Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market
The proton and ion beam therapy dosimetry and QA test equipment market is segmented by Product Type, Application, Therapy Type, End User, Verification Method, and Region. Forecast for 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Size, Market Forecast and Outlook By FMI
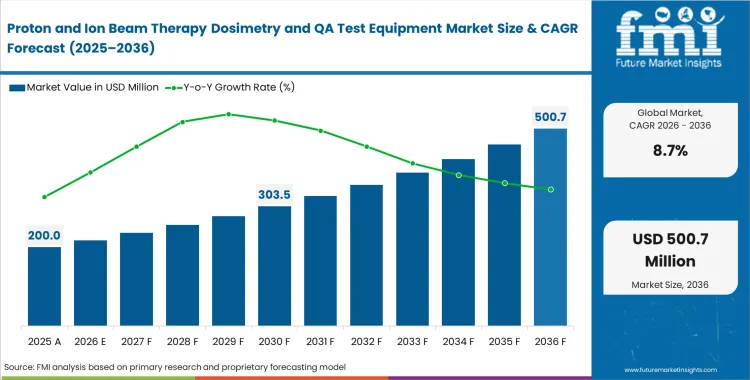
The proton and ion beam therapy dosimetry and QA test equipment market was valued at USD 180.0 million in 2025. Sales are expected to cross USD 200.0 million in 2026 at a CAGR of 8.70% during the forecast period. Demand outlook propels overall opportunity to USD 460.0 million through 2036 as clinical sites transition from commissioning single-room systems to maximizing patient throughput across multi-gantry facilities.
Summary of Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market
- The market is forecast to reach USD 460 million by 2036.
- The market is expected to grow at a CAGR of 8.7% from 2026 to 2036.
- The market was estimated at USD 180 million in 2025.
- The forecast period represents an incremental opportunity of USD 260 million.
- The market is highly specification-driven and compliance-critical, supporting commissioning, machine QA, beam modelling, and patient-specific verification in proton and ion therapy centers.
- Growth is driven by the expanding global installed base of particle therapy centers and increasing equipment intensity per facility.
- Advanced workflows such as Monte Carlo-based QA, adaptive therapy, and proton FLASH research are accelerating demand for high-precision detectors and phantoms.
- China, India, and the United States are among the fastest-growing markets, with China leading at a 10.4% CAGR through 2036.
- Water phantoms dominate the product segment, while machine QA remains the leading application area due to recurring verification needs.
- Proton therapy accounts for the majority share, supported by a larger installed base compared to ion therapy systems.
- Key players in the market include IBA Dosimetry, PTW Freiburg, Sun Nuclear, Standard Imaging, CIRS, RTsafe, and Ashland.
Chief medical physicists evaluating proton therapy QA equipment face intense pressure to compress daily machine quality assurance windows. They must simultaneously maintain the sub-millimeter precision required for complex treatments. Clinical directors demand higher patient volumes to amortize the massive capital cost of particle therapy QA devices, placing workflow efficiency squarely on the dosimetry team. Relying on legacy water tank measurements consumes hours of potential treatment time. This delay forces a pivot toward fast, high-spatial-resolution 2D and 3D detector grids. The commercial consequence of delayed modernization is a rigid cap on daily patient throughput. FMI analysts highlight an underlying reality: hospitals evaluating dosimetry upgrades focus heavily on absolute dose accuracy. The true bottleneck limiting facility revenue is the setup and teardown time of the measurement hardware itself.
Once regulatory bodies finalize standardized protocols for independent Monte Carlo dose calculation, the burden of physical patient-specific QA drops dramatically. Physicists trigger this shift when they validate secondary check software against proton beam QA test equipment for their specific beam models. Crossing this threshold allows facilities to replace exhaustive pre-treatment phantom irradiations with computational checks for standard indications. This operational pivot immediately frees up beam time. It converts mandatory compliance hours into revenue-generating clinical slots, expanding the baseline for adaptive proton therapy QA.
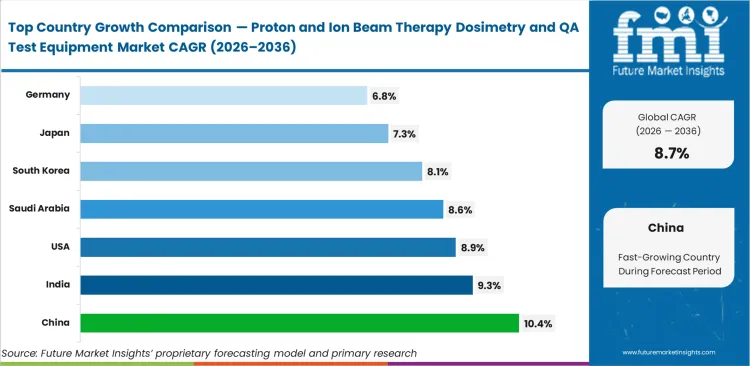
Demand for proton and ion beam therapy dosimetry and QA test equipment is estimated to grow steadily across major countries through 2036, reflecting varied stages of market maturity. China is projected to lead expansion, with market demand anticipated to rise at a 10.4% CAGR, driven by large-scale commissioning of new particle therapy centers. India is expected to follow closely, posting a 9.3% CAGR as private hospital networks invest in full baseline QA systems. The United States is likely to see the market expand at an 8.9% CAGR, supported by replacement-driven upgrades to improve clinical throughput. Saudi Arabia is forecast to record 8.6% CAGR, while South Korea is anticipated to witness 8.1% CAGR as precision requirements intensify. Japan and Germany are estimated to grow at 7.3% and 6.8% CAGR, respectively, largely supported by structured upgrade cycles rather than new installations.
