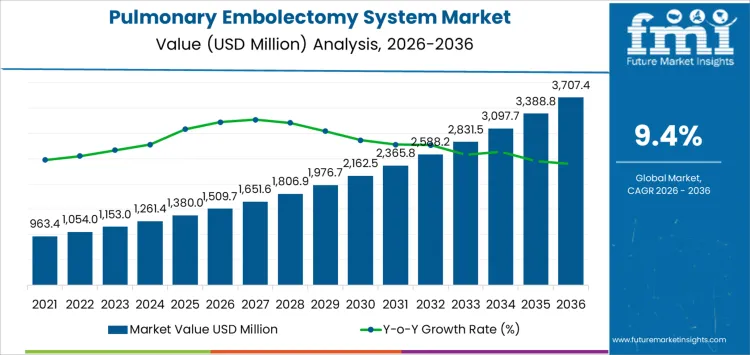
The pulmonary embolectomy system market is projected to expand from USD 1,509.72 million in 2026 to USD 3,707.4 million by 2036, progressing at a CAGR of 9.4%. Purchasing behavior in this market is shaped by high clinical risk, capital sensitivity, and the need for procedural reliability in acute care settings. Procurement decisions are typically centralized within tertiary hospitals, where technology selection is governed by multidisciplinary committees and evidence-based protocols. Preference is given to systems with established clinical data, predictable performance across clot burdens, and compatibility with existing catheterization lab infrastructure. Buyer risk aversion remains high, as device failure or workflow disruption carries significant patient and reputational consequences.
Vendor lock-in is reinforced through physician training, procedural standardization, and long-term service agreements, increasing switching costs once a platform is embedded. Pricing leverage is influenced by reimbursement clarity and bundled purchasing across cardiovascular portfolios. Outcomes are expected to favor suppliers that minimize adoption risk, reduce retraining requirements, and align closely with hospital procurement frameworks rather than those competing primarily on unit pricing.
| Metric | Value |
|---|---|
| Pulmonary Embolectomy System Market Value (2026) | USD 1,509.72 Million |
| Pulmonary Embolectomy System Market Forecast Value (2036) | USD 3,707.4 Million |
| Pulmonary Embolectomy System Market Forecast CAGR (2026-2036) | 9.4% |
The pulmonary embolectomy system market is growing as hospitals adopt faster, catheter-based solutions for managing acute and submassive pulmonary embolism. Emergency departments and intensive care units are under pressure to reduce mortality and length of stay, pushing clinicians toward mechanical and aspiration-based embolectomy systems that deliver rapid clot removal without systemic thrombolytics. For device manufacturers, value creation increasingly depends on demonstrating procedural speed, hemodynamic improvement, and reduced bleeding risk within real-world acute care settings.
Clinical adoption is also being shaped by catheter-directed thrombolysis integration. Interventional cardiologists, radiologists, and vascular specialists prefer systems that integrate smoothly into cath lab infrastructure with minimal setup complexity. Hospitals are prioritizing platforms with intuitive catheter design, predictable performance across clot burdens, and strong clinical evidence supporting safety and efficacy. Vendors offering comprehensive training programs, case support, and post-procedure outcome data are gaining stronger positioning during technology evaluations.
Procurement dynamics and reimbursement considerations are further supporting market expansion. As clinical guidelines evolve and reimbursement clarity improves, hospitals are expanding pulmonary embolism response teams and standardizing treatment protocols that include mechanical embolectomy options. Growth in minimally invasive intervention capabilities across secondary and tertiary care centers is broadening the addressable market beyond elite academic hospitals. For suppliers, competitive advantage increasingly depends on clinical validation, procedural efficiency, and the ability to support hospitals in scaling pulmonary embolism intervention programs positioning pulmonary embolectomy systems as a core component of modern acute cardiovascular care.
The pulmonary embolectomy system market is segmented by product and application, reflecting how intervention approach and disease severity guide device selection. By product, catheter-based embolectomy systems lead adoption, as minimally invasive techniques are increasingly preferred in acute care settings. Other products include surgical embolectomy systems and aspiration thrombectomy devices, which address complex clot burden and anatomical considerations. By application, massive pulmonary embolism represents the primary treatment segment, driven by the need for rapid clot removal to stabilize hemodynamics. Submassive pulmonary embolism and chronic pulmonary embolism account for additional demand across elective and staged interventions.
