The CRISPR-integrated gene editing delivery devices market was valued at USD 1.03 billion in 2025. Sales is set to reach a CRISPR delivery market size 2026 of USD 1.18 billion at a CRISPR device market CAGR of 14.10% during the forecast period. Steady funding contributes to the growth in valuation to a CRISPR delivery devices forecast 2036 of USD 4.41 billion as clinical developers shift from research-grade transfection workflows to closed-system, clinical-scale cellular modification platforms capable of meeting commercial viability metrics.
Biopharmaceutical manufacturing directors are currently forced to decide between scaling legacy viral transduction networks or qualifying non viral CRISPR delivery market architectures. This decision dictates the commercial baseline of their autologous and allogeneic pipelines for the next decade. Relying entirely on viral vectors limits manufacturing throughput and introduces significant batch-to-batch variability. Organizations that delay the integration of high-throughput electroporation or microfluidic disruption risk yielding their commercial window to competitors operating with lower cost-of-goods and faster turnaround times. Transitioning to scalable non-viral methods removes the primary bottleneck in autologous engineered cell therapy production, shifting the CRISPR delivery device trends toward highly automated solutions.
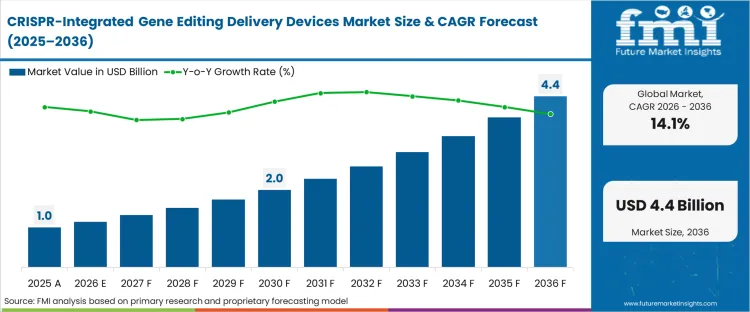
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.18 Billion |
| Industry Value (2036) | USD 4.41 Billion |
| CAGR (2026 -2036) | 14.10% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The validation of continuous-flow electroporation systems for clinical-stage ex vivo gene editing delivery systems serves as the primary inflection gate. This threshold is crossed when regulatory agencies approve pivotal trials utilizing entirely non-viral delivery modalities for critical hematology targets. Once integrated into commercial manufacturing protocols, the requirement for extensive viral vector facility outlays collapses, accelerating subsequent pipeline development.
China is estimated to advance at 15.7%, driven by aggressive translational capacity build-outs prioritizing non-viral infrastructure. India follows closely at 15.1%, leveraging a lower-base expansion in centralized biotechnology parks. South Korea tracks at 14.8% as domestic biopharmaceutical contract manufacturers aggressively install closed-system engineering lines. The United States is poised to expand at 13.8%, reflecting steady upgrades across a mature installed base of advanced therapy centers. The United Kingdom is projected to post a 13.2% rate, while Germany is likely to garner 12.9% growth as major CDMOs requalify their process workflows. Japan is estimated to register 12.4%. The divergence stems from the transition rate of ex vivo engineering shifting from academic discovery labs into fully standardized therapeutic supply chains.
The CRISPR gene editing delivery devices market encompasses the specialized hardware, electroporation platforms, microfluidic disruption systems, and targeted nanoparticle injection frameworks required to physically or chemically transport CRISPR-Cas constructs across cell membranes. It represents the interface between therapeutic nucleic acid payloads and target cellular machinery, bounded strictly by the physical delivery modalities rather than the genetic constructs themselves.
This sector includes clinical and research-grade electroporation instruments, nucleofection devices, continuous-flow cellular engineering systems, microfluidic shear-stress delivery apparatuses, and the hardware required for systemic lipid nanoparticles administration. The scope encapsulates all capital equipment and associated proprietary consumables explicitly designed to facilitate the intracellular transport of gene-editing components, forming the core of the CRISPR delivery platform market.
The genetic payloads, including custom sgRNA sequences, standalone Cas nucleases, and base-editing enzymes, are explicitly excluded. These represent the biological cargo rather than the delivery mechanism. General-purpose laboratory incubators, standard liquid handling robotics, and upstream cell isolation centrifuges are also excluded, as they do not perform the distinct function of breaching the cell membrane to facilitate construct integration.
