Skin Perfusion Pressure Testing Devices Market
Skin Perfusion Pressure Testing Devices Market Size and Share Forecast Outlook 2025 to 2035
About the Report
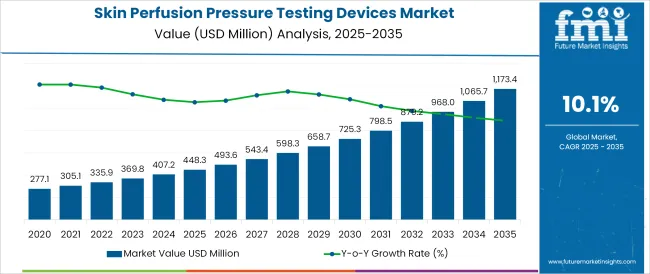
The Skin Perfusion Pressure Testing Devices Market is estimated to be valued at USD 448.3 million in 2025 and is projected to reach USD 1173.4 million by 2035, registering a compound annual growth rate (CAGR) of 10.1% over the forecast period.
Quick Stats for Skin Perfusion Pressure Testing Devices Market
- Industry Value (2025): USD 448.3 million
- Forecast Value (2035): USD 1173.4 million
- Forecast CAGR: 10.1%
- Leading Segment in 2025: Laser Doppler Skin Perfusion Pressure Testing Devices (42.5%)
- Key Growth Region: North America, Asia-Pacific, Europe
- Top Key Players: Vasamed Incorporated, Moor Instruments, Perimed AB, ELCAT GmbH, ATYS Medical, Viasonix, Promed Group
Rationale for Segmental Growth in the Skin Perfusion Pressure Testing Devices Market
Market Overview
The skin perfusion pressure (SPP) testing devices market is witnessing notable growth, fueled by increasing clinical awareness of peripheral artery disease and the need for accurate assessment of microcirculatory blood flow. Industry publications and clinical practice updates have emphasized the role of SPP testing in evaluating wound healing potential and guiding revascularization decisions.
Hospitals and vascular clinics have incorporated SPP testing into standard diagnostic protocols, particularly for diabetic foot ulcers and chronic limb ischemia management. Continuous technological advancements have led to the introduction of compact, user-friendly devices, enhancing accessibility across various healthcare settings.
Manufacturers have focused on developing non-invasive, real-time monitoring solutions, improving diagnostic accuracy and patient comfort. Future market expansion is expected through product innovation in portable and laser Doppler-based technologies and the rising demand for point-of-care diagnostic tools.
Segmental growth is anticipated to be driven by Laser Doppler Skin Perfusion Pressure Testing Devices, Portable Devices, and Hospitals as the primary end users, reflecting advancements in technology, ease of use, and the clinical necessity of these tools.
Segmental Analysis
The market is segmented by Product, Modality, and End User and region. By Product, the market is divided into Laser Doppler Skin Perfusion Pressure Testing Devices, Photoplethysmography Skin Perfusion Pressure Testing Devices, Consumables, Pressure Cuff Controller, and Fibre Optic Probe. In terms of Modality, the market is classified into Portable Devices and Cart-based Devices.
Based on End User, the market is segmented into Hospitals, Ambulatory Surgical Centers, Wound Care Centers, and Vascular Laboratories. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Product Segment: Laser Doppler Skin Perfusion Pressure Testing Devices
The Laser Doppler Skin Perfusion Pressure Testing Devices segment is projected to account for 42.5% of the market revenue in 2025, establishing itself as the leading product segment. Growth of this segment has been driven by the high sensitivity of laser Doppler technology in measuring microvascular blood flow, enabling accurate skin perfusion assessment.
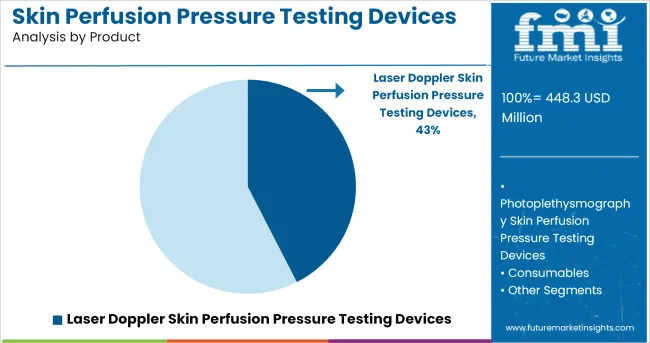
Hospitals and vascular specialists have preferred laser Doppler devices due to their ability to provide real-time, quantitative measurements, facilitating precise diagnosis and treatment planning. Continuous product enhancements have resulted in compact and ergonomic designs, improving user convenience and patient compliance.
