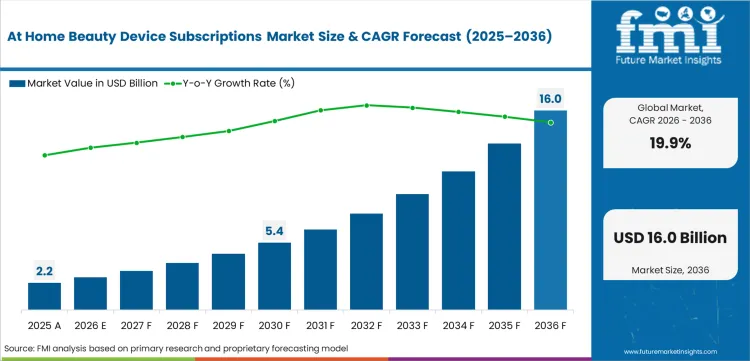
The At Home Beauty Device Subscriptions Market registered USD 2.14 billion in 2025, and is estimated to reach USD 2.68 billion in 2026 at a CAGR of 19.9%. Forward projections indicate a surge to USD 16.54 billion by 2036 as IoT-enabled skin diagnostics transform aesthetic hardware into real-time data interfaces.
According to FMI's estimates, D2C platform operators are respecifying application programming interfaces to support complex beauty subscription tiers that unlock premium treatment algorithms based on monthly fee structures. This architectural pivot eliminates the static functionality of older aesthetic tools. Biometric software architects who secure medical validation for their connected vanity applications dictate the baseline features that all subsequent entrants must match to achieve platform visibility. Suppliers that fail to establish native connected capabilities surrender recurring revenue to digitally native disruptors before the Q4 retention cycle opens.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.68 Billion |
| Industry Value (2036) | USD 16.54 Billion |
| CAGR (2026-2036) | 19.9% |
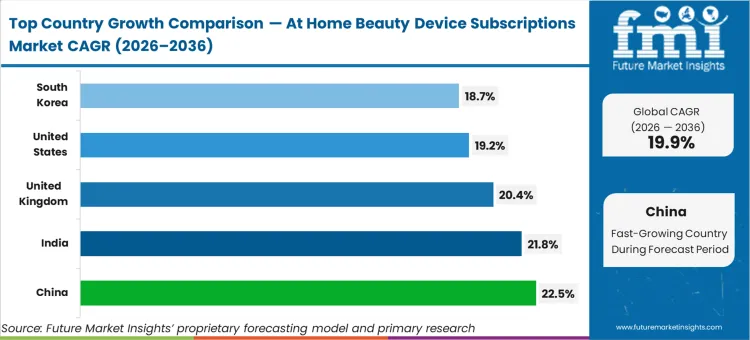
Emerging Asian markets navigate this transition with unprecedented speed, as China sets the pace with a 22.5% CAGR, followed closely by India at 21.8%, with both nations utilizing greenfield e-commerce integrations to bypass traditional physical retail distribution entirely. The United Kingdom advances at a 20.4% rate, supported by structural backlogs in public dermatology pathways that force consumers toward private clinical-grade digital interventions. The United States registers a 19.2% expansion driven by the FDA's software precertification frameworks, while South Korea tracks at 18.7% as established dermatology conglomerates extend their physical clinic presence into the home via SaaS-locked premium hardware.
At Home Beauty Device Subscriptions represent the recurring revenue business model and digital infrastructure where consumers pay periodic fees to access connected dermatological hardware and integrated biometric software. The ecosystem encompasses specialized diagnostic scanners, app-locked microcurrent tools, and serialized LED therapy masks that rely on active digital memberships to unlock professional treatment parameters. A core inclusion is the closed-loop delivery of proprietary consumables, such as conductive serums, which are algorithmically tracked and automatically replenished to ensure optimal device conductivity. Regulatory frameworks such as the FDA's Digital Health Software Precertification Program define the operational parameters for the software-as-a-medical-device platforms governing these precise treatment regimens.
The market scope includes IoT-enabled aesthetic hardware, network interface applications, software-as-a-service platforms, and embedded communication modules featuring native diagnostic capabilities designed for home use. Replenishment-linked serum cartridges, lease-to-own medical-grade laser memberships, and subscription-gated personalized treatment algorithms fall entirely within the boundaries. A concrete inclusion is a connected light-therapy mask that functions physically but requires a paid monthly app membership to activate specific wavelength sequences for acne management; both the hardware contract and the recurring software revenue are fully incorporated into the valuation.