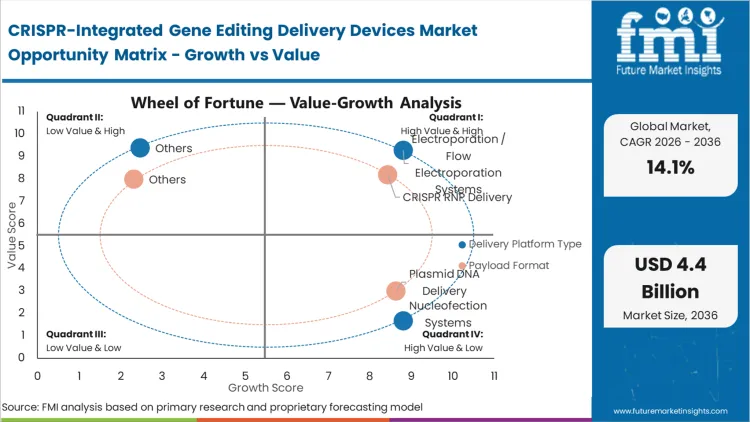
The reason electroporation systems hold 34.0% of this market comes down to a single operational reality: clinical manufacturers cannot absorb the batch failure rates associated with legacy chemical transfection at a commercial scale. This technology is not chosen solely for its biological efficiency. It is chosen because it is the only non-viral option that allows process development directors to consistently process billions of cells in a closed, GMP-compliant loop without manual intervention.
According to FMI's estimates, facilities relying on older batch methods face severe throughput constraints when pivoting from autologous to allogeneic frameworks. The integration of flow electroporation fundamentally restructures the cost-of-goods equation, enabling viable cell therapy manufacturing for larger patient populations. Organizations that attempt to scale using unvalidated experimental platforms often face forced process redesigns during late-stage clinical reviews, emphasizing the critical CRISPR delivery device market share by technology.
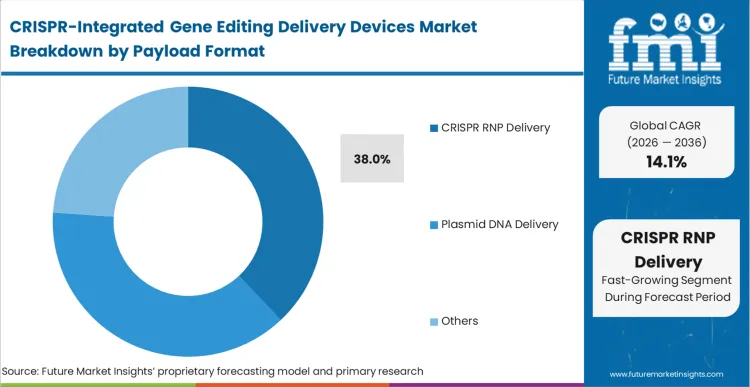
Ribonucleoprotein (RNP) complexes represent the specific payload choice facing biopharma translation directors right now, dictating an estimated 38.0% of the segment's requirements. The shift toward RNP formats forces a corresponding requirement for delivery devices capable of handling delicate protein-RNA structures without causing aggregation. FMI analysts opine that hardware configured exclusively for robust plasmid purification pipelines often fails to deliver adequate RNP integration without significant viability drops. RNP delivery ensures transient nuclease activity, sharply reducing the window for off-target chromosomal translocations compared to persistent viral or plasmid expression. Process scientists operating entirely on mRNA/sgRNA combinations eventually confront stability and translation efficiency variations. Those delaying the optimization of RNP-specific electroporation protocols encounter significant regulatory friction during long-term safety evaluations, directly impacting the CRISPR transfection systems market.
