Vitamins and Minerals Based Injectables Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Vitamins and Minerals Based Injectables Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Vitamins and Minerals Based Injectables Market Forecast and Outlook 2026 to 2036
The vitamins and minerals based injectables market stands at USD 6.5 billion in 2026, and is projected to reach USD 13.5 billion by 2036, implying a CAGR of 7.6%. Use is concentrated in hospital settings, clinics, and critical care units where rapid correction of deficiencies or nutritional support is required. Demand is shaped more by clinical protocols and patient acuity than by routine supplementation trends. Regions with larger inpatient populations and higher procedure volumes account for a disproportionate share of consumption. Manufacturing and formulation capabilities for sterile injectables remain geographically concentrated, which influences supply reliability, regulatory scrutiny, and pricing discipline across markets.
Treatment guidelines and reimbursement frameworks play a central role in determining utilization patterns. Physicians prescribe these products based on laboratory confirmation, absorption constraints, or acute clinical need rather than on patient preference. Adoption varies widely by region because access to infusion infrastructure, cold chain logistics, and trained staff is not uniform. Hospitals and large healthcare networks typically standardize suppliers to simplify procurement, quality assurance, and inventory control. The market expands through growth in hospital based care, oncology support, and intensive treatment pathways, not through substitution for oral supplements or discretionary wellness use.
Quick Stats for Vitamins and Minerals Based Injectables Market
- Vitamins and Minerals Based Injectables Market Value (2026): USD 6.5 billion
- Vitamins and Minerals Based Injectables Market Forecast Value (2036): USD 13.5 billion
- Vitamins and Minerals Based Injectables Market Forecast CAGR 2026 to 2036: 7.6%
- Leading Type in Vitamins and Minerals Based Injectables Market: Multivitamin injectable formulations
- Key Growth Regions in Vitamins and Minerals Based Injectables Market: Asia Pacific, North America, Europe
- Top Players in Vitamins and Minerals Based Injectables Market: Pfizer, Bayer, Merck & Co., Novartis, GlaxoSmithKline, B. Braun Melsungen, Fresenius Kabi
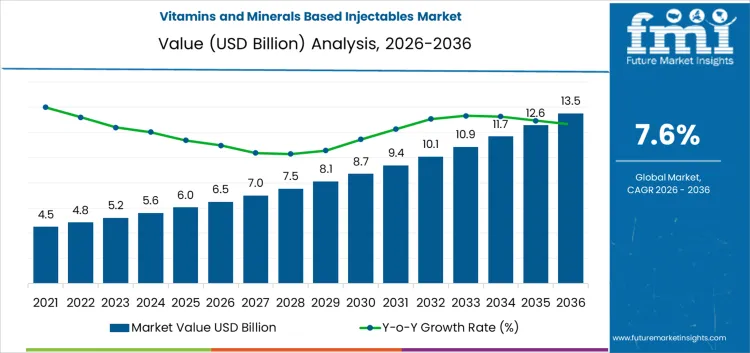
What is the Growth Forecast for Vitamins and Minerals Based Injectables Market through 2036?
The vitamins and minerals based injectables segment is expanding as clinical practice increasingly favors rapid correction of deficiencies and supportive therapy across acute, perioperative, and long-term care settings. Early growth is driven by rising use in hospitals, clinics, and infusion centers for cases where oral supplementation delivers slower outcomes, absorption is limited, or patient adherence is uncertain. Over time, injectable micronutrients become firmly established in treatment protocols for critical care, oncology support, post-surgical recovery, and malabsorption-related disorders. In this phase, expansion is driven mainly by patient throughput and procedure volumes rather than by major shifts in formulation complexity or pricing.
