Demand for Aseptic Processing in USA
Demand for Aseptic Processing in USA Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Demand for Aseptic Processing in USA 2026 to 2036
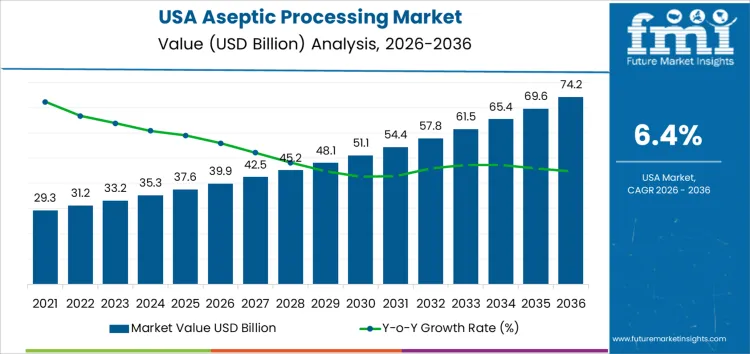
Demand for aseptic processing in USA is valued at USD 39.9 billion in 2026 and is forecasted to reach USD 74.2 billion by 2036, reflecting a CAGR of 6.4%. Demand expands due to rising production of sterile pharmaceuticals, biologics, injectable drugs, and shelf-stable food and beverage products. Regulatory emphasis on contamination control, product safety, and extended shelf life reinforces adoption of aseptic technologies across manufacturing environments.
Quick Stats for Demand for Aseptic Processing in USA
- USA Aseptic Processing Sales Value (2026): USD 39.9 billion
- USA Aseptic Processing Forecast Value (2036): USD 74.2 billion
- USA Aseptic Processing Forecast CAGR (2026 to 2036): 6.4%
- Leading Type Segment in USA: Packaging
- Key Growth Regions in USA: West USA, South USA, Northeast USA, Midwest USA
- Top Players in USA: STERIS Corporation, Getinge, Sartorius, MilliporeSigma (Merck KGaA), IMA Life
Packaging leads type-segment usage because aseptic filling and sealing protect product integrity while enabling room-temperature storage and extended distribution reach. Manufacturers prioritize advanced aseptic packaging systems that ensure sterility assurance, high throughput, and compatibility with liquid, semi-liquid, and sensitive formulations. Integration of automated filling, sterilization, and environmental monitoring improves process reliability and compliance.
West USA, South USA, Northeast USA, and Midwest USA represent key growth regions supported by strong pharmaceutical manufacturing bases, biotech clusters, and food processing infrastructure. STERIS Corporation, Getinge, Sartorius, MilliporeSigma (Merck KGaA), and IMA Life anchor competitive activity through sterilization systems, aseptic filling technologies, and integrated solutions aligned with U.S. regulatory standards and evolving production scalability requirements.
USA Aseptic Processing Key Takeaways
| Metric | Value |
|---|---|
| USA Aseptic Processing Sales Value (2026) | USD 39.9 billion |
| USA Aseptic Processing Forecast Value (2036) | USD 74.2 billion |
| USA Aseptic Processing Forecast CAGR (2026 to 2036) | 6.4% |
Why is the Demand for Aseptic Processing in USA Growing?
Demand for aseptic processing in the United States grows due to expanding production of shelf stable foods, beverages, and injectable pharmaceuticals requiring strict microbial control. Food manufacturers use aseptic systems to extend product shelf life without refrigeration while preserving nutritional quality and flavor stability. Rising consumption of ready to drink beverages, dairy alternatives, soups, and liquid nutrition supports investment in sterile filling and packaging lines. Pharmaceutical and biotechnology companies rely on aseptic processing for vaccines, biologics, and parenteral drugs that require contamination free environments.
Growth in contract manufacturing strengthens adoption as service providers standardize high capacity aseptic facilities to meet client requirements. Regulatory oversight from domestic health authorities emphasizes validated sterility assurance, which reinforces continuous system upgrades. Healthcare demand for injectable therapies increases batch volumes that depend on reliable aseptic workflows. Technological advancements improve automation, isolator systems, and real time monitoring, which supports consistency and reduces manual intervention. Sustainability goals encourage use of lightweight packaging compatible with aseptic formats. Domestic focus on supply resilience increases preference for long shelf life products produced within national facilities.