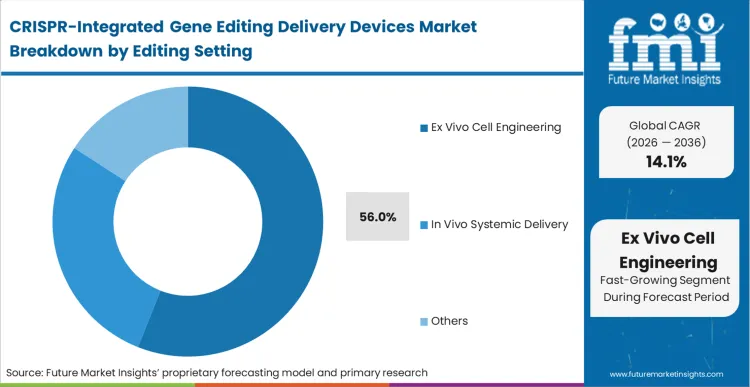
The ex vivo engineering modality holds 56.0% share because it represents the only controlled environment where massive, multiplexed genomic modifications can be verified before patient exposure. This setting necessitates highly specialized benchtop to floor-standing equipment that isolates the crispr and cas gene transfer process outside the body. As per FMI's projection, the physical separation of the editing event from the patient allows quality control managers to sequence the modified batch, ensuring off-target integrations fall below strict regulatory thresholds. This operational reality dictates that the bulk of current capital expenditure flows into closed-system isolators and continuous flow devices. Attempts to bypass ex vivo validation for complex multiplex edits using immature systemic delivery methods frequently result in clinical holds. Facilities that fail to standardize their ex vivo instrumentation face crippling bottlenecks during regulatory audits, highlighting the importance of securing the best CRISPR delivery platform for cell therapy manufacturing.
Biopharma and cell & gene therapy developers command 41.0% of device adoption, driven by the requirement to internalize critical manufacturing steps for their lead clinical assets. This cohort does not just purchase equipment; they define the qualification standards that filter down to the rest of the industry. Based on FMI's assessment, major developers are actively moving away from decentralised academic partnerships, opting instead to build vertically integrated hubs equipped with advanced cell and gene therapy manufacturing infrastructure. This internalization allows them to protect proprietary transfection protocols and maintain absolute control over production timelines. Relying entirely on external academic core facilities for pivotal-stage clinical supply invariably introduces schedule slippage and quality inconsistencies. Developers who underinvest in their own device infrastructure remain dangerously dependent on constrained CDMO capacity and third-party CRISPR delivery device suppliers.
The clinical outcome defines the hardware requirement, and for haematology and hemoglobinopathies, that requirement translates to processing massive volumes of CD34+ hematopoietic stem cells without compromising engraftment potential. This application, representing 29.0% of the segment, acts as the proving ground for all commercial-scale delivery devices. In FMI's view, the recent validation of these gene editing tools in treating sickle cell disease has established a concrete operational baseline for CRISPR delivery for hematopoietic stem cells. Delivery platforms must gently navigate the sensitivity of primary stem cells, as aggressive electroporation permanently degrades their ability to repopulate patient bone marrow. Devices optimized for resilient immortalized cell lines frequently destroy primary CD34+ populations. Therapeutic sponsors who fail to qualify their devices specifically against delicate haematological targets suffer catastrophic engraftment failures in early clinical phases.
The transition from autologous to allogeneic cell therapy frameworks forces biopharmaceutical process development directors to completely redesign their delivery architectures. Producing universally compatible "off-the-shelf" therapies requires multiple simultaneous gene knockouts to prevent graft-versus-host disease, a feat that overwhelms legacy viral transduction capacities. This compels facility heads to mandate the integration of a GMP electroporation system for genome editing capable of executing multiplexed RNP delivery in a single, high-throughput pass. The commercial stakes of delaying this transition are absolute: developers relying on sequential viral editing simply cannot achieve the cost-of-goods necessary to make allogeneic therapies financially viable.
The primary operational friction slowing device integration lies in the severe optimization curve required to translate research-scale parameters into closed-system, clinical-grade workflows. Moving from a static 100-microliter cuvette to a continuous flow line is not a linear scaling exercise; it involves entirely different fluid dynamics and electrical resistance profiles. This requires process development scientists to dedicate months to empirical recalibration, significantly delaying the progression from discovery to cell and gene therapy clinical trial phases. While newer devices offer software-driven scaling models, the inherent biological variability of primary patient cells ensures this structural bottleneck persists.
The regional breakdown indicates that the CRISPR‑Integrated Gene Editing Delivery Devices market spans over 40 countries, categorized across North America, Europe, Asia Pacific, and the rest of the world.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 15.7% |
| India | 15.1% |
| South Korea | 14.8% |
| United States | 13.8% |
| United Kingdom | 13.2% |
| Germany | 12.9% |
| Japan | 12.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research

Aggressive state-backed capitalization of regional biotechnology parks shapes the adoption pattern in Asia Pacific, accelerating the installation of advanced clinical infrastructure regardless of immediate pipeline maturity. Unlike Western markets where capital expenditure closely trails specific clinical trial approvals, this region builds capacity proactively. FMI analysts opine that this strategy pulls forward the integration of commercial-scale continuous flow systems, bypassing the interim upgrades often seen in legacy facilities. This creates an environment where contract manufacturing organizations can offer immediate, validated non-viral processing capabilities to global biopharma clients. However, the reliance on imported core hardware creates acute supply chain vulnerabilities during periods of geopolitical trade friction.