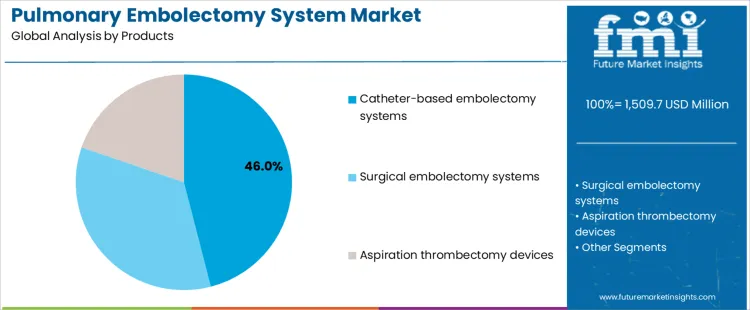
Catheter-based embolectomy systems account for 46% share of the pulmonary embolectomy system market because they enable rapid thrombus removal with lower procedural risk compared to open surgery. Interventional cardiologists and vascular specialists favor catheter-based approaches for their percutaneous access, reduced recovery time, and suitability for hemodynamically unstable patients. These systems allow precise navigation within pulmonary arteries and support aspiration or mechanical clot extraction under imaging guidance. Hospitals prioritize catheter-based devices to expand treatment options within cath lab settings. Growing clinician familiarity and protocol integration further support adoption. These clinical efficiency and workflow advantages explain why catheter-based systems remain the leading product category.
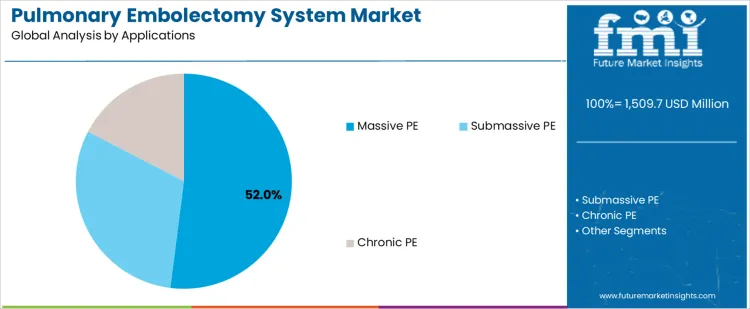
Massive pulmonary embolism accounts for 52% share of the pulmonary embolectomy system market because it presents an immediate life-threatening condition requiring urgent intervention. Patients with massive pulmonary embolism experience severe hemodynamic compromise, making rapid clot removal essential to restore pulmonary circulation. Embolectomy systems provide a direct method to reduce clot burden when thrombolytic therapy is contraindicated or ineffective. Emergency departments and intensive care units prioritize access to embolectomy technologies for high-risk cases. Higher procedure urgency and clinical severity drive concentrated device utilization. These acute care requirements explain why massive pulmonary embolism remains the dominant application segment.
The pulmonary embolectomy system market is driven by increasing incidence of pulmonary embolism (PE), a life-threatening blockage of pulmonary arteries often caused by deep vein thrombosis (DVT). Growth in diagnostic imaging use (CT pulmonary angiography), heightened clinical awareness of acute PE, and demand for minimally invasive treatment options are expanding utilization of catheter-based and surgical embolectomy systems. For OEMs, device efficacy, procedural safety, ease of use, and compatibility with hospital interventional suites influence hospital purchasing decisions. Expanded thromboprophylactic drug use aids the prevention of recurrent thromboembolism in high-risk patients.
Clinical practice evolution and technological innovation are shaping the pulmonary embolectomy system market by prioritizing rapid, targeted removal of emboli with lower risk profiles. Advances include aspiration catheters with optimized lumen design for effective clot extraction, mechanical fragmentation tools, and hybrid systems combining pharmacomechanical approaches to reduce reliance on systemic thrombolysis. Navigation support technologies (e.g., steerable catheters, imaging guidance) improve procedural accuracy and reduce fluoroscopy times. For system manufacturers, delivering devices with high navigability, low profile design, and validated safety data enhances acceptance among interventional cardiologists, radiologists, and pulmonologists in high-acuity care environments.
Clinical, regulatory, and adoption barriers restrain growth in the pulmonary embolectomy system market. Acute PE care pathways vary across hospitals, with some facilities relying on systemic anticoagulation or thrombolytics rather than mechanical intervention, reducing procedural volumes for embolectomy systems. Stringent regulatory requirements for safety and effectiveness especially in high-risk interventions necessitate extensive clinical evidence, which increases development timelines and costs. Hospital budget constraints, procedural training needs, and referral patterns limit rapid adoption outside high-volume centers. Limited reimbursement in some regions for catheter-based PE interventions further hinders uptake.
