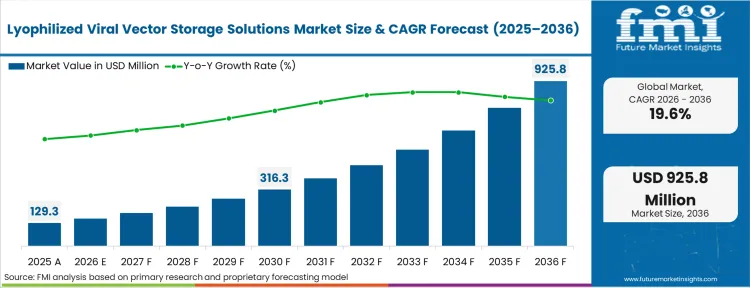
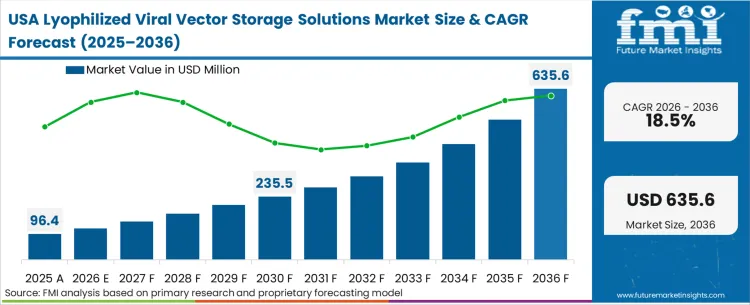
The lyophilized viral vector storage solutions market was valued at USD 129.3 million in 2025. The industry is poised to surpass USD 154.6 million in 2026 at a CAGR of 19.6% during the forecast period. Ongoing investment is expected to expand the market opportunity to USD 925.8 million by 2036, driven by gene therapy developers moving away from ultra‑cold supply chains toward more scalable thermal storage profiles that support global commercialization.
Biopharma sponsors are being forced to make a decisive supply chain transition before late-stage clinical trials. Historically, early-stage gene therapies relied on frozen storage utilizing cryogenic systems, an approach that fractures commercially once a therapy targets larger patient populations across decentralized hospital networks. The stakes of delaying the transition to a lyophilized format involve severe constraints on market access and prohibitive specialized logistics costs. An inherent constraint often missed by generalists is that viral vector development timelines are rarely gated by the drying cycle itself, but by the analytical bridging required to prove the reconstituted drug performs identically to the frozen clinical batch.
For the market to scale beyond specialized contract manufacturing and achieve broad commercial adoption, the formulation excipient challenge must be addressed. Partners with the capability to accurately characterize the glass transition behavior of complex viral capsids are critical to enabling this shift. Once robust, scalable protective matrices are validated, refrigerated or ambient storage can be implemented as a deliberate lifecycle strategy, supporting predictable commercialization rather than serving as a late-stage corrective measure.
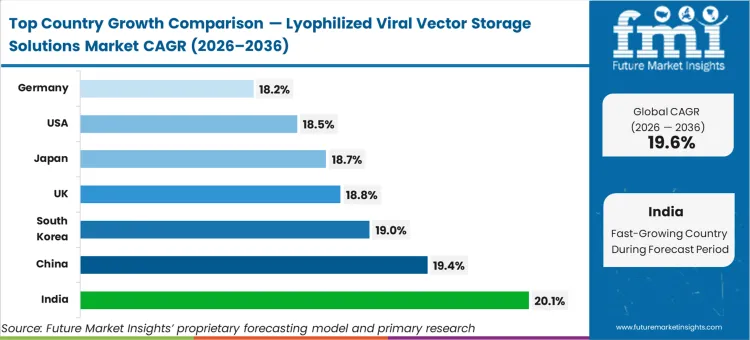
India is estimated to track at 20.1%, followed by China advancing at 19.4%, South Korea registering 19.0%, and the United Kingdom expanding at 18.8%. Japan is likely to grow at 18.7%, the United States expanding at 18.5%, and Germany posting 18.2%. The divergence across the range reflects how quickly regional manufacturing ecosystems are upgrading their analytical formulation capabilities to support global export mandates, separating advanced formulation hubs from basic fill-finish geographies.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 154.6 million |
| Industry Value (2036) | USD 925.8 million |
| CAGR (2026–2036) | 19.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
