Chain-of-Custody Packaging Solutions Market
Chain-of-Custody Packaging Solutions Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Chain-of-Custody Packaging Solutions Market Forecast and Outlook 2026 to 2036
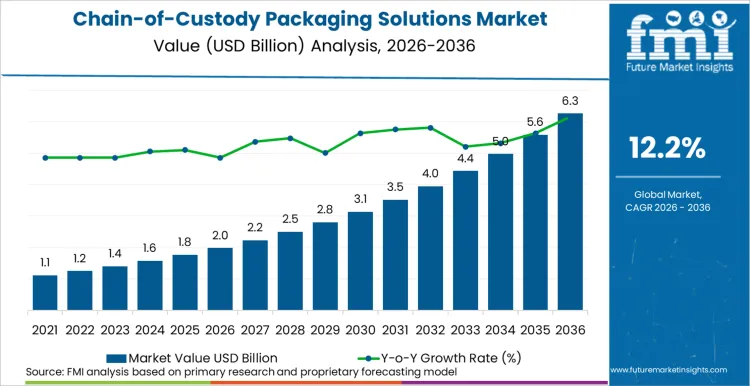
The chain-of-custody packaging solutions market is projected to total USD 1,980 million in 2026, increasing to USD 6,240 million by 2036, at a CAGR of 12.2%. FMI’s analysis indicates the market is undergoing a fundamental shift from standalone secure packaging to integrated platforms that combine specialized materials, unique identifiers, and cloud-based logging. Expansion will be defined by the global enforcement of pharmaceutical serialization mandates and the digitization of legal evidence management, creating compulsory demand for packaging that functions as a secure data conduit.
Growth is anchored in the tightening of liability and compliance standards across regulated supply chains. In pharmaceuticals, clinical research, and forensics, the integrity of an item must be defensible from origin to destination through a documented sequence of custody events. As a result, packaging is no longer treated as a passive container; it functions as a control point for tamper evidence, identification, and auditable handling. This shift from optional security to procedural requirement is accelerating demand for chain-of-custody packaging systems where the package identity and handoff documentation are integral to compliance and risk mitigation.
A verifiable, widely adopted approach in this market is the pairing of tamper-evident closures/seals with unique identifiers such as serialized labels and QR/DataMatrix codes that can be scanned at each transfer. These systems are typically supported by cloud-based event logging that timestamps handoffs and creates a traceable record for audits and investigations. In practice, legal defensibility is achieved less through blockchain by default and more through validated procedural controls, controlled access logs, and standardized documentation requirements across participants.
Technical innovation is centered on packaging formats that can carry reliable identifiers without compromising barrier performance or processability. In regulated pharmaceutical distribution, the most defensible reference point for custody traceability is alignment with established serialization and traceability requirements, particularly those embedded in pharmaceutical supply chain regulations. Packaging suppliers and integrators are therefore emphasizing print quality, code durability, tamper indication, and scan reliability across real logistics conditions, enabling event capture through existing enterprise track-and-trace systems rather than requiring proprietary ledgers.
In Europe, competition is shaped by procedural integration—packaging that supports repeatable, auditable workflows used by evidence handlers, laboratories, and logistics providers. Solutions commonly focus on evidence bags, security mailers, and tamper-evident cartons designed to be scanned at each handoff and reconciled with digital evidence logs. This emphasizes interoperability with the customer’s evidence management environment through standardized identifiers and scanning routines, rather than vendor-specific claims of “pre-integration” with law enforcement platforms.
Across Asia, chain-of-custody packaging demand is growing in export-oriented manufacturing and regulated trade lanes, where buyers require documented integrity assurance. Regional packaging suppliers are expanding security features such as tamper-evident seals, serialized labels, and RFID-enabled tracking options, particularly for high-value electronics and industrial components, while focusing on cost-efficient scalability. These solutions are generally deployed as part of the customer’s broader logistics tracking stack, with packaging acting as the physical anchor for custody events.
