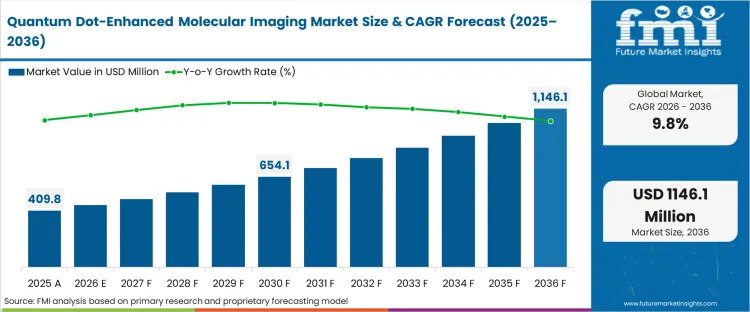
The quantum dot-enhanced molecular imaging market was valued at USD 410 million in 2025. The sector is set to cross USD 450 million in 2026 at a CAGR of 9.80% during the forecast period. Steady investment advances the valuation to USD 1,150 million through 2036, establishing a strong quantum dot molecular imaging forecast 2036 as the transition from qualitative fluorescence observation to quantifiable, long-term multi-target tracking forces researchers to abandon traditional organic dyes that suffer from rapid photobleaching.
Spatial biology and high-throughput screening protocols are forcing primary investigators to decide between maintaining legacy single-plex fluorophore workflows or upgrading to single-excitation multiplexed panels. Laboratories that delay the integration of these architectures risk losing their ability to map complex tumour microenvironments comprehensively, effectively locking themselves out of advanced oncology funding tiers. The actual barrier to adoption within the quantum dot bioimaging market is not baseline awareness but the batch-to-batch variability in quantum yield during commercial scale-up, which compromises quantitative multiplexing for translational research. Advanced quantum dot synthesis techniques are slowly resolving these disparities, creating a more predictable optical imaging environment for fluorescence molecular imaging quantum dots.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 450 million |
| Industry Value (2036) | USD 1,150 million |
| CAGR (2026 to 2036) | 9.80% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Before robust in vivo adoption accelerates, the standardization of surface passivation techniques that prevent core degradation in biological buffers over extended timeframes must be definitively achieved. Material scientists and biochemists trigger this threshold by commercializing unified silica-coating protocols that reliably mask heavy-metal toxicity without compromising the probe's hydrodynamic radius, paving the way for safer cadmium-free quantum dots for molecular imaging. Once this stability is guaranteed, the trajectory turns rapidly from basic cellular assays into real-time surgical navigation and quantum dots for in vivo imaging.
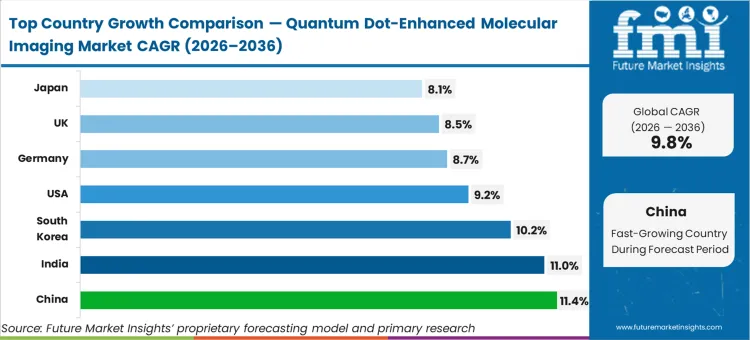
The quantum dot imaging market China leads the geographic expansion with an anticipated 11.4% CAGR. India is set to record an 11.0% CAGR. South Korea is expected to advance at 10.2%. The United States is poised to grow at a 9.2% CAGR. The quantum dot imaging market Germany is estimated to track an 8.7% CAGR. The United Kingdom is likely to follow at 8.5%. Japan is expected to expand at 8.1%. The divergence across these territories reflects a fundamental split between markets accelerating purely on massive state-backed nanomaterials funding and those constrained by stricter translational and toxicity scrutiny regarding heavy-metal clearance pathways.
