Non-Medical Grade Ophthalmic Device Market
Non-Medical Grade Ophthalmic Device Market Analysis Size and Share Forecast and Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Non-Medical Grade Ophthalmic Device Market Forecast and Outlook 2026 to 2036
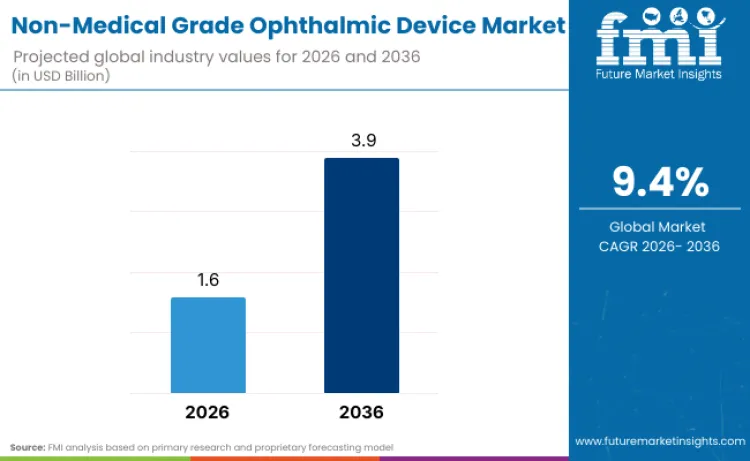
The non-medical grade ophthalmic device market is projected to reach USD 3.9 billion by 2036, rising from USD 1.6 billion in 2026 at a CAGR of 9.4%. Escalating rates of myopia and digital eye strain are fundamentally reshaping industry trajectory. Data from the China National Health Commission (November 2024) reveals that myopia now affects 52.7% of children and adolescents, creating an urgent mandate for preventive solutions. This widespread prevalence forces a shift from reactive vision correction to proactive ocular health management. Manufacturers are responding by integrating advanced technologies into everyday eyewear.
"Our strategic evolution toward med-tech and transformative industries is unlocking cross-sector innovations that will pioneer the future of technology, eye health and human connection." - Francesco Milleri, Chairman and CEO, EssilorLuxottica
As per FMI's projection, the dry eye symptom relief segment will capture substantial value, driven by an aging demographic and increased screen usage. Clinical data cited by Bruder Healthcare (August 2024) indicates that 80 percent of dry eye patients suffer from Evaporative Dry Eye, necessitating accessible home-based interventions. Bruder Healthcare capitalized on this demand by launching Eyeleve MGD in August 2024, a preservative-free solution designed for daily management. Such product evolutions highlight a lucrative opportunity for investors to fund consumer-grade medical devices that bridge the gap between clinical therapy and retail convenience.
Summary of Non Medical Grade Aesthetic Device Market
What Is Growth Outlook for Non‑Medical Grade Aesthetic Device Market as per Future Market Insights Projection?
Future Market Insights projects the non‑medical grade aesthetic device industry to expand at a CAGR of 6.5% from 2026 to 2036, increasing from USD 1.6 billion in 2026 to USD 3.9 billion by 2036.
FMI Research Approach: Based on FMI’s proprietary bottom‑up forecasting model, incorporating device shipment data, revenue estimation, normalization across online retail and D2C channels, and exclusion of clinical‑grade and physician‑operated equipment.
How Do FMI Analysts Perceive Non‑Medical Grade Aesthetic Device Market to Evolve?
FMI analysts perceive the market evolving toward clinical‑at‑home ecosystems, where consumer devices begin replicating dermatology‑clinic outcomes using regulated energy delivery, software‑guided routines, and AI‑enabled treatment personalization.
FMI Research Approach: Supported by evidence of regulatory convergence in the USA, EU, and China, increased app‑based usage tracking, and rising consumer shift from salon procedures to verified home‑use platforms.
Which Country Holds Largest Share in Global Non‑Medical Grade Aesthetic Device Market?
The United States holds the largest share of the global non‑medical grade aesthetic device market by value.
FMI Research Approach: Country‑level revenue modeling, high penetration of premium home‑use aesthetic devices, strong compliance with FDA consumer laser guidelines, and a mature online retail + D2C beauty‑tech ecosystem.
