Intravenous Contrast Agents Market Size and Share Forecast Outlook 2026 to 2036
The intravenous contrast agents market is valued at USD 11,880 million in 2026 and is projected to reach USD 20,485.9 million by 2036, expanding at a CAGR of 5.60%. Expansion in emerging markets, where imaging infrastructure is scaling rapidly, adds volume momentum but also increases execution risk related to pricing pressure and supply reliability. In mature markets, adoption friction is shaped by heightened safety scrutiny, protocol standardization, and cautious use in renal-risk populations, limiting aggressive volume acceleration.
Economic sensitivity is moderate contrast agents are essential consumables, yet procurement decisions are increasingly influenced by total cost of use, wastage control, and supplier reliability rather than brand preference alone. Recent supply disruptions have exposed vulnerabilities in manufacturing concentration and logistics dependencies, raising the importance of redundancy and regional production. Between 2026 and 2036, sustainable value creation will depend on operational resilience, regulatory compliance, and disciplined portfolio management rather than rapid penetration or pricing expansion.
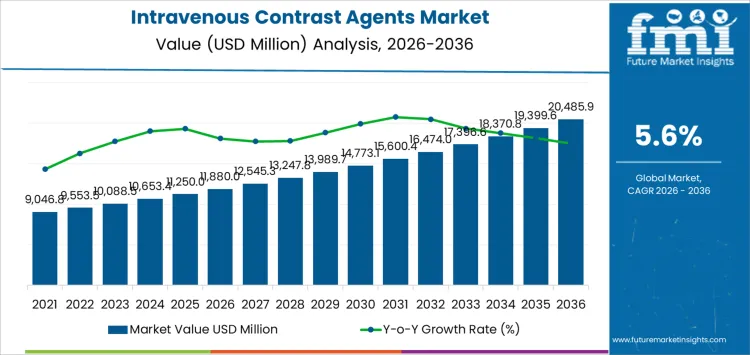
| Metric | Value |
|---|---|
| Intravenous Contrast Agents Market Value (2026) | USD 11,880 Million |
| Intravenous Contrast Agents Market Forecast Value (2036) | USD 20,485.9 Million |
| Intravenous Contrast Agents Market Forecast CAGR (2026-2036) | 5.60% |
The intravenous contrast agents market is expanding as diagnostic imaging volumes continue to rise across CT, MRI, and angiography procedures. Hospitals and imaging centers are optimizing protocols to improve diagnostic confidence while managing throughput and patient safety. For manufacturers, demand is increasingly tied to agents that offer consistent image enhancement, predictable pharmacokinetics, and compatibility with high-throughput imaging workflows. Utilization growth in emergency care, oncology staging, and cardiovascular diagnostics is sustaining steady baseline demand.
Safety considerations and protocol standardization are central to purchasing decisions. Providers are prioritizing contrast agents with favorable safety profiles, lower osmolality, and reduced risk of adverse reactions, particularly for patients with renal impairment or comorbidities. This is driving preference for products supported by robust clinical data and clear dosing guidance across diverse patient populations. Suppliers that provide protocol support, training materials, and post-market surveillance data are gaining stronger adoption among large hospital networks.
Procurement economics and supply assurance are also shaping market dynamics. Group purchasing organizations and centralized procurement teams are evaluating contrast agents based on total cost of use, including wastage, vial sizing, and logistics reliability not unit price alone. Recent supply disruptions have elevated the importance of dual sourcing, regional manufacturing, and inventory resilience. For manufacturers and distributors, competitive advantage increasingly depends on dependable supply, portfolio breadth across imaging modalities, and the ability to align product offerings with evolving clinical protocols positioning intravenous contrast agents as a stable, volume-driven segment within diagnostic imaging.
The intravenous contrast agents market is segmented by product and application, reflecting imaging modality requirements and diagnostic workflow preferences. By product, iodinated contrast agents lead the market, as they are extensively used for vascular and soft-tissue visualization. Other products include gadolinium-based agents and microbubble contrast agents, which support modality-specific imaging needs. By application, CT imaging represents the largest demand area, followed by MRI imaging and ultrasound imaging, each requiring contrast agents with tailored physicochemical and safety profiles.
