Plant Based API Market
Plant Based API Market Size and Share Forecast Outlook 2026 to 2036
Plant Based API Market Forecast and Outlook 2026 to 2036
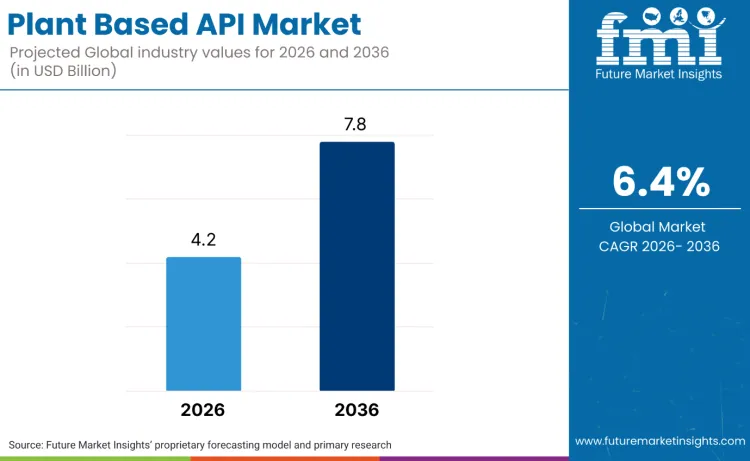
The global plant based API market is forecasted to reach USD 4.2 billion in 2026 and expand to USD 7.8 billion by 2036, advancing at a CAGR of 6.4%. This outlook shows a structural shift in pharmaceutical manufacturing, where drug development is moving from synthetic compound dependency toward natural botanical extraction and biosynthesis. Growth is anchored in the rising demand for complex therapeutic molecules, driven by oncology treatments, personalized medicine applications, and expanding use of plant-derived compounds in chronic disease management.
Two converging pressures are widening the addressable market. Regulatory agencies are increasingly favoring natural compound sourcing while environmental concerns are pushing pharmaceutical companies toward renewable raw materials. These forces are elevating the clinical and commercial value of plant-derived active pharmaceutical ingredients, encouraging manufacturers to adopt botanical extraction technologies earlier in their development pipelines. Healthcare companies and research institutions increasingly view plant based APIs as strategic assets rather than alternative inputs.
Quick Stats for Plant Based API Market
- Plant Based API Market Value (2026): USD 4.2 billion
- Plant Based API Market Forecast Value (2036): USD 7.8 billion
- Plant Based API Market Forecast CAGR (2026-2036): 6.4%
- Leading Product Type by Demand Share: Amino acids
- Fastest-Growing Countries: India, China, Germany, USA, Brazil
- Top Players in Global Demand: Alchem International Ltd., Indena S.p.A., Naturex SA, DSM Nutritional Products, Biocon Limited
The industry's value proposition is evolving from basic extraction services toward integrated and AI-based botanical pharmaceutical solutions. Advanced AI-based plant based API platforms are incorporating precision agriculture, biotechnology optimization, and AI-driven compound identification to enhance bioavailability while reducing production costs. Biotechnology companies and pharmaceutical manufacturers are gaining preference for turnkey botanical solutions, enabling standardized compound development at scale across research and commercial manufacturing settings.
Plant Based API Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.2 billion |
| Market Forecast Value (2036) | USD 7.8 billion |
| Forecast CAGR (2026-2036) | 6.4% |
Market Definition
The plant-based Active Pharmaceutical Ingredient (API) market refers to the global market for naturally derived compounds used in pharmaceutical applications. These ingredients are extracted from plants and utilized in drug development, focusing on natural, sustainable sources for therapeutic treatments. The market encompasses products derived from botanical sources like amino acids, alkaloids, glycosides, and other plant-derived compounds used in various pharmaceutical, nutraceutical, and food and beverage applications. The market is measured in USD billion and analyzed for the 2026 to 2036 period.
Market Inclusion
Included within this market are plant-based APIs sourced through conventional extraction, biotechnology synthesis, and hybrid methods. Key product categories include amino acids, alkaloids, glycosides, and others. The scope also covers applications in pharmaceuticals, nutraceuticals, and food and beverages, particularly those targeting chronic conditions, personalized medicine, and wellness.