To explain the quantum dot-enhanced molecular imaging market, it encompasses the commercial ecosystem of semiconductor nanocrystals engineered specifically for biological visualization. This boundary excludes traditional small-molecule fluorophores and bulk fluorescent proteins, focusing strictly on nanoscale particles that exhibit quantum confinement effects. These properties deliver tunable emission spectra, high molar extinction coefficients, and exceptional resistance to photobleaching within defined biological environments.
Scope inclusions cover all variations of functionalized and unfunctionalized quantum dots intended for biological tracing, including core-shell structures, cadmium-free alternatives, and carbon quantum dots for bioimaging. The sector also captures the specialized bioconjugation kits and surface-modified imaging markers designed to link these nanocrystals to antibodies, peptides, or oligonucleotides for specific target acquisition in both cellular and small-animal models.
The market explicitly excludes quantum dots utilized for consumer electronics, display panels, and photovoltaic applications, as these lack the rigorous biocompatibility and surface functionalization required for life sciences. Standard organic fluorescent dyes and standalone imaging instrumentation, such as microscopes or preclinical scanners, are also excluded because they represent separate capital equipment or legacy consumable categories rather than the specialized quantum dot imaging probes market specifically.
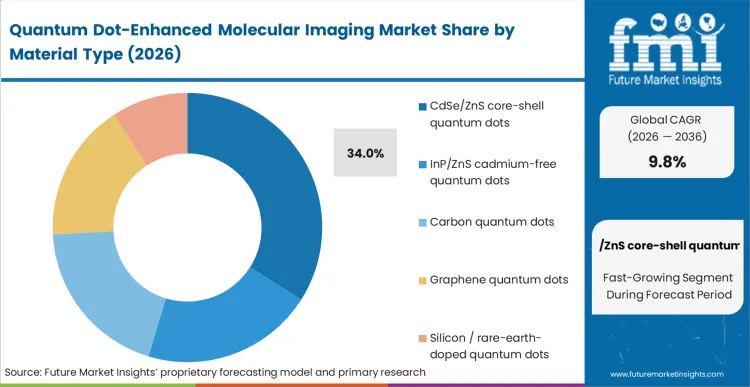
The legacy approach of utilizing standard organic dyes fails when protocols demand continuous illumination over several hours, forcing laboratories to qualify alternative architectures. CdSe/ZnS core-shell quantum dots maintain a 34.0% share because their optical brightness and narrow emission bandwidths are currently unmatched in controlled in vitro environments. According to FMI's estimates, core facility directors base their procurement primarily on these proven optical properties, provided the assays do not require in vivo safety clearances. The integration of a zinc sulfide shell effectively confines the exciton within the core, maximising quantum yield while simultaneously shielding the biological environment from immediate cadmium leakage. Laboratories that attempt long-term tracking without these robust core-shell architectures experience rapid signal degradation, necessitating repetitive dosing that alters the biological baseline. Integrating highly stable multiplex biomarker imaging frameworks effectively switches the operational bottleneck from probe stability to data analysis.
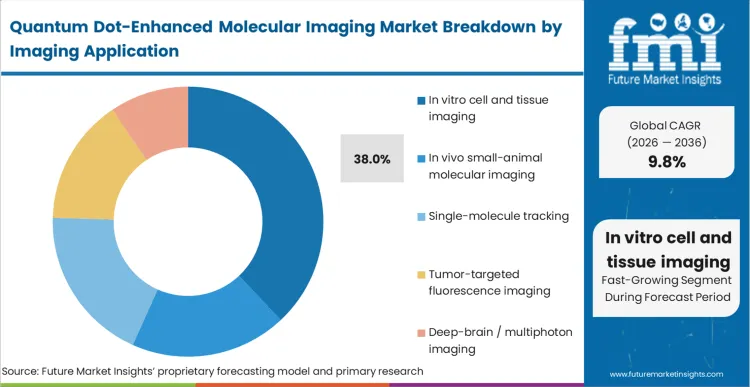
In vitro cell and tissue imaging dominates this landscape with a 38.0% share, functioning as the primary environment where the unique photophysics of quantum dots provide immediate operational superiority. FMI analysts opine that the transition away from qualitative snapshots toward quantitative spatial biology makes the extreme photostability of these probes mandatory for high-throughput screening. When multiplexing five or more targets, the broad emission tails of traditional dyes create mathematical deconvolution errors. Quantum dots eliminate this cross-talk through tightly confined emission peaks. The integration of advanced super-resolution microscope systems further amplifies this advantage, demanding probes that can withstand intense laser excitation without blinking. Facilities that rely on legacy fluorophores for complex spatial mapping face severe limitations in data reproducibility, ultimately capping their throughput capacity when executing quantum dots for single-molecule tracking.