As utilization widens, demand strengthens further through increased outpatient infusion use, aging populations, and stronger clinical focus on nutritional support in chronic disease management. Standardized care pathways and repeat dosing regimens increasingly shape consumption, supporting higher usage intensity across healthcare systems. In mature stages, value growth depends primarily on broader adoption across regions and higher treatment volumes rather than price escalation. Competitive positioning remains centered on consistent product quality, dependable supply chains, and strict regulatory compliance. Purchasing decisions continue to prioritize formulation stability, clinical reliability, and availability over product differentiation, making operational execution and manufacturing credibility critical to long-term success.
Vitamins and Minerals Based Injectables Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 6.5 billion |
| Forecast Value (2036) | USD 13.5 billion |
| Forecast CAGR 2026 to 2036 | 7.6% |
How Is the Vitamins and Minerals-Based Injectables Market Supporting Targeted Nutritional Therapy?
Vitamins and minerals-based injectables are increasingly adopted to address nutrient deficiencies, enhance clinical outcomes, and support patient wellness in medical and therapeutic settings. Historically, supplementation relied primarily on oral formulations, which faced absorption variability, gastrointestinal limitations, and delayed therapeutic effects. Modern injectable formulations deliver precise dosages directly into the bloodstream or muscle tissue, enabling rapid bioavailability, controlled dosing, and improved efficacy. Hospitals, clinics, and specialty wellness centers prioritize formulation accuracy, safety, and regulatory compliance. Early adoption focused on high-risk patient populations and clinical interventions, while current demand spans general healthcare, preventive medicine, and sports nutrition applications, driven by rising awareness of micronutrient deficiencies, personalized therapy, and targeted nutritional interventions. Formulation stability, absorption efficiency, and safety profiles influence supplier selection.
Clinical effectiveness, patient adherence, and therapeutic outcomes are shaping market growth. Compared with oral supplementation, injectable solutions emphasize rapid action, predictable bioavailability, and flexibility for individualized treatment plans. Cost structures depend on ingredient quality, formulation processes, and compliance testing, concentrating margins among suppliers capable of delivering reliable, high-quality injectables. Healthcare providers adopt these formulations to improve nutrient delivery, optimize patient outcomes, and support preventive or corrective therapy. By 2036, vitamins and minerals-based injectables are expected to become a standard option in clinical and wellness settings, supporting precise nutritional supplementation, rapid therapeutic impact, and improved patient care outcomes.
What Factors Are Shaping the Demand for Vitamins and Minerals Based Injectables, Segment wise by Product Type and Indication, in 2026?
The vitamins and minerals based injectables market in 2026 is segmented by product type and by clinical indication. By formulation type, demand is divided into multivitamin injectable formulations, vitamin B complex injectables, injectable iron and mineral complexes, and vitamin D and calcium injectable products, each serving different deficiency patterns and treatment protocols. By indication, demand is organized around nutritional deficiencies, age related metabolic disorders, post-surgical supplementation, and prenatal or maternal health, which differ in patient population size, treatment duration, and clinical urgency. These segments reflect how clinicians choose injectable supplementation when oral routes are insufficient or impractical.
Why Do Multivitamin Injectable Formulations Lead Product Demand?
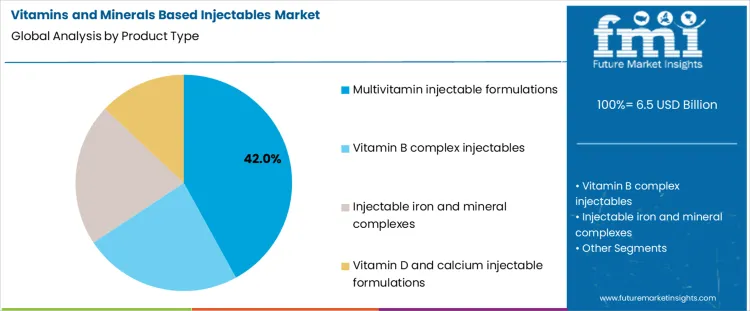
Multivitamin injectable formulations account for about 42% of demand in 2026 because they address multiple deficiencies in a single administration. In hospital and clinical settings, patients often present with complex or unclear nutritional gaps, especially after surgery, during critical illness, or in cases of malabsorption. A combined formulation allows rapid correction without waiting for detailed profiling of each vitamin level. This simplifies prescribing and inventory management. These products are also used in supportive therapy during recovery and in long term care settings. The breadth of indications and the convenience of a single injection explain why multivitamin formulations remain the most widely used product category.
