Electrophysiology Equipment and Recording Systems Market
This report outlines the electrophysiology equipment and recording systems market landscape, covering market valuation, revenue estimates, competitive framework, application landscape analysis, demand evaluation, principal growth drivers, market impediments, supply chain mechanics, and actionable growth strategies.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Electrophysiology Equipment and Recording Systems Market Size, Market Forecast and Outlook By FMI
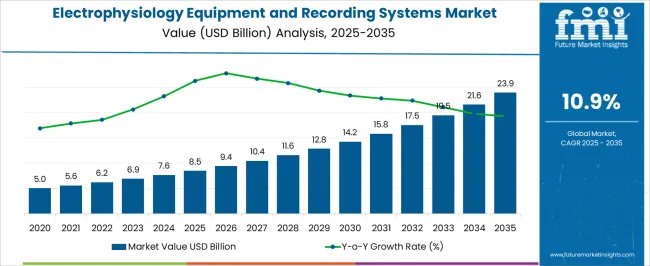
The electrophysiology equipment and recording systems market was valued at USD 8.50 billion in 2025, projected to reach USD 9.43 billion in 2026, and is forecast to expand to USD 26.53 billion by 2036 at a 10.90% CAGR. Rising global atrial fibrillation prevalence, expanding cardiac ablation procedure volumes, and the growing adoption of three-dimensional electroanatomical mapping systems are driving sustained investment in electrophysiology laboratory infrastructure. Pulsed field ablation technology adoption is generating comprehensive EP lab equipment upgrade cycles as hospitals install new energy delivery systems alongside existing radiofrequency and cryoablation platforms.
The absolute dollar opportunity of approximately USD 17.10 billion over the forecast period reflects one of the fastest-growing medical device infrastructure segments. Single-shot catheter ablation devices are expanding the addressable procedure population by reducing procedural complexity and enabling lower-volume centres to offer EP services, creating new equipment procurement at facilities that previously lacked ablation programme infrastructure. AI-integrated recording systems capable of automated arrhythmia substrate identification and real-time catheter navigation guidance are commanding premium installed system prices by reducing operator dependency on specialist electrophysiologist interpretation.
China sets the pace at a 14.8% CAGR, driven by cardiovascular intervention centre expansion across provincial hospitals and growing AF treatment access under national cardiac disease management programmes. India follows at 13.7% as cardiac catheterization laboratory investment and EP training programme expansion generate equipment procurement at tertiary hospitals. Germany registers 12.6% growth supported by established EP centre density and pulsed field ablation technology early adoption. France maintains 11.5% driven by comprehensive cardiac rhythm management programme equipment upgrades. UK expands at 10.4% anchored by NHS long-term plan cardiovascular service expansion. USA tracks at 9.3% supported by the largest installed EP lab base and PFA technology adoption driving equipment refresh. Brazil registers 8.2% with SUS cardiovascular centre expansion and EP programme establishment.
Summary of Electrophysiology Equipment and Recording Systems Market
- Electrophysiology Equipment and Recording Systems Market Definition
- Electrophysiology equipment and recording systems are capital devices, catheters, and software for diagnosing and treating cardiac arrhythmias through intracardiac signal recording, electroanatomical mapping, and catheter ablation in hospital EP laboratories.
- Demand Drivers in the Market
- Rising atrial fibrillation prevalence driven by aging populations and improved diagnostic detection rates is expanding cardiac ablation procedure volumes that generate per-procedure catheter consumption and EP lab equipment utilization.
- Pulsed field ablation technology adoption is driving comprehensive EP laboratory upgrade cycles as hospitals install new energy delivery platforms alongside existing RF and cryoablation systems.
- Single-shot catheter devices enabling lower-volume centres to offer EP services are creating new equipment procurement at facilities that previously lacked ablation programme infrastructure.
- Key Segments Analyzed in the FMI Report
- Hospital by Application: 48.9% share in 2026, as hospital-based EP laboratories house the three-dimensional mapping systems and multi-energy ablation platforms required for complex arrhythmia procedures.