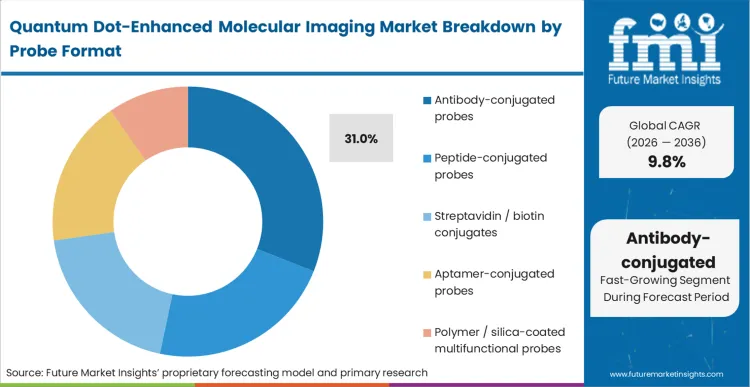
Antibody-conjugated probes secure 31.0% of the market because they bridge the gap between novel nanomaterial physics and established biological targeting protocols. As per FMI's projection, assay developers choose this format to immediately leverage existing libraries of validated antibodies, bypassing the costly and unpredictable process of discovering entirely new targeting ligands for quantum dot fluorescence probes. The challenge lies in controlling the bioconjugation stoichiometry. Attaching too many antibodies to a single nanocrystal induces aggregation, while too few reduces avidity. Overcoming this friction requires precisely engineered surface linkers that orient the antibody correctly without masking its antigen-binding site. Teams that fail to optimize this conjugation step routinely deploy probes that suffer from high non-specific binding, rendering the subsequent fluorescence data virtually useless for quantitative analysis. The deployment of nanoparticle technology specifically engineered for stable covalent linkage is slowly standardizing these outcomes.
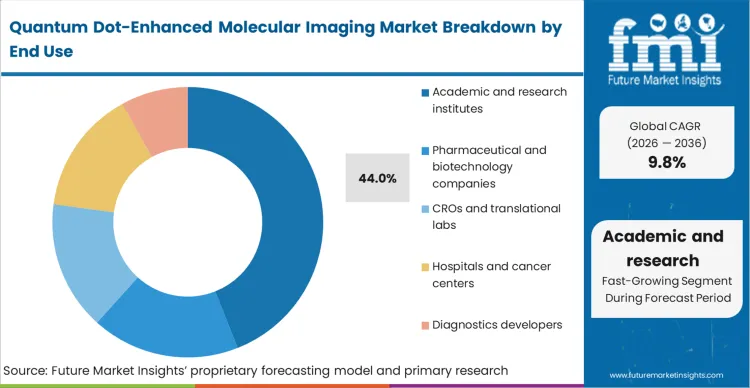
Academic and research institutes operate under a mandate to push the boundaries of spatial biology, which explains their dominant 44.0% share in this segment. Based on FMI's assessment, these entities are uniquely positioned to absorb the early-stage optimization friction inherent in adopting complex bioconjugates, as their funding models prioritize methodological innovation over immediate clinical throughput. The primary operational advantage for these institutes is the ability to construct custom multiplex panels that commercial vendors do not yet support, allowing them to map highly specific niche pathways such as those using quantum dots for deep-brain imaging. However, the custom bioconjugation processes required for these bespoke panels introduce significant batch-to-batch variability. Academic cores that fail to standardize their internal quality control metrics for these custom probes routinely produce unrepeatable data, endangering multi-year grant renewals. The integration of sophisticated pre-clinical imaging system architectures eventually forces these academic users to adopt more rigorous standardization protocols.