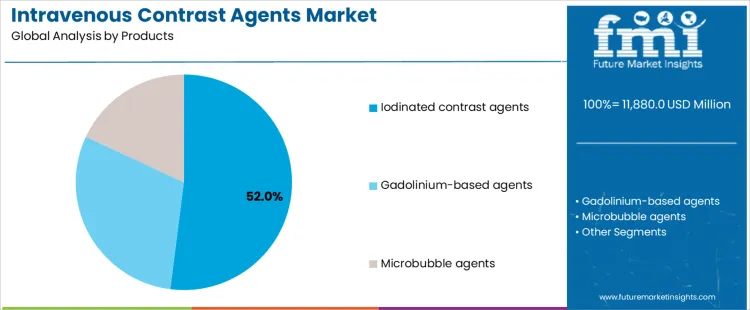
Iodinated contrast agents account for 52% share of the intravenous contrast agents market because they are the standard agents used in CT and angiographic procedures. Their high atomic number provides strong X-ray attenuation, enabling clear visualization of blood vessels, organs, and pathological lesions. Hospitals rely on iodinated agents due to broad clinical familiarity, rapid imaging capability, and compatibility with high-throughput CT scanners. These agents support emergency diagnostics, oncology imaging, and cardiovascular assessment. High procedure volumes drive consistent consumption across healthcare facilities. For manufacturers, iodinated contrast agents offer stable demand and established production scale, reinforcing their leadership.
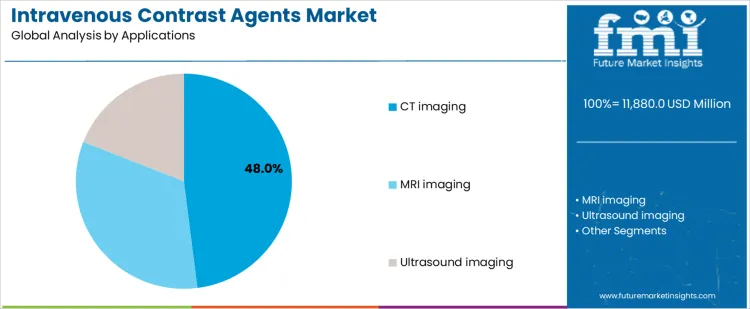
CT imaging holds 48% share of the intravenous contrast agents market because CT scans are widely used for fast, high-resolution diagnostic evaluation. Emergency medicine, trauma care, oncology staging, and cardiovascular assessment rely heavily on contrast-enhanced CT imaging. The speed of CT scanners supports high patient throughput, increasing contrast agent utilization per facility. Contrast-enhanced CT improves lesion detection, vascular mapping, and diagnostic confidence. Growing access to CT systems across hospitals and diagnostic centers further expands usage. These clinical efficiency and volume-driven factors explain why CT imaging remains the dominant application segment.
The intravenous contrast agents market is driven by increasing usage of diagnostic imaging procedures such as computed tomography (CT), magnetic resonance imaging (MRI), and angiography, where contrast agents enhance visibility of vascular structures and soft tissues. Growing prevalence of chronic diseases including cancer, cardiovascular disorders, and neurological conditions fuels demand for enhanced imaging for accurate diagnosis and treatment planning. For contrast agent manufacturers and suppliers, product safety profile, image enhancement effectiveness, cost per dose, and compatibility with a broad range of imaging systems are core factors shaping procurement by healthcare providers, imaging centers, and hospitals.
Clinical practice and imaging technology trends are shaping the intravenous contrast agents market as hospitals and imaging centers increasingly adopt advanced diagnostic modalities that require high-performance contrast media. Innovations in CT scanner resolution and MRI field strength demand contrast agents with improved relaxivity, lower toxicity, and enhanced perfusion characteristics. Clinicians are placing greater emphasis on rapid infusion, reduced adverse reaction rates, and agents with lower osmolality to improve patient comfort and safety. For suppliers, developing formulations that support both high-quality imaging and streamlined workflow such as ready-to-use liquid formats enhances adoption in busy diagnostic environments.
Safety concerns and stringent regulatory requirements restrain the intravenous contrast agents market because contrast media are administered systemically and must demonstrate minimal risk of adverse events. Risks such as contrast-induced nephropathy with iodinated agents and nephrogenic systemic fibrosis associated with certain gadolinium-based agents lead clinicians to exercise caution, especially in patients with kidney impairment. Regulatory bodies require robust clinical evidence for safety and efficacy, extending time to approval and increasing development cost. Post-marketing surveillance and evolving labeling requirements also affect prescribing patterns and hospital formularies, contributing to restrained uptake in certain patient populations.