Summary of Chain-of-Custody Packaging Solutions Market
- The chain-of-custody packaging solutions market comprises specialized packaging systems designed to provide a documented, auditable record of the possession, handling, and transfer of sensitive or high-value items. These solutions integrate tamper-evident features, unique identifiers, and often digital tracking to maintain an unbroken custody trail for pharmaceuticals, legal evidence, regulated industrial goods, and precision components.
- The defined scope follows FMI taxonomy, covering primary and secondary packaging formats where chain-of-custody tracking is a core design function, including serialized pouches, secure mailers, lockable cartons, and custom boxes with integrated logging technology. It excludes generic packaging and basic tamper-evident solutions without a dedicated custody logging component.
- The market is projected to grow at a CAGR of 12.2% from 2026 to 2036, expanding from USD 1,980 million in 2026 to USD 6,240 million by 2036, based on FMI proprietary modeling incorporating pharmaceutical serialization deadlines, legal procedural digitization rates, industrial compliance audits, and primary interviews with quality assurance and logistics compliance officers.
- The sector is transitioning from simple evidence bags to intelligent custody platforms, with solutions increasingly segmented by legal burden of proof—from FDA-compliant drug sample tracking to courtroom-admissible evidence handling and certified industrial part distribution.
- Competition is concentrated among security packaging specialists and broad-line industrial packaging firms with dedicated track-and-trace divisions, with differentiation driven by the defensibility of the custody protocol, integration with inventory management software, and speed of forensic analysis in case of a breach.
- India records the fastest growth at 15.6% CAGR through 2036, driven by its rapidly expanding GMP-certified pharmaceutical manufacturing base requiring compliant export packaging, while North America remains the highest-value market, shaped by its dense concentration of global pharma HQs, stringent legal discovery rules, and advanced forensic science infrastructure.
Chain-of-Custody Packaging Solutions Market Key Takeaways
| Metrics | Values |
|---|---|
| Expected Value (2026E) | USD 1,980 million |
| Projected Value (2036F) | USD 6,240 million |
| CAGR (2026–2036) | 12.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Chain-of-Custody Packaging Solutions Market Growing?
The global harmonization of pharmaceutical serialization and traceability laws, culminating in the full enforcement of the US Drug Supply Chain Security Act (DSCSA) and the EU Falsified Medicines Directive, is a primary legislative driver. These regulations mandate unit-level tracking, making chain-of-custody packaging not a strategic choice but a legal requirement for market access, creating a vast, captive market for compliant solutions.
The digitization of the legal system is transforming evidence handling. From crime scenes to corporate litigation, the demand for a digitally documented, unbroken chain of custody is paramount to ensure evidence admissibility. Packaging systems that automatically generate a digital audit trail with each opening or transfer are replacing paper logs, reducing human error and providing stronger defensibility in court, driving adoption across law enforcement and legal firms.
The rise of responsible sourcing and ESG-linked industrial contracts is creating demand in non-traditional sectors. Manufacturers of conflict minerals, sustainable commodities, or aerospace components must provide verifiable proof of ethical sourcing and handling to secure contracts. Chain-of-custody packaging provides the physical-digital link to certify that materials have not been tampered with or substituted during transit, turning packaging into a certificate of authenticity and compliance.
How is the Chain-of-Custody Packaging Solutions Market Segmented?
The market is segmented by the primary application, the packaging format that best enables custody tracking, and the material offering the optimal balance of security and functionality. The pharma & forensics end-use segment dominates with a 46% share, as it represents the most regulated and liability-intensive applications. Track-and-trace pouches lead the format segment (38%) due to their suitability for unit-level serialization and efficient handling of small, high-value items.
How is the Dominance of Polymer Films Sustained in Critical Tracking Applications?