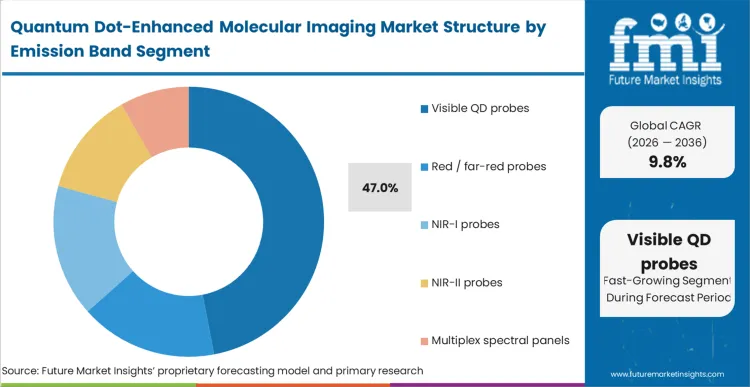
Visible QD probes hold 47.0% of the market strictly because they align seamlessly with the existing excitation and emission filter sets installed across global microscopy core facilities. In FMI's view, the decision to deploy visible-spectrum nanocrystals requires zero capital expenditure on new optical hardware, making it the most frictionless entry point for laboratories upgrading from organic dyes. The limitation of this band is the profound scattering and absorption of visible light by biological tissues, confining its utility almost entirely to thin-slice in vitro preparations or superficial cellular monolayers. The inability to penetrate deeper than a few hundred microns forces researchers investigating complex 3D organoids or whole-animal models to transition toward longer wavelengths by adopting NIR quantum dots for molecular imaging. Laboratories attempting deep-tissue visualization using visible-band probes invariably encounter overwhelming autofluorescence, rendering deep mapping impossible. The slow integration of multi-photon microscopic system hardware eventually dictates a necessary shift toward the near-infrared spectrum.
The requirement for continuous, real-time tracking of cellular events forces primary investigators to qualify optical probes that resist rapid photobleaching, answering the critical question of why are quantum dots better than fluorescent dyes. Traditional organic dyes degrade under sustained laser excitation, destroying the biological signal before complex cellular interactions can be fully mapped. This pressure compels spatial biology assay developers to integrate semiconductor nanocrystals, which offer the extreme photostability required to monitor live-cell dynamics over periods extending beyond 48 hours. Laboratories that successfully pivot to these architectures secure the ability to generate the longitudinal data sets demanded by high-tier translational oncology funding.
The single biggest friction slowing comprehensive adoption is the persistent difficulty in achieving reproducible, scalable bioconjugation without compromising the probe's hydrodynamic radius, leading many buyers to ask are quantum dots safe for biomedical imaging and effective in vivo. Adding protective shells, polymer coatings, and targeting ligands to the nanocrystal core often increases its physical size to the point where it can no longer efficiently cross cellular membranes or penetrate dense tumor microenvironments. While vendors are developing increasingly sophisticated nanoparticle technology using compact PEGylated linkers, the delicate balance between maintaining optical brightness, ensuring biological stability, and minimizing steric hindrance remains a significant operational hurdle for assay developers.
Based on the regional analysis, the Quantum Dot-Enhanced Molecular Imaging market is segmented into Asia Pacific, North America, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 11.4% |
| India | 11.0% |
| South Korea | 10.2% |
| United States | 9.2% |
| Germany | 8.7% |
| United Kingdom | 8.5% |
| Japan | 8.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Aggressive, state-backed funding initiatives focused explicitly on nanomaterials and advanced biophotonics drive this region's rapid commercial trajectory. FMI analysts opine that unlike Western markets where strict toxicity clearances often bottleneck early-stage in vivo testing, several Asian research hubs operate under translational frameworks that actively accelerate the deployment of novel optical probes into preclinical environments. This condition allows domestic research institutes to scale up custom bioconjugation pipelines at a pace unmatched globally. The integration of domestic fluorescence cell analysis technologies further localizes the supply chain.
FMI's report includes extensive coverage of emerging research nodes across Southeast Asia and Oceania. These peripheral markets consistently demonstrate an accelerated adoption of completely cadmium-free architectures, bypassing the legacy core-shell phase entirely to align immediately with the strictest international publication standards.
