Demand for Rapid Infuser in USA
Demand for Rapid Infuser in USA Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Demand for Rapid Infuser in USA 2025 to 2035
The demand for rapid infuser in the USA is valued at USD 55.5 million in 2025 and is projected to reach USD 85.2 million by 2035, reflecting a CAGR of 4.4%. Growth is driven by rising procedural volumes in trauma care, emergency medicine, cardiovascular surgeries, and obstetrics where rapid fluid resuscitation is required. Increased preparedness for mass casualty incidents and ongoing hospital investments in advanced critical care equipment further support adoption.
Trolley-mounted devices represent the leading product type due to their ease of mobility, high flow rate capability, and suitability for operating rooms and emergency departments. Their integration with warming systems, automated safety controls, and compatibility with blood components ensures reliable use in time-sensitive resuscitation scenarios. Clinicians prefer systems that reduce air embolism risk and streamline workflow through intuitive interfaces and minimal setup.
The West, South, and Northeast regions lead demand based on large healthcare infrastructure, higher trauma case volumes, and greater capital equipment budgets in tertiary care facilities. Key players include 3M Company, Smiths Medical, ZOLL Medical Corporation, Stryker Corporation, and GE Healthcare. These companies focus on innovation in infusion rate accuracy, device compactness, and interoperability with hospital monitoring ecosystems, alongside strengthening service networks to ensure operational readiness in critical environments.
Quick Stats for USA Rapid Infuser Demand
- USA Rapid Infuser Sales Value (2025): USD 55.5 million
- USA Rapid Infuser Forecast Value (2035): USD 85.2 million
- USA Rapid Infuser Forecast CAGR (2025–2035): 4.4%
- Leading Product Type in USA Rapid Infuser Demand: Trolley-Mounted Devices
- Key Growth Regions in USA Rapid Infuser Demand: West USA, South USA, Northeast USA
- Top Players in USA Rapid Infuser Demand: 3M Company, Smiths Medical, ZOLL Medical Corporation, Stryker Corporation, GE Healthcare
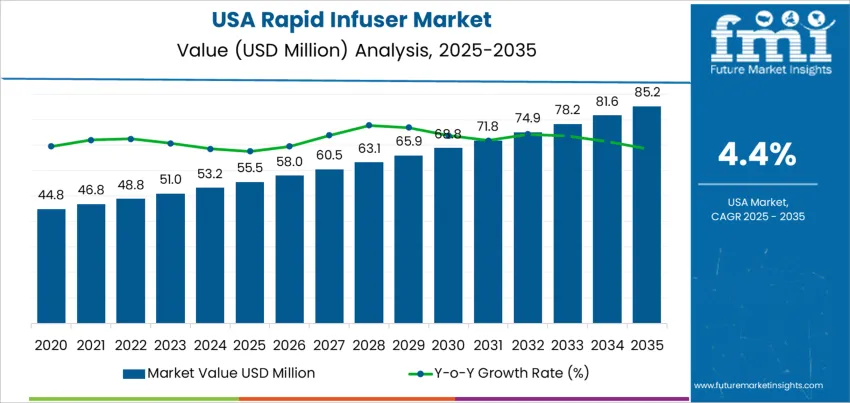
What is the Growth Forecast for Rapid Infuser Industry in USA through 2035?
Demand for rapid infusers in the United States shows a steady upward trajectory when comparing the early and later years of the projected ten-year period. Early growth is influenced by existing adoption in emergency departments, intensive care units, and trauma centers where rapid fluid delivery is already a standard clinical requirement. Hospitals maintain consistent procurement patterns driven by trauma case loads and emergency response capabilities, keeping initial growth stable rather than aggressive.
Later-period gains become stronger as healthcare facilities continue upgrading infusion technologies to improve transfusion speed, patient stabilization, and outcome efficiency. Increased preparedness for large-scale emergencies and mass-casualty events supports more accelerated expansion after the mid-period. The rising burden of critical care interventions, including severe blood loss injuries and surgical complications, reinforces demand in the final years of the timeline. Growth over the decade demonstrates a transition from equipment replacement cycles in the early stage to expanded utilization across more care units in the later stage. The comparison highlights gradual but widening adoption, with the later years contributing a larger share of total incremental demand in the United States.