How Are the Segments Classified in the Demand for Aseptic Processing in USA?
Demand for aseptic processing in USA is shaped by shelf-life extension needs, stringent food safety regulations, and preference for preservative-free products. Producers evaluate contamination control, system reliability, material compatibility, and compliance with FDA and USDA requirements. Adoption patterns show strong concentration in food and beverage manufacturing, supported by large-scale processing infrastructure and focus on ambient-stable distribution models.
Which Aseptic Processing Type Accounts for the Highest Demand in USA?
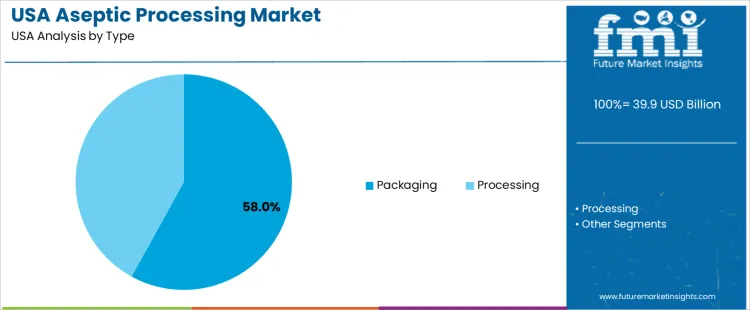
Packaging holds 58.0%, accounting for the highest demand within aseptic processing in USA. Aseptic packaging ensures sterile filling and sealing, preserving product safety throughout storage and distribution without refrigeration. Food and beverage manufacturers prioritize packaging systems to maintain final product integrity and extend shelf life. Processing holds 42.0%, covering upstream sterilization of liquids and semi-liquids before filling. Type distribution reflects stronger investment in packaging-focused systems, as final containment remains critical for aseptic assurance across USA production lines.
Key Points
- Packaging leads due to role in shelf-life and safety control.
- Sterile filling supports ambient distribution models.
- Processing systems enable upstream product sterilization.
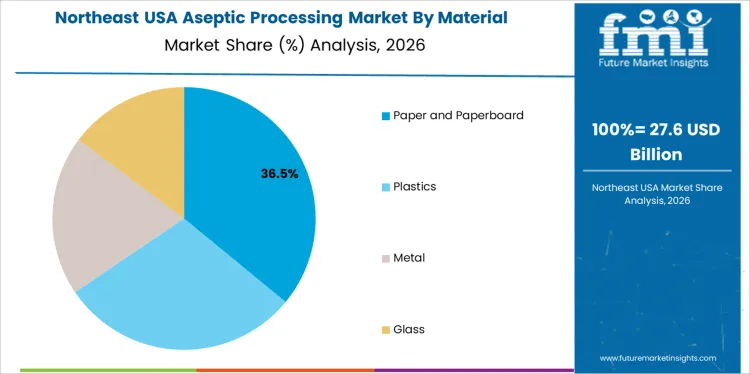
Which Material Is Most Commonly Used in Aseptic Processing Systems in USA?
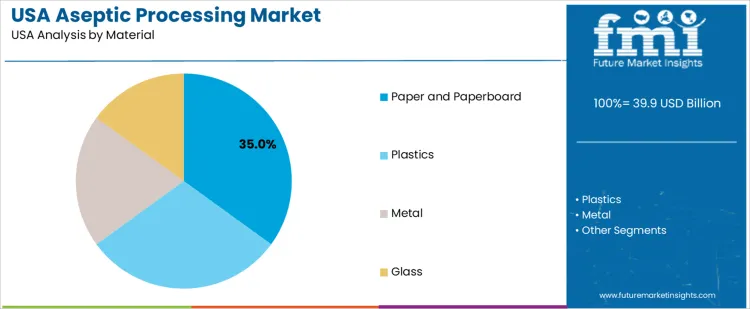
Paper and paperboard hold 35.0%, making them the most commonly used material in USA aseptic processing. These materials are widely applied in carton-based packaging for milk, juices, and liquid foods. Plastics hold 30.0%, supporting flexible and rigid containers requiring barrier performance. Metal accounts for 20.0%, used in cans and industrial applications. Glass holds 15.0%, serving premium and niche products. Material distribution reflects balance between functionality, cost efficiency, and recyclability considerations.
