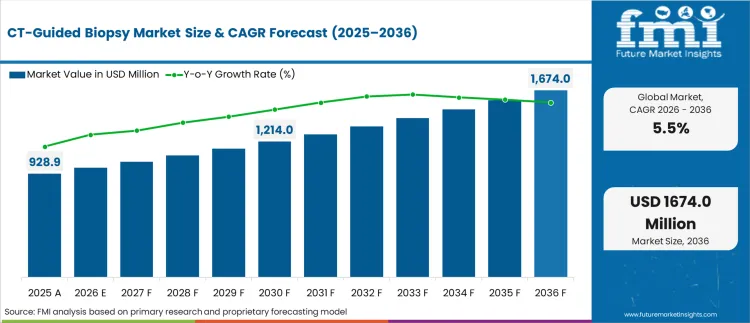
In 2026, the CT-guided biopsy market was valued at USD 980.0 million. Based on Future Market Insights analysis, demand for CT-guided biopsy is estimated to grow to USD 1,680.0 million by 2036. FMI projects a CAGR of 5.5% during the forecast period.
Absolute dollar growth of USD 700.0 million over the decade signals steady expansion rather than a rapid technology replacement cycle. As per FMI, growth is expected to be supported by rising image-guided tissue sampling volumes for lung and other internal lesions of the body, while reimbursement controls, procedure scheduling constraints, and complication-risk governance are expected to limit rapid value expansion in mature systems. “This is a major step forward compared to other possible solutions,” said Guobao Wang, professor of radiology and principal investigator. “We’re using the PET scan’s own data to create a second, high-energy CT image. When combined with the regular CT scan, it enables dual-energy imaging that provides a much clearer picture and more detailed information about tissue composition.”
Procurement behaviour is being shaped by standardisation of interventional kits and preference for reliable needle performance, with buyers prioritising sampling consistency, ergonomic handling, and compatibility with CT-fluoroscopy and conventional CT workflows. Multi-vendor sourcing is being maintained in larger hospitals to avoid supply disruption, while product selection is being narrowed to a limited set of proven coaxial and core biopsy configurations aligned to target-organ protocols.
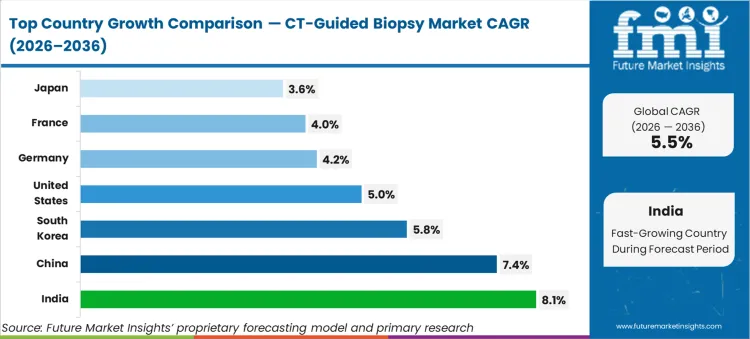
China (7.4% CAGR, supported by expanding interventional radiology capacity and rising oncology work-up volumes) and India (8.1% CAGR, driven by higher diagnostic throughput and broader access to CT-guided interventions) are expected to lead growth. South Korea (5.8% CAGR) is expected to follow through high utilisation in advanced imaging centers. The U.S. (5.0% CAGR) is expected to expand through steady procedural volumes and standardised kits. Mature markets including Germany (4.2%), France (4.0%), and Japan (3.6%) are expected to contribute more through replacement demand, constrained by scheduling capacity and conservative utilisation governance.
The market covers devices and consumables used to perform tissue sampling procedures under CT guidance for diagnostic confirmation of suspected disease. It includes needle systems and related biopsy tools used across lung, liver, kidney, bone, pancreas, and other targets, using modalities such as conventional CT, CT-fluoroscopy, cone-beam CT, and spectral or dual-energy CT. Buyers include hospitals, imaging centers, and ambulatory surgical centers that perform interventional radiology procedures requiring precise lesion targeting and controlled sampling. Demand is shaped by oncology and chronic disease work-up volumes, procedure safety protocols, and throughput requirements, with revenue generated through sales of biopsy needle systems and supporting configurations.
The report includes global and regional market sizes and a 10-year forecast for 2026 to 2036. Segment-level sizing is provided by target organ, biopsy technique, CT modality, end user, and needle system, along with country-level CAGR comparisons across key markets. Coverage includes analysis of procurement and standardisation practices in interventional radiology, kit adoption patterns, and how safety and yield targets influence needle system selection, based on FMI analysis.