Standard commercial aesthetic hardware lacking consumer-facing mobile synchronization or diagnostic tracking capabilities is excluded. Purely cosmetic topical products, standalone clinical machines designed solely for professional medical-spa deployment, and unlinked consumer gadgets fall outside the defined parameters. A borderline example explicitly omitted is a high-end microcurrent device sold with a one-time purchase model that includes a free, static instructional app; because it lacks a recurring payment structure or software-locked functionality, it does not qualify as a subscription ecosystem.
Subsidized Hardware Models Dictate the Migration Timeline for Premium Therapeutics
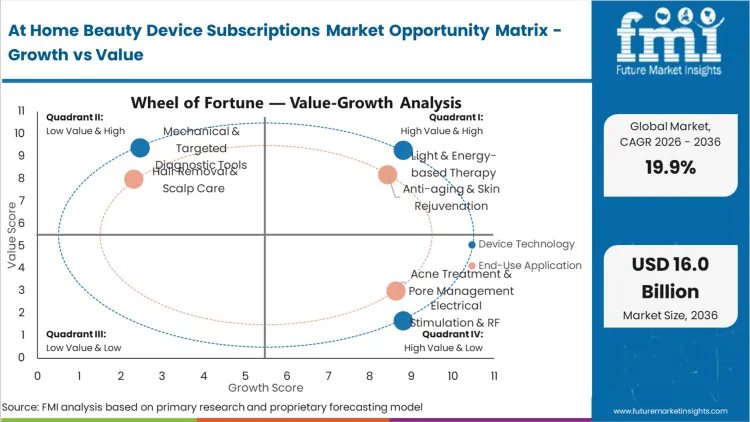
With high upfront capital costs proving inadequate for mass-market clinical tool adoption, D2C platform operators execute full-scale subsidized hardware deployment programs. Replenishment-linked Device Kits capture a dominant 46.8% share in 2026, reflecting the absolute requirement for specialized consumable serums to operate advanced wearable beauty masks safely. As per FMI's projection, this commercial integration eliminates the friction of large initial investments while locking the user into a proprietary subscription economy loop. Platform architects specifying these bundled ecosystems streamline their logistics networks and significantly reduce customer acquisition costs over the device lifetime. Diagnostic software designers failing to demonstrate seamless consumable tracking algorithms lose priority status in critical retail pharmacy upgrade cycles.
Light and Energy Delivery Systems Force Rigorous Software Safety Gates
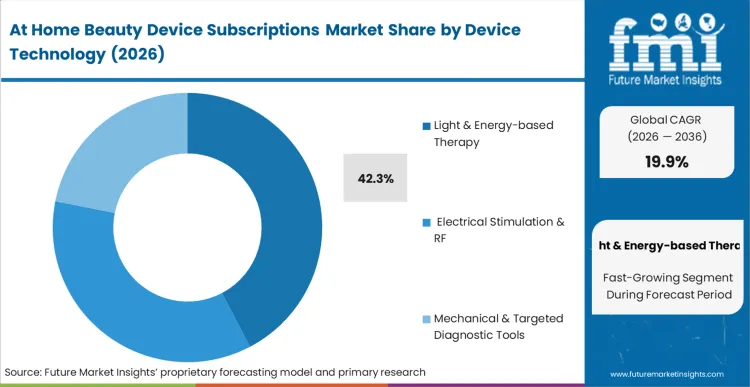
Every biometric software architect bidding on connected vanity platforms now faces strict adherence criteria for clinical energy dispersion limits. Based on FMI's assessment, Light & Energy-based Therapy modules command 42.3% share in 2026. Cosmetic dermatologists advising remote patients reject at home laser device designs that introduce uncalibrated pulse variations into the treatment protocol. Incorporating certified optical control modules guarantees that high-intensity photonic commands arrive exactly as specified by the app-based algorithmic guide. The convergence of smartphone camera diagnostics with core microcurrent facial calibration amplifies the demand for high-bandwidth deterministic links between phone and tool. Device engineers unable to supply robust biometric feedback safety switches risk exclusion from the next wave of greenfield telemedicine integration requests for proposals.