USA Rapid Infuser Key Takeaways
| Metric | Value |
|---|---|
| USA Rapid Infuser Sales Value (2025) | USD 55.5 million |
| USA Rapid Infuser Forecast Value (2035) | USD 85.2 million |
| USA Rapid Infuser Forecast CAGR (2025-2035) | 4.4% |
Why is the Demand for Rapid Infusers in the USA Growing?
Demand for rapid infusers in the USA is increasing because trauma centers, emergency departments and surgical units frequently treat patients who require immediate fluid or blood replacement. High speed delivery of warmed fluids helps stabilize patients experiencing severe blood loss, shock or major injury. Clinical teams rely on rapid infusers to maintain blood pressure and avoid complications such as hypothermia during transfusions in critical care settings.
Growing surgical volumes and increased emergency visits contribute to consistent usage in operating rooms and intensive care units. Prehospital care providers, including air and ground emergency response teams, also adopt compact rapid infusion systems to support early life-saving intervention before hospital arrival. Advances in device design provide automated flow control, air bubble detection and temperature regulation, which improve safety and reliability for both providers and patients. Hospitals value these capabilities when treating obstetric emergencies, cardiovascular surgeries and trauma incidents. Constraints include high equipment cost for advanced models and training requirements for clinical staff. Smaller facilities may delay upgrades due to budget limitations and lower frequency of critical cases.
Which Product Types, Usability Models, and End Users Lead Demand for Rapid Infusers in the USA?
Demand for rapid infusers in the United States is strengthened by the need for fast and controlled fluid administration in trauma, emergency care, and perioperative procedures. High adoption in critical care units and battlefield medicine supports steady utilization trends. Investments in advanced thermal management, pressure regulation, and battery-powered mobility enhance clinical performance. Growth is further supported by rising surgical volumes, increasing trauma cases, and emergency preparedness measures across civilian and defense healthcare networks.
By Product, Trolley-Mounted Devices Hold the Largest Share
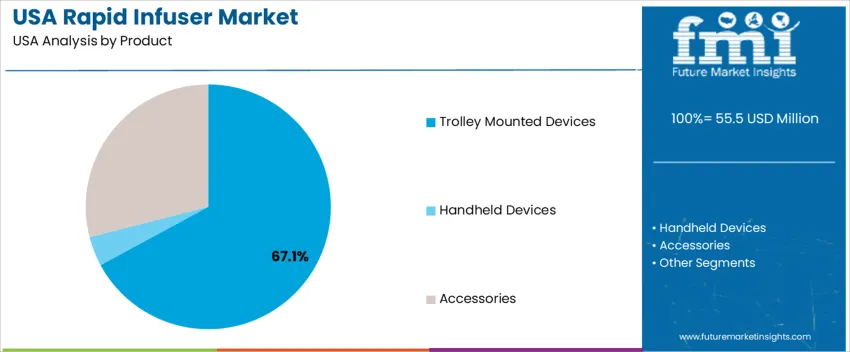
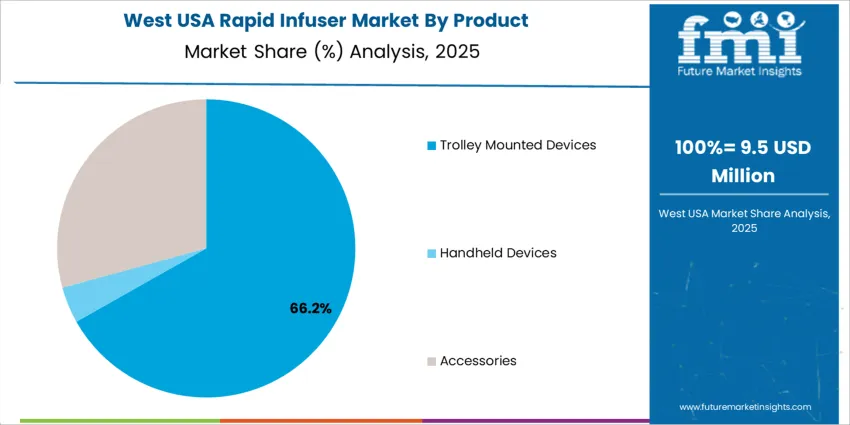
Trolley-mounted rapid infusers account for 67.1%, driven by their widespread use in operating rooms, trauma bays, and intensive care units where continuous high-volume infusion and heating are required. Their stability, integrated controls, and compatibility with large-bore IV access support rapid blood and fluid delivery protocols. Accessories represent 29.0%, including administration sets, pressure bags, and tubing used regularly for device compatibility and replacement needs. Handheld devices hold 3.9%, serving prehospital and ambulatory scenarios requiring lightweight and portable setups. Product selection aligns with acuity levels and the need for precise temperature-regulated infusion in critical medical environments.