Segmental Analysis
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by Product Type
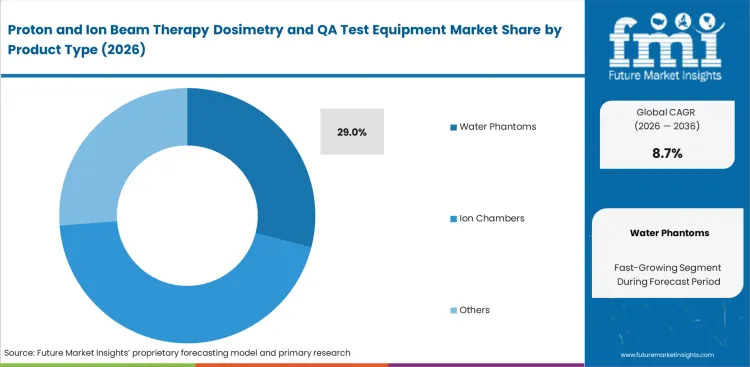
Absolute reference dosimetry depends on a medium that closely replicates the stopping power of human tissue, which is why water phantoms are expected to hold for a 29.0% share of the product segment. Chief medical physicists rely on these motorized 3D scanning tanks during the demanding commissioning phase of new particle therapy systems. Selecting a high-quality proton therapy water phantom enables faster and more reliable acquisition of integrated depth-dose curves and off-axis beam profiles. Evaluation goes beyond radiation sensors to include the mechanical stability and precision of detector positioning systems. Cost-cutting on this foundational radiotherapy device introduces systematic setup errors that carry through into patient treatment planning. Facilities that compromise on scanning tank rigidity often encounter repeated recalibration cycles, delaying clinical commissioning timelines by several weeks.
- Baseline calibration: Motorized scanning tanks establish the reference standard for absolute dose measurement and beam range. Chief medical physicists minimize systematic treatment deviations by ensuring high mechanical accuracy in detector movement.
- Ongoing constancy checks: Routine monthly measurements verify that accelerator output remains stable over time. Clinical QA teams maintain compliance by benchmarking these readings against initial commissioning data.
- Vendor dependency: Water phantom systems rely on specialized software to manage complex scan sequences. Switching vendors introduces significant operational friction, as physics teams must be retrained on new software environments and workflows.
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by Application
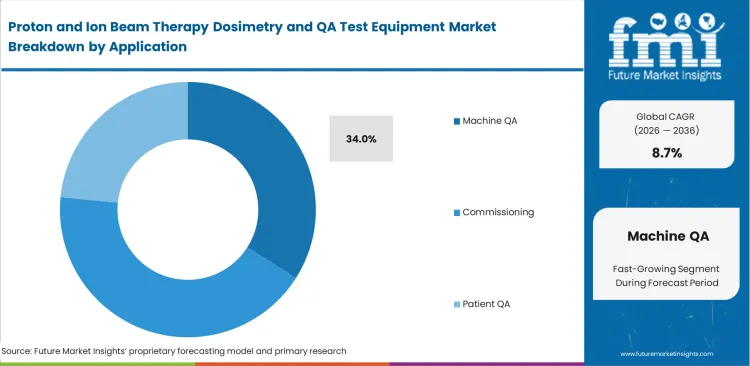
Radiation safety officers face a daily trade-off between running comprehensive QA routines that can consume up to an hour of beam time and relying on faster checks that may overlook subtle spot-positioning drift. Morning output verification and beam symmetry checks remain mandatory before any patient treatment begins. While modern proton accelerators are generally stable, beam delivery nozzles remain vulnerable to minor electromechanical misalignments that are not easily detected through basic checks. This ongoing operational constraint is a key factor driving Machine QA demand, which is expected to account for a 34.0% share in 2026. FMI’s analysis indicates that investment in multi-channel detector arrays enables dosimetrists to validate all critical dosimetric parameters within a single irradiation sequence. High-density sensor grids are essential for detecting fine deviations, particularly in spot positioning. Facilities that delay adopting automated QA systems often depend on sequential manual measurements, increasing workload and extending verification timelines.
- Daily output verification: Radiation safety officers confirm that the proton beam delivers the prescribed dose accurately at the isocenter, a mandatory requirement before initiating treatment sessions.
- Pencil beam mapping: High-resolution detector arrays capture the precise position and profile of individual scanning spots, enabling early identification of beam steering inconsistencies that could lead to localized dosing errors.
- Automated trend analysis: Advanced radiation therapy software continuously tracks machine performance over time, allowing maintenance teams to identify gradual deviations and address potential failures before they affect clinical operations.