The market includes global demand for plant-based APIs from pharmaceutical manufacturers, biotech companies, and nutraceutical developers. The geographic focus spans across Asia Pacific, Europe, North America, Latin America, and the Middle East & Africa, with specific attention to countries such as India, the USA, Germany, and China.
Market Exclusion
Excluded from the scope are synthetic pharmaceutical ingredients and non-botanical-based APIs. Additionally, APIs used solely in non-therapeutic or non-health-related industries, such as those used in industrial or agricultural applications, are not included.
Also excluded are traditional synthetic or semi-synthetic drugs that do not rely on plant-based sources. The scope does not include plant-based APIs used exclusively in cosmetics, personal care products, or food additives unless they directly support pharmaceutical or nutraceutical purposes.
What Key Structural Factors are Likely to Influence Plant-based API Market During 2026-2036?
In 2026, regulatory bodies and pharmaceutical standards organizations began mandating biotechnology-enhanced plant extraction in therapeutic compound development. This regulatory evolution established clear guidelines for acceptable botanical processing methods and required transparent validation of natural compound synthesis. As a result, plant based API solutions shifted from basic extraction to scientifically validated, standardized botanical pharmaceutical ingredients that meaningfully influence drug development and patient outcomes. Consumer and manufacturer engagement has shifted dramatically.
Traditional plant extraction competed on raw material availability, generating inconsistent therapeutic compounds that limited clinical applications. Real-time, biotechnology-optimized plant based API platforms now integrate with pharmaceutical development systems and regulatory databases, delivering standardized compound profiles and validated therapeutic benefits. These systems use advanced extraction technologies, genetic optimization, and continuous quality monitoring to produce pharmaceutical-grade ingredients, transforming plant based APIs into trusted therapeutic components rather than alternative materials.
The manufacturing and development paradigm is equally significant. Previously, seasonal cultivation cycles dominated production planning, with limited scalability and restricted compound availability. Between 2026 and 2036, controlled environment agriculture and biotechnology synthesis enable year-round production and predictable compound yields. Automated extraction systems and real-world pharmaceutical integration accelerate innovation and ensure solutions meet evolving therapeutic requirements and regulatory standards.
How is the Plant-based API Market Segmented?
The plant based API market is segmented by product type into amino acids, alkaloids, glycosides, and other botanical derived active compounds, reflecting differences in source crops, purification complexity, and end use positioning. By application, demand spans pharmaceuticals, nutraceuticals, food and beverages, and other uses, with each application class shaped by distinct regulatory expectations, dosage formats, and quality specifications. By extraction method, the market divides into conventional extraction, biotechnology synthesis, and hybrid methods, a split that captures the trade off between natural origin claims, yield consistency, impurity control, and scalability.
Regionally, the market covers Asia Pacific including China, Japan, South Korea, India, Australia and New Zealand, ASEAN, and the rest of Asia Pacific, Europe comprising Germany, the United Kingdom, France, Italy, Spain, the Nordics, BENELUX, and the rest of Europe, North America with the United States, Canada, and Mexico, Latin America across Brazil, Chile, and the rest of Latin America, and the Middle East and Africa including the Kingdom of Saudi Arabia, other GCC countries, Turkey, South Africa, other African Union countries, and the rest of the region.
Which Product Type Holds the Largest Share in Plant Based APIs?
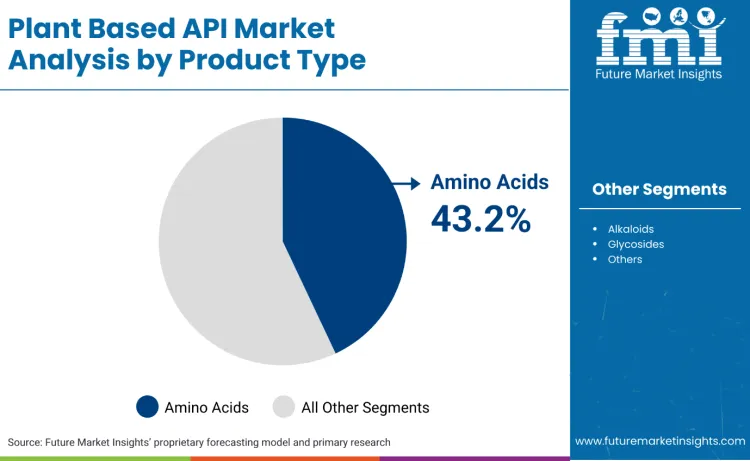
Amino acids account for 43.2% because the fastest growing part of plant based APIs is not a single blockbuster molecule, it is the expanding set of peptide and protein adjacent therapies that rely on controlled amino acid inputs and tight impurity management. FDA guidance on highly purified synthetic peptide drug products reflects how modern peptide manufacturing is built through stepwise amino acid addition, which makes amino acid quality systems and pharmacopoeial alignment a procurement priority rather than a commodity purchase.