Continuous Cellular Maintenance Outpaces Episodic Acne Intervention Models
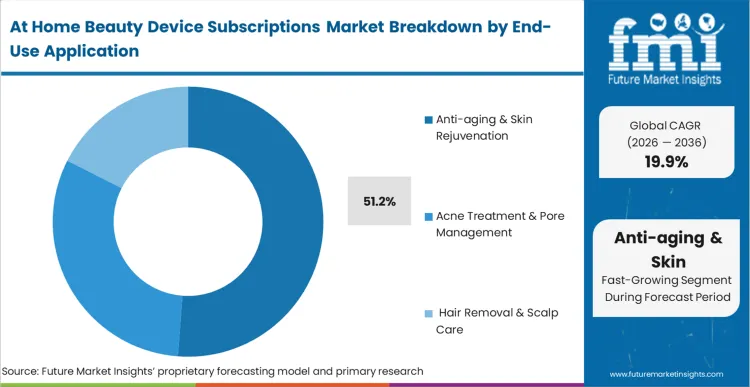
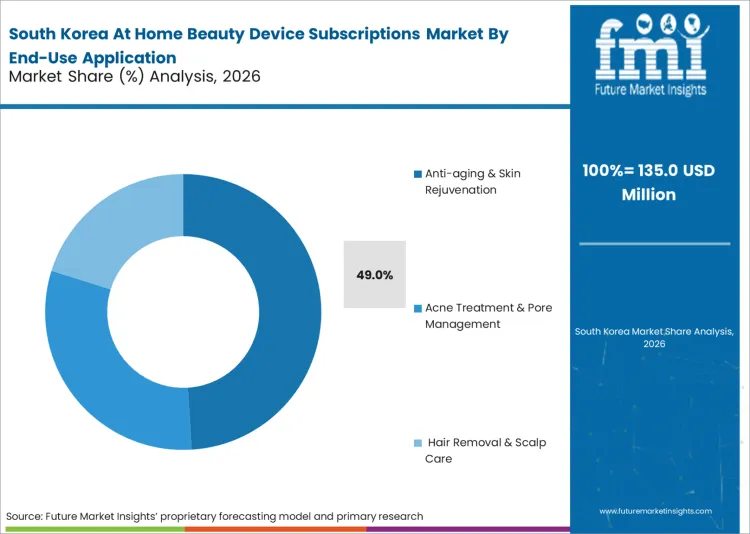
Anti-aging & Skin Rejuvenation emerges as the dominant application area, expected to represent 51.2% of total market share in 2026, as older demographic cohorts recognize the biological necessity of continuous stimulus. FMI's analysis indicates that flattening the clinical architecture directly enables real-time personalized beauty devices to manage collagen degradation on a daily rather than quarterly basis. Cosmetic dermatologists validate these continuous anti aging devices to authorize final post-procedure maintenance sign-offs. The integration of high-frequency home routines across isolated clinical procedures forces retail category managers to rethink traditional episodic skincare sales. Contract manufacturers that fail to develop precision micro-pulsation capabilities forfeit clinical validation credibility in the hyper-competitive connected vanity space.
The convergence of mobile camera diagnostics and therapeutic at home therapeutic beauty devices forces beauty tech integration engineers to extract actionable biometric data directly from the user's bathroom. This architectural requirement renders fragmented, unlinked aesthetic gadgets obsolete. D2C platform operators upgrading digital interfaces face a strict binary choice between deploying complex custom computer vision models or licensing established AI dermatologist engines. Transitioning to a unified diagnostic backbone simplifies the treatment recommendation engine and enables direct cloud connectivity for advanced longitudinal analytics. Facilities that fail to modernize their core evaluation layers risk structural margin collapse as hardware commoditizes.
The intricate configuration parameters required to optimize secure personal health information flows create steep learning curves for traditional cosmetic brands. FMI analysts opine that designing conflict-free data transmission architectures across multi-vendor hardware demands specialized cryptographic expertise that most beauty conglomerates lack internally. Retail category managers attempting to orchestrate unified consumer profiles discover that competing subscription ecosystems actively block API sharing, fragmenting the user's diagnostic history. To mitigate this skill gap, capital project directors increasingly rely on advanced middleware providers that automate the compliance process and validate data privacy designs before physical product deployment.