Key Points
- Paperboard dominates due to carton-based beverage packaging.
- Plastics provide flexible and rigid barrier solutions.
- Metal and glass support specialized applications.
Which Application Sector Drives Aseptic Processing Demand in USA?
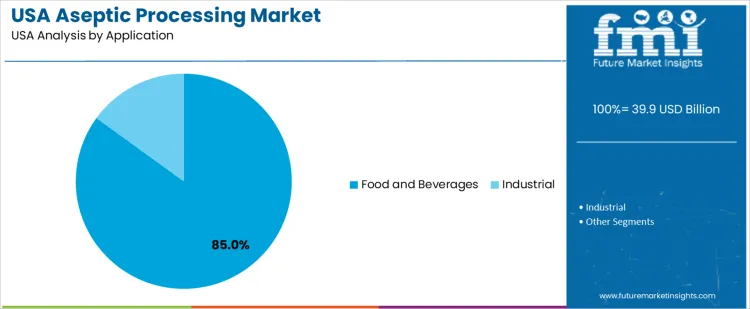
Food and beverages hold 85.0%, driving aseptic processing demand in USA. Aseptic systems support dairy products, juices, sauces, soups, and ready-to-drink beverages requiring long shelf life and safety assurance. Industrial applications hold 15.0%, covering specialty chemicals and liquid intermediates needing sterile handling. Application distribution reflects dominant reliance on aseptic technologies within food and beverage production driven by consumer demand for convenience and safety.
Key Points
- Food and beverages dominate due to shelf-stable product demand.
- Aseptic systems reduce need for preservatives.
- Industrial usage remains limited to specialty liquids.
What are the Key Dynamics in the Aseptic Processing Industry?
Demand rises as US food, beverage, and pharmaceutical manufacturers expand sterile production to meet safety, shelf-life, and quality expectations. Aseptic processing ensures microbial control, extended ambient stability, and regulatory compliance in products such as dairy, juices, biologics, and injectable. Adoption aligns with stringent FDA and USDA standards, consumer preference for fresh-taste convenience, and risk mitigation in sterile supply chains. Usage concentrates in large-scale production facilities and contract manufacturing organizations.
How do food safety requirements and pharmaceutical sterility standards drive adoption in USA?
US food processors deploy aseptic systems to extend shelf life and reduce dependence on refrigeration for products including milk, plant-based drinks, and liquid nutrition. Controlled environments, rapid sterilization, and closed-loop filling minimize contamination risk and support compliance with FSMA and relevant USDA rules. In pharmaceuticals and biologics, aseptic processing is central to sterile product manufacturing, including vaccines, monoclonal antibodies, and injectable therapies. FDA guidance dictates strict environmental monitoring, operator qualification, and process validation. Contract manufacturing organizations adopt automated aseptic platforms to serve multiple clients with consistent quality control, supporting rapid time-to-market.
How do regulatory compliance burden, capital intensity, and workforce constraints influence demand stability in USA?
Aseptic facilities require substantial capital investment in cleanrooms, controlled environments, and sterility assurance systems. Validation, qualification, and documentation demands under FDA and USDA frameworks increase implementation time and cost. Skilled operators and quality personnel are essential for environmental monitoring, change control, and deviation management, contributing to workforce competition. Small and mid-sized manufacturers face budget constraints when upgrading to aseptic capability. Complexity of sterility standards requires robust quality management systems, increasing operational overhead. Demand remains steady where product safety and shelf stability are imperative, while growth depends on balancing investment costs with regulatory compliance and throughput efficiency across US manufacturing sectors.
How Is Demand for Aseptic Processing in the USA Evolving by Region?