USP has also been modernising amino acid monographs, signalling sustained pharmaceutical relevance and tighter standardisation expectations that favour scaled suppliers with validated quality. Alkaloids hold 28.7% because they remain a proven therapeutic class in pain and neurology, yet market growth is shaped by compliance burden and risk controls tied to potency, toxicity, and variability in botanical sourcing.
EMA guidance on pyrrolizidine alkaloids illustrates how regulators treat alkaloid related safety risk as a real constraint that forces additional testing and supplier qualification. Glycosides represent 28.1% because demand is anchored in defined clinical niches such as cardiac care, with digoxin a persistent example of a plant derived cardiac glycoside still used in practice, which sustains steady but narrower API pull versus amino acid platforms.
How Do Application Requirements Influence Plant Based API Selection?
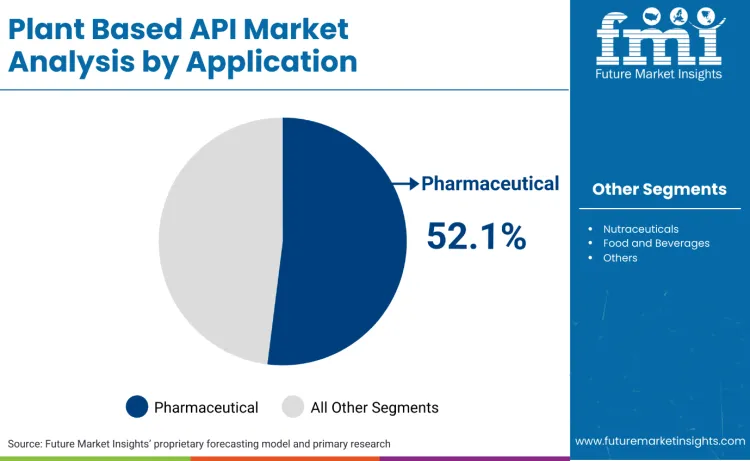
Pharmaceutical applications hold 52.1% because plant based APIs used as drug substances must survive the regulatory reality of identity, consistency, and impurity control across batches and seasons. FDA guidance on botanical drug development makes quality and CMC expectations explicit for botanical products submitted under IND and NDA pathways, which pushes sponsors toward validated compound profiles, tight specifications, and supplier discipline that favour pharmaceutical grade demand over looser extract use cases. The effect is structural. Once an API is tied to a regulated therapeutic claim, procurement shifts from price per kilogram to reproducibility, documented controls, and audit readiness, which keeps pharma as the anchor segment.
Nutraceuticals represent 31.4% because the category scales through supplements that still require formal manufacturing quality systems, yet typically face a different evidentiary burden than drugs. In the United States, dietary supplement cGMP rules in 21 CFR Part 111 require manufacturers to establish and follow manufacturing practices and quality controls, which increases demand for standardized botanical ingredients and consistent assay profiles without necessarily forcing the full drug development pathway.
Food and beverage applications account for 16.5% because use is constrained by food ingredient approval frameworks and safety substantiation norms, with many companies relying on GRAS conclusions and notifications in the USA and formal authorisation procedures in the EU for additive style use, which tends to favour lower dose functional roles over API intensity.