How Large Will Non‑Medical Grade Aesthetic Device Market Be by 2036?
The global non‑medical grade aesthetic device market is projected to reach USD 3.9 billion by 2036.
FMI Research Approach: Long‑term revenue forecasts based on evolving product and channel mix, accelerated adoption of LED and light‑therapy devices, and scaling effects of connected devices and repeat accessory purchases, validated against historical beauty‑tech spending patterns.
What Is Definition of Non‑Medical Grade Aesthetic Device Market?
The market comprises consumer‑use aesthetic devices designed for cosmetic enhancement and personal grooming that do not require medical prescription or clinical supervision.
FMI Research Approach: Based on FMI’s taxonomy validating inclusion of LED, microcurrent, RF, cleansing, and home‑use hair‑removal devices, aligned with cosmetic‑device regulatory categories and explicitly excluding prescription lasers and physician‑operated platforms.
What Are Globally Unique Trends Shaping Non‑Medical Grade Aesthetic Device Market?
Globally unique trends include medicalization of home beauty, increasing regulatory tightening, and the transition from standalone devices to connected treatment ecosystems.
FMI Research Approach: Evidenced by China’s NMPA Class III classification on RF home‑use devices, FDA’s strengthened safety guidance for consumer lasers, and rapid growth of IoT‑enabled aesthetic devices that exceed low‑cost unregulated alternatives.
Non-Medical Grade Ophthalmic Device Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.6 billion |
| Industry Value (2036) | USD 3.9 billion |
| CAGR (2026 to 2036) | 9.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Which Factors Propel Adoption of Non-Medical Grade Ophthalmic Devices?
Strict regulatory policies regarding digital consumption are catalyzing demand for protective solutions. FMI analysts opine that such regulations force parents to invest in preventive hardware like protective eyewear and smart monitoring glasses. Xiaomi validated this structural shift by expanding its AI-glass roadmap in late 2024.
How is the Non-Medical Grade Ophthalmic Device Market Segmented?
Segmentation in non-medical grade ophthalmic device sector is defined by a dichotomy between therapeutic utility for chronic conditions and lifestyle-integrated tech for prevention. Eyelid warming devices and eye massagers address the physiological burden of MGD and digital fatigue, while blue light blocking glasses serve as the primary defense against screen exposure. Vision screening devices are migrating from clinical settings to home use, democratizing access to diagnostics. Meanwhile, smart eyewear represents the convergence point, merging prescription correction with digital utility. FMI analysis indicates that while traditional sunglasses focused on UV protection, the modern landscape prioritizes "med-tech" hybrids. This evolution allows companies to capture value across two distinct user bases: patients managing chronic dry eye and digital natives enhancing productivity. Kids eyewear is similarly evolving from simple correction to integrated myopia management tools.
Why do Eyelid Warming Devices hold a Dominant Share?
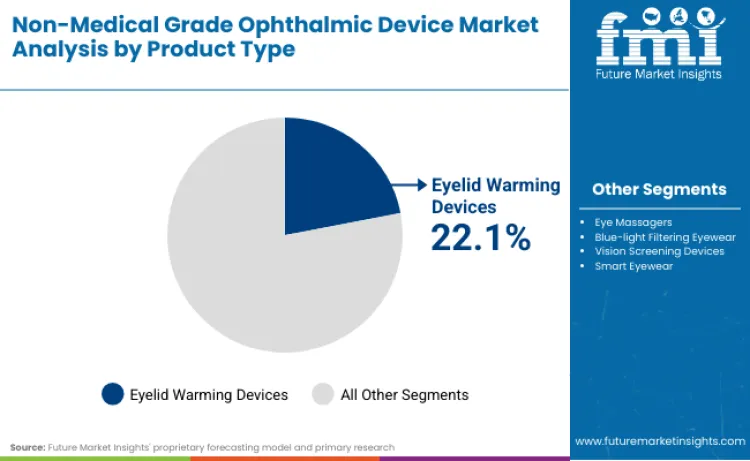
Eyelid warming devices command a leading 22.1% share, driven primarily by the chronic nature of Meibomian Gland Dysfunction (MGD). Clinical evidence confirms that over 80% of dry eye patients suffer from MGD, creating a massive, recurring need for thermal therapy. These devices offer a non-invasive alternative to pharmacological treatments, appealing to long-term sufferers. High efficacy rates in stabilizing tear films ensure consistent patient adherence, thereby securing steady revenue streams for manufacturers focusing on home-based ocular hygiene.