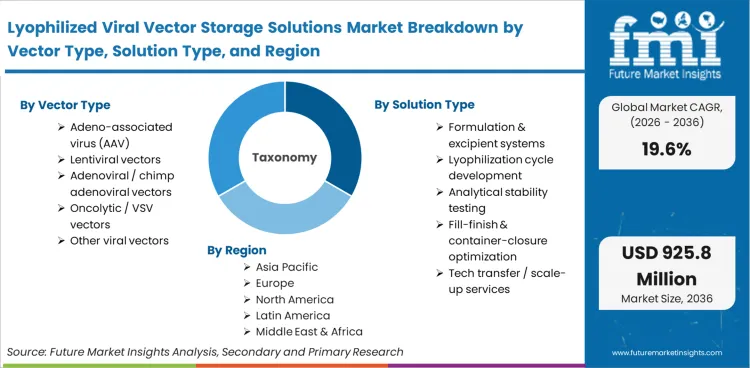
The Lyophilized Viral Vector Storage Solutions Market covers the formulation chemistries, drying cycles, and container-closure systems engineered specifically to preserve viral vector stability outside of deep-frozen states. It is functionally distinct from general biologics lyophilization due to the unique biophysical fragility of viral capsids and the complex excipient matrices required to prevent viral aggregation during the moisture removal process.
This market includes specialized excipient formulations, tailored freeze-drying cycle development services, analytical stability testing specifically for dried vectors, and primary packaging optimized for high-vacuum sealing. Revenue generated by contract development and manufacturing organizations (CDMOs) providing end-to-end cell and gene therapy manufacturing for lyophilized formats is captured within this scope, alongside specialized container-closure components designed to maintain moisture barriers over extended shelf lives.
Standard frozen storage media, general-purpose cryogenic freezers, and liquid formulation services that do not target a freeze-dried end state are excluded. These categories belong to broader logistics and traditional bioprocessing markets. Their inclusion would obscure the highly specialised value generated by moisture-removal technologies and the specific analytical techniques required to validate solid-state viral titer integrity.
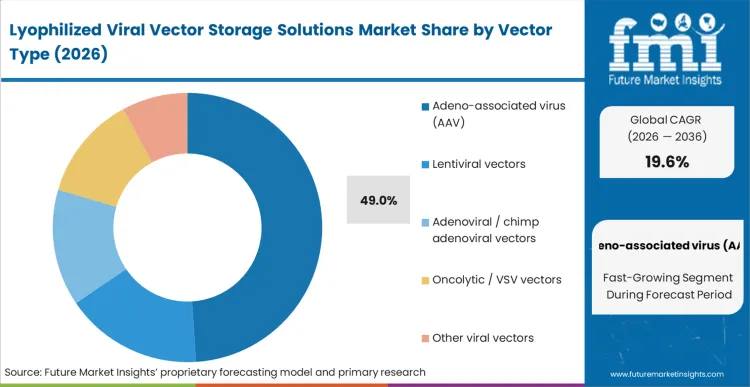
The logic behind Adeno-associated virus (AAV) holding a 49.0% share stems from its overwhelming dominance in the current pipeline of late-stage in vivo therapies. According to FMI's estimates, as developers push these therapies toward commercialization, they rely heavily on specialized aav packaging services and targeted formulation to prevent the unique aggregation tendencies of AAV capsids under stress. The biophysical characteristics of AAV require highly specific excipient matrices to maintain the integrity of the viral genome during the water-removal phase. Sponsors face intense pressure to perfect this stabilisation early; failing to do so results in unacceptable titer drops that render the final drug product sub-therapeutic. By integrating bioprocess containers and fluid transfer solutions tailored for these sensitive vectors, manufacturing teams can streamline the transition to solid-state stability. Developers who underinvest in vector-specific formulation logic face severe regulatory delays during pivotal trial reviews.