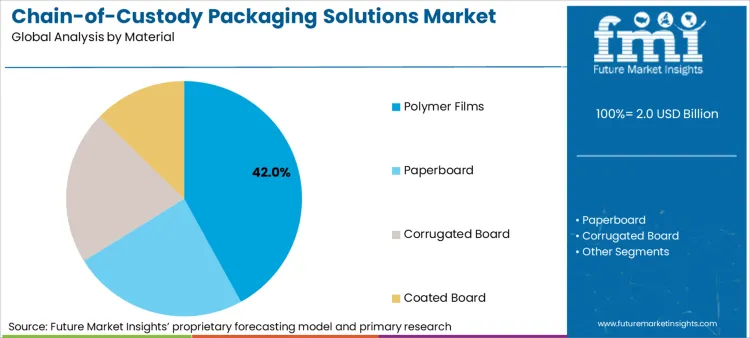
Polymer films account for a leading 42% material share, primarily due to their superior compatibility with direct-print serialization technologies, transparency for content inspection without opening, and ability to form high-integrity tamper-evident seals.
For pharmaceutical blisters, clinical trial kits, and forensic sample bags, polymer films allow for the clear printing of unique 2D codes, lot numbers, and handling instructions while providing a barrier against contamination. The material's flexibility also enables the creation of pouches that clearly exhibit any breach attempt, which is a fundamental requirement for legal and regulatory custody protocols.
Which Regulatory and Liability Factors are Concentrating Value in the Pharma & Forensics Segment?
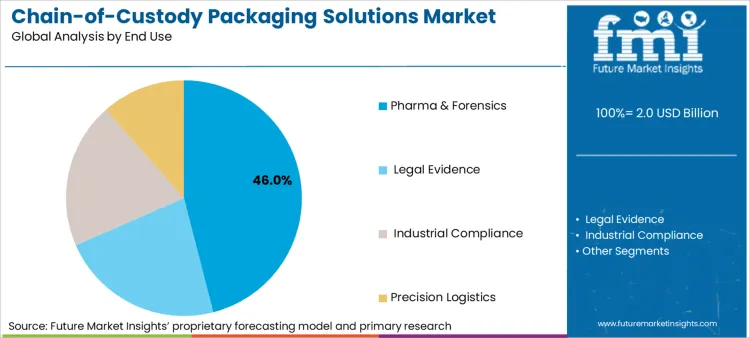
The pharma & forensics segment commands a 46% share because it operates under the highest-stakes regulatory and liability frameworks. In pharma, a broken chain of custody can invalidate billion-dollar clinical trials or lead to product recalls and regulatory action. In forensics, it can compromise criminal prosecutions.
This extreme consequence of failure justifies premium pricing for packaging systems that offer forensic-grade tamper evidence and digitally immutable logs. The segment drives innovation in features like timed-access seals, temperature logging integration, and compatibility with regulatory reporting platforms, setting the high-performance benchmark for the entire market.
What Role does the Concentration of Regulated Industries Play in North America's Market Leadership?
North America holds a dominant 34% regional share, sustained by its concentration of global pharmaceutical headquarters, advanced forensic laboratories, and a legal system with rigorous discovery and evidence rules. The region is the epicenter for the development and enforcement of track-and-trace regulations like the DSCSA.
Packaging solutions are often developed and certified first for the US market, creating a high-value, early-adopter segment. Furthermore, the litigious environment makes defensible chain-of-custody a critical risk-mitigation investment for companies across sectors, from legal cannabis to aerospace, further deepening the market.
What Drivers, Restraints, Opportunities, and Trends Prevail in the Market?
Market expansion is supported by the proliferation of IoT sensors becoming cost-effective for integration. Packaging is evolving from passive to active with embedded sensors that log temperature, shock, tilt, and even light exposure, automatically uploading this custody-condition data to the cloud. This provides irrefutable proof of proper handling, particularly crucial for temperature-sensitive pharmaceuticals and sensitive electronics.
While demand is robust, a key restraint is the high total cost of ownership and integration complexity. Implementing a full chain-of-custody system requires not just specialized packaging but also scanners, software, employee training, and process redesign. This can be prohibitive for smaller organizations or for applications with thin margins, slowing adoption outside of highly regulated or high-value segments.