- Electrophysiology Laboratory System by Types: 39.3% share in 2026, reflecting the high capital value of integrated EP lab recording, mapping, and ablation delivery platform installations.
- China: 14.8% compound growth driven by cardiovascular intervention centre expansion across provincial hospitals and AF treatment access expansion under national cardiac disease programmes.
- Analyst Opinion at FMI
- The EP equipment market is entering an accelerated technology refresh cycle where pulsed field ablation adoption compels hospitals to invest in new energy delivery platforms while maintaining existing RF and cryo systems, expanding the installed equipment base per laboratory. Facilities that delay PFA capability investment face referral pattern shifts as patients and referring physicians migrate to centres offering the latest ablation technology. The critical market access risk centers on clinical evidence generation timelines, as PFA devices with limited long-term efficacy data face payer coverage uncertainty that delays institutional procurement approvals.
- Strategic Implications / Executive Takeaways
- EP equipment manufacturers must develop multi-energy platform architectures accommodating RF, cryo, and PFA ablation modalities within unified recording and mapping environments to capture comprehensive lab upgrade contracts.
- Hospital cardiovascular service line directors must plan PFA technology capital budgets alongside existing ablation programme operations to maintain referral competitiveness as ablation technology standards evolve.
- EP catheter companies must invest in clinical trial programmes generating long-term PFA efficacy data to support payer coverage determination submissions that unlock institutional procurement budgets.
Electrophysiology Equipment and Recording Systems Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 9.43 Billion |
| Industry Value (2036) | USD 26.53 Billion |
| CAGR (2026-2036) | 10.90% |
Source: Future Market Insights, 2026
Electrophysiology Equipment and Recording Systems Market Definition
Electrophysiology equipment and recording systems encompass the capital devices, disposable catheters, and software platforms used to diagnose and treat cardiac arrhythmias through intracardiac electrical signal recording, electroanatomical mapping, and catheter-based ablation procedures. These systems include EP laboratory recording platforms, three-dimensional mapping systems, diagnostic and ablation catheters, stimulation devices, and integrated workflow management software for hospital EP laboratories and cardiac catheterization suites.
Electrophysiology Equipment and Recording Systems Market Inclusions
Market scope includes EP laboratory recording systems, 3D electroanatomical mapping platforms, diagnostic EP catheters, ablation catheters (radiofrequency, cryoablation, pulsed field), cardiac stimulators, and EP laboratory information management software. Coverage spans hospital, clinic, and ambulatory surgery centre applications.
Electrophysiology Equipment and Recording Systems Market Exclusions
Implantable cardiac devices (pacemakers, defibrillators), external cardiac monitoring devices, standalone ECG machines, and cardiac imaging equipment without EP-specific functionality are excluded.
Electrophysiology Equipment and Recording Systems Market Research Methodology
- Primary Research: Analysts engaged with EP laboratory directors, hospital cardiovascular service line administrators, and EP equipment manufacturer clinical affairs managers to map procurement triggers for lab establishment and technology upgrade decisions.
- Desk Research: Data collection phases aggregated cardiac ablation procedure volume registries, EP laboratory establishment statistics, and pulsed field ablation regulatory approval timelines across key national markets.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of EP equipment system installations and per-procedure catheter consumption multiplied by average prices, applying region-specific procedure volume growth curves.
- Data Validation and Update Cycle: Projections are tested against publicly reported cardiac rhythm management and EP device segment revenues from leading medical device manufacturers.
Why is the Electrophysiology Equipment and Recording Systems Market Growing?
The electrophysiology equipment and recording systems market is experiencing steady growth, driven by increasing prevalence of cardiac arrhythmias, neurological disorders, and other electrophysiological conditions worldwide. Growing investments in advanced diagnostic and therapeutic technologies, coupled with rising awareness of early disease detection, are encouraging healthcare providers to adopt sophisticated equipment for accurate patient monitoring. Continuous advancements in electrode technology, signal acquisition systems, and real-time data processing are enhancing the precision and reliability of electrophysiological measurements.
Hospitals and specialized diagnostic centers are increasingly deploying integrated systems that support multi-patient monitoring, automated reporting, and seamless electronic health record integration. Regulatory initiatives emphasizing patient safety and standardized diagnostic protocols are also supporting the market by fostering trust in high-quality equipment.