FMI's report includes secondary markets across Southeast Asia and Oceania. These peripheral zones are currently establishing initial regulatory definitions for advanced therapies, delaying major hardware procurement until clear localized compliance pathways are published.
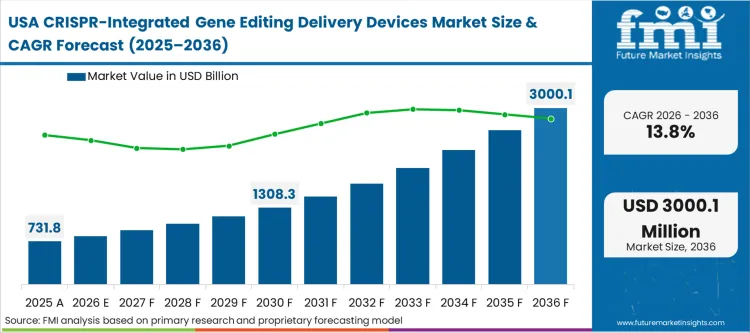
The massive, pre-existing installed base of academic and clinical research infrastructure shapes how new delivery technologies penetrate the North American landscape. This is not a market building from scratch; it is a market executing complex requalification cycles. Process development directors must justify the capital expense of novel microfluidic or advanced electroporation systems against the operational disruption of replacing functional, albeit slower, legacy hardware. According to FMI's estimates, these dynamic forces device manufacturers to compete heavily on seamless integration and software compatibility rather than base specifications. The region dictates global standards for GMP compliance, meaning hardware validated here rapidly becomes the baseline requirement for international crispr based gene editing operations.
FMI's report includes detailed assessments of the Canadian biotechnology corridor. Facilities in these hubs heavily leverage cross-border research consortiums to offset the initial capital burden of high-end clinical delivery systems.
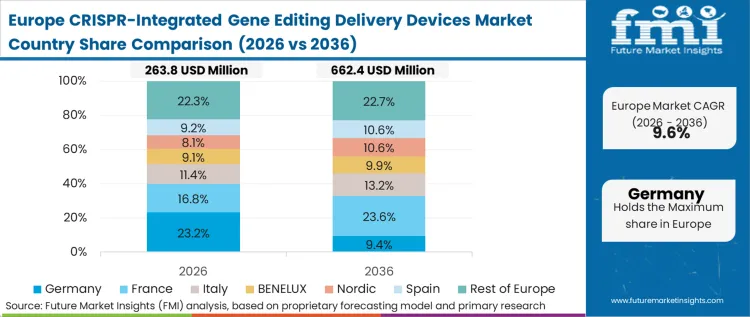
Stringent, centralized regulatory directives governing advanced therapy medicinal products (ATMPs) dictate the procurement patterns across the European manufacturing network. The European Medicines Agency's rigorous demands for process consistency and extensive characterization heavily penalize experimental or unvalidated delivery modalities. As per FMI's projection, this regulatory environment forces CDMOs to coalesce around a very narrow range of proven, closed-system electroporation devices that already possess robust master files. This risk-averse climate lengthens initial sales cycles but creates deep, unshakeable vendor lock-in once a platform is embedded into a pivotal trial protocol.
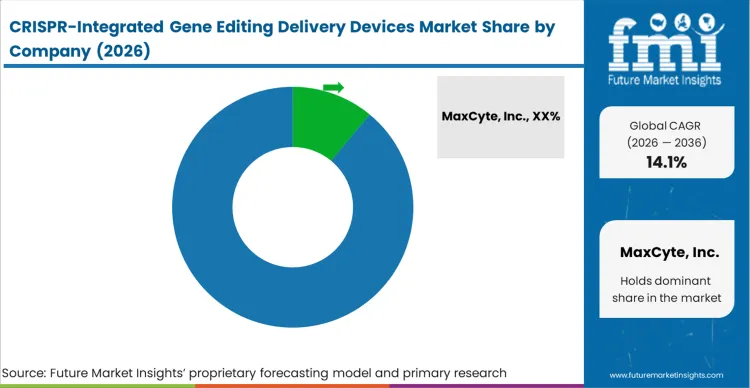
The highly concentrated nature of the clinical-grade delivery device sector stems directly from the immense regulatory friction associated with validating hardware for human therapeutics. Major biopharmaceutical companies do not select platforms based purely on peak transfection efficiency; they select based on the existence of comprehensive regulatory master files and a proven history of clinical success when looking for CRISPR delivery device suppliers. Incumbents like MaxCyte, Inc. and Lonza Group AG dominate because they provide facility directors with immediate access to these established regulatory pathways, effectively de-risking the entire manufacturing process.