Single vitamin or mineral focused injectables serve more targeted needs. Vitamin B complex is used in specific neurological or metabolic conditions. Iron and mineral complexes address anemia and trace element deficiencies but require careful dosing and monitoring. Vitamin D and calcium injections are used in defined bone health or maternal care protocols. These products are essential in their niches, yet their use is episodic and condition specific. This keeps their overall volumes below those of broad spectrum multivitamin injections that fit into many different treatment pathways.
Why Do Nutritional Deficiencies Dominate Clinical Indications?
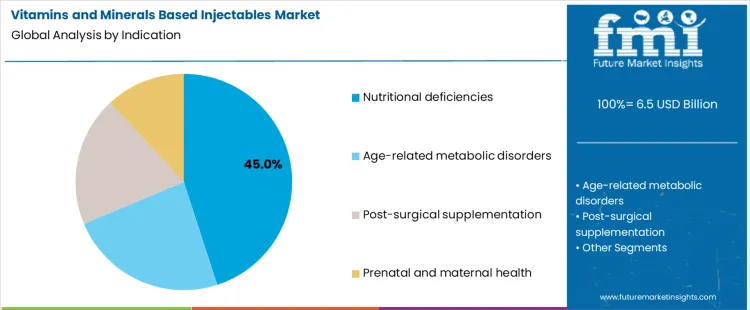
Nutritional deficiencies represent about 45% of demand in 2026 because they cover a wide and growing patient population across age groups. Hospitalized patients, the elderly, and individuals with chronic gastrointestinal or metabolic disorders frequently require rapid replenishment of vitamins and minerals. In many cases, oral supplementation is ineffective or too slow, which makes injectable routes necessary. Screening and treatment of deficiencies are also routine parts of clinical protocols in many settings. This creates a steady flow of prescriptions rather than isolated treatment events.
Age related metabolic disorders, post-surgical care, and prenatal support follow different demand patterns. Metabolic disorders require ongoing management but involve smaller patient cohorts. Post-surgical supplementation is intensive but time limited. Prenatal and maternal use is tied to specific stages of pregnancy and clinical guidelines. These indications are important and clinically significant, yet they do not match the broad, cross setting prevalence of general nutritional deficiency treatment, which keeps it as the leading indication segment.
How Are the Key Dynamics Reshaping the Vitamins and Minerals Injectables Market?
The category is being shaped by clinical protocol standardization, cautious adoption barriers, and the growing role of protocol driven supplementation. Injectable vitamins and minerals are used where absorption limits, acute deficiency, or clinical workflows justify parenteral delivery. Demand grows through hospital pathways and outpatient infusion settings, but uptake is tempered by evidence expectations, safety governance, and reimbursement alignment. At the same time, providers are standardizing treatment bundles and order sets, which creates scale for products that are embedded in protocols. The result is a market driven by care pathway inclusion and institutional governance rather than by consumer awareness or retail supplementation trends.
Why Are Clinical Protocols Driving Injectable Vitamin Demand?
Demand is anchored in situations where oral supplementation is inadequate or impractical. In critical care, oncology support, malabsorption, perioperative care, and severe deficiency states, injectables provide predictable and rapid correction. Hospitals increasingly rely on standardized order sets and treatment pathways, which specify when parenteral supplementation is required. Once a product is written into these protocols, usage becomes routine and repeatable across patients and departments. This ties volume to case mix and protocol adoption rather than to promotional activity. Growth follows expansion of protocol based care and the spread of outpatient infusion and day care treatment models.
