The non-medical grade aesthetic devices market is poised to reach USD 30.6 billion by 2036, climbing from a 2026 baseline of USD 17.2 billion at a CAGR of 6.5%. As per Future Market Insights, growth is primarily fueled by a paradigm shift where consumers increasingly demand clinical-grade efficacy in home-use formats, effectively bridging the gap between professional dermatology and daily self-care. As per FMI's projection, adoption rates of these sophisticated home-use gadgets will eclipse professional salon equipment sales through 2036.
Nicolas Hieronimus, CEO of L'Oréal, stated, “We are writing the future of beauty and this future is clearly augmented by technology. Future beauty has to be more inclusive, more sustainable, and more personalized.” Manufacturers are increasingly pressured to provide robust clinical evidence as regulators aggressively close gaps between mere toys and medical tools. LED and light therapy segments represent a high-growth frontier, expanding at 13.8% CAGR, more than double the base market rate.
Investors, in 2026, observe that companies capable of navigating these complex compliance landscapes in Asia and North America will secure sustainable competitive advantages.
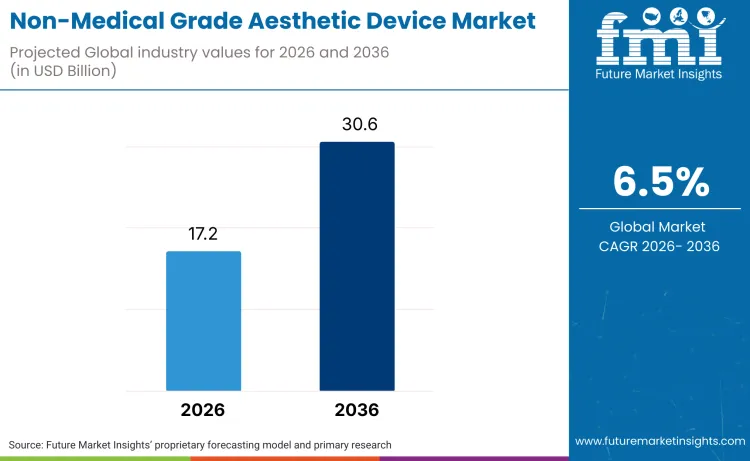
Future Market Insights projects the non‑medical grade aesthetic device market to expand at a CAGR of 6.5% from 2026 to 2036, increasing from USD 17.2 billion in 2026 to USD 30.6 billion by 2036.
FMI Research Approach: Based on FMI’s proprietary bottom‑up forecasting model using shipment and revenue estimation, channel‑wise normalization across online retail, specialty stores, and D2C sales, and exclusion of clinical‑grade or physician‑operated equipment.
FMI analysts perceive the market shifting toward clinical‑at‑home ecosystems, where home‑use devices replicate professional dermatology outcomes using regulated energy delivery and software‑guided routines.
FMI Research Approach: Validated through evidence of regulatory convergence across the US, EU, and China, increasing app‑based treatment guidance, consumer migration from salon procedures to home‑use platforms, and rising requirements for clinical efficacy substantiation.
The United States holds the largest share of the global non‑medical grade aesthetic device market by value.
FMI Research Approach: Derived from FMI’s country‑level revenue modeling, high penetration of premium home‑use devices, strong adherence to FDA consumer laser safety guidelines, and a mature online and D2C beauty‑tech ecosystem.
The global non‑medical grade aesthetic device market is projected to reach USD 30.6 billion by 2036.
FMI Research Approach: Long‑term forecast based on evolving product and distribution mix, accelerated growth of LED/light therapy devices, expansion of connected devices and accessory cycles, and cross‑validation with historical beauty‑tech spending patterns.
The market comprises consumer‑use devices for cosmetic enhancement and personal grooming that do not require medical prescriptions or clinical supervision.
FMI Research Approach: Grounded in FMI’s taxonomy validating inclusion of LED, microcurrent, RF, cleansing, and hair‑removal devices, aligned with cosmetic‑device regulatory frameworks and explicitly excluding prescription lasers and physician‑operated systems.
Key global trends include the medicalization of home beauty, tightening regulatory standards, and a shift from standalone devices toward connected, ecosystem‑based treatment platforms.
FMI Research Approach: Evidenced by China’s NMPA Class III classification for RF home‑use devices, FDA safety reinforcement for consumer lasers, rapid growth of IoT‑enabled beauty devices, and widening gap between verified and unregulated low‑cost products.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 17.2 Billion |
| Industry Value (2036) | USD 30.6 Billion |
| CAGR (2026-2036) | 6.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Rising dermatological prevalence is fundamentally reshaping markets, manifesting as a surge in demand for daily management tools that offer professional-grade results. Connected, guided treatment regimens are rapidly becoming the new standard for users seeking consistent results. FMI analysis suggests that integrating software guidance with personal care appliances increases user retention rates by approximately 40%. Post-pandemic consumer behavior continues to favor preventative self-care, fueling sustained investment in anti-aging technologies.