Additionally, the rising adoption of minimally invasive and catheter-based procedures has highlighted the need for advanced electrophysiology recording systems As healthcare infrastructure expands globally, and with an increasing focus on patient-centered care, the market is expected to sustain growth, with manufacturers prioritizing technological innovation, training, and service support to meet evolving clinical demands.
Segmental Analysis
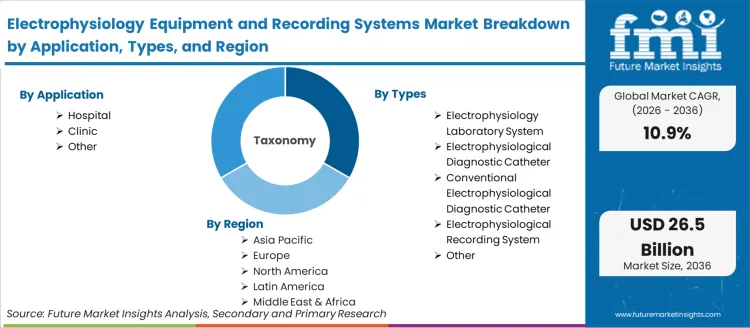
The electrophysiology equipment and recording systems market is segmented by application, types, and geographic regions. By application, electrophysiology equipment and recording systems market is divided into Hospital, Clinic, and Other. In terms of types, electrophysiology equipment and recording systems market is classified into Electrophysiology Laboratory System, Electrophysiological Diagnostic Catheter, Conventional Electrophysiological Diagnostic Catheter, Electrophysiological Recording System, and Other. Regionally, the electrophysiology equipment and recording systems industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Hospital Application Segment
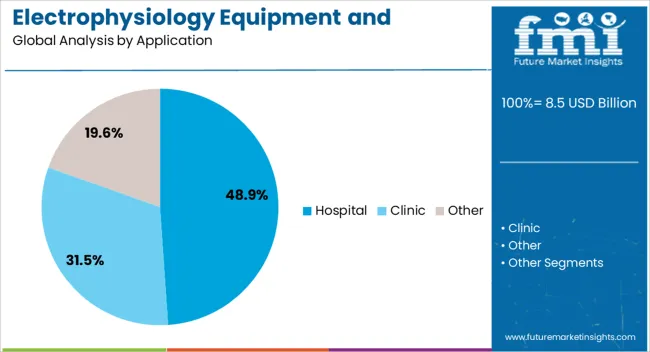
The hospital application segment is projected to account for 48.9% of the electrophysiology equipment and recording systems market revenue share in 2026, establishing it as the leading end-use segment. This dominance is driven by the increasing reliance on hospitals as central hubs for complex diagnostic and therapeutic procedures, including cardiac and neurological interventions. Hospitals benefit from the ability to integrate electrophysiology systems with other monitoring and imaging technologies, enhancing workflow efficiency and enabling comprehensive patient management.
Growing patient inflow for arrhythmia treatment and neurological disorder management has further reinforced the need for advanced monitoring solutions. The segment is also supported by the adoption of multi-patient and multi-procedure systems that allow scalable utilization of equipment, improving return on investment for healthcare facilities.
Continuous upgrades in recording systems, including enhanced signal resolution, automated data interpretation, and real-time analytics, have strengthened adoption With healthcare infrastructure improving in both developed and emerging markets, hospital-based deployment remains the dominant growth driver, ensuring high market share and ongoing investment in equipment and training.