Why does E-commerce lead the Distribution Landscape?
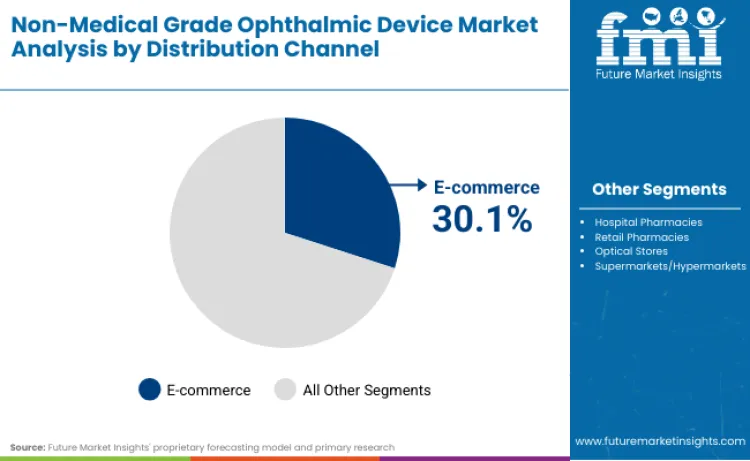
E-commerce channel has secured a leading 30.1% market share, fundamentally disrupting traditional optical retail models. this dominance is driven by the proliferation of virtual try-on technologies and the aggressive direct-to-consumer (DTC) strategies of players like Lenskart and Xiaomi, who are bypassing brick-and-mortar intermediaries. FMI predicts that the convenience of browsing vast inventories of contact lens solution and protective devices from home will continue to appeal strongly to the digital-native demographic. Unlike prescription-heavy medical devices requiring in-person fitting, non-medical grade products face fewer regulatory barriers to online sales, allowing for rapid scaling of cross-border trade.
How is Smart Eyewear gaining Momentum?
Smart eyewear is rapidly expanding beyond novelty, finding legitimate utility in vision correction and health monitoring. Global shipments surged 210% in 2024, driven by seamless integration of audio, AI, and prescription lenses. Xiaomi’s collaboration with GoerTek (Late 2024) to mass-produce AI glasses democratizes access to this technology, challenging the premium positioning of competitors like EssilorLuxottica. Consumers are increasingly viewing these devices as "wearable tech" rather than just medical necessities. This segment attracts significant investment from tech giants, effectively merging the contact lenses logic of correction with digital connectivity.
What Trends Are Shaping Industry Evolution?
- Convergence of Vision and Tech
Manufacturers are integrating audio and AI directly into vision correction frames. EssilorLuxottica extended its partnership with Meta (September 2024) to develop multi-generational smart eyewear. Mark Zuckerberg noted, "We have the opportunity to turn glasses into the next major technology platform." This trend transforms passive lenses into active digital interfaces. As these smart glasses become more mainstream, they may lead to a future where vision correction is seamlessly integrated into daily activities, making life more convenient and connected.
- Digitization of Diagnostics
Landscape of medical diagnostics is rapidly evolving, with a distinct shift from traditional clinics to cloud-connected home devices. By enabling remote monitoring of ocular health through sophisticated technology, patients can now track their conditions from the comfort of their homes. This advancement not only reduces the need for frequent in-person visits, minimizing the burden on healthcare systems, but also leads to better long-term outcomes as data can be gathered continuously and analyzed in real-time.
- Preservative-Free Home Therapy
Demand for chemical-free management of chronic conditions is rising. Bruder Healthcare launched Eyeleve MGD (August 2024), a preservative-free ophthalmic emulsion. Rise of "clean" medical solutions reflects broader consumer preferences for natural and non-toxic options, as individuals seek alternatives that do not compromise their health or well-being. With Eyeleve MGD, users can manage their symptoms confidently, utilizing a product they can incorporate into their daily routines without fear of adverse reactions.