Energy-based devices command significant market shares, driven by 'clinical-at-home' trends where verified efficacy is paramount. Current segmentation allows consumers to access radio frequency beauty equipment and LED treatments that were previously restricted to clinical settings. Cleansing and microcurrent tools are also expanding rapidly, reflecting demographic shifts toward preventative aging among younger users. FMI analysts opine that regulatory classifications in major economies are creating distinct tiers between medical-grade and purely cosmetic gadgets. Strategic outlooks suggest manufacturers must now choose between mass-market accessibility or premium medical certification pathways to define future market positions.
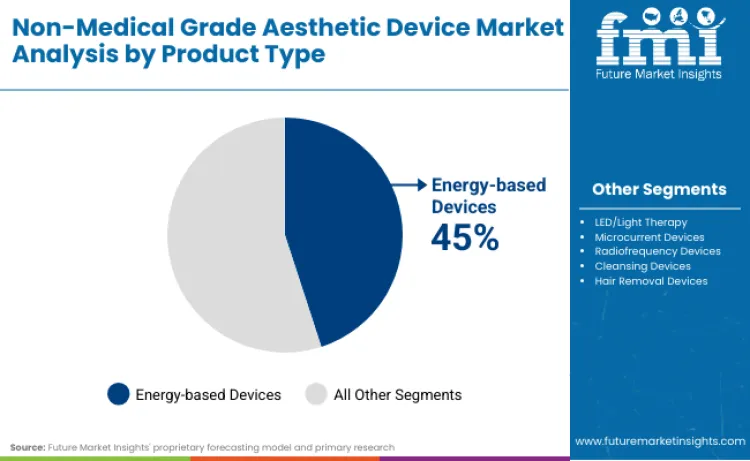
Energy-based devices command a 45% share, driven by consumer desire for professional-grade anti-aging results within home environments. Regulatory shift heavily favors established players like Philips and Panasonic, who possess resources to navigate complex compliance requirements. Clinical efficacy claims are becoming mandatory, compelling brands to invest significantly in trials. Technologies offering tangible collagen stimulation or acne reduction justify higher price points, securing revenue dominance. Consumers increasingly view laser hair removal devices as long-term investments in skin health rather than discretionary beauty spending.
Facial toning segments are expanding at an 11.1% CAGR, reflecting strong uptake within 'preventative aging' demographics. Foreo's expansion into 'preventative' microcurrents specifically targets 25-35 age brackets, a group actively seeking non-invasive alternatives to injectables. NuFACE remains a dominant incumbent in US microcurrent facial spaces, leveraging strong brand equity and FDA clearances to maintain leadership. Usage data indicates high frequency of operation, driving consistent replacement cycles for conductive gels and accessories. Devices offering visible, immediate lifting effects align perfectly with social media trends, fueling viral growth. Innovation in portable, app-connected electric facial cleansing brush tools continues to attract tech-savvy consumers globally.
Regulatory bodies are aggressively closing gaps between 'toy' and 'tool', forcing manufacturers to provide transparency regarding efficacy data. Philips emphasized this shift by highlighting AI-driven diagnostics in its 2024 Future Health Index, moving industry conversations beyond simple mechanics. Tera Peterson, Co-founder of NuFACE, noted that “As we get older, we need to exercise more to keep our bodies toned and tight,” reinforcing the need for active treatment. This medicalization compels brands to invest in clinical trials akin to pharmaceutical standards. Consequently, markets are bifurcating into verified, high-efficacy devices and low-cost, unregulated alternatives, with value rapidly migrating to photorejuvenation equipment.
Devices are evolving into IoT endpoints that collect skin health data to create personalized routines. NuFACE launched its app-connected 'Fitness for Your Face' platform in January 2024, indicating strategic shifts from one-off device sales to subscription-based retention models. Consumers now expect real-time feedback and progress tracking from personalized beauty devices, much like they do from fitness wearables. FMI predicts that data-driven personalization will become the primary mechanism for brand loyalty. Future competitive advantages will likely hinge on the sophistication of accompanying software ecosystems rather than hardware specifications alone.
Growth is pivoting from saturated Western markets to high-potential regions like India and LATAM. Rising disposable incomes in these regions are creating a new class of consumers eager for advanced phototherapy lamps. Manufacturers are thus reorienting supply chains to serve these burgeoning hubs of demand.