Additionally, clinical publications have highlighted the role of laser Doppler in improving the prognosis of patients undergoing limb salvage procedures, reinforcing its clinical relevance. As demand increases for evidence-based vascular assessments, the Laser Doppler Skin Perfusion Pressure Testing Devices segment is expected to maintain its leadership through continuous technological evolution and expanding clinical adoption.
Insights into the Modality Segment: Portable Devices
The Portable Devices segment is projected to contribute 56.3% of the skin perfusion pressure testing devices market revenue in 2025, solidifying its leadership in modality adoption. This segment’s growth has been driven by the demand for point-of-care vascular assessment tools, allowing clinicians to perform diagnostics in outpatient, home care, and remote clinical settings.
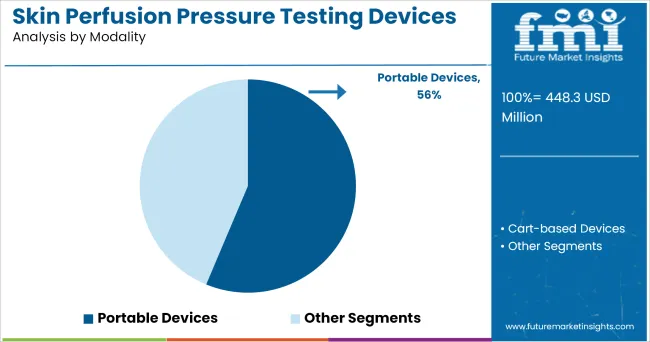
Portable SPP devices have been favored for their lightweight design, ease of transport, and rapid setup, enabling vascular assessments outside traditional hospital environments. Manufacturers have focused on developing user-friendly interfaces and wireless data transmission capabilities, supporting telehealth integration and remote monitoring.
Additionally, the portability of these devices has facilitated their use in wound care clinics and community health initiatives aimed at early detection of peripheral vascular disease. As healthcare delivery continues to shift toward decentralized and home-based care models, the Portable Devices segment is expected to retain its dominant position, supported by increasing demand for accessible diagnostic solutions.
Insights into the End User Segment: Hospitals
The Hospitals segment is projected to account for 39.70% of the skin perfusion pressure testing devices market revenue in 2025, sustaining its leadership among end users. Growth of this segment has been driven by the clinical need for comprehensive vascular diagnostics in acute and chronic care settings.
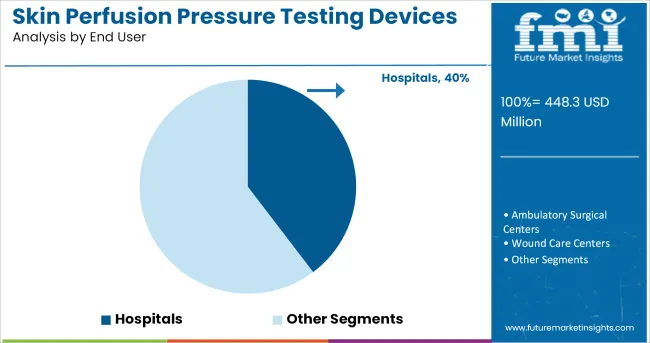
Hospitals have integrated SPP testing into multidisciplinary vascular care pathways, particularly in diabetic foot clinics, wound care centers, and vascular surgery units. Institutional procurement of advanced diagnostic equipment has prioritized devices with high diagnostic accuracy and patient safety, further boosting the adoption of SPP testing systems.
Hospitals have remained the preferred environment for managing complex vascular conditions, where immediate diagnostic and interventional capabilities are required. As reimbursement frameworks evolve to support preventative diagnostics and clinical guidelines recommend SPP testing for wound healing evaluation, the Hospitals segment is expected to remain the primary user base, reinforced by ongoing investments in advanced vascular diagnostics.
How Can Historic and Future Outlook for Skin Perfusion Pressure Testing Devices Market Be Compared?
Peripheral artery disease (PAD) is a progressive disorder affecting more than 200 million people, worldwide. It has a significant impact on mortality in hemodialysis patients. Approximately 6.5 million people aged 40 years and older in the United States have PAD. The increasing PAD population has propelled the growth of the skin perfusion pressure testing market.
According to FMI's analysis, skin perfusion pressure testing devices sales have grown at 5.3% CAGR between 2020 and 2024.