Insights into the Electrophysiology Laboratory System Types Segment
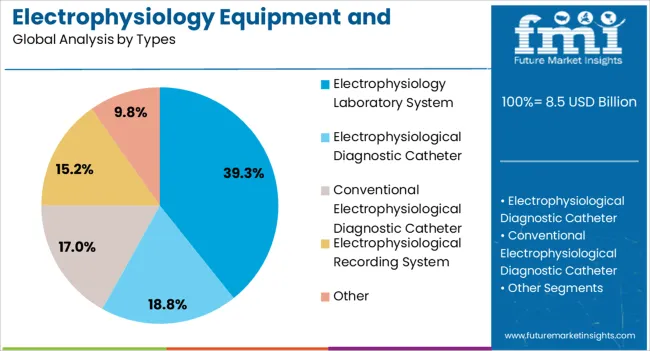
The electrophysiology laboratory system types segment is expected to hold 39.3% of the electrophysiology equipment and recording systems market revenue share in 2026, making it the leading system type. This leadership is being driven by the system’s ability to provide precise measurement, mapping, and analysis of electrical activity in cardiac and neural tissues. The integration of advanced software for signal acquisition, filtering, and real-time analysis allows clinicians to perform accurate diagnosis and therapeutic planning.
The segment benefits from the adoption of catheter-based and minimally invasive procedures, where laboratory systems play a critical role in guiding interventions. Growing investment in hospital and specialty clinics for electrophysiology laboratories, coupled with demand for high-throughput, multi-patient capable systems, has strengthened market adoption.
Enhanced reliability, real-time reporting, and interoperability with hospital IT infrastructure are supporting the segment’s growth The increasing emphasis on standardization of procedures and quality control in electrophysiology diagnostics is further reinforcing the preference for sophisticated laboratory systems, ensuring the segment maintains its leading position in the market.
Market Overview
An electrophysiology device are used to study the electrical properties of biological cells and tissues by measuring the voltage change of various biological cells, organs, tissues and the body as a whole.
These devices mainly used for the diagnosis as well as treatment of effected system of the body due to abnormal electrical properties of cells and tissues.
Electrophysiology devices and recoding system are used in the process of diagnosis and treatment of those disease that occur due to abnormal electrical properties of biological cells and tissues. These devices have wide application in the management of cardiac arrhythmias.
Abbot Laboratories Growing Market Share
A sizable portion of the medical devices division's bottom line comes from sales of electrophysiology equipment and recording systems by the leading company. The organization's goals include bettering its research and development, production, quality, and regulatory assurance departments. They put a lot of money into their medical devices division to boost their overall growth and profits. Sales of electrophysiology equipment and recording systems category are expected to fall to USD 1,578 million in 2026 from USD 1,721 million in 2026, largely as a result of the COVID - 19 pandemic's impact on the distribution channel and the flow of patients to hospitals.
Biosense Webster, Medtronic, Boston Scientific, and Microport Scientific Corporation are Abbott's main rivals in the electrophysiology equipment and recording systems market. In addition to its sensor-enabled catheters, the company also provides a cutting-edge ensuite precision cardiac mapping system. The company is ahead of the curve because of how it uses its size in high-growth emerging countries like China, Japan, and India.
How are Companies Strategizing to Conquer the Market?
Companies operating in the electrophysiology equipment and recording systems market are sometimes prevented from developing internationally due to a lack of uniformity in the rules governing the industry. New electrophysiological devices and equipment availability might be significantly impacted by regulatory restrictions in different countries.
In some regions, rules stipulate that manufacturers must conduct all or some of their clinical trials (including pre-and post-market) within the country. However, firms must pay for the trials themselves in countries like Brazil, Japan, India, and China, where local regulations are still in their infancy. As it becomes more challenging for manufacturers to provide adequate devices for running trials, the value and accuracy of test results suffer for trials of devices like cardiac rhythm management (CRM) in several nations.
Electrophysiology Equipment and Recording Systems Market: Drivers and Restraints
Presently, electrophysiology equipment and recording systems global market is driven by technological advancement in healthcare industry, rapid growth in geriatric population, increasing incidence of arrhythmia cases across the globe.
Global electrophysiology equipment and recording system market is drives by the aging population, technological advancement in healthcare industry, growing focus of key market players to expand their geographic presence, increasing incidence of arrhythmia cases across the globe. However, lack of skilled and experienced electrophysiologists in developing countries and unfavorable healthcare reforms are some of the major barrier for this market.
Electrophysiology Equipment and Recording Systems Market: Overview
With rapid technological advancement, rapid growth of aging population and increasing incidence of arrhythmia cases across the globe, the global electrophysiology equipment and recording system market is expected to have healthy growth rate in the forecast period (2026 to 2026).