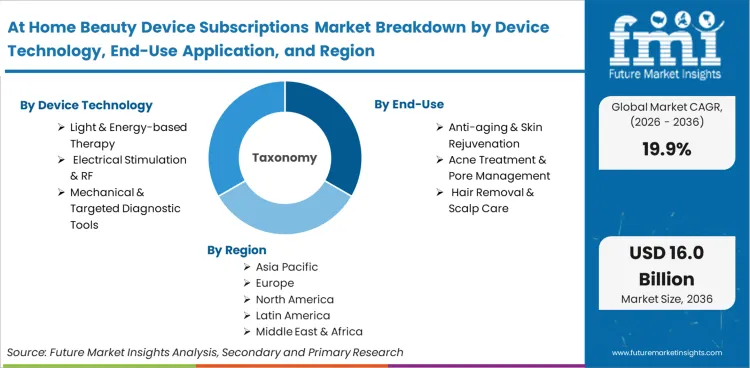
Based on the regional analysis, the At Home Beauty Device Subscriptions market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 22.5% |
| India | 21.8% |
| United Kingdom | 20.4% |
| United States | 19.2% |
| South Korea | 18.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Greenfield digital infrastructure development across the Asia Pacific region accelerates the bypass of legacy retail cosmetic constraints. D2C platform operators constructing new super-app ecosystems specify unified diagnostic algorithms in their initial consumer funnels. As FMI's research confirms, this clean-slate approach entirely eliminates the costly physical footprint phase that plagues older western beauty distribution models. By building native AI capabilities into the foundation, regional brands establish highly flexible engagement environments capable of rapid algorithm retooling.
FMI's report includes detailed analysis and country-wise assessment for the Asia Pacific region. The coverage spans Japan, Indonesia, and Australia. A primary structural condition across these nations involves the rapid deployment of cross-border data privacy localization mandates, forcing cosmetic companies to establish distinct national cloud infrastructures to legally process biometric facial scans.
Stringent medical device classifications across North America target the systematic eradication of unsubstantiated clinical claims in the consumer vanity sector. Biometric software architects leading platform overhauls face strict directives to unify their diagnostic software under regulated software-as-a-medical-device frameworks. FMI's proprietary forecasting model projects that venture-backed digital health entities actively drive this consolidation to secure their recurring revenue streams against sudden regulatory reclassification. Implementing a standardized, FDA-cleared backend enables brands to deploy advanced prescriptive algorithms without compromising legal compliance.
FMI's report includes comprehensive evaluation of the North American digital wellness sector. It features specific analysis of the Canadian and Mexican subscription markets. A defining dynamic in these countries involves the alignment of digital health reimbursement codes, which increasingly dictate whether specific connected therapeutic memberships can be subsidized through consumer health savings accounts.
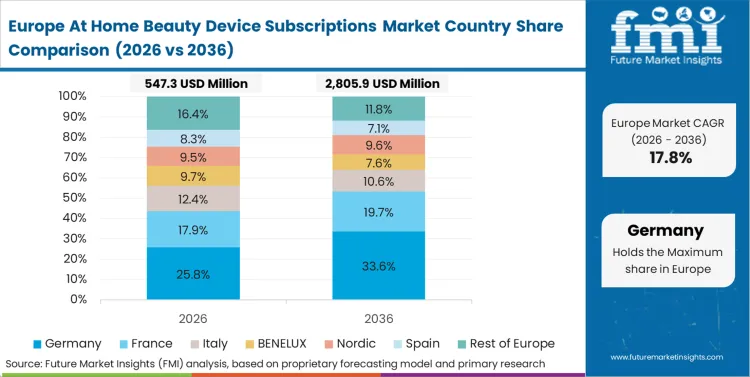
European digital health policy actively penalizes the unauthorized cross-border transmission of unanonymized facial diagnostic data. Biometric software architects redesigning app infrastructures must integrate continuous local-device processing capabilities to satisfy the General Data Protection Regulation requirements for biometric information. This compliance requirement forces the rapid adoption of edge-computing chips within the subscription hardware itself to guarantee analysis stability without relying on external cloud processing. The transition requires a complete overhaul of existing communication topologies, shifting from centralized servers to decentralized, app-based architectures.