How Will Non-medical Grade Ophthalmic Device Expansion Unfold Across Key Global Regions?
The regional outlook reflects a sharp divergence between emerging economies prioritizing access and developed nations focusing on premiumization. Emerging regions like India and China are witnessing double-digit growth driven by first-time adoption and government health mandates regarding myopia. Conversely, Western markets such as Germany and the USA are characterized by replacement cycles involving high-value "med-tech" devices for aging populations. FMI predicts that this bi-modal growth pattern will compel global manufacturers to maintain dual product portfolios: affordable volume drivers for Asia and sophisticated digital platforms for the West.
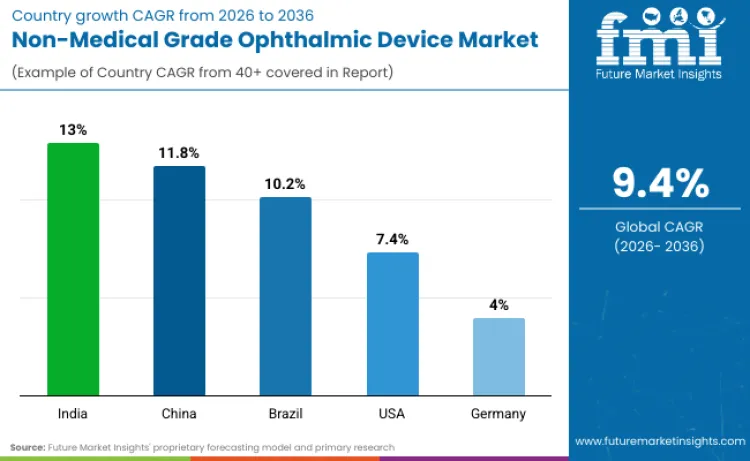
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 13.0% |
| China | 11.8% |
| Brazil | 10.2% |
| USA | 7.4% |
| Germany | 4.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How is the National Programme for Control of Blindness reshaping Indian demand?
Demand for ophthalmic devices in India is projected to expand at a robust 13% CAGR. Growth is catalyzed by the National Programme for Control of Blindness, which targeted 27 lakh surgeries in early 2025 to clear massive backlogs. The Ministry of Health (July 2025) reported over 11.7 million beneficiaries, validating the sheer scale of public health focus. Lenskart filed its RHP for an IPO targeting USD 7.9 billion (October 2025), signaling a massive shift from unorganized to organized retail.
"Going public may be a nice milestone, but it is not the goal. The goal is to build a brand that delivers consistently in India or globally." - Peyush Bansal, Founder and CEO, Lenskart
Strategic maneuvering of this magnitude indicates India is rapidly graduating from a volume-based ecosystem to a value-driven hub. Investors are consequently pivoting to capture this expanding middle-class demographic before market saturation occurs.
Why are myopia prevention mandates driving the China market?
Demand for non-medical grade ophthalmic devices in China are set to rise at an 11.8% CAGR, driven by intense government focus on youth myopia prevention. Xiaomi responded by preparing to launch new AI glasses (Late 2024), directly addressing the tech-savvy urban consumer.
"Everyone wants to be China's answer to Ray-Ban Meta. Xiaomi is gearing up to introduce a new generation of artificial intelligence-powered glasses in collaboration with GoerTek." - Industry Expert, KrASIA
Development of such high-tech compliance solutions suggests the China sector is decoupling from traditional optical models. Domestic tech giants are therefore poised to seize market share previously held by legacy international eyewear brands.
What structural gaps are fueling device adoption in Brazil?
Sales of non-medical grade ophthalmic device in Brazil is anticipated to grow at a 10.2% CAGR, serving as the regional anchor. Regional disparities drive demand; Census data (December 2024) shows ophthalmologist density is high in cities but negligible in rural areas, creating a vacuum for home-use devices. Economic stabilization is clearly empowering Brazilian consumers to prioritize premium ocular health investments. Long-term modernization of the South American optical infrastructure appears inevitable as these consumption patterns solidify.