Technical innovation is defined by the convergence of physical seals with digital trust anchors. The most advanced systems now use cryptographic seals where a unique digital code is revealed only upon opening. This code must be scanned to validate the break event, creating a two-factor authentication process for custody transfer. This makes fraudulent resealing virtually impossible and is becoming a gold standard for high-security evidence transport.
The emergence of decentralized digital ledgers (blockchain) for custody logging represents a disruptive opportunity. While most systems use centralized databases, pilots are using permissioned blockchains to create a shared, immutable custody record accessible to all authorized parties in a supply chain (e.g., manufacturer, logistics, hospital). This could increase transparency and reduce disputes, potentially reshaping how multi-party custody is managed.
Analysis of Chain-of-Custody Packaging Market by Key Countries
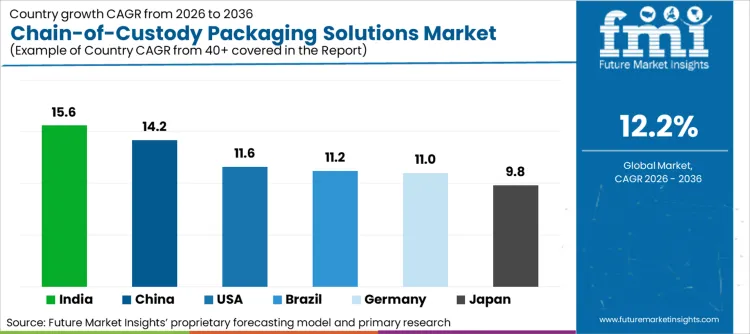
| Country | CAGR (2026–2036) |
|---|---|
| USA | 11.6% |
| Germany | 11.0% |
| China | 14.2% |
| Japan | 9.8% |
| India | 15.6% |
| Brazil | 11.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How do Pharmaceutical Serialization and Legal Discovery Rules define USA’s Market?
Two powerful forces, pharmaceutical regulation and legal procedural rules dominate USA, growing at an 11.6% CAGR. The final implementation phase of the DSCSA is driving massive investment in unit-level, serialized packaging across the drug supply chain.
Federal Rules of Evidence and civil discovery procedures mandate stringent evidence handling. US packaging suppliers like Sentry Systems must develop solutions that are compliant with both FDA technical guidelines and the procedural standards admissible in federal and state courts, creating a deeply specialized and compliance-driven market.
What Role does EU-Wide Regulation and Forensic Standardization Play in Germany?
Germany’s 11.0% CAGR is fueled by its central role in shaping EU-wide regulations and forensic standards. Germany is a hub for pan-European clinical trials and a leader in forensic science. Packaging solutions must therefore comply not just with German law but with the European Medicines Agency (EMA) requirements and the standards of the European Network of Forensic Science Institutes (ENFSI).
German suppliers like Bong Group focus on creating systems that meet these harmonized, high-level requirements, ensuring their products are pre-validated for use across the EU’s single market, giving them a significant export advantage within Europe.
How is China's Market Driven by Supply Chain Integrity for Global Exports?
China’s high growth rate of 14.2% CAGR is primarily driven by its need to guarantee supply chain integrity for its massive export economy. As Western regulators and corporations demand more transparency from Chinese suppliers of pharmaceuticals, electronics, and industrial components, documented chain-of-custody becomes a competitive necessity. Chinese manufacturers are adopting secure packaging not primarily for domestic regulation but as a credential to maintain access to lucrative, regulated export markets. This drives demand for cost-optimized yet technically robust solutions that can scale to millions of units.
How are Japan's Precision Logistics and Anti-Tampering Culture Influencing Packaging?
Japan’s market, growing at a 9.8% CAGR, is shaped by its world-leading precision logistics and a cultural aversion to tampering. The focus is on the secure transport of high-value, miniature electronic components, semiconductor wafers, and diagnostic reagents.
Japanese packaging, from suppliers like Rengo, emphasizes extreme precision in fit, multi-layered tamper indication often using delicate, proprietary or tear materials, and seamless integration with just-in-time, highly automated logistics systems. The market values flawless, predictable performance and discreet, integrated security over overt, bulky solutions.