What Is Slowing Wider Use of Injectable Supplements?
The main barrier is governance and evidence burden. Injectables carry higher safety expectations, require sterile handling, and involve administration time and monitoring. Many indications still rely on clinician judgment rather than strong guideline mandates, which limits routine use. Payers and hospital committees scrutinize cost, necessity, and outcome evidence, especially where oral options exist. There are also concerns around overuse, adverse reactions, and inconsistent practices across departments. In regulated settings, any new product addition requires formulary review and training. These factors keep adoption conservative and focused on clearly justified clinical scenarios rather than broad preventive use.
How Are Care Pathways Changing Market Structure?
The trend is toward protocol embedded demand. Health systems are consolidating formularies and standardizing treatment pathways across sites to control variation and cost. Injectable vitamins and minerals that are included in these pathways benefit from predictable, system wide usage. Procurement shifts to framework agreements and long term supply contracts tied to hospital networks. Packaging, compatibility, and documentation become selection criteria alongside price. Over time, competition moves from individual product promotion to being specified within approved clinical pathways and bundled treatment regimens used across multiple departments and facilities.
What is the Demand for Vitamins and Minerals Based Injectables by Country?
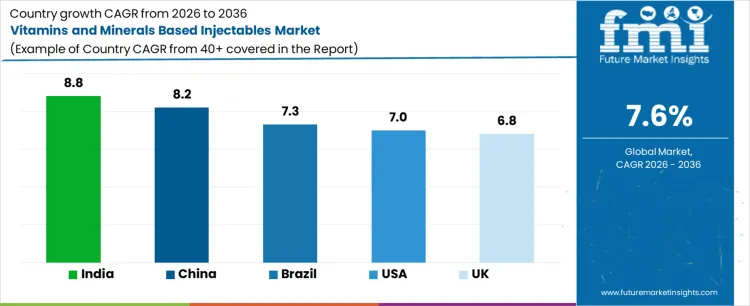
| Country | CAGR (%) |
|---|---|
| US | 7.0% |
| UK | 6.8% |
| China | 8.2% |
| India | 8.8% |
| Brazil | 7.3% |
Demand for vitamins and minerals based injectables is rising as healthcare providers and clinics adopt nutritional therapies for deficiency management, wellness programs, and patient-specific treatment plans. India leads with an 8.8% CAGR, driven by rising awareness of micronutrient deficiencies, growing healthcare services, and increasing adoption of injectable supplementation. China follows at 8.2%, supported by expanding healthcare infrastructure and preventive nutrition programs. Brazil records 7.3% growth, shaped by demand in hospitals and wellness clinics. The US grows at 7.0%, influenced by adoption in clinical and outpatient settings. The UK shows 6.8% CAGR, reflecting steady use of vitamins and minerals injectables in healthcare and wellness services.
How is the United States experiencing growth in the vitamins and minerals based injectables market?
United States is experiencing growth at a CAGR of 7%, driven by an established healthcare infrastructure and high prevalence of nutritional supplementation programs across states such as California, New York, and Florida. Market concentration exists among hospital networks, specialty clinics, and outpatient care facilities. Investments focus on high-quality formulations, regulatory compliance, and integration with patient care protocols. Growth reflects rising awareness of nutritional deficiencies, aging population needs, and routine preventive healthcare initiatives.
- Hospitals and specialty clinics drive adoption.
- Urban healthcare hubs concentrate demand.
- Regulatory compliance and formulation quality guide investment.
- Preventive care initiatives support growth.
How is the United Kingdom witnessing growth in the vitamins and minerals based injectables market?
United Kingdom is witnessing growth at a CAGR of 6.8%, supported by increasing patient spending on nutritional supplements and preventive healthcare services in London, Manchester, and Birmingham. Market concentration is moderate, with adoption across NHS clinics and private healthcare providers. Investments focus on standardized injectable formulations and patient monitoring programs. Growth reflects increasing health awareness, demand for age-related supplementation, and integration with routine healthcare services.