The advantage held by these incumbents persists because the cost of switching devices mid-clinical trial is commercially ruinous. A challenger must build an entirely closed, GMP-compliant hardware and consumable ecosystem, and then convince a developer to risk their pivotal trial on an unproven physical delivery mechanism. To break this lock-in, emerging device manufacturers must offer a categorically distinct capability, such as high-efficiency lyophilized viral vector storage solutions integration or acoustic disruption that preserves 99% of primary cell viability,that fundamentally alters the cost-of-goods equation.
Heading toward 2036, the competitive pressure between CDMOs demanding open, flexible manufacturing suites and device vendors enforcing proprietary, closed consumable ecosystems will intensify. Large contract manufacturers actively resist being tethered to a single vendor's pricing model for critical disposable pathways, driving procurement teams to closely compare CRISPR electroporation vendors for commercial manufacturing. This dynamic will likely force a gradual unbundling, where established players are pressured to license their core electroporation or microfluidic architectures for integration into broader, automated facility-wide genomics processing lines.

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.18 Billion to USD 4.41 Billion, at a CAGR of 14.10% |
| Market Definition | The market defines the specialized physical and electro-chemical instrument ecosystem required to transport nucleases and guide RNAs into target cells. It constitutes the bridge between raw genetic tools and viable therapeutic cell products. |
| Delivery platform type Segmentation | Electroporation / flow electroporation systems, Nucleofection systems, Lipid nanoparticle delivery platforms, Viral vector-assisted delivery systems, Microfluidic / membrane-disruption delivery devices, Hybrid / exosome-enabled delivery platforms |
| Payload format Segmentation | CRISPR RNP delivery, Cas9 mRNA + sgRNA delivery, Plasmid DNA delivery, Base-editing payload delivery, Prime-editing payload delivery, Viral construct delivery |
| Editing setting Segmentation | Ex vivo cell engineering, In vivo systemic delivery, Localized / organ-targeted in vivo delivery, Research-scale screening and discovery workflows |
| End user Segmentation | Biopharma and cell & gene therapy developers, CDMOs / process development organizations, Academic and translational research institutes, Hospital-based advanced therapy centers, Biotechnology platform startups |
| Application Segmentation | Hematology and hemoglobinopathies, Oncology cell therapy engineering, Rare monogenic disease programs, Immunology / autoimmune editing programs, Ophthalmology and liver-targeted editing |
| Regions Covered | North America, Europe, Asia Pacific, Rest of the World |
| Countries Covered | United States, China, Germany, United Kingdom, Japan, South Korea, India, and 40 plus countries |
| Key Companies Profiled | MaxCyte, Inc., Lonza Group AG, Thermo Fisher Scientific Inc., Bio-Rad Laboratories, Inc., Cytiva, Takara Bio Inc., Miltenyi Biotec B.V. & Co. KG |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews were conducted with process development directors and vector core managers to establish baseline hardware utilization. The baseline anchors to the active installed base of clinical-grade electroporation and closed-system transfection units. Forecasts were cross-validated against reported capital expenditure patterns and utilization rates from leading cell therapy contract manufacturers. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the CRISPR-Integrated Gene Editing Delivery Devices Market in 2026?
The gene editing delivery devices market reaches USD 1.18 billion in 2026, anchoring a massive shift in capital expenditure as biopharmaceutical developers transition from legacy batch transfection to scalable, closed-system delivery networks.
What will it be valued at by 2036?
It is projected to hit USD 4.41 billion by 2036, signalling the complete integration of non-viral continuous-flow devices into standardized commercial manufacturing protocols.
What CAGR is projected?
The 14.10% CAGR reflects the urgency with which CDMOs are forced to upgrade their physical infrastructure to accommodate the throughput requirements of incoming multiplexed allogeneic trials.
What are CRISPR-integrated gene editing delivery devices?
CRISPR gene editing delivery devices are specialised physical and electro-chemical instruments, such as electroporators, microfluidic systems, and LNP mixers, required to transport nucleases and guide RNAs into target cells, forming the critical bridge between raw genetic tools and viable cell therapies.