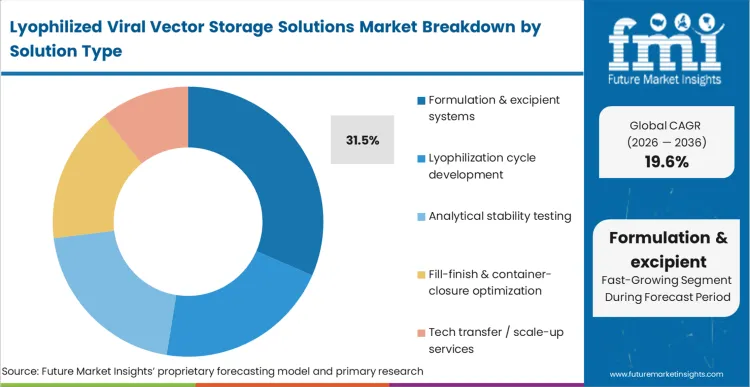
The operational consequence of a failed drying cycle almost always traces back to inadequate chemical protection, positioning Formulation & excipient systems to capture 31.5% of the market. Rather than competing purely on equipment capacity, leading CDMOs differentiate by offering proprietary stabilisation matrices that protect the vector from freeze-concentration stresses.
FMI analysts opine that this upstream chemical architecture dictates the success of the entire downstream manufacturing process, directly impacting healthcare cold chain logistics. Buyers must prioritize partners with deep biophysical analytical capabilities to custom-design these matrices. A misaligned excipient strategy ultimately forces sponsors to restart time-consuming stability studies from scratch.
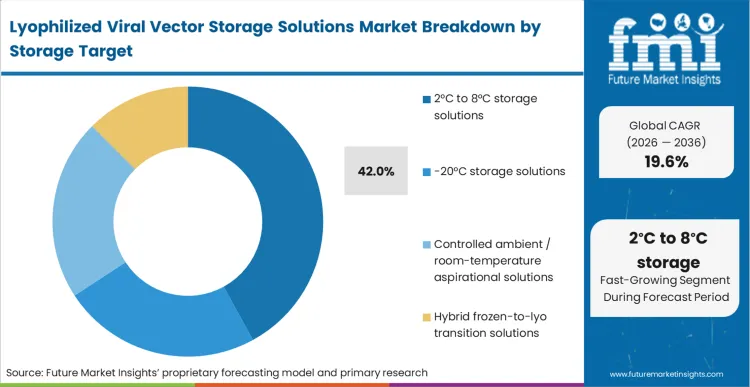
The specific choice facing a CMC director right now is whether to push for aggressive ambient stability or settle for 2°C to 8°C targets, which command a 42.0% share. Refrigerated storage represents the pragmatic middle ground, aligning perfectly with existing hospital pharmacy infrastructure while still bypassing the severe limitations of -80°C networks.
As per FMI's projection, this target thermal profile allows developers to utilize established pharmaceuticalutilisehain packaging formats without requiring bespoke ambient stabilization chemistries that often complicate regulatory reviews. Integration with standard automated compound storage and retrieval systems further solidifies refrigerated targets as the commercial standard. Organisations that aim exclusively for ambient stability without a 2°C to 8°C fallback often find their clinical timelines extended by years due to formulation instability.
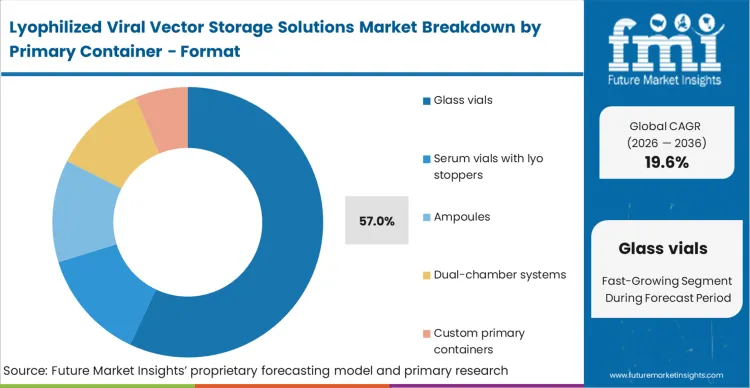
The 57.0% share held by Glass vials is rooted in the necessity of maintaining an absolute moisture barrier over a multi-year shelf life. While polymers advance, Type 1 borosilicate glass remains the undisputed standard for deep-vacuum sealing required in sterile solid-state biologics.
Based on FMI's assessment, the interaction between the freeze-dried cake, the glass surface, and specialised cryogenic vials utilized in earlier stages dictates the long-term viability of the drug product. Switching to alternative materials introduces an immense regulatory comparability burden that most sponsors refuse to shoulder. Consequently, biopharmaceuticals packaging decisions heavily favor established glass formats to streamline the path to market. Procurement teams that deviate from validated glass systems face severe delays as container-closure integrity failures emerge during long-term stability trials.