Skin perfusion pressure has become a potential tool for wound healing in patients with chronic limb ischemia. Compared to other non-invasive methods such as toe blood pressure and ankle-brachial index, skin perfusion pressure provides more accurate results, thus, its usage is gaining popularity among physicians.
This technique also remains a clinically relevant and objective tool of local perfusion in the setting of chronic limb ischemia, particularly among higher-risk patients with arterial calicifications and comorbidities that tend to microvascular disease. FMI expects global skin perfusion pressure testing devices market to grow at 4.5% CAGR through 2035.
How is Shift to Non-invasive Procedures Affecting Skin Perfusion Pressure Testing Devices Sales?
Non-invasive procedures have continued to grow in popularity as compared to surgeries. The measurement of skin perfusion pressure by a laser doppler is a non-invasive method that is widely used by professionals. Due to high reproducibility, accuracy, specificity, and sensitivity, skin perfusion pressure testing has become one of the soundest methods.
For instance, non-invasive VMS-VASC system (Moor) Instruments), including the laser Doppler monitor moorVMS-LDF, low profile fibre optic probe (VP-SPP), and pressure cuff controller moorVMS-PRES have been specifically designed to aid micro vascular assessments in a variety of clinical applications and physiological research.
What is the Commercial Potential for Laser Doppler Skin Perfusion Pressure Testing Devices?
Laser dopplers have proven to be effective devices for the measurement of skin perfusion pressure. Laser dopplers are among the regularly utilized gadgets for the appraisal of skin perfusion pressure, they are seen as the ideal instrument for the location of peripheral artery disease as well as diagnosis of wound healing potential in limb ischemia patients.
Furthermore, the accuracy in a laser doppler is much higher than any other traditional method. The high accuracy, simplicity of operability and non-invasiveness feature of the devices are likely to fuel the utilization of laser dopplers by the physicians, boosting the demand of the skin perfusion testing devices.
What is the Impact of FDA Regulatory Approvals on Global Sales?
Due to an increase in the need for assessment of wound healing in peripheral artery disease and limb ischemia, many manufacturers are seeking 510(k) Premarket Notification to FDA which demonstrates the device to be safe and effective as equivalent to a legally-marketed device
For example, Perimed Inc. seeks 510(k) Premarket Notification for PeriFlux System 5000, which is a laser doppler system consisting of main units and functional units
However, according to FDA Modernization Act, devices that fall under class I and II are exempted from premarket approval, thus, the time required for regulatory approval is reduced, which brings down the costs. The increasing FDA approvals attracts the end users, and this expected to boost the demand for skin perfusion pressure testing devices.
How is Lack of Skilled Professionals Restraining Growth?
Skin pressure perfusion is the most advanced method for the assessment of wound healing in limb ischemia and peripheral artery diseases. This method requires highly trained or skilled professionals to operate the device.
Since it is fully-automated and provides quantitative evaluation of microcirculation, its operability requires skilled people, which as a result may limit the use of skin perfusion pressure testing devices.
Thus, reliability and reproducible measurement is highly dependent on a skilled technician or operator who interprets the results of skin perfusion pressure measurement. Lack of skilled professionals therefore can create significant challenges for the market.
Country-wise Insights
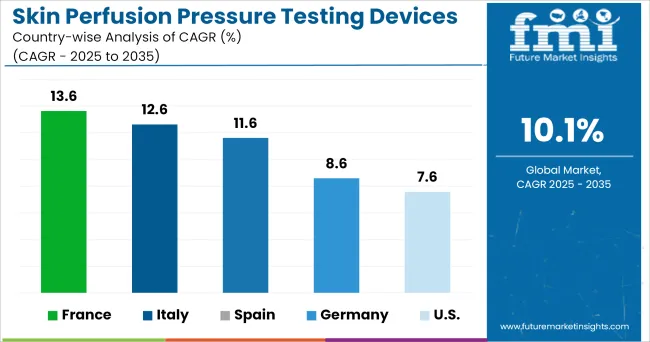
How Big is the Opportunity in the USA?
The healthcare system of the USA is well renowned for its advanced infrastructure and expenditure. The care regime of the care centers and hospitals are monitored by various regulatory rules and insurance policies. The massive hospital bills incurred other than the Medicare have been influencing the market in the country.
Government and regulatory bodies of the USA are adopting several cost-containment plans to reduce their healthcare burden. The shift from a volume to a value-based system is majorly driven by the various measures undertaken by providers, governments, biotechnology companies, and insurance payers.
Some of the measures are outcome-based profits, pricing, risk sharing, competitive tendering, and price control.