Key Points:
- High-acuity settings prefer trolley-mounted units for volume and control
- Accessories support recurring replacement demand
- Handheld devices serve mobility-focused emergency response
By Usability, Reusable Systems Dominate
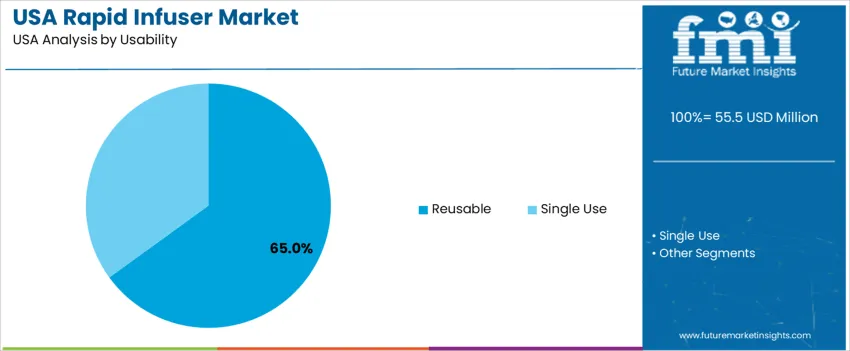
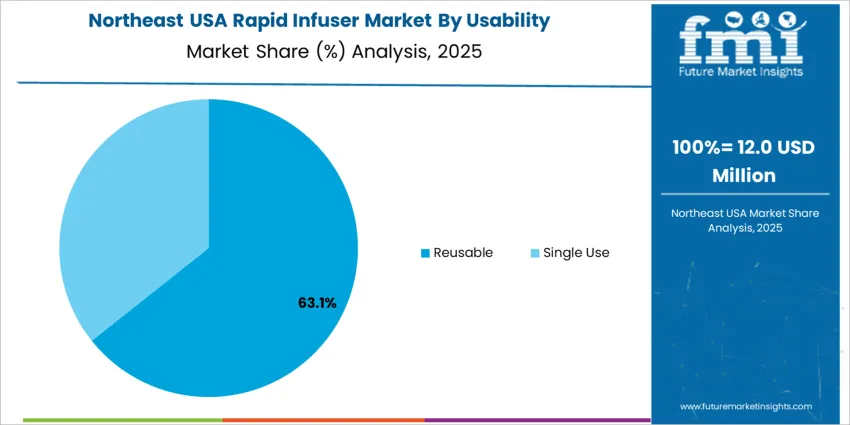
Reusable rapid infusers hold 65.0%, supported by lower lifetime cost and integration into hospital sterilization workflows. Facilities prioritize durability, calibration stability, and long-term reliability across repeated high-pressure cycles. Single-use systems account for 35.0%, used in field operations, infection control scenarios, and community care expansions. Growing focus on contamination prevention and simplified logistics drives adoption in portable care settings. Selection depends on sterilization access, operational mobility, and cost framework across procurement cycles.