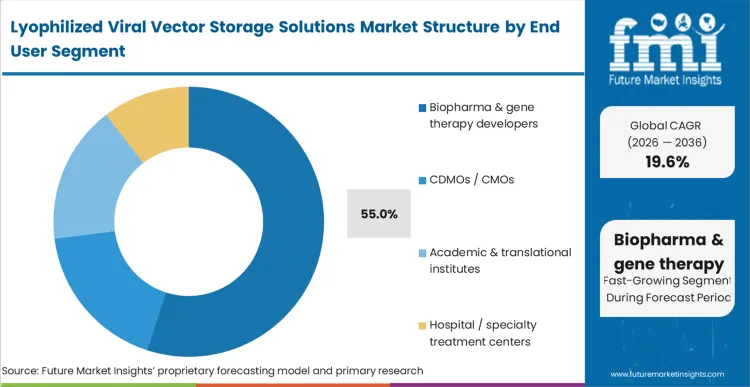
A contradiction defines the Biopharma & gene therapy developers segment, which holds 55.0% share: while manufacturing is heavily outsourced, the core intellectual property of the formulation must be tightly controlled by the sponsor. These developers are the primary risk-bearers, forced to fund the extensive analytical bridging studies required to move a therapy from a liquid clinical format to a commercial lyophilized state.
In FMI's view, integrating upstream gene synthesis with downstream stabilization planning is crucial for these entities to maintain product viability. Their decisions dictate the protocols used in cell and gene therapy clinical trial phases. Biopharma teams that fail to lock in their lyophilization strategy early in the clinical pathway suffer severe commercialization delays, effectively forfeiting market exclusivity windows to faster competitors.
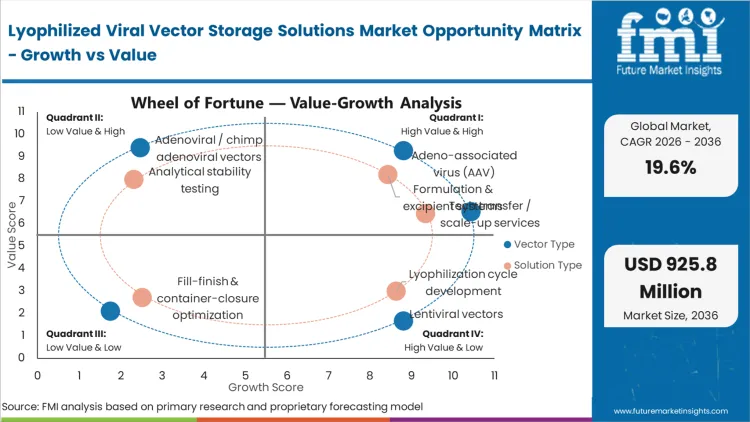
The pressure of scaling from localized clinical trials to global commercial distribution compels CMC directors to eliminate active deep-freeze logistics networks. A therapy that requires -80°C storage, limits its patient reach to a handful of elite treatment centers equipped with specialized handling infrastructure. By transitioning to a lyophilized format, sponsors remove the burden from complex clinical transit and onto the upfront formulation architecture, drastically widening the addressable patient population. The commercial stakes are absolute: therapies that successfully bridge to 2°C to 8°C profiles capture the mass market, while those tethered to cryogenic requirements remain niche, low-volume assets.
The single biggest buyer friction is the biophysical degradation of the viral capsid during the freeze-drying process itself. Unlike simple monoclonal antibodies, complex viral vectors are highly susceptible to aggregation, physical shearing, and irreversible titer loss when subjected to the extreme physical stresses of freezing and sublimation. While predictive thermal modeling offers a partial solution for identifying optimal excipient matrices, it cannot replace the lengthy, empirical stability trials required to validate the process. The complexity is compounded by strict clinical trial packaging regulations, which mandate precise documentation of stability profiles under various thermal excursions.