A value-based healthcare system will not only provide improved outcomes through integrated care pathways, but also levy pricing pressure on medical device companies, and this is likely to impact investments on the R&D of advanced medical treatment technologies.
Furthermore, due to the higher prevalence of critical limb ischemia and peripheral arterial disease in USA, several manufactures are conducting campaigns to raise general awareness among people related to these diseases.
This is likely to support the overall market growth in the country. For example, Amputee Coalition of America (NGO) launched a campaign in April 2020 to raise limb loss awareness among people.
Why Innovation is Key to Success in German Medical Device Market?
The Federal Statistical Office has reported that healthcare spending in Germany rose by 4% in 2020 from 2020. In 2020, the country’s healthcare spending accounted for 11.7% of the GDP. Furthermore, health expenditure was expected to surge to 277.1 billion Euros by 2020, presenting a lucrative landscape for the expansion of the skin perfusion pressure testing devices.
Also presence of companies such as ELCAT GmbH is supporting the expansion in Germany. The company has been investing significantly in expansion efforts. Growth strategies adopted by these companies are expected to aid the expansion in Germany.
Since 2009, health insurance has been made mandatory for all citizens of the country. According to National Center for Biotechnology Information (NCBI), a total of 70 million people, which roughly covers 85% of the population are covered by statutory health insurance.
Another 11% population of Germany is covered by substitutive private health insurance. Conducive reimbursement policies and regulatory scenario are presenting attractive opportunities for growth.
How is Rising Disposable Income in China Affecting Growth?
Increasing disposable income can have significant impact in development of economy in emerging countries which results in affordability of proper healthcare treatment. China’s per capita disposable income reached (about 4,961 USA dollars) in 2024, more than double the level in 2010.
Potential for high healthcare insurance and rising disposable income will contribute to increase in affordability of healthcare services. This will also allow improved accessibility to expensive equipment for treatment of diseases. The market in China is expected to witness significant growth at a CAGR of around 5.4%.
How Can Players Boost Skin Perfusion Pressure Testing Devices Sales in South Africa?
Both chronic limb ischemia and peripheral artery disease impose serious threats to the population. Lack of awareness of diseases and lack of proper treatment leads to high prevalence of these diseases. Due to limited campaigning about the assessment of these diseases, there seems to occur less usage of these devices in hospitals in South Africa.
Moreover, poor healthcare facilities in some countries restrain the growth of the skin perfusion pressure testing market. Despite financial burden, government in South Africa is adopting various initiatives to spread create awareness among population, which is expected to enable steady growth in the market. The market in South Africa is expected to witness sluggish growth at a CAGR of just around 3.8%.
Category-wise Insights
How Has Laser Doppler Skin Perfusion Testing Devices Emerged Dominant?
Laser dopplers are widely used for skin perfusion pressure testing. Due to easy operability and noninvasiveness, most physicians opt for laser dopplers. Since laser dopplers are costly, manufacturers provide reimbursements for such devices.
However, the use of laser dopplers is not only limited to the measurement of skin perfusion pressure. These can be used in a wide range of testing parameters such as toe blood pressure (TBP), post occlusive reactive hyperaemia (PORH), ankle brachial pressure index (ABPI), and others.
For example, Moor Instrument’s new moorVMS-VASC 2.0 monitoring system is designed to test pulse volume recording (PVR) and other parameters apart from skin perfusion pressure.
What’s the Market Share of Portable Devices?
The portable devices segment will continue to grow and dominate the market during the forecast period. According to FMI, the segment is expected to hold around 73% of the market share, in terms of modality, by the end of 2035. A portable device is a wearable or hand-held device. This is designed for easy usage, to be able to move or carry it from one place to another, thus its ease of operability, makes the device most widely adopted devices in the market.
Why are Hospitals Considered Leading End Users?
Hospitals have been dominating the market as key end users because it receives higher footfall of patients. Besides this, hospitals have more advanced infrastructure to support advance equipment. Backed by these factors, FMI forecasts hospitals to continue dominating the market and account for 34.7% of the overall share in 2024.
Competitive Landscape
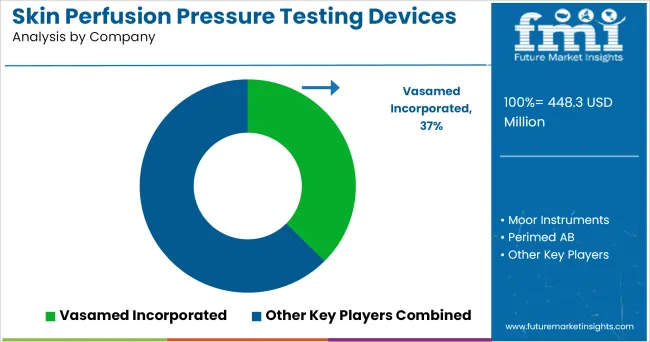
Most of the medical device companies are currently focusing on designing laser doppler since it has gain much popularity in assessment of wound healing.