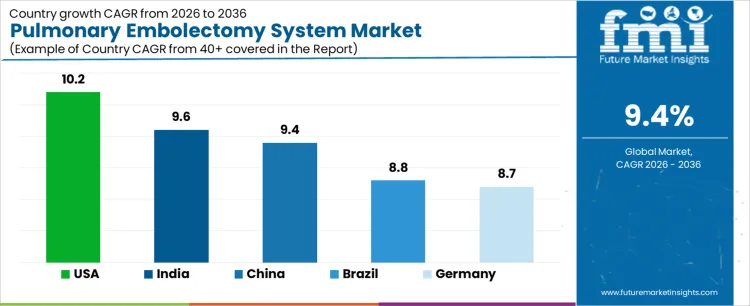
The pulmonary embolectomy system market is expanding as hospitals increasingly adopt catheter-based interventions to manage acute and high-risk pulmonary embolism cases. These systems enable rapid clot removal while reducing reliance on open surgical procedures and systemic thrombolysis. Growth is supported by rising pulmonary embolism incidence, coagulation marker diagnostics, and expansion of interventional cardiology and radiology capabilities. Country-level growth varies based on healthcare infrastructure, reimbursement frameworks, physician training, and adoption of multidisciplinary pulmonary embolism response protocols. Mature markets emphasize clinical evidence, safety, and guideline alignment, while emerging markets focus on expanding access, affordability, and procedural capacity. Market value is driven by procedural efficiency, clinical outcomes, and ease of integration into existing catheterization lab workflows.
| Country | CAGR (%) |
|---|---|
| United States | 10.2 |
| India | 9.6 |
| China | 9.4 |
| Brazil | 8.8 |
| Germany | 8.7 |
The pulmonary embolectomy system market in the United States is expanding at a CAGR of 10.2% during 2026-2036, driven by increasing use of catheter-based interventions for acute and submassive pulmonary embolism. USA hospitals emphasize rapid clot removal to reduce ICU stay, right ventricular strain, and mortality risk. Adoption is led by tertiary care centers and cardiac catheterization labs with multidisciplinary pulmonary embolism response teams. Buyers prioritize clinical evidence, procedural safety, ease of use, and compatibility with existing imaging and catheterization infrastructure. Demand for thromboprophylactic drugs in the United States reflects broader preventive strategies impacting PE care. Qualification cycles focus on demonstrated reduction in complications and improved patient outcomes. Market value is created through systems that shorten procedure time, reduce bleeding risk, and integrate smoothly into established interventional workflows. The USA represents a protocol-driven market where clinical validation and physician acceptance determine commercial success.
Brazil’s pulmonary embolectomy system market is growing at a CAGR of 8.8% during 2026-2036, shaped by expanding interventional cardiology capabilities in major urban hospitals. Adoption is concentrated in private hospitals and advanced public centers managing high-risk pulmonary embolism cases. Buyers focus on affordability, procedural simplicity, and adaptability to existing catheterization lab setups. Use of embolectomy systems is selective, often reserved for severe or life-threatening cases where thrombolysis is contraindicated. Procurement decisions are influenced by budget constraints, physician training availability, and distributor support. Qualification cycles are moderate, with emphasis on clinical familiarity and post-sale service. Market growth is supported by gradual expansion of specialist training and awareness. Brazil represents a developing market where success depends on cost-effective systems, strong clinical education, and reliable local distribution rather than broad-based procedural adoption.
China’s pulmonary embolectomy system market is expanding at a CAGR of 9.4% during 2026-2036, driven by rising incidence of cardiovascular disease and growing interventional treatment capacity. Adoption is led by large public hospitals and regional medical centers upgrading their catheter-based therapy offerings. Buyers emphasize throughput efficiency, procedural reliability, and cost control. Embolectomy systems are increasingly used as alternatives to surgical intervention in high-risk patients. Procurement decisions favor suppliers with localized manufacturing, strong regulatory compliance, and scalable distribution networks. Qualification cycles are relatively short, with clinical acceptance supported by rapid deployment in high-volume centers. Market value is driven by system robustness and ability to operate efficiently under high patient loads. China represents a scale-oriented market where growth depends on hospital expansion, procedural standardization, and supplier capacity to meet rising demand.