The scope excludes CT scanners and associated capital imaging hardware, radiology information systems, and navigation software sold as standalone categories. It also omits pathology testing revenue, procedure fees, and hospital service income linked to biopsy delivery. Ultrasound-guided and MRI-guided biopsy devices are excluded unless used specifically for CT-guided workflows. The focus remains on CT-guided biopsy needle systems and related consumables aligned to the stated target organs, techniques, modalities, and end users.
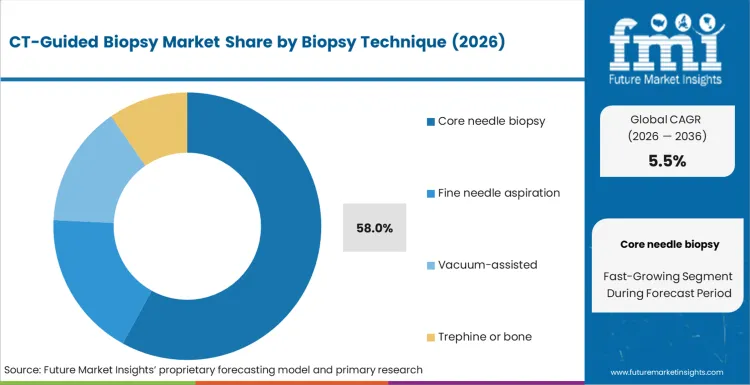
Based on FMI’s report, core needle biopsy is estimated to hold 58.0% share in 2026, followed by fine needle aspiration at 22.0% share. Core needle leadership is supported by its ability to provide tissue architecture for histology, which reduces repeat procedures in many oncology pathways and supports downstream testing needs. Fine needle aspiration remains important where cytology is sufficient or a less invasive approach is preferred, yet share is lower because diagnostic sufficiency can vary by lesion type and sampling adequacy.
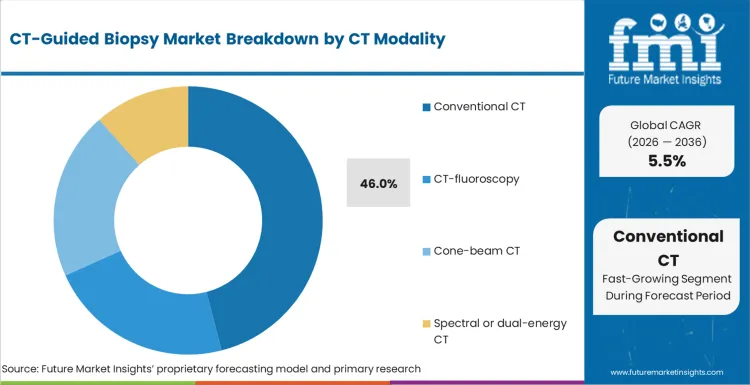
Conventional CT accounts for 46.0% share in 2026, followed by CT-fluoroscopy at 32.0% share, based on FMI’s report. Conventional CT leadership reflects wide availability and established workflow familiarity across interventional radiology, which supports stable adoption across hospitals. CT-fluoroscopy demand is supported by real-time guidance needs in technically challenging access pathways, where procedural control and needle placement precision can reduce adjustments and shorten procedure time, yet uptake is paced by training requirements and radiation management practices.
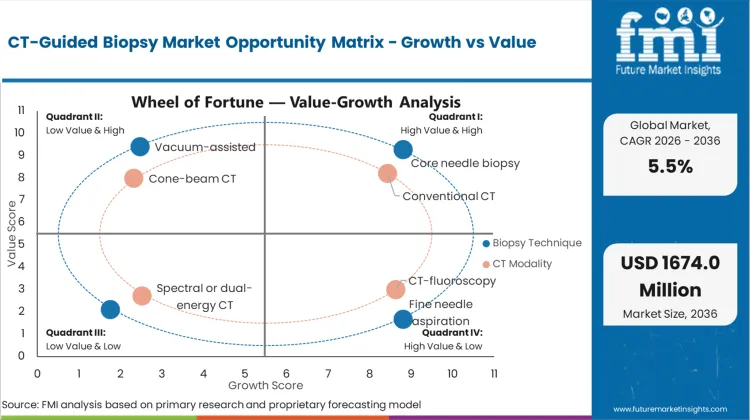
Future Market Insights analysis that historical patterns point at a procedure-driven interventional diagnostics category where demand is anchored in imaging-led lesion detection and oncology confirmation pathways. Estimated valuation in 2026 is being supported by repeat referrals for tissue confirmation and preference for minimally invasive sampling under CT guidance, as per FMI.