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by Therapy Type
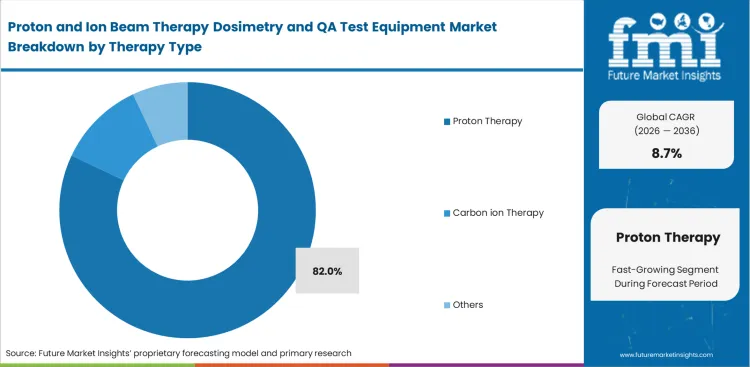
Legacy passive scattering systems are being replaced by pencil beam scanning, requiring facilities to overhaul their measurement hardware. Transition across the installed base is a major reason proton therapy is anticipated to hold an 82.0% share of the market in 2026. For hospital administrators, proton systems remain the standard entry point into heavy particle oncology. Adapting to proton beam scanning dosimetry requires medical physicists to map thousands of individual Bragg peaks across complex target volumes. Despite proton therapy being the most established particle modality, QA practices in dosimetry are still not uniformly standardized. Clinics that do not upgrade their radiotherapy positioning devices to meet pencil beam scanning requirements face unacceptable dose uncertainty, limiting their ability to safely treat pediatric and skull-base cases.
- Pencil beam validation: Medical physicists measure spot size and position across complex, multi-energy delivery sequences to confirm sub-millimeter targeting accuracy and preserve the clinical advantages of scanning beams.
- Range verification: Precise measurement of Bragg peak depth ensures the dose falls off before reaching critical organs, helping dosimetrists control proton penetration depth during routine verification.
- FLASH protocol testing: Research teams are pushing proton cyclotrons to deliver ultra-high dose rates to study normal tissue sparing. Under these conditions, physicists working on proton FLASH dosimetry require specialized electrometers, since conventional ion chambers cannot perform reliably at FLASH dose rates.
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by End User
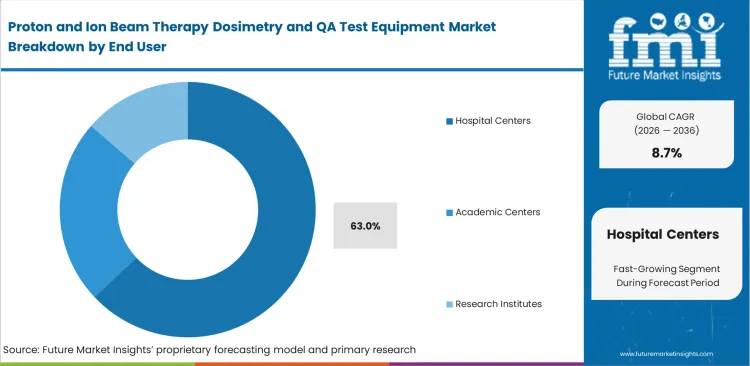
Shifting particle therapy from a research-driven discipline to a routine clinical service has forced hospitals to focus heavily on patient throughput as a core operational priority. Oncology leaders align technology investments with the goal of reducing the time patients remain immobilized during treatment. Hospital physics teams place greater value on seamless software integration and automated reporting than on incremental gains in detector resolution. When procurement decisions overlook system interoperability between image-guided radiotherapy and QA platforms, the resulting manual data handling places a significant burden on already stretched physics teams. This adaptive underpinning is estimated to capture 63.0% share held by hospital-based centers, with the segment expected to expand at a steady CAGR over the forecast period. Adopting rapid QA solutions enables clinical teams to manage forty or more patients per day within a single treatment room.
- Throughput optimization: Hospital oncology directors prioritize QA systems that complete daily checks in under twenty minutes, ensuring that treatment systems remain available for patient use for the majority of the day.
- Staffing efficiency: Automated QA platforms reduce the time senior physicists spend on routine verification tasks, allowing hospitals to manage labor costs by delegating standard data collection to junior staff.
- Regulatory reporting: Integrated data systems compile QA results into standardized audit-ready formats, enabling clinical teams to meet accreditation requirements while minimizing the risk of manual reporting errors.
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by Verification Method
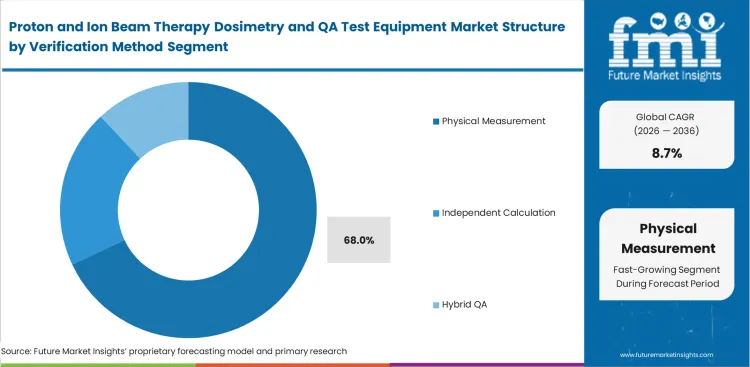
Theoretical confidence in Monte Carlo algorithms often collides with the clinical reality that software cannot identify issues such as a miscalibrated steering magnet. This limitation continues to reinforce the role of physical verification, giving measurement-based is anticipated to hold a 68.0% share in 2026. In measurement-based vs calculation-based proton QA, chief medical physicists remain accountable for treatment accuracy and are reluctant to rely solely on simulation without experimental confirmation. Placing a detector array directly in the beam path provides clear evidence that the machine output matches the planned dose. In routine practice, calculation-based tools are used to flag complex plans, while final validation still depends on physical measurement. Facilities that attempt to move away from measurement entirely typically encounter resistance from clinical physics teams.