Key Points:
- Reusable devices preferred in major hospitals with sterilization capability
- Single-use formats support military and prehospital safety demands
- Cost efficiency influences facility-specific selection
By End User, Hospitals Represent the Largest Share
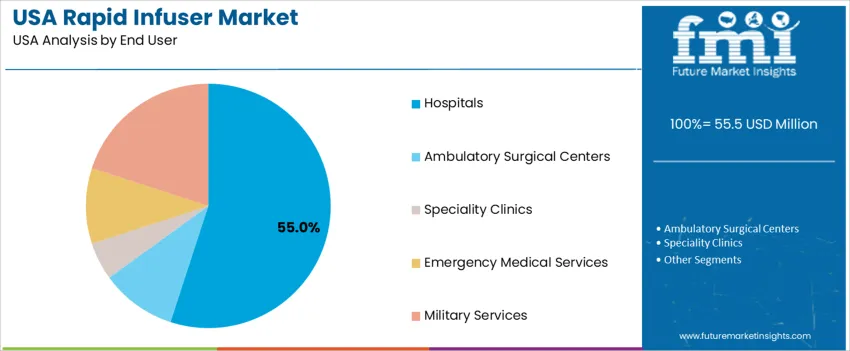
Hospitals contribute 55.0%, reflecting their central role in trauma care, emergency surgery, and transfusion-heavy procedures. Military services hold 20.0%, where rapid infusers are critical for combat casualty care and remote medical operations. Emergency medical services represent 10.0%, requiring portable and ruggedized devices for prehospital stabilisation. Ambulatory surgical centers also account for 10.0%, while specialty clinics hold 5.0%, serving limited infusion needs in controlled settings. End-user demand distribution aligns with varying clinical urgency, patient throughput, and intervention complexity across healthcare environments.
Key Points:
- Hospitals are the primary deployment zone for high-volume cases
- Military forces drive tactical readiness applications
- EMS adoption supports fast-response patient stabilization
What are the Drivers, Restraints, and Key Trends of the Demand for Rapid Infusers in the USA?
Growth of trauma and emergency response demand, increased surgical procedure volumes and improved blood-management protocols drive demand.
In the United States, rapid infusers are essential for delivering warm fluids or blood products in critical situations such as trauma, organ transplantation and cardiovascular surgery. Emergency departments, trauma centers and air-medical services rely on these devices to stabilize patients experiencing severe blood loss. Hospitals with high surgical caseloads require multiple units to support parallel operating-room capacity and reduce delays during complex procedures. National efforts to improve sepsis management and maternal hemorrhage response increase training and procurement of rapid infusion systems in both urban and regional hospitals. Wider adoption of massive transfusion protocols reinforces clinical use and replacement purchases of equipment that supports faster, controlled fluid delivery.
High capital cost, specialized user training and limited deployment in smaller facilities restrain demand.
Rapid infusers require significant investment per unit, and budget-limited hospitals may prioritize other life-support equipment with broader usage. Devices require proper staff training to ensure safe use during fast-paced clinical events, which can be challenging in facilities with lower emergency throughput or frequent personnel turnover. Smaller community hospitals often maintain limited inventories and may rely on manual warming and infusion methods when critical events are less frequent. These constraints reduce uniform adoption and result in concentration of rapid infusers within large trauma systems and academic health networks.
Shift toward portable systems, increased automation in fluid warming and monitoring and rising use in pre-hospital care define key trends.
Manufacturers are developing compact rapid infusers suitable for air-ambulance and emergency-transport teams to enable early intervention before hospital arrival. Automated temperature control and real-time pressure monitoring improve safety and reduce operator workload in high-stress environments. Disposable components tailored for infection-control standards support expanded use in emergency and surgical suites. Hospital systems standardize equipment fleets across multiple campuses to improve staff familiarity and facilitate centralized maintenance. These developments indicate consistent, acuity-driven demand for rapid infusers across the United States healthcare system, strengthening critical-care readiness and trauma response performance.
Analysis of the Demand for Rapid Infuser in the USA by Region
Demand for rapid infusers in the United States is increasing as hospitals prioritize faster fluid resuscitation for trauma, obstetric emergencies, sepsis, and perioperative blood loss. Emergency departments and operating rooms seek devices capable of delivering warmed blood and crystalloids at controlled high flow rates. Integration with massive transfusion protocols, patient-warming strategies, and standardized critical-care pathways shapes purchasing policies. Training, ease of use, and safety features such as air-detection and pressure monitoring remain important. West USA shows the fastest expansion at 5.0% CAGR, followed by South USA at 4.5%, Northeast USA at 4.0%, and Midwest USA at 3.5%, reflecting varying trauma volumes and capital-investment capacity.