While growth is being restrained by CT suite scheduling constraints, operator dependence, and complication risk management that can limit throughput, expansion is being supported by wider access to CT-guided interventions and continued preference for core biopsy techniques that reduce repeat sampling. Based on FMI’s report, procurement is increasingly being filtered by kit completeness, ease of use, and ability to standardize technique and modality workflows across sites.
Based on the regional analysis, CT-guided biopsy market is segmented into North America, Latin America, Western Europe, Eastern Europe East Asia, South Asia, and Middle East & Africa across 40+ countries. Regional performance is assessed using country-level demand signals linked to cancer diagnosis volumes, interventional radiology capacity, reimbursement design, and availability of CT and cone-beam CT suites, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 5.0% |
| China | 7.4% |
| Germany | 4.2% |
| France | 4.0% |
| Japan | 3.6% |
| India | 8.1% |
| South Korea | 5.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America is shaped by high interventional radiology throughput and guideline-led cancer workups, where procurement is driven by hospital standardisation and device preference for coaxial systems in lung and liver biopsies. BD (Bard) holds strong positioning in biopsy needles and coaxial access systems used in routine CT-guided procedures. Boston Scientific participates through interventional portfolios that align with image-guided access workflows. Cook Medical competes through specialty needles and procedural tools supported by hospital accounts. FMI analysts note that hospitals remain the primary end user, with imaging centers contributing selective volume.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Asia Pacific remains the highest growth region, supported by expanding cancer screening and diagnosis activity, rising availability of CT and CT-fluoroscopy in tertiary hospitals, and increasing investment in interventional radiology infrastructure. BD (Bard) maintains relevance through broad biopsy access portfolios. Terumo and Merit Medical participate through interventional device ecosystems used in hospital procedure suites. Cook Medical remains present through specialty needles and accessories. Future Market Insights analysis links growth to higher lung and liver biopsy volumes and increasing adoption of semi-automated biopsy devices in high-throughput centers.
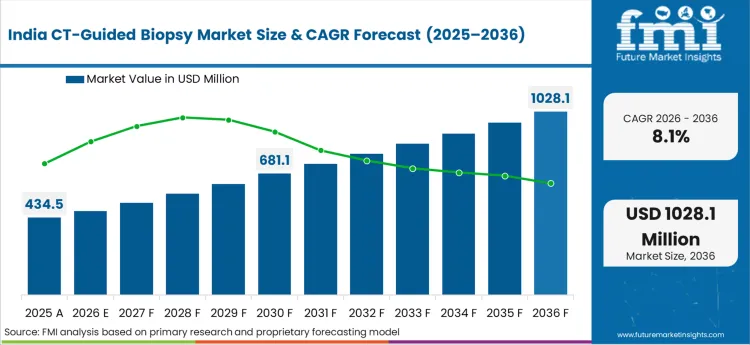
The full report analyze the CT-guided biopsy market across East and South Asia from 2021-2036, covering pricing, trends, and growth drivers in China, Japan, South Korea, India, Indonesia, Malaysia, and Thailand. The assessment highlights trends that dictate regional demand and procurement behaviour.
Europe is tender-led and protocol-driven, where CT-guided biopsy demand is shaped by diagnostic pathways, hospital procurement, and increasing emphasis on minimally invasive tissue diagnosis. BD (Bard) remains strongly placed through biopsy needles and access systems. B. Braun and Hologic maintain relevance through procedural devices and related portfolios used in interventional suites. Boston Scientific competes through interventional offerings aligned to hospital demand. FMI opines that demand remains anchored in hospitals, with imaging centers contributing selective case volume.