- Ground-truth validation: Physical detectors capture the delivered dose under real conditions, providing the assurance required for approving treatments near critical structures such as the brainstem or spinal cord.
- Algorithm benchmarking: Measured dose distributions serve as reference data for refining Monte Carlo models, allowing dosimetrists to improve the accuracy of simulation outputs through continuous comparison.
- Hardware anomaly detection: Measurement-based QA identifies mechanical issues that software assumes are functioning correctly, enabling early detection of component degradation or beamline misalignment.Top of Form
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Drivers, Restraints, and Opportunities
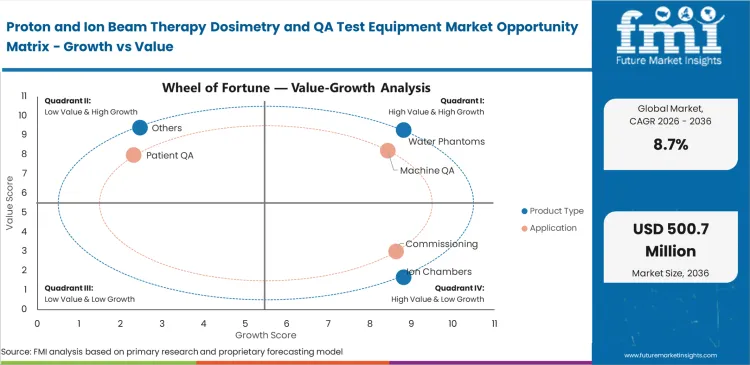
Clinical directors managing multi-room proton facilities face an unavoidable math problem where a single accelerator must serve three or four treatment gantries. This extreme capital amortization pressure compels chief medical physicists to completely redesign their quality assurance workflows. Every minute spent irradiating a physical phantom for patient-specific QA is a minute denied to a revenue-generating patient treatment. This urgency drives the immediate procurement of fast 3D detector arrays and integrated radiotherapy patient positioning accessories that verify complex dose distributions rapidly. Delaying workflow modernization essentially caps the facility's earning potential. Executive teams evaluating the ROI of proton therapy QA automation recognize that idle treatment rooms cost far more than premium measurement arrays.
The binding operational friction slowing the adoption of advanced independent calculation software is the lack of universally accepted regulatory guidelines for replacing physical measurements. Even when clinical physics teams want to transition to log-file analysis, national radiation safety regulators often mandate physical phantom irradiations for every single patient plan. This compliance gap forces facilities to maintain dual workflows. They purchase expensive computational tools while still bearing the labor costs of manual physical QA. Until standardization task groups provide legally defensible protocols for software-only verification, hospitals will remain reluctant to fully abandon legacy proton beam range verification tools.
Opportunities in the Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market
- Proton therapy end to end QA: Research physicists require specialized phantoms capable of accurately recording full delivery chains to satisfy rigorous proton therapy beam modelling data requirements.
- Automated log-file analysis: Software developers can capture contracts by creating platforms that instantly compare machine delivery logs against intensity-modulated radiation therapy or proton pencil beam plans.
- MR-compatible QA phantoms: The emergence of MRI-guided proton therapy forces procurement managers to acquire non-ferrous phantoms and detectors that function perfectly inside strong magnetic fields.
Regional Analysis
Based on regional analysis, Proton and Ion Beam Therapy Dosimetry and QA Test Equipment is segmented into North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 10.4% |
| India | 9.3% |
| United States | 8.9% |
| Saudi Arabia | 8.6% |
| South Korea | 8.1% |
| Japan | 7.3% |
| Germany | 6.8% |
Source: Future Market Insights (FMI) analysis, based on a proprietary forecasting model and primary research
East Asia Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis
State-backed infrastructure initiatives across East Asia have transformed the region from a technology importer into the most aggressive builder of heavy particle oncology centers globally. The expansion is heavily weighted toward carbon-ion facilities and massive multi-room proton installations. Unlike mature markets focused on efficiency upgrades, procurement directors here are outfitting empty bunkers. Buying deep-water phantoms, full detector suites, and electrometers simultaneously. The practitioner reality in these new centers is a severe shortage of experienced medical physicists. Talent gap drives a strong preference for highly automated, turnkey QA systems that minimize manual data interpretation.
- China: A dense wave of recent proton and carbon-ion center openings mandates massive upfront purchases of commissioning hardware. Hospital administrators authorize these comprehensive proton therapy QA bundles in China to secure operational licenses swiftly. Demand is expected to rise at a 10.4% CAGR through 2036, driving a decade-long pipeline of maintenance revenue for vendors that win these greenfield contracts.