How does the 'med-tech' shift characterize USA market maturity?
According to FMI's estimates, the USA non-medical grade ophthalmic device sector is projected to expand at a 7.4% CAGR. The high prevalence of Dry Eye Disease, affecting 50 million adults, acts as a primary structural driver. Continued FDA clearance of new categories supports this premiumization trend. Bruder Healthcare’s launch of Eyeleve MGD (August 2024) directly capitalizes on this, offering clinical-grade relief for the aging demographic.
"We are thrilled to introduce Eyeleve MGD to our portfolio of dry eye treatments. Adding Eyeleve MGD strengthens our product line and better equips eyecare professionals." - Brent Jones, Global Head of Vision Care Solutions, Bruder Healthcare
Innovation of this caliber confirms the USA landscape is successfully transitioning from basic correction to comprehensive therapeutic care. Future growth will likely hinge on the seamless integration of diagnostics into consumer retail channels.
Why does precision engineering sustain Germany's market position?
The non-medical grade ophthalmic device sector in Germany is poised to register a 4% CAGR, reflecting a stable, high-tech driven marketplace. The German photonics sector remains robust, with SPECTARIS (October 2025) projecting 7% annual sales growth for the broader industry. Zeiss Meditec’s financial results (December 2025) highlighted Germany as a strong contributor, emphasizing the demand for integrated diagnostic workflows. Sustained focus on precision engineering ensures Germany remains the undisputed quality benchmark for the European continent. Manufacturers here are expected to continue prioritizing high-margin clinical devices over mass-market consumer optics.
What Strategic Moves Are Defining Competitive Landscape?
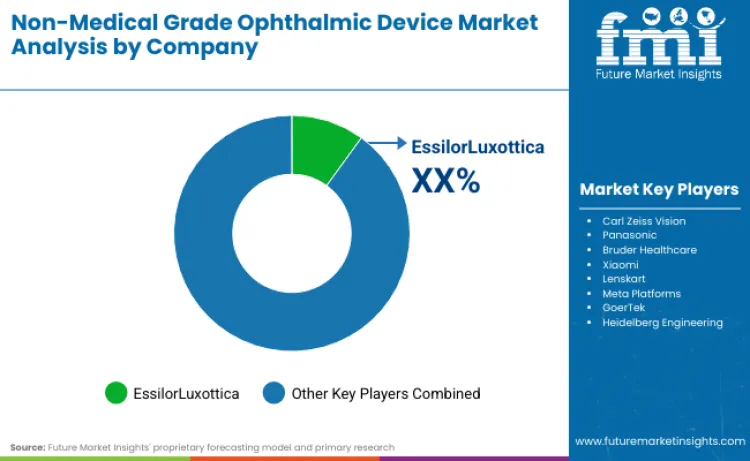
The competitive landscape in non-medical grade ophthalmic device market is defined by the race to dominate "med-tech" therapeutics while defending legacy eyewear franchises. EssilorLuxottica leads with a 14.2% share, leveraging vertical integration of frames, lenses, and new technological capabilities. Market consolidation is accelerating, evidenced by EssilorLuxottica's partnership with Meta (September 2024) to secure smart eyewear dominance. Major players, including Carl Zeiss Vision and Panasonic, control substantial market portions through early-mover advantages in digital optics. Their competitive edge rests on massive R&D investment and established distribution networks that emerging players cannot easily replicate.
Companies are aggressively pivoting toward Direct-to-Consumer (DTC) models and smart integration. Xiaomi (Late 2024) expanded its roadmap for AI glasses, signaling a bold move to challenge traditional eyewear players in the Asian market. Carl Zeiss Meditec responded by integrating the Dutch Ophthalmic Research Center (DORC) to strengthen its surgical and diagnostic footprint. These maneuvers ensure companies remain relevant in a market rapidly moving away from simple vision correction. Patent portfolios and strategic tech alliances are becoming the primary currency for future market leadership.
Recent Developments:
- In August 2024, Advancing Eyecare acquired Eyefficient, a provider of affordable diagnostic ophthalmic instruments. This acquisition expands Advancing Eyecare's portfolio and nationwide distribution, improving access to non-invasive diagnostic tools for eye care professionals.