Demand for aseptic processing in the USA is increasing due to shelf-stable food consumption, pharmaceutical manufacturing expansion, and focus on contamination control. West USA leads with a 7.3% CAGR, supported by biopharmaceutical production and advanced food processing facilities. South USA follows at 6.6%, driven by beverage processing, contract manufacturing, and capacity expansion. Northeast USA records a 5.9% CAGR, shaped by pharmaceutical aseptic filling, clinical manufacturing, and regulatory compliance needs. Midwest USA posts 5.1%, reflecting large-scale food processing and packaged dairy production. Regional variation reflects industry mix, regulatory intensity, capital investment patterns, and adoption of sterile manufacturing practices across the USA.
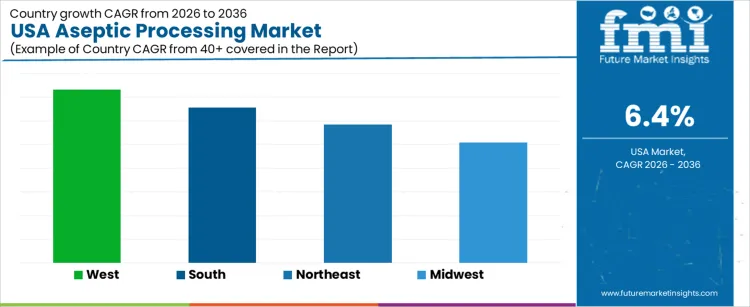
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 7.3% |
| South USA | 6.6% |
| Northeast USA | 5.9% |
| Midwest USA | 5.1% |
What factors are driving aseptic processing adoption in West USA?
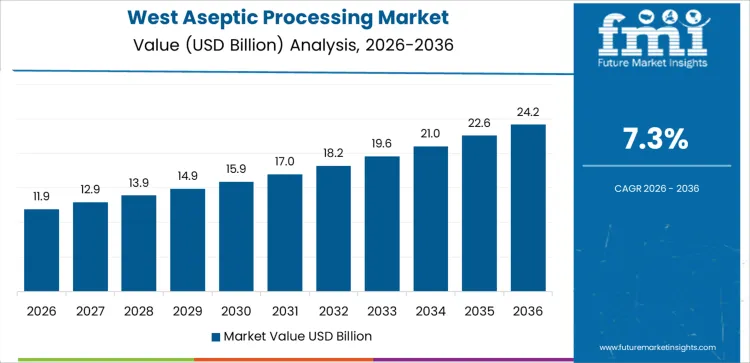
West USA drives demand through strong biopharmaceutical manufacturing presence, specialty food processing, and innovation-led production facilities. Region’s CAGR of 7.3% reflects extensive use of aseptic processing for injectable drugs, biologics, and shelf-stable nutritional products. Manufacturers prioritize contamination control, extended product shelf life, and compliance with FDA sterile processing standards. Advanced automation and closed-system technologies support high-throughput operations. Companies invest in aseptic filling lines to support product launches and pipeline expansion. Demand favors flexible systems capable of handling multiple formulations. Growth remains innovation-led and compliance-focused, aligned with high-value product manufacturing and strict quality requirements.
- Biopharmaceutical and sterile injectable production
- Advanced automation and closed-system adoption
- Shelf-stable nutrition and specialty foods
- High regulatory compliance requirements
Why is South USA experiencing steady growth in aseptic processing demand?
South USA demand is shaped by beverage manufacturing, food processing expansion, and growth of contract manufacturing organizations. Region’s CAGR of 6.6% reflects rising adoption of aseptic processing for juices, dairy alternatives, and liquid nutritional products. Manufacturers value aseptic technology for reduced preservative use and extended distribution reach. Lower operating costs and available industrial space support new facility investments. Contract manufacturers adopt aseptic lines to serve multiple brand owners efficiently. Demand favors scalable and standardized systems supporting high-volume production. Growth remains capacity-led and efficiency-driven, aligned with expanding packaged food and beverage output.
- Beverage and liquid food processing growth
- Contract manufacturing expansion
- Cost-efficient large-scale production facilities
- High-volume aseptic filling demand
How do regulatory and clinical needs shape aseptic processing demand in Northeast USA?