The regional assessment segments the Lyophilized Viral Vector Storage Solutions Market across more than 40 countries, spanning North America, Europe, Asia Pacific, and the rest of the world.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 20.1% |
| China | 19.4% |
| South Korea | 19.0% |
| United Kingdom | 18.8% |
| Japan | 18.7% |
| United States | 18.5% |
| Germany | 18.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How the regulatory environment shapes buyer behaviour fundamentally dictates the North American market trajectory. The FDA’s increasingly stringent demands for long-term commercial stability data force regional biopharma sponsors to aggressively pursue lyophilization earlier in the clinical pathway. FMI analysts opine that North American CDMOs are pivoting from capacity-building to capability-building, prioritizing advanced biophysical characterization labs over sheer bioreactor volume. This concentration of analytical expertise ensures that the initial proof-of-concept drying cycles for most global gene therapies are established here, heavily influencing the specification of downstream united states and canada cold chain packaging requirements.
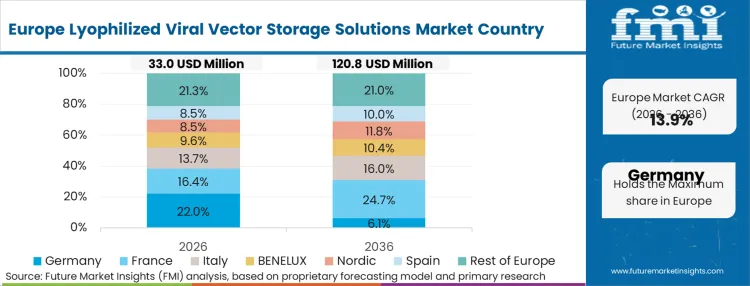
How cost structures and margin dynamics drive decisions heavily influences the European landscape. Public health technology assessment (HTA) bodies aggressively scrutinize the total lifecycle cost of advanced therapies, heavily penalizing assets that require specialized cryogenic handling at the hospital level. To maintain pricing viability, European developers utilize advanced cell therapy manufacturing frameworks to build thermal stability directly into their product profiles. According to FMI's estimates, this economic pressure forces local CDMOs to perfect low-cost, highly scalable drying cycles that meet stringent EMA quality directives without inflating unit costs.
How physical and technical infrastructure conditions shape adoption defines the Asia Pacific dynamic. The lack of reliable -80°C deep-freeze logistical networks outside of major metropolitan hubs mandates that any broadly distributed therapy must achieve ambient or refrigerated stability. Based on FMI's assessment, local CDMOs are responding by rapidly scaling their formulation suites to serve both domestic pipelines and Western sponsors seeking cost-effective secondary manufacturing sites. This necessitates massive investments in controlled environments, including specialized cryo box systems to handle intermediate clinical batches before drying.
FMI's report includes secondary Asian markets and Latin American pharmaceutical hubs actively attempting to build domestic gene therapy capabilities. The recurring pattern across these emerging markets is an intense reliance on imported formulation technology and analytical equipment, creating a lucrative export market for established Western and tier-1 Asian solution providers. Integration of automated sample storage systems across these regions further facilitates the transition to modern commercial formats.
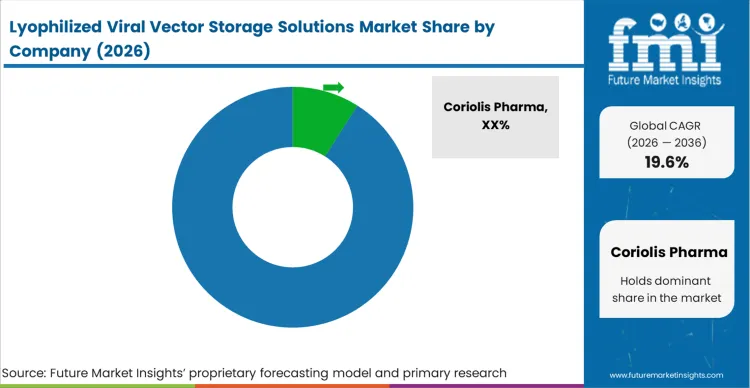
The high technical barrier to successfully stabilising viral capsids creates a highly concentrated market at the premium end. Buyers do not select vendors based on available freeze-dryer shelf space; they qualify partners based exclusively on their biophysical analytical capabilities and proprietary excipient libraries. Companies like Coriolis Pharma and Leukocare dominate because they possess the specialised assays required to prove that the vector has not aggregated during the sublimation phase. Standard fill-finish operators attempting to enter this space routinely fail qualification because they treat viral vectors like resilient monoclonal antibodies, leading to catastrophic batch losses.