FMI's report includes thorough investigation of the European connected aesthetic framework. The analysis encompasses Germany, France, Italy, and the Nordics. A prevailing structural condition across these nations is the mandatory compliance with the EU Artificial Intelligence Act, forcing biometric software architects to expose the decision-making logic of their skin-analysis algorithms to independent third-party regulatory audits.
The introduction of unified biometric privacy standards under the European Artificial Intelligence Act forces D2C platform operators to completely restructure their algorithmic data pipelines. Independent device manufacturers that previously relied on black-box diagnostic engines must now implement transparent, verifiable logic chains that explain exactly why a specific treatment intensity was recommended to the subscriber. This compliance burden structurally advantages massive clinical conglomerates possessing the capital to re-engineer their software from the ground up, effectively pricing out smaller hardware startups from launching competitive diagnostic tiers. Biometric software architects who fail to secure algorithmic transparency certifications lose primary placement in heavily regulated retail pharmacy ecosystems before the 2027 enforcement deadlines.
Most venture capital firms evaluate beauty technology by measuring physical hardware unit economics, but the deployment of over-the-air firmware updates proves the actual competitive moat resides entirely in software upgrade cycles. The capacity to push newly discovered radio frequency beauty equipment modulation patterns to existing subscriber devices instantly depreciates the value of static, non-connected competitor tools sitting on retail shelves. This architectural superiority allows connected brands to launch "new" products via digital updates rather than manufacturing new plastic, radically compressing innovation timelines. Retail category managers without active SaaS-enabled hardware portfolios surrender recurring revenue to digitally native disruptors before the Q4 retention cycle.
The normalization of health savings account eligibility for FDA-cleared aesthetic software dictates a mandatory pivot toward clinical interoperability standards across the subscription ecosystem. Establishing direct API connections with regional electronic health record networks enables connected beauty platforms to export verified dermatological progress imagery directly to the user's primary care physician. This institutional integration converts vanity applications into legitimate telehealth nodes, dramatically lowering user churn rates as the subscription becomes a recognized component of formal medical treatment plans. Cosmetic dermatologists who ignore these unified API protocols face structural margin collapse as hardware commoditizes into unlinked, low-margin retail products.
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.68 Billion to USD 16.54 Billion, at a CAGR of 19.9% |
| Market Definition | At Home Beauty Device Subscriptions represent a recurring revenue business model where consumers pay periodic fees to access dermatological hardware, proprietary consumable replenishments, and integrated diagnostic software. |
| Subscription Model Type Segmentation | Replenishment-linked Device Kits, Lease-to-Own / Rental Memberships, Software-as-a-Service (SaaS) Connected Subscriptions |
| Device Technology Segmentation | Light & Energy-based Therapy, Electrical Stimulation & RF, Mechanical & Targeted Diagnostic Tools |
| End-Use Application Segmentation | Anti-aging & Skin Rejuvenation, Acne Treatment & Pore Management, Hair Removal & Scalp Care |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, United Kingdom, United States, South Korea, and 40 plus countries |
| Key Companies Profiled | L'Oréal Group, Nu Skin Enterprises, Inc., Carol Cole Company (NuFACE), FOREO AB, Perfect Corp., CurrentBody, Koninklijke Philips N.V., Home Skinovations Ltd (Silk’n), YA-MAN Ltd., LYMA Life |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of active subscription accounts, applying region-specific adoption curves to project future membership velocity and average revenue per user. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
How large is the demand for At Home Beauty Device Subscriptions in the global market in 2026?
Demand for At Home Beauty Device Subscriptions in the global market is estimated to be valued at USD 2.68 Billion in 2026.
What will be the market size of At Home Beauty Device Subscriptions in the global market by 2036?
Market size for At Home Beauty Device Subscriptions is projected to reach USD 16.54 Billion by 2036.
What is the expected demand growth for At Home Beauty Device Subscriptions in the global market between 2026 and 2036?
Demand for At Home Beauty Device Subscriptions is expected to grow at a CAGR of 19.9% between 2026 and 2036.
Which Subscription Model Type is poised to lead global sales by 2026?