Northeast USA demand reflects pharmaceutical manufacturing, clinical trial production, and regulatory-driven quality standards. Region’s CAGR of 5.9% is supported by aseptic processing for injectable, ophthalmic products, and clinical-grade biologics. Manufacturers emphasize validation, documentation, and contamination risk management. Facilities invest in isolators, cleanrooms, and environmental monitoring systems. Demand favors flexible aseptic setups suitable for small-batch and high-value products. Growth aligns with clinical development pipelines and compliance requirements rather than large-scale food production.
- Pharmaceutical and clinical manufacturing focus
- Strong emphasis on validation and documentation
- Small-batch and high-value product processing
- Regulatory-driven technology adoption
What explains measured aseptic processing growth in Midwest USA?
Midwest USA demand is anchored in large-scale food processing, dairy production, and packaged meal manufacturing. Region’s CAGR of 5.1% reflects adoption of aseptic processing to improve shelf life and reduce cold-chain dependency. Manufacturers focus on operational reliability, cost control, and consistent product quality. Investment decisions align with plant modernization and equipment replacement cycles. Demand favors robust systems optimized for continuous production. Growth remains steady and process-driven, aligned with long production runs and conservative capital planning across established food manufacturing facilities.
- Dairy and packaged food processing usage
- Shelf-life extension without preservatives
- Plant modernization and replacement investments
- Cost-controlled and reliable aseptic systems
What is the competitive landscape of demand for aseptic processing in the USA?
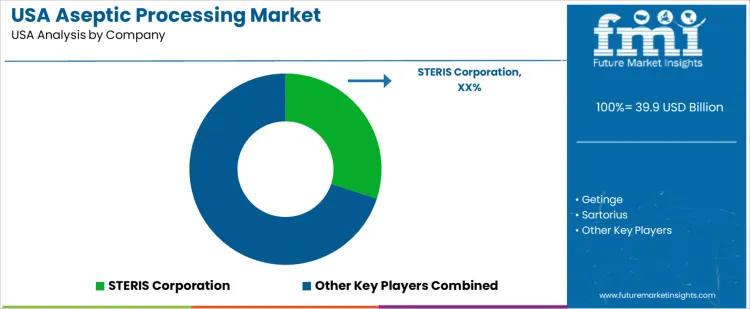
Demand for aseptic processing in the USA is driven by pharmaceutical manufacturing, biopharmaceutical fill-finish operations, food and beverage sterilization, and cell and gene therapy production requiring contamination-free environments. Aseptic systems support sterile liquid and lyophilized drug products, parenteral nutrition, biologics, and high-value injectable therapies. Buyers evaluate isolator design, cleanroom integration, sterility assurance levels, single-use compatibility, and compliance with FDA current good manufacturing practices. Procurement teams prioritize suppliers with validated technologies, strong U.S. service presence, and comprehensive qualification support for regulatory submissions. Trend in the USA market reflects increasing clinical biologics production, expanded sterile contract manufacturing, and automation to reduce contamination risk while improving throughput.
STERIS Corporation maintains a strong position in the USA through comprehensive aseptic processing platforms, isolators, and sterilization systems integrated with local service and validation support. Getinge USA participates with aseptic containment and filling solutions used in pharmaceutical and biotech facilities requiring predictable sterility performance. Sartorius USA supports demand with single-use aseptic systems, bioprocessing equipment, and expertise aligned with sterile manufacturing needs. MilliporeSigma (Merck KGaA in the USA) contributes with aseptic filtration, sterile connectors, and process equipment used across biologics and sterile product lines. IMA Life North America supplies automated aseptic filling and inspection systems tailored to U.S. pharmaceutical operations. Competitive positioning in the USA reflects regulatory alignment, service infrastructure, technology validation capability, and ability to support high-throughput, contamination-free production.