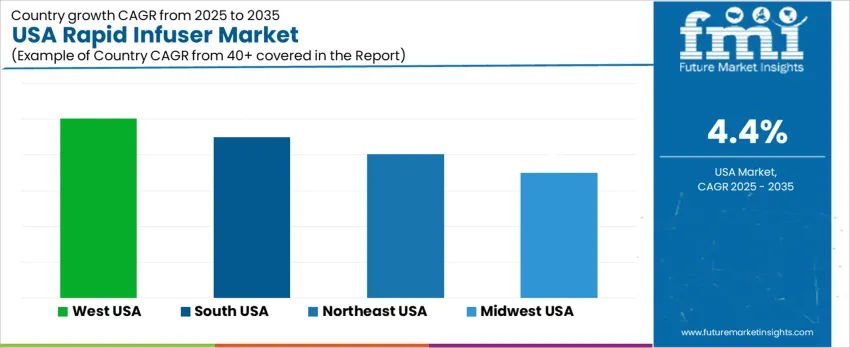
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 5.0% |
| South USA | 4.5% |
| Northeast USA | 4.0% |
| Midwest USA | 3.5% |
How is the West USA driving demand for rapid infusers?
In the West USA, demand for rapid infusers is projected to expand at a 5.0% CAGR, supported by high trauma volumes in California and robust tertiary-care networks across Western states. Level I and Level II trauma centers maintain significant needs for equipment that supports rapid transfusion during multi-system injury and severe hemorrhage. Operating rooms use rapid infusers in complex cardiovascular, transplant, and oncology surgeries where blood loss can escalate quickly. Procurement teams look for interoperability with existing blood-bank workflows and fluid-warming infrastructure. Large health systems emphasize standardized device fleets to simplify staff training and maintenance. Prehospital and air-medical organizations also evaluate compact rapid-infusion solutions for transfer stabilization. Buyers focus on devices with intuitive user interfaces, reliable disposables, and strong post-installation service contracts that minimize downtime.
- Strong trauma-center presence requiring high-throughput resuscitation capability
- Complex surgical programs reinforcing intraoperative rapid-infusion usage
- System-wide standardization improving training and support efficiency
- Service and maintenance responsiveness guiding vendor preference
How is the South USA shaping demand for rapid infusers?
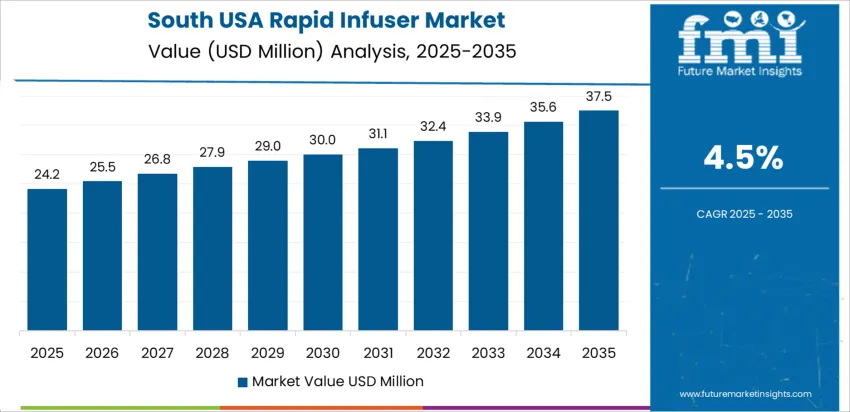
In the South USA, demand for rapid infusers is estimated to grow at around a 4.5% CAGR, driven by large regional trauma populations and extensive emergency-care networks in Texas, Florida, and neighboring states. Hospitals manage high case volumes related to traffic injuries, occupational accidents, and weather-related events, increasing reliance on rapid fluid-resuscitation equipment. Facilities caring for high-risk obstetric populations adopt rapid infusers to support hemorrhage management protocols in labor and delivery units. Many health systems extend device access to satellite hospitals as referral patterns consolidate complex cases into central hubs. Procurement strategies emphasize durable systems that tolerate continuous use in busy emergency departments. Staff education and simulation-based training are prioritized to maintain clinical readiness and adherence to transfusion protocols during peak workloads.
- High emergency-case loads sustaining continuous device utilization
- Obstetric-hemorrhage preparedness increasing demand beyond trauma units
- Regional referral networks distributing equipment across multiple hospitals
- Durability and ease of training shaping equipment selection
How is the Northeast USA influencing usage of rapid infusers?