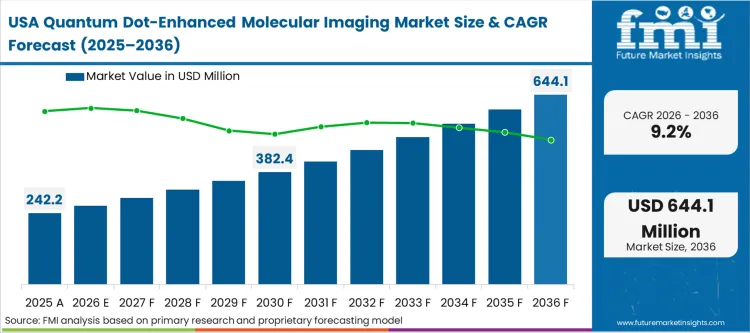
The presence of deeply established, highly concentrated pharmaceutical research infrastructure and massive oncology grant funding shapes the adoption pattern in this region. The primary force is not the speed of new material synthesis, but the rigorous integration of these advanced probes into highly standardized, high-throughput commercial screening workflows. Procurement directors prioritize extreme batch-to-batch consistency over novel emission bands, as variable quantum yields completely disrupt automated data acquisition pipelines. The deployment of advanced single molecule tracking platforms dictates a demand for highly stable, pre-conjugated reagents.
FMI's report includes analysis of the expanding Canadian biotechnology corridor. Facilities in this adjacent market demonstrate a preference for collaborative procurement frameworks, frequently pooling reagent testing data to accelerate the qualification of novel cadmium-free architectures across multiple institutional nodes.
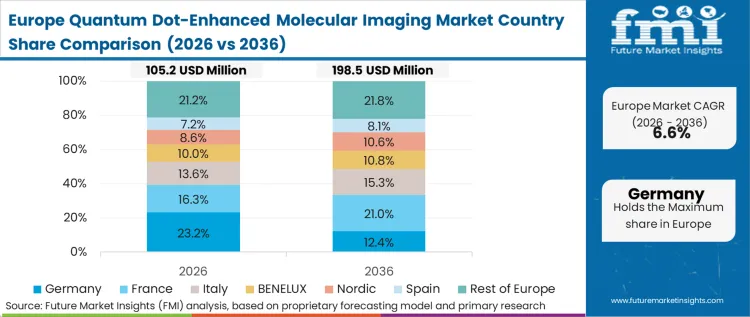
Stringent regulatory frameworks regarding heavy-metal toxicity and material clearance pathways dictate the unique operational reality for European researchers. Unlike regions that rapidly deploy cadmium-based probes for in vivo small-animal tracking, European translational laboratories operate under severe restrictions that force the premature adoption of early-generation cadmium-free alternatives. As per FMI's projection, this constraint compels European assay developers to compensate for the lower absolute quantum yield of cadmium-free dots by heavily optimizing their optical detection hardware and signal processing algorithms. The focus is heavily toward refining photoacoustic microscopy and advanced deconvolution techniques.
FMI's report includes comprehensive tracking of the Nordic and Benelux research environments. These highly integrated clusters show a pronounced move toward the adoption of sophisticated near-infrared emitting architectures, driven by specialized funding for non-invasive neuroimaging and deep-tissue mapping.
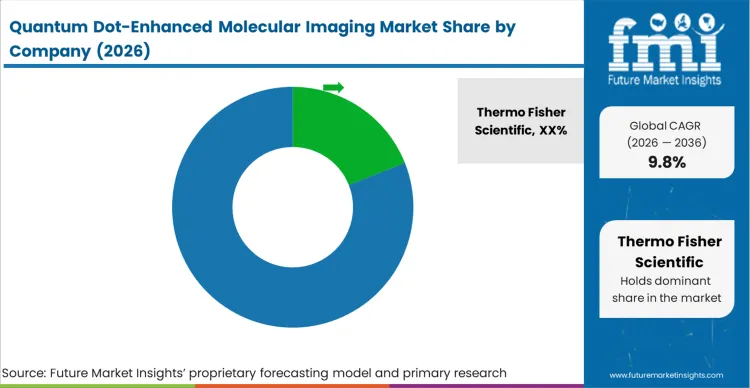
The market exhibits a high degree of concentration at the top tier, answering the question of which companies are active in quantum dot molecular imaging. This concentration is driven strictly by the complex intellectual property surrounding uniform nanocrystal synthesis and stable surface passivation. Procurement directors at major pharmaceutical firms and centralized academic cores cannot risk assay failure due to batch variability, forcing them to consolidate their purchasing around established giants. Buyers distinguish qualified vendors entirely on the reproducibility of the probe's quantum yield and the stability of its hydrodynamic radius post-conjugation, systematically excluding smaller quantum dot bioimaging reagent manufacturers who cannot guarantee commercial-scale consistency.