Global aesthetic device market reflects a fundamental divergence between developed and emerging economies, with the former prioritizing 'smart' upgrades and the latter witnessing first-time adoption. North America currently holds 45% of market value, driven by and broad tech acceptance. Asia-Pacific serves as the primary volume growth engine, fueled by massive population bases and rising middle-class aspirations. FMI predicts that regulatory harmonization across borders will accelerate global trade flows for at-home laser device manufacturers. Strategic implications involve balancing premium portfolio innovation with accessible entry-level product lines to capture diverse consumer bases.
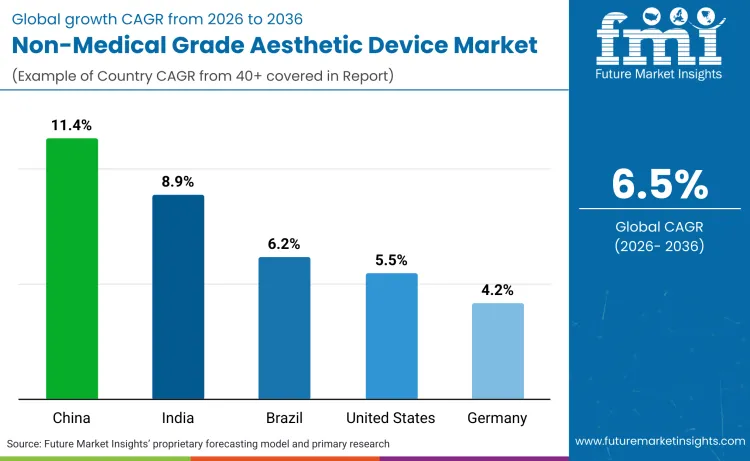
| Country | CAGR (2026-2036) |
|---|---|
| India | 8.9% |
| China | 11.4% |
| Brazil | 6.2% |
| United States | 5.5% |
| Germany | 4.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Sales of non-medical aesthetic devices in India are set to rise at a robust 8.9% CAGR. Growth is catalyzed by 'democratization of dermatology', where rising middle classes gain access to premium care previously out of reach. CDSCO's mandatory Import License requirement, implemented in October 2024, has standardized quality, removing gray-market competitors efficiently. Ministry of Health officials further validated safety protocols for light-based devices in 2024, boosting consumer trust. As per Rami Itani, Director at L'Oréal, “India’s dermatological beauty market has been very dynamic for the past few years and is one of the fastest-growing categories within beauty and personal care”. Such rapid evolution suggests a maturation of consumer palates. India is rapidly transforming into a volume-centric powerhouse, with market valuations expected to exceed USD 2.56 billion by 2030.
Demand for home-use beauty devices in China is anticipated to grow at an 11.4% CAGR. Markets are undergoing 'regulatory shock' phases where strict compliance is redefining competitive hierarchies. NMPA's implementation of Class III medical device classification for RF devices in September 2024 has consolidated market share toward compliant majors. Value growth in China is expected to outpace volume as the market matures into a highly regulated ecosystem.
Aesthetic device industry in Brazil are poised to register a 6.2% CAGR. Brazil remains the aesthetic capital of LATAM, driven by cultures placing immense value on body-focused grooming. Male aesthetics sectors are surging at 13.7% CAGR, creating entirely new demographics for home-use gadgets. Operational efficiencies are evidently paving ways for broader distribution. Operational improvements position Brazil to solidify its dominance as the central hub for LATAM's microdermabrasion devices trade by 2029.
Non-medical grade aesthetic device sector in the United States are forecast to grow at solid rates, maintaining a 45% global share. Drivers include massive shifts toward non-invasive preventative care, with 85% of young adults actively treating acne. Patrick Urban, President of Merz Aesthetics North America, noted “Patients are seeking more sustainable solutions to aging, and regenerative aesthetics treatments have the potential to deliver long-lasting results by working to improve the function of the cells for healthier and more youthful-looking skin”. This strategic focus aligns with demand for cutting-edge solutions. The US industry is thus destined to drive the premium segment, particularly for high-end hair removal wax pen units and laser systems.
Sales of beauty tech in Germany are set to rise at a steady 4.2% CAGR. Germany prioritizes engineering and efficacy, with 70% of consumers considering sustainability a primary purchase factor. EU AI Act regulatory pressures in 2024 and 2025 are actively shaping device software compliance standards. Beiersdorf AG focused heavily on 'digital skin analysis' in Q4 2024 strategies to meet these new demands. Vincent Warnery, CEO of Beiersdorf, explained “We are focusing on our strong brands, delivering breakthrough innovations and expanding into new markets, categories, and channels”. Such clear objectives validate the region's appetite for high-quality skin technology. Germany is therefore emerging as Europe's central 'Quality Control' market, establishing rigorous benchmarks for device durability and efficacy.