Replenishment-linked Device Kits commands 46.8% in 2026 as D2C platform operators execute full-scale subsidized hardware deployment programs to lock in consumable retention.
How significant is the role of Anti-aging & Skin Rejuvenation in driving At Home Beauty Device Subscriptions adoption in 2026?
Anti-aging & Skin Rejuvenation represents 51.2% of segment share as older demographic cohorts standardize on continuous, high-frequency home treatments to sustain collagen production.
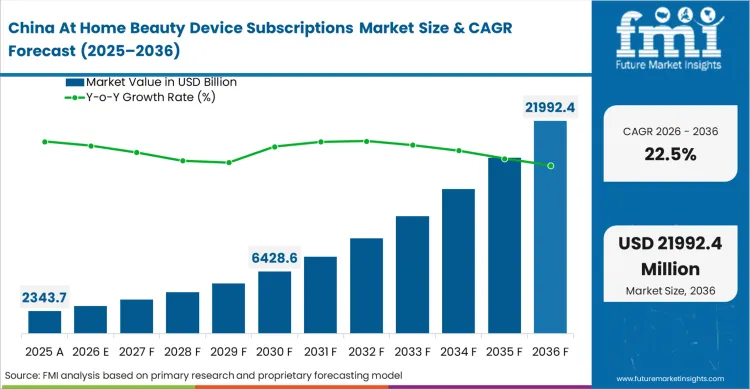
What is driving demand in China?
Total integration of consumer wellness portals into dominant social messaging platforms effectively mandates that all new aesthetic hardware possess native mini-program compatibility.
What compliance standards or regulations are referenced for China?
Tencent and Alibaba's published facial scanning SDKs set the de facto baseline for acceptable diagnostic accuracy across the mainland digital ecosystem.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 22.5% during 2026 to 2036.
Why is North America described as a priority region in this report?
Stringent medical device classifications across North America target the systematic eradication of unsubstantiated clinical claims in the consumer vanity sector.
What type of demand dominates in North America?
Demand heavily focuses on replacing unlinked aesthetic gadgets with standardized, FDA-cleared software-as-a-medical-device architectures that comply with strict biometric data mandates.
What is India's growth outlook in this report?
India is projected to expand at a CAGR of 21.8% during 2026 to 2036.
Does the report cover the United States in its regional analysis?
Yes, the United States is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing the United States?
FDA Digital Health Software Precertification Program guidelines and national biometric privacy regulations form the analytical basis.
What is the main demand theme linked to the United States in its region coverage?
The shift from fighting physical hardware clearance battles toward clearing continuous-update software engines compels the adoption of unified algorithmic ecosystems.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
What is the main United Kingdom related demand theme in its region coverage?
Severe operational backlogs within public dermatology pathways force consumers to adopt private, clinical-grade digital interventions for chronic conditions.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Subsidized diagnostic scanners embedded with localized AI algorithms directly linked to established e-commerce payment ecosystems are critical.
What is At Home Beauty Device Subscriptions and what is it mainly used for?
At Home Beauty Device Subscriptions represent hardware connected to specialized diagnostic apps via recurring payments. They are primarily used to execute and monitor clinical-grade skincare routines, such as microcurrent toning and LED therapy, directly from the home.
What does At Home Beauty Device Subscriptions mean in this report?
The scope encompasses aesthetic hardware, proprietary active serums, and the integrated biometric software required to unlock professional clinical treatment modes.
What is included in the scope of this At Home Beauty Device Subscriptions report?
The market covers IoT-enabled skin scanners, app-locked electrical stimulation tools, and replenishment-linked mask systems featuring native deterministic diagnostic capabilities.
What is excluded from the scope of this report?
Standard commercial aesthetic hardware without consumer-facing mobile synchronization, standalone clinic machines, and downstream finished cosmetics not bundled into a device mechanism are explicitly excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined technology integration and digital payment adoption assumptions for strategic planning purposes.
How does FMI build and validate the At Home Beauty Device Subscriptions forecast?
The model applies a bottom-up methodology starting with global active subscriber metrics and cross-validates projections against publicly reported recurring revenue guidance.
What does zero reliance on speculative third-party market research mean here?
Primary interviews, verified FDA regulatory filings, and official digital health compliance registries are used exclusively instead of unverified syndicated estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.