What Market Dynamics are Influencing Plant-based API Market?
| Cause | What changes in the market | Effect on plant based APIs |
|---|---|---|
| Regulators define a credible drug pathway for botanicals | Sponsors treat botanical APIs as developable drug substances, not folk ingredients | Higher pharma grade demand, higher spend on identity control, specifications, batch consistency. |
| Growing set of approved botanical drugs creates precedent | Quality systems, comparability expectations, and review patterns become clearer | More investors fund botanical API programs with defensible regulatory logic, fewer science projects with weak CMC. |
| Quality guidance tightens for herbal active substances | Dossiers require deeper control of variability, impurities, contaminants | Suppliers with validated assays and traceability win, smaller extract houses lose share in regulated demand. |
| Starting material risk becomes a regulator focus | Good agricultural and collection practice expectations formalise critical steps | Procurement shifts toward contracted cultivation, controlled harvesting, documented primary processing. |
| High value APIs depend on narrow plant chemotypes | Raw material supply becomes a constraint, not a sourcing task | Long term supply agreements, geographic diversification, stock strategies, higher working capital. |
| Demand rises for complex molecules where synthesis is hard | More programs target plant derived scaffolds and mixtures | Plant based APIs retain share in niches where structure complexity blocks economical full synthesis. |
| Analytical science improves for botanical quality control | Fingerprinting, marker based assays, multi component control become normal | Higher reproducibility, fewer batch failures, faster scale up for regulated applications. |
| Biotech routes mature for plant molecule supply | Plant cell culture, bioconversion, hybrid routes become viable for selected APIs | Reduced dependence on wild harvest, improved yield stability, scalable production for specific actives. |
| Cost base stays structurally higher than commodity synthetics | More unit operations, more testing, more supplier qualification | Adoption concentrates in indications where clinical value supports cost, weaker pull in commoditised categories. |
| Regulatory burden extends timelines | CMC documentation, consistency evidence, clinical substantiation take longer | Fewer launches from small firms, more partnering with experienced pharma quality operators. |
| Recent shift: tighter agriculture guidance in Europe | 2025 update strengthens practical expectations for herbal starting materials | Accelerates professionalisation of upstream supply, pushes marginal suppliers out of EU grade chains. |
| Recent shift: renewed FDA clarity on what counts as botanical drugs | FDA summarises approved botanical NDAs and a licensed botanical BLA in 2025 | Confidence effect for boards and BD teams, more pipeline formation around compliant botanical APIs.) |
How Is the Plant Based API Market Evolving Globally?
Global demand for plant based API solutions is increasing as pharmaceutical companies expand therapeutic development while addressing regulatory compliance, therapeutic efficacy, and compound standardization application requirements. Growth reflects rising use of biotechnology-enhanced platforms, advanced extraction systems, and quality-optimized production specifications across pharmaceutical research, biotechnology companies, and herbal nutraceutical manufacturers. Platform selection focuses on extraction efficiency, regulatory compliance, and cost performance under various therapeutic scenarios. India records 8.7% CAGR, China records 7.2% CAGR, Germany records 6.8% CAGR, USA records 6.1% CAGR, and Brazil records 5.9% CAGR. Adoption remains driven by therapeutic requirements and regulatory compliance rather than agricultural expansion alone.
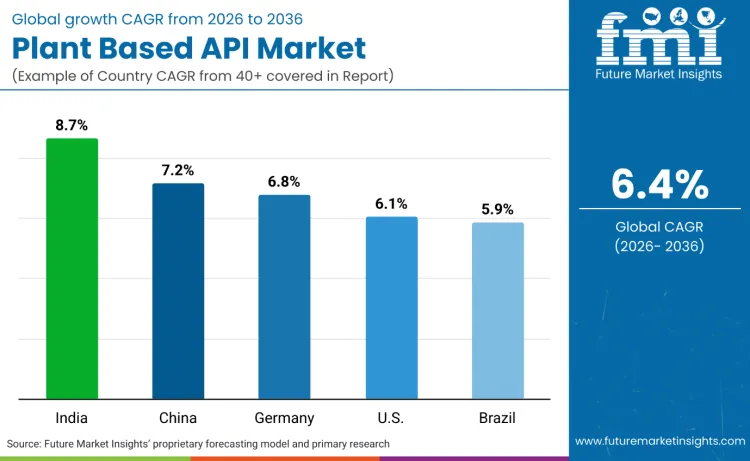
| Country | CAGR (2026-2036) |
|---|---|
| India | 8.7% |
| China | 7.2% |
| Germany | 6.8% |
| USA | 6.1% |
| Brazil | 5.9% |
India plant based API market: Can Ayurveda quality infrastructure convert tradition into export grade drug inputs?