The intravenous contrast agents market is expanding as diagnostic imaging volumes increase across CT, MRI, and angiography procedures worldwide. These agents are essential for enhancing image clarity and improving detection of cardiovascular, neurological, and oncological conditions. Growth varies by country based on imaging infrastructure expansion, disease burden, reimbursement frameworks, and hospital procurement practices. Mature markets emphasize safety profiles, protocol standardization, and long-term supplier relationships, while higher-growth regions are driven by rapid installation of imaging equipment and broader access to diagnostic services. Market value is supported by repeat procedure usage, rising diagnostic demand, and gradual adoption of advanced imaging protocols across hospital and outpatient settings.
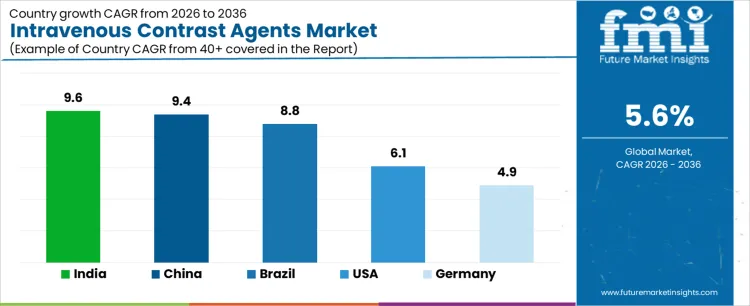
| Country | CAGR (%) |
|---|---|
| India | 9.6 |
| China | 9.4 |
| Brazil | 8.8 |
| United States | 6.1 |
| Germany | 4.9 |
The intravenous contrast agents market in the United States is growing at a CAGR of 6.1% during 2026-2036, reflecting steady expansion within a highly developed diagnostic imaging ecosystem. Demand is driven by widespread use of CT, MRI, and angiography procedures across hospitals and outpatient imaging centers. Radiologists prioritize image clarity, patient safety, and compatibility with advanced imaging equipment. Iodinated contrast agents dominate CT applications, while gadolinium-based agents are widely used in MRI. Procurement decisions are influenced by clinical evidence, safety profiles, reimbursement alignment, and long-term supplier relationships. Generic competition places pressure on pricing, but high procedure volumes support consistent demand. Market value is driven by repeat usage, protocol standardization, and incremental formulation improvements. The USA represents a mature market where growth depends on imaging utilization rates, product reliability, and integration with high-throughput diagnostic workflows.
Brazil’s intravenous contrast agents market is expanding at a CAGR of 8.8% during 2026-2036, supported by growing access to advanced diagnostic imaging in both public and private healthcare facilities. Expansion of CT and MRI infrastructure in urban centers is increasing procedure volumes. Buyers focus on affordability, reliable supply, and ease of administration. Iodinated contrast agents account for a significant share of demand, with growing use of MRI contrast in tertiary hospitals. Procurement decisions are influenced by public healthcare budgets, distributor reach, and clinician familiarity. Adoption is strongest in large hospitals and diagnostic chains. Market value is driven by increasing imaging referrals, improved diagnostic coverage, and expanding private healthcare investment. Brazil represents a growth-oriented market where competitive success depends on cost-effective portfolios, strong local distribution, and consistent product availability.
China’s intravenous contrast agents market is growing at a CAGR of 9.4% during 2026-2036, driven by rapid expansion of diagnostic imaging capacity and rising demand for advanced disease detection. Large public hospitals and regional medical centers are increasing CT and MRI procedure volumes. Buyers emphasize throughput efficiency, safety compliance, and availability of high-volume supplies. Iodinated contrast agents dominate demand, while MRI contrast usage is increasing as imaging access expands. Procurement decisions favor suppliers with local manufacturing, regulatory compliance, and scalable logistics. Qualification cycles are relatively short due to high patient volumes. Market value is driven by population scale, hospital expansion, and integration of imaging into routine diagnostics. China represents a volume-driven market where reliability, capacity, and regulatory alignment determine competitiveness.