Incumbents maintain their dominant position through massive portfolios of highly validated, pre-conjugated antibody panels that immediately drop into existing spatial biology workflows. This advantage persists because the cost of independently verifying a novel targeting ligand far exceeds the premium charged for a commercial, ready-to-use kit. Challengers must build robust, proprietary bioconjugation pipelines that reliably attach complex biologicals without inducing aggregation, a capability that requires deep biochemistry expertise rather than just material science proficiency. The deployment of advanced fluorescence guided surgery reagents specifically demands this level of flawless conjugation from quantum dot imaging probe suppliers.
Large institutional buyers actively resist vendor lock-in by aggressively funding the internal development of custom bioconjugation protocols, attempting to separate the purchase of the raw nanocrystal from the targeting ligand. However, the tension heavily favours the dominant vendors. The sheer mathematical complexity of deconvoluting highly multiplexed spatial data strongly incentivizes researchers to utilize closed, end-to-end proprietary panels that guarantee optical compatibility. Through 2036, the market will increasingly bifurcate into high-volume commodity raw material suppliers and specialized, high-margin functionalized panel developers.
| Metric | Value |
|---|---|
| Quantitative Units | USD 450 million to USD 1,150 million, at a CAGR of 9.80% |
| Market Definition | This market encompasses semiconductor nanocrystals engineered specifically for biological visualization, excluding traditional organic dyes to focus strictly on probes delivering tunable emission spectra and extreme photostability. |
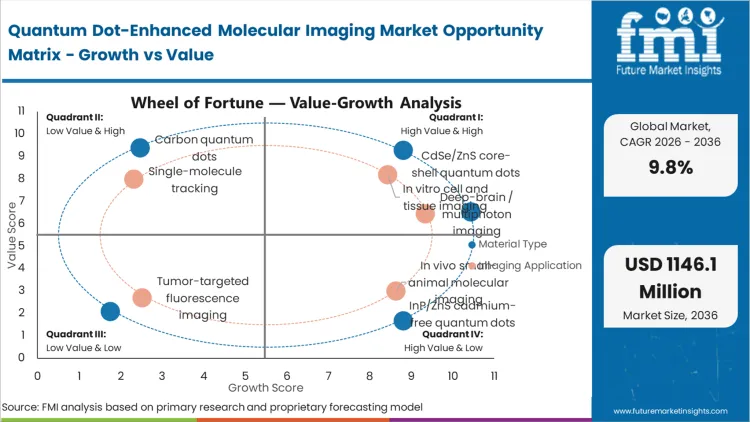
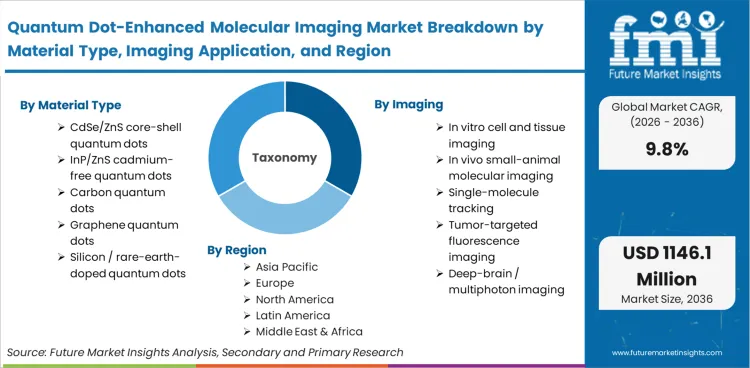
| Material Type Segmentation | CdSe/ZnS core-shell quantum dots, InP/ZnS cadmium-free quantum dots, carbon quantum dots, graphene quantum dots, silicon / rare-earth-doped quantum dots |
| Imaging Application Segmentation | In vitro cell and tissue imaging, in vivo small-animal molecular imaging, single-molecule tracking, tumor-targeted fluorescence imaging, deep-brain / multiphoton imaging |
| Probe Format Segmentation | Antibody-conjugated probes, peptide-conjugated probes, streptavidin / biotin conjugates, aptamer-conjugated probes, polymer / silica-coated multifunctional probes |
| End User Segmentation | Academic and research institutes, pharmaceutical and biotechnology companies, CROs and translational labs, hospitals and cancer centers, diagnostics developers |
| Emission Band Segmentation | Visible QD probes, red / far-red probes, NIR-I probes, NIR-II probes, multiplex spectral panels |
| Regions Covered | Asia Pacific, North America, Europe |
| Countries Covered | China, India, South Korea, United States, Germany, United Kingdom, Japan, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Merck KGaA (Sigma-Aldrich), Revvity, Inc., Bruker Corporation, Vector Laboratories, Leica Microsystems, Cytodiagnostics Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted directors of preclinical imaging core facilities and spatial biology assay developers. Baseline data anchored to the verifiable volume of specialized multiplexing kits shipped to institutional networks. Forecasts were cross-validated against institutional grant disbursements for spatial transcriptomics infrastructure. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Quantum Dot-Enhanced Molecular Imaging Market in 2026?