- South Korea: Academic medical centers push aggressively into complex pediatric and skull-base particle treatments. Chief medical physicists demand high-density 3D arrays to validate these tight margins. The sector in South Korea is expected to capture at a 8.1% CAGR, the market is also seeing faster movement through 2036. This is directed toward independent calculation software as facilities seek to maximize patient throughput on their limited number of operational gantries.
- Japan: Having pioneered carbon ion therapy, the national installed base in Japan requires modernization rather than initial outfitting. Radiation safety officers are discarding legacy passive-scattering QA tools in favor of equipment optimized for rapid pencil beam scanning. Vendors compete fiercely on software usability and data integration to displace entrenched local equipment suppliers, with the market expected to follow a steady 7.3% CAGR through 2036. This keeps competition centered on upgrade value, workflow fit, and replacement efficiency rather than greenfield volume.
FMI's report includes Taiwan and other emerging East Asian territories. The competitive baseline in this region heavily favors global QA manufacturers who can establish robust local service laboratories to support the rapidly expanding installed base. Malaysia is also identified as a growing influence, as sustained investments in semiconductor back‑end manufacturing, electronics assembly, and test and measurement infrastructure are increasing demand for localized quality assurance and calibration services.
South Asia & Pacific Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis
Private healthcare conglomerates and large corporate hospital networks dictate the adoption curve across this region. Unlike state-funded systems, these corporate operators evaluate multi-million dollar particle therapy investments through a strict return-on-investment lens. Clinical directors face immediate pressure to bring new gantries online and achieve maximum patient throughput within months of installation. This commercial environment forces physics teams to purchase the fastest integrated QA hardware available. They prioritize equipment that links directly with existing oncology information systems to avoid manual data transcription delays.
- India: In India, corporate hospital chains constructing new proton centers require complete baseline dosimetry portfolios to initiate clinical operations. India proton therapy QA equipment sector is expected to expand at a 9.3% CAGR through 2036, making the country a critical expansion frontier for suppliers. Procurement managers leverage their scale to negotiate bundled hardware and software deals that lock in predictive maintenance costs over the equipment's lifecycle.
FMI's report includes Australia, Thailand, and other regional markets. Developing a comprehensive local physicist training program remains the most effective strategy for QA manufacturers to embed their proprietary measurement protocols into these growing hospital networks. Indonesia is also highlighted as an emerging opportunity market, where rapid expansion of tertiary care hospitals and rising investments in radiation therapy and advanced imaging systems are increasing demand for certified QA expertise.
North America Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis
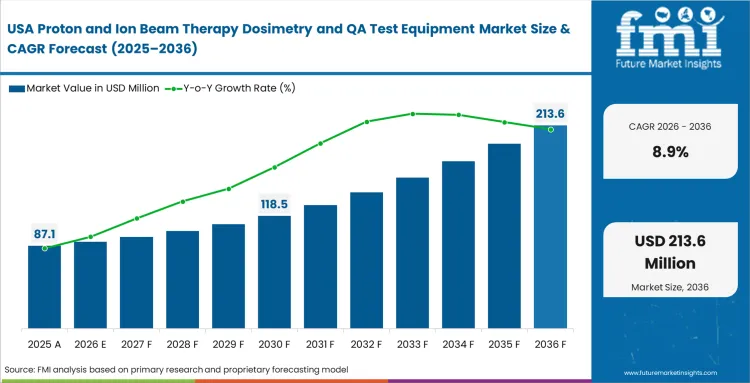
The structural reality of the North American landscape is defined by the high density of mature operational single-room proton installations. Chief medical physicists at these established facilities are not buying water phantoms for initial commissioning. They are actively removing older measurement arrays that bottleneck their daily schedules. The operational constraint driving the United States proton dosimetry sector is entirely throughput-driven. Hospitals must process more patients through their existing gantries to maintain financial viability amid shifting reimbursement rates. This forces a rapid transition toward highly automated daily QA devices that can verify pencil beam scanning accuracy in minutes.
- United States: Established proton centers aggressively adopt workflow upgrades to push higher patient volumes through aging single-room systems. US market is expected to rise at a CAGR of 8.9% through 2036, physics teams here prioritize secondary check software and high-resolution detector arrays. Moving away from manual phantom irradiations allows these centers to reclaim hours of beam time, directly increasing daily clinical revenue capacity.
FMI's report includes Canada. Cross-border collaboration among major academic oncology centers ensures that rigorous, standardized QA protocols rapidly become the baseline requirement for any new equipment vendor entering the space. Japan is also identified as a market with strong growth influence, driven by its aging population, expanding investment in advanced radiotherapy technologies, and strict national quality and safety standards.
Europe Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis
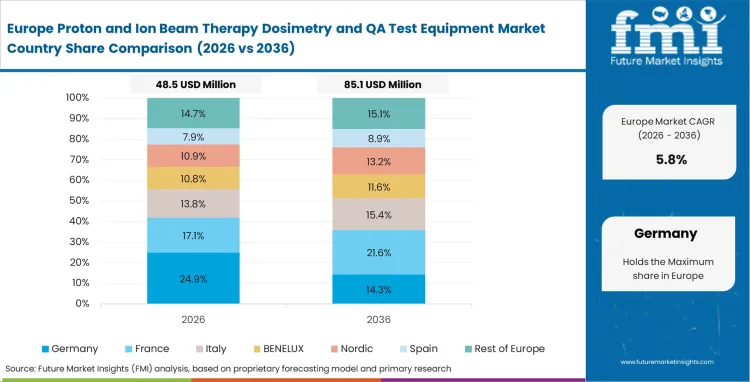
Europe's particle therapy demand is shaped by deeply embedded academic and clinical research traditions, supported by a mature installed base of proton and carbon ion centers. Chief medical physicists across the region prioritize dosimetric absolute accuracy and active participation in continental standardization groups over sheer patient throughput. Steady demand continues for high-end reference dosimetry and sophisticated stereotactic radiation therapy validation tools. Practitioner workflows often remain mixed, with facilities balancing heavy clinical loads alongside meaningful machine time allocated to physics research and beam modeling refinement.