How is India's Pharmaceutical Manufacturing Boom Catalyzing Specialized Export Packaging?
India exhibits the highest CAGR at 15.6%, directly linked to its position as the pharmacy of the world. The growth of its USFDA- and EU-GMP-certified manufacturing base creates an enormous, captive demand for chain-of-custody packaging that meets international regulatory standards for drug exports. Indian suppliers like TCPL Secure are scaling up production of serialized pouches and security cartons that comply with DSCSA and EU FMD requirements.
The market opportunity lies in providing high-quality, cost-competitive solutions locally, reducing dependency on imports and securing the integrity of the country's vital pharmaceutical export supply chain.
Which Commodity Certification and Regional Logistics Demands Shape Brazil's Market?
Brazil’s market expansion at an 11.2% CAGR is influenced by its role as a major exporter of certified commodities (like organic coffee, premium beef) and complex domestic logistics. Exporters use chain-of-custody packaging to provide verifiable proof that certified products have not been commingled with non-certified goods during storage and transport.
Domestically, the need to securely transport legal documents, forensic samples, and financial instruments across vast distances with multiple handoffs drives demand for packaging with clear custody logging. Suppliers like Ranpak develop solutions that are durable enough for Brazil's logistics environment while providing the audit trail needed for both export certification and internal compliance.
What is the Competition Outlook for Chain-of-Custody Packaging Solutions Market?
Competitive intensity is rising as the value proposition moves from supplying packaging to enabling verifiable custody assurance. The market increasingly separates into technology-led traceability platforms, which provide serialization, event logging, and data integrity, and packaging specialists, which focus on tamper-evident materials, secure seals, and process reliability. Competitive advantage depends on interoperability, particularly the ability to integrate custody data into enterprise quality, compliance, and ERP systems rather than operating as a standalone packaging add-on.
A clear strategic direction is the development of end-to-end custody systems that combine physical security with procedural validation. Leading solutions increasingly bundle packaging formats, unique seals or identifiers, scanning workflows, and documentation protocols designed to withstand regulatory and legal scrutiny. This positions suppliers not just as packaging vendors, but as risk-management partners in regulated supply chains such as pharmaceuticals, clinical trials, legal evidence handling, and high-value industrial logistics.
Strategic leadership is also shifting toward analytics derived from custody events. Aggregated handoff data from serialized and tracked packaging is being used to identify failure points, optimize logistics processes, and support compliance audits. Providers that can convert custody logs into actionable operational insights are expanding their role from compliance support into supply-chain optimization.
Key Developments:
- Sealed Air continues to expand tamper-evident and protective packaging solutions for regulated supply chains, including pharmaceutical and clinical trial logistics, with a focus on seal integrity, validation support, and compatibility with external track-and-trace systems used by sponsors and logistics providers. Public disclosures emphasize integration readiness rather than proprietary custody ledgers.
- Mondi Group has strengthened its portfolio of secure and tamper-evident paper-based packaging for sensitive documents and regulated goods across Europe. These solutions focus on controlled opening behavior, material integrity, and process reliability to support documented chain-of-custody requirements without relying on unverified forensic pattern technologies.
- TCPL Packaging remains active in secure and pharmaceutical packaging, supplying printed, laminated, and specialty packaging formats used in regulated distribution environments. Public information highlights ongoing investment in pharma-grade capabilities and compliance readiness, though without disclosure of contract-specific or capacity-expansion figures tied to custody applications.
Key Players Profiled
- Sentry Systems
- Sealed Air Corporation
- Bong Group
- Mondi Group
- Zhejiang Zijiang Enterprise Group
- Greatview Aseptic Packaging Co., Ltd.
- Rengo Co., Ltd.
- TCPL Secure (A part of TCPL Packaging Ltd.)
- Ranpak Holdings Corp.
- Smurfit Kappa Group
Market Definition
The chain-of-custody packaging solutions market comprises revenue generated from the design, manufacture, and sale of packaging systems specifically engineered to establish and maintain a documented, sequential record of the possession, handling, and transfer of an item. These solutions incorporate features such as tamper-evidence, unique serialization, and often digital tracking capabilities to provide auditable proof that an item has been controlled and protected from unauthorized access or alteration throughout its journey.