India’s advantage is not folklore. It is a state backed quality stack that converts plant supply into auditable pharmaceutical inputs. Pharmacopoeia Commission for Indian Medicine and Homoeopathy develops pharmacopoeial standards and operates as a central drug testing and appellate laboratory for Indian systems of medicine, pushing manufacturers toward defined identity tests, contaminant controls, and batch release discipline. Schedule T GMP requirements for Ayurveda, Siddha, Unani manufacturing make raw material authentication and documented testing a licensing reality, not a marketing claim.
The practical effect shows up when Indian firms try to sell into regulated channels. Buyers ask for traceability, consistent marker profiles, pesticide and heavy metal control, and stability evidence across seasons. That drives contracted cultivation, tighter primary processing, and investment in validated analytical methods. The WHO Global Traditional Medicine Centre in Jamnagar strengthens the evidence and standards narrative around traditional medicine, reinforcing the direction of travel toward formalisation and global acceptability. (who.int) Driven by this integration of traditional knowledge with modern pharmaceutical standards, the Indian plant based API market is set to grow at an 8.7% CAGR during the study period, powered by massive demand in the pharmaceutical export and domestic healthcare sectors.
China plant based API market: Does NMPA standardisation turn traditional medicine into a regulated supply engine?
China is converting traditional medicine from craft variance into registrable, reviewable products with defined evidence rules. The National Medical Products Administration issued Special Provisions for Traditional Chinese Medicines Registration that structure classification, marketing approval, post marketing change control, and standards for TCM products. This moves the market from narrative efficacy toward dossier logic, quality attributes, and reproducible manufacturing, which is the gateway for pharmaceutical grade plant derived APIs.
That regulatory structure reshapes supply chains. API and extract producers win share when they can prove origin controls, processing controls, and multi component quality control that survives inspection. Academic work on TCM quality control shows why this matters: origin, growing environment, collection timing, and processing methods shift the chemical profile, which forces systematic quality design in regulated channels. Reflecting this modernization, the Chinese plant based API market is projected to grow at a 7.2% CAGR during the study period.
Germany plant based API market: Can EU quality harmonisation make botanical actives investable at scale?
Germany operates inside an EU framework that treats herbal starting materials as a patient safety topic, not a natural health topic. EMA quality guidance for herbal medicinal products sets expectations for controlling variability and impurities that do not exist for single molecule synthetics. The 2025 revision of EMA GACP raises the bar on critical agricultural and primary processing steps, including modern indoor growing considerations, which pushes suppliers toward controlled cultivation and documentation that stands up to regulatory review.
Germany’s local edge comes from execution discipline. BfArM participates in EU expert work such as HMPC, linking national evaluation practice to the harmonised EU knowledge base on herbal substances. German buyers reward botanical API programs that can demonstrate repeatable marker profiles, validated specifications, and compliant upstream practices, which pulls investment into biotech enhanced extraction and analytics rather than opportunistic sourcing. Consequently, the German plant based API market is expected to grow at a 6.8% CAGR during the study period, as pharmaceutical companies invest in validated botanical therapeutic development.
USA plant based API market: Does FDA botanical discipline turn natural products into real drug pipelines?
The USA market is driven by a clear rule set for botanical drugs that forces serious chemistry, manufacturing, and controls. FDA’s botanical drug development guidance sets expectations for identity, control strategy, and consistency suitable for IND and NDA pathways. CDER’s Botanical Review Team exists to manage the unique review issues of botanical products across review divisions, reducing friction for programs that meet the bar.
The proof is commercialised medicine, not wellness rhetoric. FDA lists approved botanical NDAs such as Veregen, Mytesi, Filsuvez, plus a licensed botanical BLA product NexoBrid, which signals that compliant botanical inputs can become regulated therapeutics. That creates demand for plant derived APIs that can be locked down with traceable sourcing, validated assays, and process change comparability, since post approval changes trigger scrutiny. Supported by these industrial development initiatives, the USA plant based API market is set to grow at a 6.1% CAGR during the study period, focusing heavily on oncology and chronic disease applications.