Key Players in USA Aseptic Processing Demand
- STERIS Corporation
- Getinge
- Sartorius
- MilliporeSigma (Merck KGaA)
- IMA Life
Scope of the Report
| Items | Details |
|---|---|
| Quantitative Units | USD billion |
| Type | Packaging; Processing |
| Material | Paper and Paperboard; Plastics; Metal; Glass |
| Application | Food and Beverages; Industrial |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | STERIS Corporation; Getinge; Sartorius; MilliporeSigma (Merck KGaA); IMA Life |
| Additional Attributes | Demand is supported by extended shelf-life requirements, food safety regulations, and sterile manufacturing needs. Packaging solutions dominate beverage and dairy segments, while processing systems see higher uptake in pharmaceuticals and biologics. Adoption is influenced by automation, contamination control standards, and capital investment cycles across US manufacturing facilities. |
USA Aseptic Processing Demand by Segments
Type:
- Packaging
- Processing
Material:
- Paper and Paperboard
- Plastics
- Metal
- Glass
Application:
- Food and Beverages
- Industrial
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Frequently Asked Questions
How big is the demand for aseptic processing in USA in 2026?
The demand for aseptic processing in USA is estimated to be valued at USD 39.9 billion in 2026.
What will be the size of aseptic processing in USA in 2036?
The market size for the aseptic processing in USA is projected to reach USD 74.2 billion by 2036.
How much will be the demand for aseptic processing in USA growth between 2026 and 2036?
The demand for aseptic processing in USA is expected to grow at a 6.4% CAGR between 2026 and 2036.
What are the key product types in the aseptic processing in USA?
The key product types in aseptic processing in USA are packaging and processing.
Which material segment is expected to contribute significant share in the aseptic processing in USA in 2026?
In terms of material, paper and paperboard segment is expected to command 35.0% share in the aseptic processing in USA in 2026.
Table of Content
- Executive Summary
- USA Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- USA Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- USA Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- USA Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Packaging
- Processing
- Packaging
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- USA Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material, 2026 to 2036
- Paper and Paperboard
- Plastics
- Metal
- Glass
- Paper and Paperboard
- Y to o to Y Growth Trend Analysis By Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Material, 2026 to 2036
- USA Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Food and Beverages
- Industrial
- Food and Beverages
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Material
- By Application
- Competition Analysis
- Competition Deep Dive
- STERIS Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Getinge
- Sartorius
- MilliporeSigma (Merck KGaA)
- IMA Life
- STERIS Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: USA Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: USA Market Value (USD Million) Forecast by Type , 2020 to 2035
- Table 3: USA Market Value (USD Million) Forecast by Material, 2020 to 2035
- Table 4: USA Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 5: USA Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: USA Market Value (USD Million) Forecast by Type , 2020 to 2035
- Table 7: USA Market Value (USD Million) Forecast by Material, 2020 to 2035
- Table 8: USA Market Value (USD Million) Forecast by Application, 2020 to 2035
List of Figures
- Figure 1: USA Market Pricing Analysis
- Figure 2: USA Market Value (USD Million) Forecast 2020-2035
- Figure 3: USA Market Value Share and BPS Analysis by Type , 2025 and 2035
- Figure 4: USA Market Y-o-Y Growth Comparison by Type , 2025-2035
- Figure 5: USA Market Attractiveness Analysis by Type
- Figure 6: USA Market Value Share and BPS Analysis by Material, 2025 and 2035
- Figure 7: USA Market Y-o-Y Growth Comparison by Material, 2025-2035
- Figure 8: USA Market Attractiveness Analysis by Material
- Figure 9: USA Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 10: USA Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 11: USA Market Attractiveness Analysis by Application
- Figure 12: USA Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: USA Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 14: USA Market Attractiveness Analysis by Region
- Figure 15: USA Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: USA Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: USA Market Value Share and BPS Analysis by Type , 2025 and 2035
- Figure 18: USA Market Y-o-Y Growth Comparison by Type , 2025-2035
- Figure 19: USA Market Attractiveness Analysis by Type
- Figure 20: USA Market Value Share and BPS Analysis by Material, 2025 and 2035
- Figure 21: USA Market Y-o-Y Growth Comparison by Material, 2025-2035
- Figure 22: USA Market Attractiveness Analysis by Material
- Figure 23: USA Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 24: USA Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 25: USA Market Attractiveness Analysis by Application
- Figure 26: USA Market - Tier Structure Analysis
- Figure 27: USA Market - Company Share Analysis