- Patient spending on nutritional supplements drives adoption.
- NHS and private clinics concentrate demand.
- Standardized formulations guide investment.
- Preventive healthcare integration supports growth.
How is China experiencing growth in the vitamins and minerals based injectables market?
China is experiencing growth at a CAGR of 8.2%, fueled by the rising prevalence of nutritional deficiencies and expansion of hospital networks in Beijing, Shanghai, and Guangzhou. Market concentration is high in urban hospitals and specialty clinics, while regional dispersion is growing. Investments prioritize formulation efficacy, quality control, and distribution logistics. Growth reflects increasing health awareness, governmental health campaigns, and rising demand for injectable supplementation in both urban and semi-urban populations.
- Hospital networks and specialty clinics drive adoption.
- Urban centers concentrate demand.
- Formulation quality and distribution guide investment.
- Health awareness campaigns support growth.
How is India witnessing growth in the vitamins and minerals based injectables market?
India is experiencing growth at a CAGR of 8.8%, supported by exposure to both domestic healthcare demand and export-driven pharmaceutical production in Mumbai, Delhi, and Bengaluru. Market concentration is highest in urban hospitals and clinical centers, while regional demand grows with expanding healthcare infrastructure. Investments focus on cost-effective injectable formulations, quality compliance, and logistics. Growth reflects rising awareness of nutritional deficiencies, increasing urban healthcare access, and a growing middle-class consumer base seeking preventive care.
- Hospitals and clinical centers drive adoption.
- Urban healthcare hubs concentrate demand.
- Cost-effective formulations guide investment.
- Middle-class preventive care initiatives support growth.
How is Brazil experiencing growth in the vitamins and minerals based injectables market?
Brazil is experiencing growth at a CAGR of 7.3%, driven by healthcare facilities in São Paulo, Rio de Janeiro, and Brasília that cater to growing preventive care and nutritional supplementation demand. Market concentration exists in urban hospitals, clinics, and wellness centers. Investments focus on maintaining injectable quality, supply chain reliability, and compliance with ANVISA regulations. Growth reflects increasing awareness of micronutrient deficiencies, adoption of preventive healthcare practices, and integration of nutritional injectables into clinical protocols.
- Hospitals and wellness centers drive adoption.
- Urban healthcare hubs concentrate demand.
- Injectable quality and regulatory compliance guide investment.
- Preventive care awareness supports growth.
How Are Leading Manufacturers Defining the Vitamins and Minerals Based Injectables Market?
The vitamins and minerals based injectables market is defined by formulation accuracy, sterility assurance, and clinical utility across deficiency treatments and supportive care. Pfizer offers injectable formulations designed with stringent sterility and dosing precision, targeting deficiency correction in hospitalized and outpatient settings. Bayer supplies vitamin-based injectables with well-established safety profiles, often used in perioperative and nutritional therapy contexts. Merck & Co. develops micronutrient injections integrated into broader therapeutic regimens, with quality controls aligned to global pharmacopeial standards. Novartis provides vitamin and mineral solutions tailored for clinical needs where oral administration is insufficient. GlaxoSmithKline produces injectable nutritional components used in clinical nutrition programs. B.BraunMelsungen supplies parenteral vitamin and trace element solutions engineered for compatibility with infusion systems. FreseniusKabi offers a range of injectable micronutrients formulated for stability and integration into complex intravenous nutrition protocols.
Manufacturers differentiate through product range, regulatory compliance, and supply chain reliability. High-purity vitamin and mineral injectables require validated manufacturing environments that meet sterility and particulate limits. Pfizer’s portfolio emphasizes robust quality systems across global production sites. Bayer’s offerings are supported by clinical evidence for targeted deficiency states. Merck & Co. and Novartis integrate micronutrient injectables into broader care pathways, addressing patient populations with complex therapeutic needs. GlaxoSmithKline and B.Braun focus on products that align with standardized infusion practices in hospitals. FreseniusKabi designs formulations for use in parenteral nutrition therapy, with attention to compatibility with other intravenous components. Other regional producers supply niche micronutrient injections for specific markets or regulatory environments. Key differentiators include formulation stability, traceability of ingredients, packaging formats that support safe handling, and documented compliance with regional and international pharmacopoeias. Clinical adoption depends on both evidence of efficacy in deficiency correction and confidence in manufacturing quality.