The sector is anticipated to reach USD 450 million in 2026. This baseline reflects the current volume of specialized multiplexing kits shipping directly into academic and translational core facilities attempting to upgrade from traditional organic fluorophores.
What will it be valued at by 2036?
The valuation is projected to hit USD 1,150 million by the end of 2036. This cumulative buildup indicates the expected transition where functionalized nanocrystals become pre-integrated into standardized spatial biology and high-throughput screening platforms.
What CAGR is projected?
A 9.80% compound annual growth rate is estimated over the forecast period. This pace is constrained by the complex biochemistry required to achieve reproducible bioconjugation, rather than a lack of underlying demand for highly photostable imaging agents.
Which material type segment leads?
CdSe/ZnS core-shell quantum dots dominate with a 34.0% share. Facilities rely on this established architecture because the zinc sulfide shell maximizes the exciton confinement, delivering the highest absolute brightness required to detect scarce cellular receptors.
What are quantum dots used for in molecular imaging?
They are engineered to replace traditional dyes for complex quantum dots for tumor imaging and prolonged cellular mapping. Their extreme photostability prevents rapid signal degradation, allowing researchers to track live-cell events continuously for days instead of hours.
Which imaging application segment leads?
In vitro cell and tissue imaging captures a 38.0% share. The segment leads because standard high-content screening environments do not require the rigorous heavy-metal toxicity clearances that severely bottleneck in vivo small-animal applications.
Which probe format segment leads?
Antibody-conjugated probes secure 31.0% of the market. Assay developers overwhelmingly choose this format to immediately leverage decades of validated immunoglobulin research, completely bypassing the massive cost of discovering novel targeting ligands from scratch.
How do quantum dots vs organic fluorophores compare structurally?
Traditional organic dyes are small molecules that suffer from broad emission tails and rapid photobleaching under laser light. Semiconductor nanocrystals offer narrow, tunable emission peaks and crystalline structures that resist photon-induced degradation, mathematically eliminating cross-talk errors during deconvolution.
What is the primary restraint?
The inability to consistently conjugate probes without increasing their hydrodynamic radius severely restricts cellular penetration. If the addition of protective coatings and targeting ligands makes the dot too large, it cannot efficiently cross biological membranes or enter dense tissue microenvironments.
Which country grows fastest?
China expands at an 11.4% CAGR, noticeably outpacing the United States at 9.2%. This divergence exists because Chinese institutes benefit from aggressive state funding explicitly targeting domestic nanomaterial synthesis, bypassing the prolonged early-phase translational scrutiny typical of Western regulatory environments.
How do European chemical regulations impact adoption?
Strict directives regarding heavy-metal clearance force European facilities to prematurely move away from highly efficient cadmium cores. Core directors in Germany and the UK must optimize their optical hardware extensively to compensate for the inherently lower quantum yield of the permitted InP/ZnS alternatives.