- Germany: Operating one of the oldest and most diverse particle therapy installed bases, clinical QA directors focus heavily on replacement cycles and workflow optimization. Demand for ion beam QA devices in Germany is expected to rise at a 6.8% CAGR through 2036, with spending centered on upgrading legacy mechanical phantoms to faster 2D and 3D detector arrays. Facilities improve their operational resilience by integrating predictive maintenance software that tracks machine output constancy over years of operation.
FMI's report includes the United Kingdom, France, and Italy. The close collaboration between clinical sites and local European QA hardware manufacturers ensures a highly competitive environment where software interoperability often dictates vendor selection. Sweden is also gaining importance, supported by sustained investment in advanced radiotherapy centers, digital health infrastructure, and strong public funding for oncology modernization.
Middle East & Africa Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis
Heavy investment in localized advanced oncology infrastructure defines the trajectory of particle therapy across this region. State health ministries are moving away from sending patients abroad and are instead building major regional cancer hubs. Greenfield projects of this kind require entire dosimetry ecosystems to be purchased and commissioned simultaneously. Early-stage center operations also often depend on expatriate physics teams, which increases buyer preference for comprehensive single-vendor QA platforms. Procurement managers therefore favor systems that guarantee frictionless hardware and software integration out of the box.
- Saudi Arabia: Construction of advanced regional oncology centers demands immediate procurement of baseline water phantoms and full machine QA suites. Reliance on turnkey solutions remains strong because new installations need seamless hardware-software integration, fast commissioning support, and continued physicist training from the same vendor. Demand for proton therapy equipment in Saudi Arabia is expected to expand at a 8.6% CAGR. Long-cycle supplier support will remain a decisive factor as these centers move from installation to routine clinical use.
FMI's report includes South Africa and the UAE. Establishing reliable, fast‑response local calibration and repair laboratories is a mandatory prerequisite for any QA manufacturer seeking to win contracts in this expanding geography. Egypt is also identified as an emerging growth market, as public investment in oncology capacity, new radiotherapy installations, and hospital modernization programs accelerates across major urban centers.
Competitive Aligners for Market Players
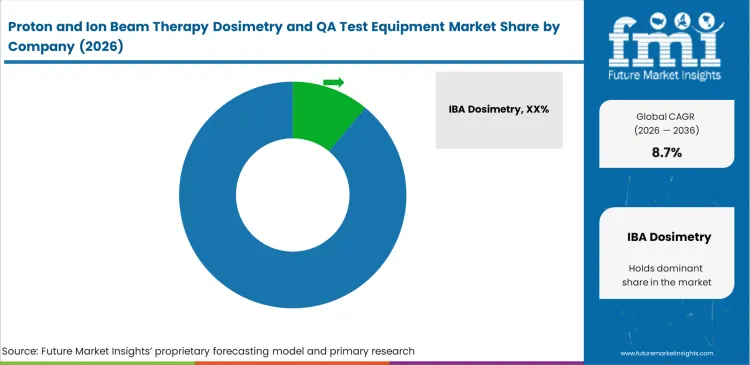
The competitive landscape in particle therapy dosimetry remains tightly concentrated, with a small group of global manufacturers securing most hospital contracts. Conventional photon radiotherapy, where standardized QA tools are often sufficient, Bragg peak measurement demands highly specialized detector designs. When hospitals compare proton QA hardware vendors, pricing is rarely the deciding factor. Chief medical physicists place greater emphasis on software compatibility and the long-term stability of detectors operating under high linear energy transfer conditions. Vendors that offer end-to-end portfolios from initial water tank commissioning through daily patient-specific QA reduce the risk of data inconsistencies across systems and are generally preferred.
Incumbent suppliers benefit from a strong structural advantage built over decades, supported by extensive beam data libraries and deeply integrated software platforms. In practical evaluations such as IBA versus PTW proton QA systems, once a vendor’s analysis software is embedded within a hospital’s oncology information infrastructure, switching even a single component like an electrometer becomes operationally complex. Established proton QA equipment suppliers also maintain validated Monte Carlo beam models and have their systems referenced in international QA protocols. New entrants face a high barrier, as they must demonstrate equivalence to these established standards while ensuring compatibility with existing hospital data environments.
Large academic hospital networks are beginning to exert greater influence by pushing for open data access and reduced reliance on proprietary calibration models. Physics teams increasingly resist factory-dependent recalibration cycles that take essential QA equipment offline for extended periods. Growing tension between closed vendor ecosystems and the need for transparent, independent validation is reshaping procurement expectations. Vendors that do not offer flexible calibration options or hybrid verification approaches are likely to face challenges in securing contracts from large, multi-room treatment centers.