India’s intravenous contrast agents market is projected to expand at a CAGR of 9.6% during 2026-2036, driven by rapid growth in diagnostic imaging capacity and improving access to advanced diagnostics. The installation of CT and MRI scanners is accelerating across private hospitals, diagnostic chains, and tier-two cities, increasing contrast-enhanced procedure volumes. Demand is supported by rising detection of cardiovascular, neurological, and oncological conditions. Buyers place strong emphasis on affordability, patient safety, and ease of administration, given high patient throughput and cost sensitivity. Iodinated contrast agents dominate usage due to their widespread application in CT imaging, while MRI contrast adoption is gradually increasing as MRI access improves. Procurement cycles are relatively short, allowing hospitals and imaging centers to onboard new suppliers quickly. Market value is driven by large patient volumes, expanding referral networks, and competitive pricing.
Germany’s intravenous contrast agents market is growing at a CAGR of 4.9% during the forecast period, reflecting a cautious and highly regulated adoption environment. Hospitals operate within standardized imaging protocols that prioritize patient safety, clinical evidence, and strict adherence to diagnostic guidelines. Contrast agent selection is heavily influenced by efficacy data, adverse event profiles, and long-term clinical outcomes. Procurement decisions typically involve multidisciplinary hospital committees and health insurance providers, resulting in extended evaluation and approval timelines. Generic penetration is well established, placing pressure on pricing while maintaining stable procedure volumes. Growth remains steady rather than accelerated, as technology adoption is incremental. Germany represents a mature market where supplier success depends on clinical credibility, regulatory compliance, and alignment with reimbursement and hospital governance frameworks rather than rapid market expansion.
Competition in the intravenous contrast agents market is driven by validated imaging performance, safety profiles, and documented clinical acceptance that matter to radiologists, hospital procurement teams, and imaging centers. GE HealthCare positions its contrast media portfolio with product materials that emphasize high iodine concentration agents offering improved vascular and tissue contrast. Official literature highlights robust safety data, reduced incidence of adverse reactions, and compatibility with CT and MRI platforms that support wide clinical adoption across diagnostic workflows.
Bayer competes by offering a diversified range of iodinated and gadolinium-based contrast agents tailored for CT, MRI, and angiographic applications. Its brochures stress documented pharmacokinetics, osmolality profiles, and broad label approvals including special population guidance. Bracco differentiates through specificity in neurological and vascular imaging agents, with official product information underlining enhanced lesion detectability and safety margins supported by extensive clinical trial evidence.
Specialty contrast providers focus on niche performance claims. Guerbet positions its agents with materials highlighting unique formulations with lower viscosity and tailored organ targeting. Product datasheets emphasize patient tolerance and detailed adverse event profiles that guide clinical decision making. Lantheus competes by aligning contrast agents with hybrid imaging systems such as PET/CT and SPECT/CT, offering agents with documented multi-modality efficacy and procedural reliability.
Others in the market include regional pharmaceutical manufacturers and emerging biologically-derived contrast solutions that focus on cost-effective alternatives and enhanced safety claims. Across all players, competitive differentiation is built on brochure-verified imaging performance, safety documentation, and workflow compatibility rather than price alone.
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Million |
| Products | Iodinated Contrast Agents, Gadolinium-Based Agents, Microbubble Agents |
| Applications | CT Imaging, MRI Imaging, Ultrasound Imaging |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | GE HealthCare, Bayer AG, Bracco Imaging S.p.A., Guerbet Group, Lantheus Holdings, Inc., Others |
| Additional Attributes | Dollar sales by product type and imaging application are evaluated across hospital and diagnostic imaging workflows. The report includes country-level demand assessment, growth projections for 2026-2036, analysis of imaging procedure volumes and protocol standardization, evaluation of safety and tolerability profiles, competitive benchmarking of contrast agent manufacturers, and assessment of procurement and reimbursement trends influencing adoption. |
How big is the intravenous contrast agents market in 2026?
The global intravenous contrast agents market is estimated to be valued at USD 11,880.0 million in 2026.
What will be the size of intravenous contrast agents market in 2036?
The market size for the intravenous contrast agents market is projected to reach USD 20,485.9 million by 2036.
How much will be the intravenous contrast agents market growth between 2026 and 2036?
The intravenous contrast agents market is expected to grow at a 5.6% CAGR between 2026 and 2036.
What are the key product types in the intravenous contrast agents market?
The key product types in intravenous contrast agents market are iodinated contrast agents, gadolinium-based agents and microbubble agents.
Which applications segment to contribute significant share in the intravenous contrast agents market in 2026?
In terms of applications, ct imaging segment to command 48.0% share in the intravenous contrast agents market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.