- In January 2025, Norlase received FDA 510(k) clearance for LYNX, the world’s first battery-powered pattern scanning laser indirect ophthalmoscope. This portable device offers untethered treatment options, enhancing efficiency in ophthalmic practices with its ergonomic design and wireless features.
- In June 2025, Johnson & Johnson Vision Care launched Acuvue Oasys Max 1-Day, the first daily disposable contact lens designed for both astigmatism and presbyopia. This innovation addresses the needs of patients with multiple vision corrections, enhancing comfort and visual clarity in the consumer ophthalmic device segment.
Key Players in Non-Medical Grade Ophthalmic Device Market
- EssilorLuxottica
- Carl Zeiss Vision
- Panasonic
- Bruder Healthcare
- Xiaomi
- Lenskart
- Meta Platforms
- GoerTek
- Heidelberg Engineering
Market Definition
The non-medical grade ophthalmic device market represents revenue generated from consumer-use ocular devices designed to support eye comfort, visual protection, and preventive eye care without requiring medical prescription or clinical supervision. As operationally defined in the article, the market measures commercially sold ophthalmic devices positioned for daily, home, or lifestyle use that address conditions such as digital eye strain, dry eye symptoms, myopia risk mitigation, and visual fatigue, while remaining outside regulated therapeutic or surgical treatment pathways. Market sizing reflects manufacturer and brand revenues from these devices, analysed by product category, distribution channel, and region, and expressed in USD billion.
The market includes non-medical grade ophthalmic devices explicitly referenced in the article, including eyelid warming devices, dry-eye relief devices, eye massagers, blue-light filtering eyewear, protective glasses, vision screening tools for home use, and smart eyewear with non-therapeutic monitoring or digital functionality. It covers products marketed for preventive eye health, comfort enhancement, fatigue reduction, and screen-related ocular protection rather than disease treatment. Revenue generated through e-commerce platforms, direct-to-consumer brands, optical retail chains, and online marketplaces is included, with e-commerce identified as the leading sales channel. The scope also counts connected and AI-enabled eyewear, audio-integrated glasses, and consumer smart vision devices that blend lifestyle technology with ocular wellness. Geographic coverage spans North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa.
The market excludes medical-grade ophthalmic devices such as prescription contact lenses, intraocular lenses, surgical instruments, diagnostic imaging systems, and clinician-operated therapeutic devices. Professional eye care services, ophthalmology consultations, surgeries, and prescription treatments are not included. Pharmaceutical products such as eye drops, medicated ointments, and prescription dry-eye therapies are excluded unless sold strictly as accessories without revenue attribution. Clinical trials, public health programs, reimbursement payments, and regulatory funding initiatives referenced contextually are not counted as market revenue. Devices requiring physician oversight, formal diagnosis, or medical device treatment claims fall outside the scope of the non-medical grade ophthalmic device market.
Scope of Report
| Items | Values |
|---|---|
| Quantitative units (2026) | USD 1.6 Billion |
| Solution Type | Eyelid Warming Devices, Eye Massagers, Blue-light Filtering Eyewear, Smart Glasses, Vision Screening |
| Deployment Model | Home-use, Clinical, Hybrid |
| End User | Dry Eye Patients, Digital Users, Geriatric Population, Kids (Myopia Control) |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, China, India, Brazil, Japan, United Kingdom, and 40+ countries |
| Key Companies Profiled | EssilorLuxottica, Carl Zeiss Vision, Panasonic, Bruder Healthcare, Xiaomi, Lenskart |
| Additional Attributes | Dollar sales by solution type and deployment, regulatory-driven adoption analysis (China Myopia Mandates), supply chain assessment, patent landscape evaluation, and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
Non-Medical Grade Ophthalmic Device Market by Segments
By Product Type:
- Eyelid Warming Devices
- Eye Massagers
- Blue-light Filtering Eyewear
- Vision Screening Devices
- Smart Eyewear
By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Optical Stores
- E-commerce
- Supermarkets/Hypermarkets
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- World Health Organization. (2024). Integrated people-centred eye care: Global implementation report. World Health Organization.