Electrophysiology Equipment and Recording Systems Market: Region-wise Outlook
Depending on geographic region, the global electrophysiology equipment and recording systems market is segmented into seven key regions: North America, South America, Eastern Europe, Western Europe, Asia Pacific, Japan, and Middle East & Africa.
Due to the rapid increase in aging population and increasing focus on emerging market by key players the North America has the largest share of market followed by Europe.
Electrophysiology Equipment and Recording Systems Market: Key Players
Some of the key participating major players in global electrophysiology equipment and recording systems market are Boston Scientific Corp., GE healthcare Inc., Koninklijke Philips N.V., Medtronic Inc., Siemens AG, Biosense Webster Inc., Microport Scientific Corp., Abbot Laboratories and Others.
The research report presents a comprehensive assessment of the market and contains thoughtful insights, facts, historical data, and statistically supported and industry-validated market data. It also contains projections using a suitable set of assumptions and methodologies. The research report provides analysis and information according to categories such as market segments, geographies, types, technology and applications.
The report covers exhaustive analysis on
- Market Segments
- Market Dynamics
- Market Size
- Supply & Demand
- Current Trends/Issues/Challenges
- Competition & Companies involved
- Technology
- Value Chain
Regional analysis includes
- North America (USA, Canada)
- Latin America (Mexico. Brazil)
- Western Europe (Germany, Italy, France, UK, Spain, Nordic countries, Belgium, Netherlands, Luxembourg)
- Eastern Europe (Poland, Russia)
- Asia Pacific (China, India, Australia & New Zealand)
- Japan
- Middle East and Africa (S. Africa, N. Africa)
The report is a compilation of first-hand information, qualitative and quantitative assessment by industry analysts, inputs from industry experts and industry participants across the value chain. The report provides in-depth analysis of parent market trends, macro-economic indicators and governing factors along with market attractiveness as per segments. The report also maps the qualitative impact of various market factors on market segments and geographies.
Analysis of Electrophysiology Equipment and Recording Systems Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 14.8% |
| India | 13.7% |
| Germany | 12.6% |
| France | 11.5% |
| UK | 10.4% |
| USA | 9.3% |
| Brazil | 8.2% |
Source: FMI analysis based on primary research and proprietary forecasting model
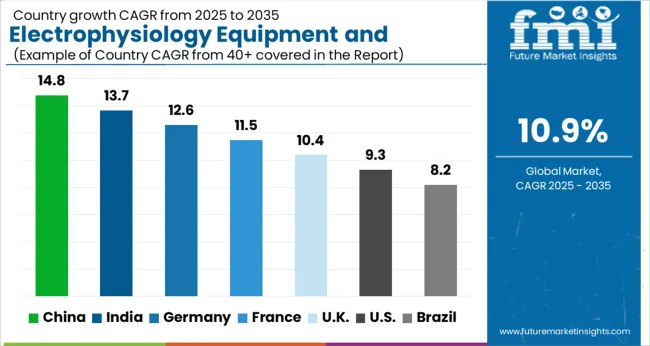
Country-wise Analysis
The Electrophysiology Equipment and Recording Systems Market is expected to register a CAGR of 10.9% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 14.8%, followed by India at 13.7%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates.
Brazil posts the lowest CAGR at 8.2%, yet still underscores a broadly positive trajectory for the global Electrophysiology Equipment and Recording Systems Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 12.6%.
The USA Electrophysiology Equipment and Recording Systems Market is estimated to be valued at USD 2.9 billion in 2026 and is anticipated to reach a valuation of USD 7.1 billion by 2036. Sales are projected to rise at a CAGR of 9.3% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 456.5 million and USD 232.3 million respectively in 2026.