The non-medical grade aesthetic device industry is fragmented but consolidating at the top as regulation tightens. FMI observes that Philips and Panasonic leverage global supply chains to dominate 'reliable utility' segments like hair removal. FOREO and NuFACE dominate 'viral social' segments, leveraging heavy influencer marketing to capture youth demographics. M&A activity is accelerating, as seen in L'Oréal's tech-hub investments to secure digital capabilities. Top players control significant shares through early-mover advantages in microcurrent and IPL technologies. Competitive advantages rest on R&D investment and established retail distribution networks that emerging players cannot easily replicate.
Strategies have shifted from selling standalone hardware to building connected ecosystems. Philips emphasized in 2024 that 92% of leaders see automation as critical for health technology. Foreo responded by launching BEAR in July 2024 as a connected skincare regimen rather than just a tool. FMI analysis indicates that competitive escalation suggests future winners will be determined by software quality as much as hardware durability. Companies like ReFa are launching specific LED products to align with new compliance rules.
Recent Developments:
The non-medical grade aesthetic device market represents revenue generated from consumer-use aesthetic devices designed for cosmetic enhancement, skin care, and personal grooming without requiring clinical supervision or medical prescription. As operationally defined in the article, the market measures commercially sold, home-use aesthetic devices that deliver light-based, energy-based, or mechanical cosmetic benefits while remaining outside formal medical device treatment pathways. Market sizing reflects manufacturer and brand revenues from non-medical aesthetic devices sold through retail and direct-to-consumer channels, analysed by product type, distribution channel, end user, and region, and expressed in USD billion.
The market includes non-medical grade aesthetic devices explicitly segmented and quantified in the article, including LED and light therapy devices, microcurrent devices, radiofrequency devices, cleansing devices, and hair removal devices intended for home or personal use. It covers products marketed for skin rejuvenation, acne management, anti-aging, facial toning, hair removal, and general cosmetic maintenance where claims remain cosmetic rather than therapeutic. Revenue generated through online retail platforms, specialty beauty stores, supermarkets and hypermarkets, and direct-to-consumer brand channels is included, with online retail identified as the leading distribution route. The scope counts connected and app-enabled beauty devices, IoT-integrated tools, and software-guided treatment ecosystems sold alongside hardware. Market coverage spans North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa.
The market excludes medical-grade aesthetic devices used in clinics or dermatology practices, including prescription lasers, injectable delivery systems, and physician-operated energy-based platforms. Professional salon equipment requiring licensed operators is excluded unless explicitly sold and certified for consumer home use. Cosmetic consumables such as creams, serums, masks, gels, and topical formulations are not included unless bundled as accessories without revenue attribution. Clinical procedures, aesthetic treatments, service fees, and practitioner revenues are outside scope. Government healthcare spending, regulatory approval fees, public safety programs, and clinical research funding referenced contextually are not counted as market revenue.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 17.2 Billion |
| Product Type | LED/Light Therapy, Microcurrent Devices, Radiofrequency Devices, Cleansing Devices, Hair Removal Devices |
| Distribution Channel | Online Retail, Specialty Stores, Supermarkets/Hypermarkets, Direct Sales |
| End User | Home-Use, Commercial/Salon-Use |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, China, India, Brazil, Japan, and 20+ others |
| Additional Attributes | Dollar sales by product and channel, regulatory-driven adoption analysis (NMPA Class III, FDA Laser Guidelines), connectivity and app-integration assessment, clinical efficacy validation, and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
What is the projected 2036 market size for non-medical grade aesthetic devices?
Global market is projected to reach USD 30.6 billion by 2036, growing from a 2026 baseline of USD 17.2 billion.
Which country is the fastest growing in the aesthetic device sector?
India and China are leading growth, with India's aesthetic device segment growing at 8.9% and home-use niches in China growing at 11.4%.
What are the new regulatory hurdles impacting manufacturers?
Significant barriers include China's NMPA Class III classification for RF devices (Sept 2024) and India's CDSCO Import License mandate.
Which product segment is expected to lead growth?
LED and light therapy segments are projected to expand at 13.8% CAGR due to rising acne prevalence and home-use adoption.
Who are the leading suppliers in the global landscape?
Philips, Panasonic, Foreo, and NuFACE lead markets through pioneering development of connected and compliant home-use therapies.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.