- National Institutes of Health, National Eye Institute. (2024). Vision and eye health surveillance update. USA Department of Health and Human Services.
- Ministry of Education, People’s Republic of China. (2024). National action plan for comprehensive myopia prevention and control among children and adolescents. Government of the People’s Republic of China.
Frequently Asked Questions
What is the current global market size for Non-Medical Grade Ophthalmic Devices?
Global sales are valued at USD 1.6 billion in 2026, with significant expansion driven by med-tech convergence.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
Demand is projected to grow at a 9.4% CAGR from 2026 to 2036.
Which regions are experiencing the fastest expansion?
India leads with a 13% CAGR, followed closely by China at 11.8%, driven by government health initiatives.
What are the primary market drivers?
Digital eye strain regulations and rising dry eye prevalence (50 million U.S. adults) are fueling adoption.
Who are the leading suppliers in the industry?
EssilorLuxottica, Carl Zeiss Vision, and Xiaomi lead the market through pioneering development of smart therapeutic eyewear.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Eyelid Warming Devices
- Eye Massagers
- Blue-light Filtering Eyewear
- Vision Screening Devices
- Smart Eyewear
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacies
- Retail Pharmacies
- Optical Stores
- E-commerce
- Supermarkets/Hypermarkets
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Mexico
- Rest of Latin America
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- France
- Italy
- Spain
- Rest of Europe
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- South Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Rest of South Asia
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- GCC Countries
- South Africa
- Rest of Middle East & Africa
- By Product Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- EssilorLuxottica
- Carl Zeiss Vision
- Panasonic
- Bruder Healthcare
- Xiaomi
- Lenskart
- Meta Platforms
- GoerTek
- Heidelberg Engineering
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Non-Medical Grade Ophthalmic Device Market Key Takeaways (2026 and 2036)
- Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 and 2036
- Non-Medical Grade Ophthalmic Device Market CAGR (%), 2026 to 2036
- Eyelid Warming Devices Share (%), 2026
- E-commerce and Virtual Try-ons Share (%), 2026
- Smart Eyewear Growth Indicator Table (Shipments/Index), 2024 to 2026
- Country-wise CAGR Table (%), 2026 to 2036
- Global Non-Medical Grade Ophthalmic Device Market Size (USD Billion) by Product Type, 2026 to 2036
- Global Non-Medical Grade Ophthalmic Device Market Size (USD Billion) by Distribution Channel, 2026 to 2036
- Global Non-Medical Grade Ophthalmic Device Market Size (USD Billion) by Region, 2026 to 2036
- North America Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- Europe Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- East Asia Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- South Asia Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- Latin America Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- Middle East & Africa Non-Medical Grade Ophthalmic Device Market Size (USD Billion), 2026 to 2036
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Product Type, 2026
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Distribution Channel, 2026
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Region, 2026
- Competitive Landscape Snapshot Table (Selected Players and Positioning)
- Recent Developments Timeline Table (2024 to 2025)
List of Figures
- Non-Medical Grade Ophthalmic Device Market Growth Framework
- Non-Medical Grade Ophthalmic Device Market Value Chain Overview
- Non-Medical Grade Ophthalmic Device Market Product Landscape Map
- Non-Medical Grade Ophthalmic Device Market Distribution Channel Landscape
- Non-Medical Grade Ophthalmic Device Market Opportunity Map
- Non-Medical Grade Ophthalmic Device Market Scenario Forecast Model
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Product Type, 2026
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Distribution Channel, 2026
- Global Non-Medical Grade Ophthalmic Device Market Share (%) by Region, 2026
- North America Non-Medical Grade Ophthalmic Device Market Structure
- Europe Non-Medical Grade Ophthalmic Device Market Structure
- East Asia Non-Medical Grade Ophthalmic Device Market Structure
- South Asia Non-Medical Grade Ophthalmic Device Market Structure
- Latin America Non-Medical Grade Ophthalmic Device Market Structure
- Middle East & Africa Non-Medical Grade Ophthalmic Device Market Structure
- Competitive Landscape Mapping (Key Players)
- Strategic Positioning Matrix (Med-tech Therapeutics vs Lifestyle Prevention)
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