Key Players in the Electrophysiology Equipment and Recording Systems Market
- Boston Scientific
- GE Healthcare
- Koninklijke Philips
- Medtronic
- Siemens
- Biosense Webster
- Microport Scientific
- Abbott Laboratories
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 9.43 Billion to USD 26.53 Billion, at a CAGR of 10.90% |
| Market Definition | Electrophysiology equipment and recording systems are capital devices, catheters, and software for diagnosing and treating cardiac arrhythmias through intracardiac signal recording, electroanatomical mapping, and catheter ablation in hospital EP laboratories. |
| Segmentation | Application: Hospital, Clinic, and Other; Types: Electrophysiology Laboratory System, Electrophysiological Diagnostic Catheter, Conventional Electrophysiological Diagnostic Catheter, Electrophysiological Recording System, and Other |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | Boston Scientific, GE Healthcare, Koninklijke Philips, Medtronic, Siemens, Biosense Webster, Microport Scientific, Abbott Laboratories |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and projecting conversion rates to technology adoption. |
Electrophysiology Equipment and Recording Systems Market by Segments
Application:
- Hospital
- Clinic
- Other
Types:
- Electrophysiology Laboratory System
- Electrophysiological Diagnostic Catheter
- Conventional Electrophysiological Diagnostic Catheter
- Electrophysiological Recording System
- Other
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. European Heart Rhythm Association. (2024). Atrial Fibrillation Management Guidelines: EP Laboratory Equipment Standards. EHRA.
- 2. Heart Rhythm Society. (2024). EP Laboratory Standards: Equipment and Facility Requirements for Catheter Ablation. HRS.
- 3. World Heart Federation. (2024). Global Atrial Fibrillation Prevalence: Epidemiological Assessment Report. WHF.
- 4. National Health Commission of China. (2024). Cardiovascular Intervention Centre Establishment Standards. NHC.
- 5. USA Food and Drug Administration. (2024). Pulsed Field Ablation Device Premarket Approval: Guidance for Industry. FDA.
- 6. National Institute for Health and Care Excellence. (2024). Interventional Procedure Guidance: Catheter Ablation for Atrial Fibrillation. NICE.
- This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Frequently Asked Questions
How large is the demand for Electrophysiology Equipment and Recording Systems in 2026?
Demand is estimated to be valued at USD 9.43 billion in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 26.53 billion by 2036.
What is the expected CAGR between 2026 and 2036?
Demand is expected to grow at a CAGR of 10.90% between 2026 and 2036.
Which Application is poised to lead global sales by 2026?
Hospital accounts for 48.9% share housing the 3D mapping and multi-energy ablation platforms required for complex procedures.
How is Electrophysiology Laboratory System positioned by Types in 2026?
EP Laboratory System represents 39.3% share reflecting high capital value of integrated recording, mapping, and ablation installations.
What is driving demand in China?
CV intervention centre expansion and AF treatment access under national cardiac programmes fuel the fastest growth at 14.8% CAGR through 2036.
What is included in the scope of this report?
The market covers EP recording systems, 3D mapping platforms, diagnostic and ablation catheters, cardiac stimulators, and lab management software.
What is excluded from the scope of this report?
Implantable cardiac devices, external monitors, standalone ECG machines, and cardiac imaging equipment without EP functionality are excluded.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2026 to 2036
- Hospital
- Clinic
- Other
- Hospital
- Y to o to Y Growth Trend Analysis By Application , 2021 to 2025
- Absolute $ Opportunity Analysis By Application , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Types
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Types, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Types, 2026 to 2036
- Electrophysiology Laboratory System
- Electrophysiological Diagnostic Catheter
- Conventional Electrophysiological Diagnostic Catheter
- Electrophysiological Recording System
- Other
- Electrophysiology Laboratory System
- Y to o to Y Growth Trend Analysis By Types, 2021 to 2025
- Absolute $ Opportunity Analysis By Types, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Types
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Types
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Types
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Types
- Competition Analysis
- Competition Deep Dive
- Boston Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- GE Healthcare
- Koninklijke Philips
- Medtronic
- Siemens
- Biosense Webster
- Microport Scientific
- Abbott Laboratories
- Boston Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Types, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Types, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Types
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Application
- Figure 23: North America Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Types
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Application
- Figure 30: Latin America Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Types
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Application
- Figure 37: Western Europe Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Types
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Application
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Types
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Application
- Figure 51: East Asia Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Types
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Types
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Types, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Types, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Types
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