In the Northeast USA, demand for rapid infusers is expected to rise at a 4.0% CAGR, supported by academic medical centers and large integrated health networks in New York, New Jersey, and Massachusetts. These institutions formalize massive transfusion protocols that specify rapid infuser availability for major trauma, complex surgery, and gastrointestinal bleeding. Emphasis on data-driven quality improvement encourages monitoring of time-to-transfusion metrics and outcomes associated with rapid fluid delivery. Procurement teams evaluate system compatibility with point-of-care warming, pressure limits, and air-elimination safeguards. Smaller community hospitals adopt a limited number of devices to manage infrequent but high-risk emergencies. Training programs ensure cross-coverage capacity among anesthesia, emergency, and critical-care staff, reducing dependence on a small set of super-users.
- Academic centers codifying rapid infuser use in clinical pathways
- Quality metrics driving consistent access in critical units
- Community hospitals maintaining core capacity for rare critical events
- Safety and monitoring features influencing purchasing decisions
How is the Midwest USA contributing to demand for rapid infusers?
In the Midwest USA, demand for rapid infusers is projected to increase at a 3.5% CAGR, reflecting steady but measured adoption across Illinois, Michigan, Ohio, and surrounding states. Regional trauma centers and larger community hospitals maintain rapid infusers primarily in emergency departments, operating rooms, and intensive-care units where high-acuity cases cluster. Rural facilities and critical-access hospitals often rely on shared devices or transfer agreements, limiting broader diffusion. Budget-conscious procurement approaches focus on long device lifespans, straightforward servicing, and predictable disposable costs. Standard operating procedures define when rapid infusion is indicated versus conventional pressure bags or gravity methods. Health systems integrate device competencies into regular education cycles to maintain staff familiarity despite variable event frequency.
- Trauma and surgical hubs anchoring most device placement
- Rural and smaller hospitals adopting targeted, limited deployments
- Cost management influencing device and disposable selection
- Ongoing training sustaining competency for low-frequency, high-risk use
How is competitive positioning structured among rapid infuser suppliers in the United States?
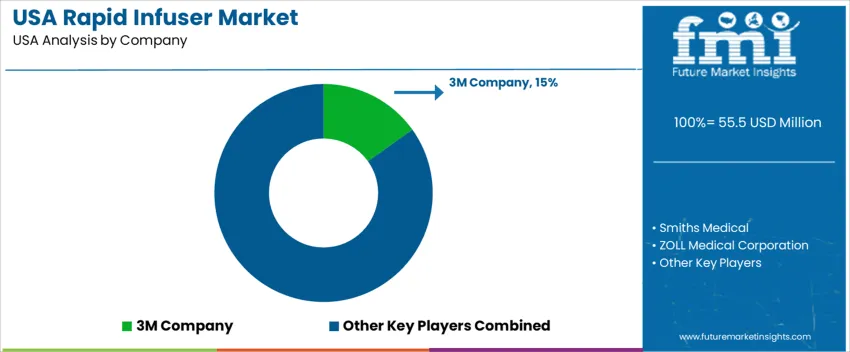
Demand for rapid infusers in the United States is driven primarily by trauma care, emergency medicine, and surgical centers requiring quick-volume fluid resuscitation. Key selection criteria include infusion rate, device reliability under high throughput, compatibility with blood and electrolyte solutions, and support for hospital workflow protocols. Regulatory compliance (FDA, hospital standards) and availability of validated disposables also heavily influence adoption decisions. 3M Company holds an estimated 15.1% share. Its position reflects long-established hospital relationships and a portfolio that includes infusion accessories trusted across critical-care settings. Reliability in supply chains and service support underlie its share.
Smiths Medical competes with integrated infusion-management systems, offering rapid-infuser solutions used in emergency departments and surgical suites. Its breadth across infusion and vascular-access technologies strengthens its acceptance. ZOLL Medical Corporation maintains presence through advanced monitoring and defibrillation systems often bundled by trauma centers; its infusion solutions gain traction where bundled emergency-response capability is preferred.
Stryker Corporation participates in large-hospital procurement, mainly in settings combining patient support equipment and fluid-management devices contributing to adoption when broader capital-equipment contracts are in place. GE HealthCare serves major hospital networks needing rapid infuser deployment across multiple units, leveraging its large-scale equipment relationships to facilitate standardization. Competitive advantage depends on device robustness, supply of sterile disposables, integration with emergency workflows, and scalable support. Suppliers combining these capabilities retain stronger positioning in the USA rapid-infuser segment.