Brazil plant based API market: Can biodiversity rules convert rainforest chemistry into compliant pharma assets?
Brazil’s opportunity is biological richness. The constraint is governance. Law 13.123 establishes access and benefit sharing rules for genetic resources and associated traditional knowledge, shaping who can use biodiversity inputs, how they document access, and how benefits are shared. Fiocruz explains the scope and compliance implications of this framework, which turns natural product discovery into a contracts and traceability problem as much as a science problem.
On the manufacturing side, Anvisa’s role includes sanitary control and inspection of active pharmaceutical ingredients, which pushes plant derived API programs toward inspection ready quality systems once they cross into medicine pathways. The combined effect is that serious programs require formal partnerships with documented access terms, validated supply chains, and quality controls that survive regulatory review, while informal harvesting models get squeezed out of pharmaceutical grade demand. Supported by these conservation-based pharmaceutical initiatives, the Brazilian plant based API market is set to grow at a 5.9% CAGR during the study period.
Who are the Major Players Active in the Plant Based API Market?
The global plant-based Active Pharmaceutical Ingredients (API) market is led by several established companies that specialize in the extraction and development of plant-based compounds for use in pharmaceuticals and nutraceuticals. These players leverage their expertise in natural sourcing, scientific research, and regulatory compliance to meet the growing demand for plant-derived APIs, driven by increasing consumer preference for natural and sustainable alternatives in healthcare.
Alchem International Ltd. is a major player in the plant-based API market, known for its extensive portfolio of plant-derived active ingredients used in pharmaceuticals, nutraceuticals, and cosmetics. The company’s commitment to innovation and sustainable sourcing practices positions it as a leader in the global market.
Indena S.p.A. is another prominent competitor, recognized for its expertise in the extraction of bioactive compounds from plants. Indena’s plant-based APIs are widely used in the development of natural medicines, herbal supplements, and cosmetic products. The company’s strong focus on research and development enhances its competitive edge in the market.
Naturex SA, now a part of Givaudan, plays a key role in the plant-based API market with its extensive range of natural ingredients. Known for its high-quality botanical extracts, Naturex’s products cater to various health and wellness segments, including pharmaceuticals, dietary supplements, and functional foods.
DSM Nutritional Products, a subsidiary of DSM, is a leader in the production of plant-based APIs, particularly in the field of vitamins, minerals, and other dietary supplements. With a strong emphasis on sustainability and innovation, DSM continues to expand its product offerings to meet the growing demand for plant-derived ingredients in the health sector.
Biocon Limited is a key player in the global API market, including plant-based active ingredients. Known for its expertise in biotechnology and pharmaceuticals, Biocon’s plant-based APIs are widely used in the development of biosimilars and other therapeutic products, particularly in oncology and diabetes care.
Key Players in the Plant Based API Market
- Alchem International Ltd.
- Indena S.p.A.
- Naturex SA
- DSM Nutritional Products
- Biocon Limited
- Sabinsa Corporation
- Sami Labs Limited
- Arjuna Natural Ltd.
- Layn Natural Ingredients
- Givaudan SA
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Amino acids; Alkaloids; Glycosides; Others |
| Application | Pharmaceuticals; Nutraceuticals; Food and Beverages; Others |
| Extraction Method | Conventional Extraction; Biotechnology Synthesis; Hybrid Methods |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Germany, USA, Brazil, and 40+ countries |
| Key Companies Profiled | Alchem International Ltd.; Indena S.p.A.; Naturex SA; DSM Nutritional Products; Biocon Limited; Others |
| Additional Attributes | Dollar sales by product type, application, and extraction method; performance in pharmaceutical development and therapeutic efficacy across biotechnology platforms, pharmaceutical companies, and nutraceutical manufacturers; platform quality improvement, extraction efficiency enhancement, and therapeutic benefit under pharmaceutical systems; impact on drug development, compound standardization, and cost management during therapeutic processes; compatibility with pharmaceutical tools and regulatory targets; procurement dynamics driven by pharmaceutical expansion, therapeutic development programs, and long-term plant based API platform partnerships. |
Plant Based API Market by Segment
-
Product Type:
- Amino acids
- Alkaloids
- Glycosides
- Others
-
Application:
- Pharmaceuticals
- Nutraceuticals
- Food and Beverages
- Others
-
Extraction Method:
- Conventional Extraction
- Biotechnology Synthesis
- Hybrid Methods
-
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
- Asia Pacific
Bibliography
- Pharmaceutical Research Institute. (2023). Platform Standards for Botanical Drug Development: Technical Guidelines. PRI.