India’s pulmonary embolectomy system market is projected to grow at a CAGR of 9.6% during 2026-2036, supported by increasing diagnosis of pulmonary embolism and expansion of interventional cardiology services. Venous thromboembolism treatment adoption in India reflects rising procedural volumes influencing device uptake. Buyers prioritize affordability, procedural safety, and training support due to variability in clinical expertise across institutions. Embolectomy systems are typically used in severe cases where rapid intervention is required. Procurement cycles are relatively short, enabling quicker adoption when clinical need is established. Market value is driven by systems that balance performance with cost efficiency. India represents a growing market where success depends on clinical education, scalable pricing models, and strong technical support to drive wider procedural acceptance.
Germany’s pulmonary embolectomy system market is growing at a CAGR of 8.7% during the forecast period, characterized by cautious, guideline-driven adoption. German hospitals emphasize evidence-based decision-making and strict adherence to clinical protocols when introducing new interventional devices. Use of embolectomy systems is concentrated in specialized centers managing complex pulmonary embolism cases. Buyers prioritize procedural safety, clinical documentation, and long-term outcomes. Procurement decisions involve multidisciplinary evaluation committees, resulting in longer qualification timelines. Market value accrues to suppliers offering robust clinical data, system reliability, and compatibility with established workflows. Germany represents a maturity-driven market where adoption is steady but selective, and success depends on scientific validation and clinician confidence rather than rapid expansion.
Competition in the pulmonary embolectomy system market centers on clot retrieval efficacy, catheter innovation, and documented clinical outcomes that matter to interventional cardiologists, vascular surgeons, and hospital procurement teams. Inari Medical positions its systems with official product literature highlighting advanced embolectomy catheters designed for large clot burden removal without the need for thrombolytics. Brochures emphasize integrated aspiration technology, intuitive blood clot retrieval device innovation, and reduced procedural time supported by clinical case data.
Penumbra competes through a portfolio of aspiration-based embolectomy tools backed by documentation of continuous lumen aspiration and large vessel access dynamics. Its materials underscore system versatility across acute pulmonary embolism (PE) severities and highlight controlled suction flow rates calibrated for safe, efficient thrombus extraction. Boston Scientific differentiates via combination systems that integrate steerable catheter platforms with aspiration and mechanical disruption features. Product guides focus on ergonomic control handles and support for complex pulmonary arterial anatomies, with clinical evidence cited for improved navigation and extraction performance.
Large OEMs bring broad cardiovascular system expertise to the competitive landscape. Medtronic positions its embolectomy systems with official brochures that emphasize delivery precision, support in tortuous vasculature, and integration with imaging guidance systems. Documentation highlights device torque response and scaffold compatibility where applicable. Cook Medical competes by integrating embolectomy solutions into a wider portfolio of interventional support products, with technical materials that focus on reliable trackability and kink resistance under clinical loads.
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Million |
| Products | Catheter-Based Embolectomy Systems, Surgical Embolectomy Systems, Aspiration Thrombectomy Devices |
| Applications | Massive Pulmonary Embolism (PE), Submassive Pulmonary Embolism (PE), Chronic Pulmonary Embolism (PE) |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Inari Medical, Penumbra, Boston Scientific, Medtronic, Cook Medical, Others |
| Additional Attributes | Dollar sales by product type and clinical application are evaluated across acute and chronic pulmonary embolism treatment settings. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of minimally invasive adoption trends, evaluation of procedural outcomes and hospital utilization, competitive benchmarking of device manufacturers, and assessment of guideline-driven adoption across interventional cardiology and vascular surgery centers. |
How big is the pulmonary embolectomy system market in 2026?
The global pulmonary embolectomy system market is estimated to be valued at USD 1,509.7 million in 2026.
What will be the size of pulmonary embolectomy system market in 2036?
The market size for the pulmonary embolectomy system market is projected to reach USD 3,707.4 million by 2036.
How much will be the pulmonary embolectomy system market growth between 2026 and 2036?
The pulmonary embolectomy system market is expected to grow at a 9.4% CAGR between 2026 and 2036.
What are the key product types in the pulmonary embolectomy system market?
The key product types in pulmonary embolectomy system market are catheter-based embolectomy systems, surgical embolectomy systems and aspiration thrombectomy devices.
Which applications segment to contribute significant share in the pulmonary embolectomy system market in 2026?
In terms of applications, massive pe segment to command 52.0% share in the pulmonary embolectomy system market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.