Key Players in Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market
- IBA Dosimetry
- PTW Freiburg
- Sun Nuclear
- Standard Imaging
- CIRS
- RTsafe
- Ashland
Scope of the Report
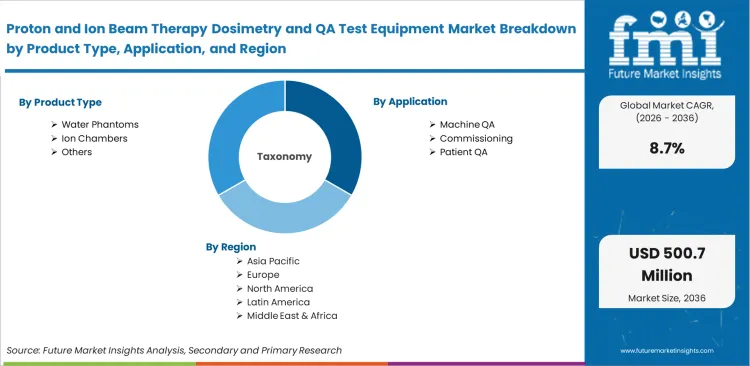
| Metric | Value |
|---|---|
| Quantitative Units | USD 200.0 million to USD 460.0 million, at a CAGR of 8.70% |
| Market Definition | Proton and Ion Beam Therapy Dosimetry and QA Test Equipment encompasses the specialized measurement instruments required to characterize particle beams and verify delivered radiation doses. |
| Segmentation | Product Type, Application, Therapy Type, End User, Verification Method |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | China, India, United States, Saudi Arabia, South Korea, Japan, Germany |
| Key Companies Profiled | IBA Dosimetry, PTW Freiburg, Sun Nuclear, Standard Imaging, CIRS, RTsafe, Ashland |
| Forecast Period | 2026 to 2036 |
| Approach | The global count of operational and under-construction particle therapy treatment rooms serves as the baseline metric for hardware procurement models. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Proton and Ion Beam Therapy Dosimetry and QA Test Equipment Market Analysis by Segments
Product Type
- Water phantoms
- Ion chambers
- Detector arrays
- Electrometers
- Phantoms
- Film dosimetry
Application
- Machine QA
- Commissioning
- Patient QA
- Beam modelling
- End-to-end testing
Therapy Type
- Proton therapy
- Carbon ion therapy
- Heavy ion therapy
End User
- Hospital centers
- Academic centers
- Research institutes
- QA labs
Verification Method
- Physical measurement
- Independent calculation
- Hybrid QA
Region
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- Australia
- Thailand
- Middle East & Africa
- Saudi Arabia
- South Africa
- UAE
Bibliography
- Particle Therapy Co-Operative Group. (2026, February). Particle therapy facilities in clinical operation. PTCOG.
- National Association for Proton Therapy. (2024). 2024 NAPT Member Survey Executive Summary.
- Gomà, C., Henkner, K., Jäkel, O., Lorentini, S., Magro, G., Mirandola, A., Placidi, L., Togno, M., Vidal, M., Vilches-Freixas, G., Wulff, J., & Safai, S. (2024). ESTRO-EPTN radiation dosimetry guidelines for the acquisition of proton pencil beam modelling data. Physics and Imaging in Radiation Oncology, 31, 100621.
- Gambetta, V., Stützer, K., & Richter, C. (2025). Current status and upcoming developments for online adaptive proton therapy enabling a closed feedback loop for near real-time adaptation. Frontiers in Oncology, 15, 1660605.
- Zhang, Q., Yang, W., Tan, L., Guo, X., Wang, T., Zhu, P., Jing, Z., Ma, L., & Hou, J. (2026). Advances in proton therapy technology and global clinical applications. Frontiers in Oncology, 16, 1718677.
- American Society for Radiation Oncology. (2024, September 30). IMRT and proton therapy offer equally high quality of life and tumor control for people with low- and intermediate-risk prostate cancer.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
Frequently Asked Questions
What is the baseline valuation of the Proton and Ion Beam Therapy Dosimetry and QA Test Equipment sector?
The sector reached USD 180.0 million in 2025. This establishes the financial baseline as clinical particle therapy centers worldwide modernize their verification hardware to handle higher daily patient volumes.
What is the revenue outlook for 2026?
Sales are poised to hit USD 200.0 million in 2026. This reflects immediate hospital procurement of high-resolution detector arrays to support the transition to complex pencil beam scanning delivery.
How big is the proton and ion beam therapy dosimetry equipment sector?
Demand expands at an 8.70% CAGR to reach USD 460.0 million by 2036. This trajectory is sustained not merely by new facility construction, but by existing sites replacing manual phantom routines with faster automated QA suites.
What equipment is used for proton therapy QA?
Facilities rely on specialized water phantoms, multi-channel detector arrays, and high-precision electrometers. These devices capture sharp dose gradients to verify that pencil beam scanning systems are accurately targeting tumor volumes.
Why is proton therapy dosimetry different from photon dosimetry?
Particle beams deposit their maximum radiation dose at a specific depth known as the Bragg peak. Measuring this sharp drop-off requires specialized hardware that conventional photon QA tools cannot accurately capture.
Why does proton therapy need special QA?