The market scope covers packaging where the creation of a legally or regulatorily defensible custody record is a primary function. It excludes standard protective packaging and basic tamper-evident solutions that do not include provisions for documenting a sequential chain of possession and handling events.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 1,980 million |
| End Use | Pharma & Forensics, Legal Evidence, Industrial Compliance, Precision Logistics, Others |
| Packaging Format | Track-and-Trace Pouches, Secure Mailers, Security Cartons, Custom Security Boxes, Others |
| Material | Polymer Films, Paperboard, Corrugated Board, Coated Board, Others |
| Regions Covered | North America, Europe, East Asia, Japan, South Asia, Latin America, Middle East & Africa |
| Countries | USA, Germany, China, Japan, India, Brazil and 40+ countries |
| Key Companies | Sentry Systems, Sealed Air, Bong Group, Mondi, Zijiang, Greatview, Rengo, TCPL Secure, Ranpak, Smurfit Kappa |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Chain-of-Custody Packaging Solutions Market Key Segments
By End Use
- Pharma & Forensics
- Legal Evidence
- Industrial Compliance
- Precision Logistics
- Others
By Packaging Format
- Track-and-Trace Pouches
- Secure Mailers
- Security Cartons
- Custom Security Boxes
- Others
By Material
- Polymer Films
- Paperboard
- Corrugated Board
- Coated Board
- Others
By Region
- North America
- Europe
- East Asia
- Japan
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- U.S. Food and Drug Administration (FDA). (2025). Drug Supply Chain Security Act (DSCSA): Package-Level Security and Serialization Guidance. FDA.
- National Institute of Standards and Technology (NIST). (2025). NIST IR 8512: Guide to Digital Forensic Evidence Handling and Chain of Custody. NIST.
- European Network of Forensic Science Institutes (ENFSI). (2025). Best Practice Manual for the Packaging, Transport, and Storage of Forensic Evidence. ENFSI.
- World Customs Organization (WCO). (2026). Secure Trade and Chain-of-Custody Controls for Regulated and High-Risk Goods. WCO.
- European Medicines Agency (EMA). (2025). Good Distribution Practice: Safety Features and Tamper-Evident Requirements for Medicinal Products. EMA.
Frequently Asked Questions
How big is the chain-of-custody packaging solutions market in 2026?
The global chain-of-custody packaging solutions market is estimated to be valued at USD 2.0 billion in 2026.
What will be the size of chain-of-custody packaging solutions market in 2036?
The market size for the chain-of-custody packaging solutions market is projected to reach USD 6.3 billion by 2036.
How much will be the chain-of-custody packaging solutions market growth between 2026 and 2036?
The chain-of-custody packaging solutions market is expected to grow at a 12.2% CAGR between 2026 and 2036.
What are the key product types in the chain-of-custody packaging solutions market?
The key product types in chain-of-custody packaging solutions market are polymer films , paperboard, corrugated board and coated board.
Which end use segment to contribute significant share in the chain-of-custody packaging solutions market in 2026?
In terms of end use, pharma & forensics segment to command 46.0% share in the chain-of-custody packaging solutions market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material , 2026 to 2036
- Polymer Films
- Paperboard
- Corrugated Board
- Coated Board
- Polymer Films
- Y to o to Y Growth Trend Analysis By Material , 2021 to 2025
- Absolute $ Opportunity Analysis By Material , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharma & Forensics
- Legal Evidence
- Industrial Compliance
- Precision Logistics
- Pharma & Forensics
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material
- By End Use
- Competition Analysis
- Competition Deep Dive
- Sentry Systems
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sealed Air Corporation
- Bong Group
- Mondi Group
- Zhejiang Zijiang Enterprise Group
- Greatview Aseptic Packaging Co., Ltd.
- Sentry Systems
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Material
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Material
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Material
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Material
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Material
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