Key Players in USA Rapid Infuser Demand
- 3M Company
- Smiths Medical
- ZOLL Medical Corporation
- Stryker Corporation
- GE Healthcare
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product | Trolley Mounted Devices, Handheld Devices, Accessories |
| Usability | Reusable, Single Use |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Emergency Medical Services, Military Services |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | 3M Company, Smiths Medical, ZOLL Medical Corporation, Stryker Corporation, GE Healthcare |
| Additional Attributes | Dollar sales by product category, usability type, and end-user segments; regional adoption influenced by trauma care infrastructure, emergency preparedness, and surgical volume; competitive offerings in handheld vs. trolley-mounted infusion systems; integration with warming systems, high-flow settings, and rapid blood/IV fluid delivery technologies; procurement trends in EMS and military-grade critical care equipment across the USA. |
USA Rapid Infuser Demand by Segments
Product:
- Trolley Mounted Devices
- Handheld Devices
- Accessories
Usability:
- Reusable
- Single Use
End User:
- Hospitals
- Ambulatory Surgical Centers
- Speciality Clinics
- Emergency Medical Services
- Military Services
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Frequently Asked Questions
How big is the demand for rapid infuser in USA in 2025?
The demand for rapid infuser in USA is estimated to be valued at USD 55.5 million in 2025.
What will be the size of rapid infuser in USA in 2035?
The market size for the rapid infuser in USA is projected to reach USD 85.2 million by 2035.
How much will be the demand for rapid infuser in USA growth between 2025 and 2035?
The demand for rapid infuser in USA is expected to grow at a 4.4% CAGR between 2025 and 2035.
What are the key product types in the rapid infuser in USA?
The key product types in rapid infuser in USA are trolley mounted devices, handheld devices and accessories.
Which USAbility segment is expected to contribute significant share in the rapid infuser in USA in 2025?
In terms of USAbility, reusable segment is expected to command 65.0% share in the rapid infuser in USA in 2025.
Table of Content
- Executive Summary
- USA Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- USA Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- USA Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2025 to 2035
- Trolley Mounted Devices
- Handheld Devices
- Accessories
- Y to o to Y Growth Trend Analysis By Product , 2020 to 2024
- Absolute $ Opportunity Analysis By Product , 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Usability
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Usability, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Usability, 2025 to 2035
- Reusable
- Single Use
- Y to o to Y Growth Trend Analysis By Usability, 2020 to 2024
- Absolute $ Opportunity Analysis By Usability, 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Ambulatory Surgical Centers
- Speciality Clinics
- Emergency Medical Services
- Military Services
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Usability
- By End User
- Competition Analysis
- Competition Deep Dive
- 3M Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Smiths Medical
- ZOLL Medical Corporation
- Stryker Corporation
- GE Healthcare
- 3M Company
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: USA Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: USA Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 3: USA Market Value (USD Million) Forecast by Usability, 2020 to 2035
- Table 4: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 5: USA Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: USA Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 7: USA Market Value (USD Million) Forecast by Usability, 2020 to 2035
- Table 8: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: USA Market Pricing Analysis
- Figure 2: USA Market Value (USD Million) Forecast 2020-2035
- Figure 3: USA Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 4: USA Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 5: USA Market Attractiveness Analysis by Product
- Figure 6: USA Market Value Share and BPS Analysis by Usability, 2025 and 2035
- Figure 7: USA Market Y-o-Y Growth Comparison by Usability, 2025-2035
- Figure 8: USA Market Attractiveness Analysis by Usability
- Figure 9: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 11: USA Market Attractiveness Analysis by End User
- Figure 12: USA Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: USA Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 14: USA Market Attractiveness Analysis by Region
- Figure 15: USA Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: USA Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: USA Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 18: USA Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 19: USA Market Attractiveness Analysis by Product
- Figure 20: USA Market Value Share and BPS Analysis by Usability, 2025 and 2035
- Figure 21: USA Market Y-o-Y Growth Comparison by Usability, 2025-2035
- Figure 22: USA Market Attractiveness Analysis by Usability
- Figure 23: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 24: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 25: USA Market Attractiveness Analysis by End User
- Figure 26: USA Market - Tier Structure Analysis
- Figure 27: USA Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