Why are visible QD probes currently dominant over NIR architectures?
Visible band probes capture 47.0% share because they map perfectly onto the 488nm and 561nm laser lines already installed in global microscopy cores. Upgrading to NIR-II dots requires massive capital expenditure on specialized InGaAs cameras and longer-wavelength excitation sources.
How do major buyers distinguish between competing probe vendors?
Institutional procurement directors do not base decisions on minor differences in theoretical brightness. They evaluate vendors strictly on batch-to-batch consistency and the guaranteed absence of probe aggregation upon delivery, systematically excluding suppliers that introduce variability into automated data pipelines.
What makes custom bioconjugation a significant operational risk?
Academic cores attempting to link their own custom antibodies to raw nanocrystals frequently struggle with stoichiometry. Improper conjugation either masks the antigen-binding site or causes the particles to clump, completely ruining the quantification of the subsequent spatial assay.
How do quantum dots improve deep-tissue imaging?
Visible light scatters aggressively within biological tissue, restricting analysis to the absolute surface or requiring destructive physical sectioning. Probes engineered to emit in the 1000 to 1700 nm range evade this scattering, allowing researchers to track real-time events deep within intact animal models.
When evaluating carbon quantum dots vs cadmium quantum dots, what dictates choice?
The choice is strictly dictated by the translational stage of the assay. In vitro researchers prefer cadmium cores for maximum absolute brightness, while groups planning immediate in vivo models choose carbon dots to completely bypass the rigorous heavy-metal clearance hurdles.
What changes when silica-coating protocols become standardized?
Uniform silica passivation definitively seals the heavy-metal core and standardizes the surface chemistry for easy ligand attachment. Once this occurs, the primary operational bottleneck shifts from probe stability and toxicity management entirely over to advanced image data processing.
Why do academic institutes lead in end-user adoption?
Holding a 44.0% share, academic centers are positioned to absorb the early-stage friction of utilizing complex, custom-built bioconjugates. Their grant funding models reward methodological innovation, allowing them to experiment with novel multiplex panels before commercial vendors standardize them.
What is the future of quantum dots in imaging?
The necessity for flawless integration into high-throughput screening will force the market to bifurcate. A few major chemical suppliers will provide the commodity nanocrystal cores, while value will concentrate in specialized vendors delivering closed, highly functionalized, and mathematically validated multiplex panels.
What role do preclinical CROs play in India's 11.0% growth?
Indian contract research organizations are rapidly upgrading their imaging capabilities to secure complex longitudinal oncology studies from Western pharmaceutical clients. Offering robust quantum dot-enhanced multiplexing allows these CROs to deliver data quality that standard dye-based assays cannot match.
How are diagnostic developers utilizing aptamer-conjugated probes?
Developers leverage aptamers because these synthetic oligonucleotides can be engineered to be significantly smaller than full-length antibodies. This dramatic reduction in the overall probe radius heavily mitigates the steric hindrance issues that traditionally block tissue penetration.
Why is the United States growth constrained compared to Asia?
The 9.2% US expansion reflects a highly mature, heavily consolidated procurement environment. Massive pharmaceutical operators demand extreme validation and consistency before integrating a new probe into their automated pipelines, creating a slower, more deliberate adoption curve than in heavily subsidized emerging research hubs.
What is the functional advantage of multi-photon excitation with these probes?
Multi-photon microscopy utilizes pulsed near-infrared lasers to excite visible-emitting quantum dots only at the precise focal point. This virtually eliminates out-of-focus background fluorescence and further minimizes phototoxicity in delicate live-tissue preparations.
Can quantum dots be used in vivo effectively?
Yes, but effective in vivo utilization is entirely dependent on the standardization of surface passivation and polymer coatings. Uncoated cores rapidly degrade and leak heavy metals, restricting true in vivo applications to those using carefully engineered, non-toxic, or cadmium-free architectures.
Which quantum dots are cadmium-free?
InP/ZnS (indium phosphide/zinc sulfide) structures, carbon quantum dots for bioimaging, and silicon-based nanocrystals completely remove cadmium from the equation. These materials sacrifice a marginal amount of absolute optical brightness in exchange for immediate compliance with stringent European toxicity frameworks.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.