The sub-millimeter precision of proton beams means that even microscopic steering magnet misalignments can result in severe normal tissue toxicity. Specialized QA confirms that these highly conformal doses land exactly where intended.
What is the difference between proton QA and photon QA?
Proton QA emphasizes precise range verification and depth-dose profiles, whereas photon QA focuses primarily on 2D intensity modulation. The instruments differ significantly in their required spatial resolution and linear energy transfer capabilities.
Which companies lead proton QA hardware?
Major global providers include IBA Dosimetry, PTW Freiburg, and Sun Nuclear. These vendors secure contracts by integrating their hardware platforms smoothly with existing hospital oncology information systems.
What should a hospital compare before buying proton QA equipment?
Procurement directors must evaluate software interoperability, calibration cycle downtime, and absolute detector stability. A system's value is often determined by its ability to export data directly into the hospital's native database.
What is the role of water phantoms in proton commissioning?
Water perfectly simulates human tissue stopping power. Chief medical physicists rely on motorized 3D scanning tanks to establish the absolute baseline calibration required before any patient treatments begin.
What are proton therapy water phantoms used for daily?
While essential for baseline commissioning, they are also deployed for strict monthly constancy checks. These deep-water readings validate that the accelerator energy output has not drifted from its initial baseline.
How often do proton centers perform machine QA?
Regulatory mandates require exhaustive daily and monthly testing of beam output and electromechanical positioning. Radiation safety officers must pass these checks every morning before initiating any clinical workflows.
How is patient-specific QA performed in proton therapy?
Traditionally, physicists irradiate a physical phantom using the patient's specific treatment plan. Modern centers increasingly adopt hybrid workflows that combine selective physical measurements with advanced secondary calculation software.
What is changing in adaptive proton therapy QA?
Facilities are migrating toward real-time log-file analysis to confirm dose delivery continuously. This shift aims to reduce the beam time consumed by pre-treatment phantom scanning, unlocking higher daily patient throughput.
How does proton FLASH affect dosimetry requirements?
Research teams push cyclotrons to deliver ultra-high dose rates to investigate normal tissue sparing. Physicists must acquire specialized electrometers because conventional ion chambers saturate completely under FLASH conditions.
Why do hospital centers capture the majority of end-user demand?
Particle therapy has matured from a specialized physics experiment into a routine clinical service. Oncology directors at major hospitals acquire QA tools optimized for rapid patient throughput.
Why does physical measurement maintain its dominance?
Monte Carlo algorithms cannot physically detect a misaligned steering magnet. Medical physicists insist on placing a detector array in the beam path to confirm the absolute physical truth of the delivery.
What countries are adding the most proton therapy QA capacity?
China expands at 10.4% annually driven by state-backed greenfield projects. India follows at 9.3% as large corporate hospital networks acquire baseline QA equipment for their new advanced regional oncology hubs.
What drives demand in the United States?
The United States tracks at 8.9%. Mature single-room centers are actively replacing older measurement arrays with highly automated systems to maximize patient throughput and clinical revenue.
Why does Saudi Arabia show strong momentum?
Advancing at 8.6%, Saudi Arabia is building extensive regional cancer hubs. Procurement directors favor comprehensive turnkey QA platforms that minimize reliance on expatriate physics teams.
What shapes the South Korean landscape?
South Korea expands at 8.1%. Academic medical centers require ultra-precise 3D arrays to safely validate the complex dosimetric margins involved in pediatric and skull-base treatments.
How does the Japanese market behave?
Japan is estimated to yield a 7.3% trajectory through 2036. Possessing a mature carbon and proton installed base, clinical directors focus on discarding legacy passive-scattering tools in favor of pencil beam scanning hardware.
What defines the German dynamic?
Germany grows at 6.8%. Highly experienced physics teams balance clinical patient loads with deep dosimetric research, driving demand for complex end-to-end testing phantoms and stable reference electrometers.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Water Phantoms
- Ion Chambers
- Others
- Water Phantoms
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Machine QA
- Commissioning
- Patient QA
- Machine QA
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Type, 2026 to 2036
- Proton Therapy
- Carbon ion Therapy
- Others
- Proton Therapy
- Y to o to Y Growth Trend Analysis By Therapy Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital Centers
- Academic Centers
- Research Institutes
- Hospital Centers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Verification Method
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Verification Method, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Verification Method, 2026 to 2036
- Physical Measurement
- Independent Calculation
- Hybrid QA
- Physical Measurement
- Y to o to Y Growth Trend Analysis By Verification Method, 2021 to 2025
- Absolute $ Opportunity Analysis By Verification Method, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By Therapy Type
- By End User
- By Verification Method
- Competition Analysis
- Competition Deep Dive
- IBA Dosimetry
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- PTW Freiburg
- Sun Nuclear
- Standard Imaging
- CIRS
- RTsafe
- IBA Dosimetry
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Therapy Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Verification Method, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Therapy Type
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Verification Method
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Therapy Type
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Verification Method
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Application
- Figure 51: Latin America Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Therapy Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Verification Method
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Application
- Figure 67: Western Europe Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Therapy Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Verification Method
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Application
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Therapy Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Verification Method
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Application
- Figure 99: East Asia Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Therapy Type
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Verification Method
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Therapy Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Verification Method
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Therapy Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Therapy Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Verification Method, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Verification Method, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Verification Method
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