Key Players in the Vitamins and Minerals Based Injectables Market
- Pfizer
- Bayer
- Merck & Co.
- Novartis
- GlaxoSmithKline
- B. Braun Melsungen
- Fresenius Kabi
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Multivitamin injectable formulations, Vitamin B complex injectables, Injectable iron and mineral complexes, Vitamin D and calcium injectable formulations |
| Indication | Nutritional deficiencies, Age-related metabolic disorders, Post-surgical supplementation, Prenatal and maternal health |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Pfizer, Bayer, Merck & Co., Novartis, GlaxoSmithKline, B. Braun Melsungen, Fresenius Kabi |
| Additional Attributes | Dollar sales by product type and indication; multivitamin injectables and deficiency treatment as leading segments; demand driven by hospital protocols, oncology and critical care use, and malabsorption cases; growth tied to inpatient volumes and outpatient infusion expansion; supplier selection based on sterility assurance, formulation stability, regulatory compliance, and supply reliability. |
Vitamins and Minerals Based Injectables Market Segmentation
Product Type:
- Multivitamin injectable formulations
- Vitamin B complex injectables
- Injectable iron and mineral complexes
- Vitamin D and calcium injectable formulations
Indication:
- Nutritional deficiencies
- Age-related metabolic disorders
- Post-surgical supplementation
- Prenatal and maternal health
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- United States Food and Drug Administration. (2025). Small volume parenteral drug products and pharmacy bulk packages for parenteral nutrition: Aluminum content and labeling recommendations (Draft guidance). U.S. Department of Health and Human Services.
- United States Food and Drug Administration. (2024). Temporary policies for compounding certain parenteral drug products: Guidance for industry. U.S. Department of Health and Human Services.
- Wischmeyer, P. E., et al. (2024). Parenteral nutrition in clinical practice. National Institutes of Health / PubMed Central.
Frequently Asked Questions
How big is the vitamins and minerals based injectables market in 2026?
The global vitamins and minerals based injectables market is estimated to be valued at USD 6.5 billion in 2026.
What will be the size of vitamins and minerals based injectables market in 2036?
The market size for the vitamins and minerals based injectables market is projected to reach USD 13.5 billion by 2036.
How much will be the vitamins and minerals based injectables market growth between 2026 and 2036?
The vitamins and minerals based injectables market is expected to grow at a 7.6% CAGR between 2026 and 2036.
What are the key product types in the vitamins and minerals based injectables market?
The key product types in vitamins and minerals based injectables market are multivitamin injectable formulations , vitamin b complex injectables, injectable iron and mineral complexes and vitamin d and calcium injectable formulations.
Which indication segment to contribute significant share in the vitamins and minerals based injectables market in 2026?
In terms of indication, nutritional deficiencies segment to command 45.0% share in the vitamins and minerals based injectables market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Multivitamin injectable formulations
- Vitamin B complex injectables
- Injectable iron and mineral complexes
- Vitamin D and calcium injectable formulations
- Multivitamin injectable formulations
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Nutritional deficiencies
- Age‑related metabolic disorders
- Post‑surgical supplementation
- Prenatal and maternal health
- Nutritional deficiencies
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- Competition Analysis
- Competition Deep Dive
- Pfizer
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bayer
- Merck & Co.
- Novartis
- GlaxoSmithKline
- B. Braun Melsungen
- Fresenius Kabi
- Pfizer
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Indication
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Indication
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Indication
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Indication
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