Which delivery platform type segment leads?
Electroporation / flow electroporation systems lead because they currently provide the only regulatory-validated pathway for processing billions of cells without the batch failures inherent to manual cuvette methods.
Which payload format segment leads?
CRISPR RNP delivery leads because clinical researchers must strictly limit nuclease exposure time to prevent off-target translocations, an operational mandate that mRNA or plasmid vectors cannot reliably satisfy.
Which editing setting segment leads?
Ex vivo cell engineering leads because regulatory frameworks absolutely require external verification and karyotyping of modified therapeutic cells before they are cleared for patient infusion.
what is the best delivery method for CRISPR therapeutics?
The optimal method depends entirely on the clinical target: continuous-flow electroporation is the gold standard for ex vivo cellular engineering, while lipid nanoparticles are emerging as the preferred vehicle for targeted in vivo hepatic delivery.
why is delivery the bottleneck in CRISPR gene editing?
The biological variability of primary patient cells creates a severe optimization bottleneck, requiring process scientists to spend months empirically recalibrating shear stress and electrical parameters to transport large RNP complexes without destroying the target cells.
How is Casgevy delivered ex vivo?
Casgevy is a first-of-its-kind therapy that relies on the non-viral delivery of a CRISPR/Cas9 RNP complex into patient-derived CD34+ hematopoietic stem cells using clinical-scale electroporation.
Which country grows fastest?
China outpaces India by pulling its translational expansion directly into commercial-grade non-viral infrastructure, bypassing the legacy batch architectures that constrain older Western facilities.
How does the ATMP regulatory framework impact device selection?
Strict European directives penalize experimental device modifications, forcing CDMOs to universally adopt platforms that already possess robust regulatory master files to avoid crippling audit delays.
electroporation vs LNP for CRISPR: Are lipid nanoparticles replacing electroporation in gene editing?
While LNPs are rapidly capturing share for in vivo applications due to their systemic administrability, they are not replacing electroporation in complex ex vivo autologous workflows where immediate, transient RNP expression is required.
Why do developers resist vendor lock-in with closed-system consumables?
Tethering an entire commercial manufacturing pipeline to a single vendor's proprietary flow kits exposes developers to unmanageable pricing leverage and acute supply chain disruption risks during peak clinical phases.
What limits the adoption of in vivo systemic delivery devices?
The inability to consistently direct complex nanoparticle payloads to specific organ targets at therapeutic titers prevents clinical directors from shifting capital away from proven ex vivo engineering hardware.
Which end users buy CRISPR delivery devices the most?
Biopharma and cell & gene therapy developers account for the majority of the market, driven by the requirement to internalize critical manufacturing steps for their lead clinical assets.
Why are RNP formats incompatible with legacy plasmid delivery workflows?
RNP complexes require highly specialized chilling and rapid-mixing protocols to prevent protein aggregation, overwhelming standard fluidic channels designed for robust DNA stability.
What characterizes Japan's approach to adopting new delivery platforms?
A highly risk-averse regenerative medicine framework lengthens the procurement cycle, requiring exhaustive off-target characterization data before researchers can authorize the capital transition.
Why are Tier-1 biopharma developers internalizing their device infrastructure?
Relying on external academic cores for crucial transfection steps introduces schedule slippage, prompting developers to build vertically integrated hubs to maintain absolute control over pivotal trial timelines.
What operational metric dictates hardware selection for hematology programs?
Preservation of CD34+ cell stemness and engraftment potential is the absolute priority, automatically disqualifying any platform that relies on aggressive membrane permeabilization.
How does South Korea's CDMO concentration affect device procurement?
The requirement for absolute standardization across massive production suites favors established, GMP-certified devices that integrate seamlessly into existing digital batch recording systems.
What triggers the inflection point for non-viral clinical dominance?
The commercial scale-up of multiplexed therapies crosses the threshold when pivotal trials secure approval utilizing entirely non-viral delivery modalities for critical targets.
How are CDMOs in Germany maintaining their margin structure?
By phasing out labor-intensive batch transfection and installing automated lines capable of concurrent processing, German facilities drastically reduce manual intervention requirements and cost-of-goods.
Why is the United States growth rate closer to the global average?
The mature installed base of early-generation devices forces capital deployment toward strategic requalification and phased process upgrades, moderating the raw expansion rate seen in emerging Asian hubs.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.