For example, Moor Instruments has design moorVMS-VASC 2.0 laser doppler system. Moreover, the manufactures are focusing on getting stamp of approvals for their product and also entering into partnership and collaboration agreement to expand their product portfolio in different countries globally.
For instance, in April 2020, Perimed AB announced FDA approval for PeriFlux 6000.
Some of the leading companies operating in the market are:
- Vasamed Incorporated
- Moor Instruments
- Perimed AB
- ELCAT GmbH
- ATYS Medical
- Viasonix
- Promed Group
Scope of the Report
| Attribute | Details |
|---|---|
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2020 to 2020 |
| Market Analysis | USD Million for Value |
| Key Regions Covered | North America; Latin America; Europe; East Asia; South Asia; Oceania Middle East & Africa |
| Key Countries Covered | USA, Canada, Germany, UK, France, Italy, Spain, Poland, Russia, China, Japan, South Korea, India, Thailand, Malaysia, Indonesia, Australia, New Zealand, GCC Countries, Turkey, Northern Africa, South Africa |
| Key Segments Covered | Product Type, Modality, End User, and Region |
| Key Companies Profiled | Vasamed Incorporated; Moor Instruments; Perimed AB; ELCAT GmbH; ATYS Medical; Viasonix; Promed Group |
| Report Coverage | Market Forecast, Competition Intelligence, DROT Analysis, Market Dynamics and Challenges, Strategic Growth Initiatives |
| Customization & Pricing | Available upon Request |
Skin Perfusion Pressure Testing Devices Market by Category
By Product:
- Laser Doppler Skin Perfusion Pressure Testing Devices
- Photoplethysmography Skin Perfusion Pressure Testing Devices
- Consumables
- Pressure Cuff Controller
- Fibre Optic Probe
By Modality:
- Portable Devices
- Cart-based Devices
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Wound Care Centers
- Vascular Laboratories
By Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Middle East and Africa (MEA)
- Oceania
Frequently Asked Questions
How big is the skin perfusion pressure testing devices market in 2025?
The global skin perfusion pressure testing devices market is estimated to be valued at USD 448.3 million in 2025.
What will be the size of skin perfusion pressure testing devices market in 2035?
The market size for the skin perfusion pressure testing devices market is projected to reach USD 1,173.4 million by 2035.
How much will be the skin perfusion pressure testing devices market growth between 2025 and 2035?
The skin perfusion pressure testing devices market is expected to grow at a 10.1% CAGR between 2025 and 2035.
What are the key product types in the skin perfusion pressure testing devices market?
The key product types in skin perfusion pressure testing devices market are laser doppler skin perfusion pressure testing devices, photoplethysmography skin perfusion pressure testing devices, consumables, pressure cuff controller and fibre optic probe.
Which modality segment to contribute significant share in the skin perfusion pressure testing devices market in 2025?
In terms of modality, portable devices segment to command 56.3% share in the skin perfusion pressure testing devices market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2025 to 2035
- Laser Doppler Skin Perfusion Pressure Testing Devices
- Photoplethysmography Skin Perfusion Pressure Testing Devices
- Consumables
- Pressure Cuff Controller
- Fibre Optic Probe
- Y-o-Y Growth Trend Analysis By Product , 2020 to 2024
- Absolute $ Opportunity Analysis By Product , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Modality
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Modality, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Modality, 2025 to 2035
- Portable Devices
- Cart-based Devices
- Y-o-Y Growth Trend Analysis By Modality, 2020 to 2024
- Absolute $ Opportunity Analysis By Modality, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Ambulatory Surgical Centers
- Wound Care Centers
- Vascular Laboratories
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- U.S.
- Canada
- Mexico
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- U.K.
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Modality
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Modality
- By End User
- Key Takeaways
- Key Countries Market Analysis
- U.S.
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- U.K.
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Modality
- By End User
- U.S.
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Modality
- By End User
- Competition Analysis
- Competition Deep Dive
- Vasamed Incorporated
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Moor Instruments
- Perimed AB
- ELCAT GmbH
- ATYS Medical
- Viasonix
- Promed Group
- Vasamed Incorporated
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020 to 2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025 to 2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025 to 2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025 to 2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025 to 2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025 to 2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025 to 2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