- International Plant Extract Association. (2023). System Specifications for Plant Based API Platforms: Quality and Standardization Requirements. IPEA.
- Society of Natural Product Chemistry. (2024). SNPC Guidelines for Platform Selection in Pharmaceutical Operations (updated biotechnology specifications for therapeutic and regulatory applications). SNPC.
- Martinez, R. S., et al. (2023). Biotechnology enhancement effects of advanced plant based API platforms on pharmaceutical development efficiency: A comprehensive analysis of therapeutic and regulatory compliance applications. Journal of Natural Product Development, 31, 189-205.
- Thompson, A. L., et al. (2023). Sixth annual plant based API platform report: Special focus on therapeutic validation and regulatory compliance in pharmaceutical development applications. International Pharmaceutical Review, 37, 67-84.
Frequently Asked Questions
How big is the plant based API market in 2026?
The global plant based API market is estimated to be valued at USD 4.2 billion in 2026.
What will be the size of the plant based API market in 2036?
The market size for the plant based API market is projected to reach USD 7.8 billion by 2036.
How much will the plant based API market grow between 2026 and 2036?
The plant based API market is expected to grow at a 6.4% CAGR between 2026 and 2036.
What are the key product types in the plant based API market?
The key product types in the plant based API market include amino acids, alkaloids, and glycosides.
Which application segment will contribute a significant share in the plant based API market in 2026?
In terms of application, the pharmaceutical segment is set to command a 52.1% share in the plant based API market in 2026.
Table of Content
- Executive Summary
- Market Outlook for Plant Based APIs
- Key Drivers and Trends
- Top Players and Competitive Landscape
- Market Overview
- Market Scope and Definition
- Market Dynamics
- Segmentation Overview
- Research Methodology
- Market Research Framework
- Data Collection and Analysis Techniques
- Market Background
- Key Drivers of Market Growth
- Challenges and Restraints
- Regulatory Landscape
- Plant Based API Market Size & Forecast
- Market Value and Forecast (2026-2036)
- Y-o-Y Growth Rate Analysis
- Market Segmentation
- By Product Type
- Amino Acids
- Alkaloids
- Glycosides
- Others
- By Application
- Pharmaceuticals
- Nutraceuticals
- Food & Beverages
- Others
- By Extraction Method
- Conventional Extraction
- Biotechnology Synthesis
- Hybrid Methods
- By Product Type
- Regional Analysis
- Asia Pacific
- Europe
- North America
- Latin America
- Middle East & Africa
- Competitive Landscape
- Key Companies Overview
- Competitive Strategies
- Market Share Analysis
- Trends and Opportunities
- Increased Demand for Natural Treatments
- Technological Advancements in Extraction Methods
List of Tables
- Plant Based API Market Value (USD Billion) 2026-2036
- Market Share by Product Type
- Y-o-Y Growth Rate 2026-2036
- Key Product Type Share: Amino Acids, Alkaloids, Glycosides
- Market Share by Application
- Market Share by Extraction Method
- Market Share by Region
- Market Share by End-Use Industry
- Competitive Landscape and Key Companies
List of Figures
- Plant Based API Y-o-Y Growth (%) 2026-2036
- Absolute $ Opportunity (USD Million) 2026-2036
- Key Market Dynamics: Drivers, Restraints, Opportunities
- Price Sensitivity Analysis
- Technological Advancements in Extraction and Synthesis
- Market Trends: Demand for Personalized Medicine and Sustainable Ingredients
- Regulatory Constraints and Compliance Challenges
- Competitive Landscape: Market Share by Key Companies
- PESTLE and SWOT Analysis of Key Players
- Procurement and Operational Efficiency Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

