigns by organizations such as Sepsis Trust UK are propelling market growth in the region. The UK Sepsis Trust has reported that around 200,000 cases of sepsis occur in adults yearly in the country.
The UK has the presence of cutting-edge academic research facilities and diagnostic laboratories which is contributing to innovations in pancreatic stone protein testing.
Significant innovation is taking place in the market and major players such as Thermo Fisher Scientific, Ray Biotech, Inc., and Lifespan Biosciences Inc. are heavily investing in R&D to improve the sensitivity, specificity, and turnaround time of PSP diagnostic kits. Moreover, multiplex assays and point-of-care (POC) devices are being focused on by companies to fulfill the growing demand for rapid, bedside diagnostics.
Furthermore, few companies are dealing with research institutions and academics to discover new applications of PSP testing for critical care, such as chronic inflammation monitoring and oncology diagnostics. These deals are boosting innovation and helping companies to expand their product portfolios.
The USA FDA, European Medicines Agency (EMA), and counterparts in Asia-Pacific regions are using different strategies to increase the uptake of advanced diagnostic devices.
Despite this, receiving regulatory approvals is a lengthy process and requires huge documentation. Different market players, particularly in Europe are focusing on regulatory harmonization efforts to enter the market. However, issues including changing reimbursement policies, along with the slow inclusion of PSP testing in public healthcare programs hamper bulk adoption.
| Company | Area of focus |
|---|---|
| Thermo Fisher Scientific | Diagnostic testing capabilities |
| Ray Biotech | Collaborations with regional distributors |
| Abnova Corporation | Forming alliances with biotechnology firms |
| Creative Diagnostics | Forming alliances with biotechnology firms |
Market players are using different strategies including acquisitions, partnerships, and mergers to enhance their position. For instance, Thermo Fisher Scientific’s acquisition of PPD has improved its diagnostic testing capabilities. Additionally, Ray Biotech has collaborated with regional distributors to expand its presence in emerging markets.
Similarly, Creative Diagnostics and Abnova Corporation are focusing on alliance formation with biotechnology companies to jointly develop cost-effective testing solutions for underserved regions. Moreover, smaller companies and start-ups with niche expertise in PSP biomarker technologies are being used by large companies to increase their market touch and technological capabilities.
Recent Developments
The pancreatic stone protein testing market represents revenue generated from in-vitro diagnostic assays and point-of-care testing platforms that measure pancreatic stone protein (PSP) concentrations in blood samples for the early detection, risk stratification, and monitoring of sepsis and systemic infection.
Inclusions cover IVD cartridge-based PSP assays, nanofluidic point-of-care PSP platforms, laboratory-based PSP immunoassays, and associated reader hardware. It includes consumables such as test cartridges and calibration materials. Both adult and pediatric PSP testing applications are included.
Exclusions include general procalcitonin (PCT) and C-reactive protein (CRP) assays not specific to PSP measurement, broad-spectrum sepsis panel tests that do not isolate PSP as a distinct biomarker, and research-use-only PSP reagents not cleared for clinical diagnostic use.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 2.30 Billion |
| Product Type | Point-of-Care PSP Assays, Laboratory PSP Immunoassays, Reader Hardware, Consumables |
| Application | Sepsis Early Detection, ICU Monitoring, Emergency Department Screening, Pediatric Sepsis |
| End User | Hospitals, Emergency Departments, ICUs, Pediatric Units |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Abionic |
In terms of product type, the pancreatic stone protein testing market is segmented into PSP testing kits, laboratory-based PSP assays, and point-of-care (POC) PSP devices.
In terms of application, the market is segmented into sepsis diagnosis, critical care, systemic inflammatory response syndrome (SIRS) diagnosis, and acute pancreatitis monitoring.
In terms of technology, the pancreatic stone protein testing market is segmented into immunoassay-based testing, point-of-care diagnostic devices, multiplex assays, and ELISA-based testing.
In terms of end-user, the pancreatic stone protein testing market is segmented into hospitals, diagnostic laboratories, academic and research institutions, and specialty clinics.
What is the current global market size for Pancreatic Stone Protein Testing?
The global market is valued at USD 2.30 Billion in 2026, driven by the FDA 510(k) clearance of the first rapid PSP test and growing clinical validation of PSP as a sepsis biomarker.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a CAGR of 2.3% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
North America leads following FDA clearance, with Europe maintaining strong adoption driven by existing IVDR-compliant clinical workflows.
What are the primary market drivers?
FDA clearance of the first rapid PSP diagnostic and the extension of PSP testing into pediatric populations are the primary catalysts.
Who are the leading suppliers in the industry?
Abionic is the primary supplier, differentiating through its nanofluidic abioSCOPE platform that delivers PSP results from a single drop of blood in five minutes.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.