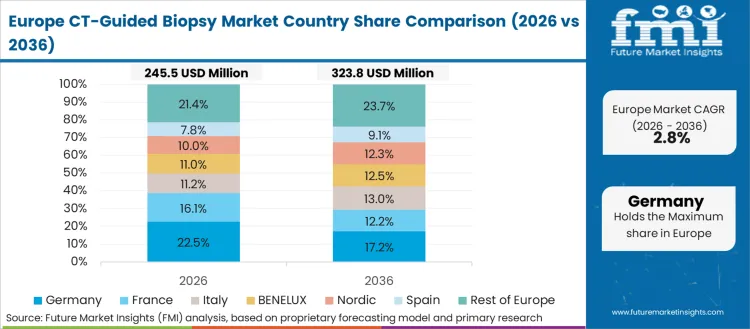
FMI’s analysis of CT-guided biopsy market in Europe consists of country-wise assessment that includes the Germany, Italy, France, UK, Spain, Russia, Nordic, Benelux and Rest of Europe. Readers can know various regulations and latest trends in the regional market.
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can deliver qualification-ready systems, maintain repeatable low temperature process control, and support device maker audit needs across regulated production. Competition is shaped by validation depth, material compatibility evidence, and throughput reliability, rather than short-cycle price movement, because switching into this modality requires documented change control and performance acceptance. Providers that can support both equipment deployment and service-based access tend to influence adoption by reducing transition burden for medical device manufacturers, as per FMI.
Companies with structural advantages typically combine installed base reach, proven sterilization engineering support, and the capability to produce consistent sterility assurance outcomes across polymers, elastomers, and complex device configurations. Suppliers that can demonstrate tight residue management, repeatable cycle outcomes, and stable process parameters are better positioned when customers expand from pilot runs into routine production. Manufacturers with narrower platform maturity or limited validation support tend to face slower adoption because customers must carry more internal qualification work, which can delay scale-up and increase program risk, based on FMI’s report.
Customer concentration reinforces buyer leverage. Medical device manufacturers often manage supplier dependency by qualifying more than one route or provider, using staged validations, and writing performance clauses tied to cycle acceptance, lead times, and deviation closure. In-house use is preferred where schedule control and confidentiality are required, while outsourced access is selected to avoid capex and to tap validated capacity for complex devices. This behavior constrains pricing power for standard programs, while measured premiums are retained mainly where low temperature compatibility reduces rework risk and protects high value device yields, Future Market Insights analysis.
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:

| Metric | Value |
|---|---|
| Quantitative units | USD 980.0 Mn (2026) to USD 1,680.0 Mn (2036), at a CAGR of 5.5% |
| Market definition | The CT-guided biopsy market comprises global production and trade of percutaneous biopsy devices and procedure enabling consumables used under CT guidance to obtain tissue samples for diagnosis, staging, and therapy planning, where demand is tied to imaging guided intervention volumes, lesion accessibility, and protocol driven sampling practices in hospitals and imaging centers. |
| Target organ segmentation | Lung, Liver, Kidney, Bone, Pancreas, Other |
| Biopsy technique segmentation | Core needle biopsy, Fine needle aspiration, Vacuum-assisted, Trephine or bone |
| CT modality coverage | Conventional CT, CT-fluoroscopy, Cone-beam CT, Spectral or dual-energy CT |
| End user coverage | Hospitals, Imaging centers, ASCs |
| Needle system coverage | Coaxial systems, Non-coaxial, Semi-automated, Fully automated |
| Regions covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key companies profiled | Becton, Dickinson and Company, Cook Medical LLC, Boston Scientific Corporation, Argon Medical Devices, Inc., Merit Medical Systems, Inc., Terumo Corporation, Medtronic plc, B. Braun Melsungen AG, Hologic, Inc., Cardinal Health, Inc. |
| Forecast period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with interventional radiology stakeholders and device suppliers, supported by procedure volume triangulation and regulatory pathway review, as per FMI. |
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
How large is the demand for CT-guided biopsy in the global market in 2026?
Demand for CT-guided biopsy in the global market is estimated to be valued at USD 980.0 Million in 2026, as per FMI.
What will be the market size of CT-guided biopsy in the global market by 2036?
Market size for CT-guided biopsy is projected to reach USD 1,680.0 Million by 2036.
What is the expected demand growth for CT-guided biopsy in the global market between 2026 and 2036?
Demand for CT-guided biopsy in the global market is expected to grow at a CAGR of 5.5% between 2026 and 2036.
Which biopsy technique is poised to lead global demand by 2026?
Core needle biopsy is expected to be the dominant technique, capturing 58.0% share in 2026.
How significant is the role of hospitals in driving CT-guided biopsy adoption in 2026?
Hospitals are projected to hold 72.0% share of end user demand in 2026.
Which target organ contributes the largest share of CT-guided biopsy demand in 2026?
Lung is projected to account for 34.0% share of the target organ split in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.