To compete, challengers must build comprehensive analytical bridging frameworks rather than simply purchasing newer drying equipment. Incumbents maintain their advantage because they possess historical datasets mapping the glass transition behaviors of multiple AAV and Lentiviral serotypes. Replicating this requires challengers to invest heavily in predictive thermal modeling and high-throughput biophysical characterization. The integration of robust reusable biologics cold chain shippers for handling pre-lyophilized clinical material demonstrates the end-to-end competency that sponsors demand during the vendor selection process.
Large biopharma sponsors actively resist vendor lock-in by attempting to internalize the core formulation intellectual property, treating CDMOs purely as execution partners for the drying cycle. However, the sheer complexity of custom excipient matrices often forces a reliance on the specialized formulation provider. Through 2036, the market will likely see increased vertical integration, with massive generalist CDMOs acquiring specialized boutique formulation labs to close their analytical capability gaps and offer fully integrated viral vector commercialization pathways.
| Metric | Value |
|---|---|
| Quantitative Units | USD 154.6 million to USD 925.8 million, at a CAGR of 19.6% |
| Market Definition | The market covers the specialized formulation, drying cycles, and container systems engineered to preserve viral vector stability outside of deep-frozen states. |
| Vector Type Segmentation | Adeno-associated virus (AAV), Lentiviral vectors, Adenoviral / chimp adenoviral vectors, Oncolytic / VSV vectors, Other viral vectors |
| Solution Type Segmentation | Formulation & excipient systems, Lyophilization cycle development, Analytical stability testing, Fill-finish & container-closure optimization, Tech transfer / scale-up services |
| Storage Target Segmentation | 2°C to 8°C storage solutions, -20°C storage solutions, Controlled ambient / room-temperature aspirational solutions, Hybrid frozen-to-lyo transition solutions |
| Primary Container / Format Segmentation | Glass vials, Serum vials with lyo stoppers, Ampoules, Dual-chamber systems, Custom primary containers |
| Regions Covered | North America, Europe, Asia Pacific, Rest of the World |
| Countries Covered | United States, United Kingdom, Germany, Japan, South Korea, China, India, and 40 plus countries |
| Key Companies Profiled | Coriolis Pharma, Leukocare, Thermo Fisher Scientific (Patheon Viral Vector Services), Recipharm, Curia (formerly AMRI), Sharp Sterile Manufacturing (formerly Berkshire Sterile Manufacturing), Azenta Life Sciences |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews were conducted with CMC and formulation directors at gene therapy developers. The baseline was anchored to active late-stage vector clinical trials. Forecasts were cross-validated using specialized CDMO capacity utilization rates. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Lyophilized Viral Vector Storage Solutions Market in 2026?
The industry valuation reaches USD 154.6 million in 2026. This metric signals that the transition from clinical-scale liquid frozen formats to commercial-scale solid states is actively underway across tier-1 biopharma pipelines.
What will it be valued at by 2036?
The market is expected to expand to USD 925.8 million by 2036. This long-term baseline reflects that the global distribution networks will systematically refuse to absorb the logistics overhead of active cryogenic cooling for mass-market therapies.
What CAGR is projected?
The sector records a 19.6% CAGR over the forecast period. This rate is heavily tied to the volume of late-stage in vivo assets progressing toward regulatory review, where definitive commercial stability profiles must be established.
Which Vector Type segment leads?
Adeno-associated virus (AAV) commands 49.0% of the market. Its dominance is purely a function of its current status as the primary delivery mechanism for systemic gene therapies, forcing sponsors to solve its specific thermal fragility before commercial launch.
Which Solution Type segment leads?
Formulation & excipient systems account for 31.5% of the total share. The intellectual property and technical complexity reside almost entirely in the chemical matrix that protects the vector during water removal, rather than in the drying equipment itself.
Which Storage Target segment leads?
The 2°C to 8°C target represents 42.0% of the market. This profile provides the most pragmatic alignment with standard hospital pharmacy infrastructure without incurring the massive regulatory delays often associated with proving absolute ambient stability.
What drives rapid growth?
The commercial imperative to eliminate -80°C supply chains compels the growth. Therapies locked into ultra-cold infrastructure face severely restricted patient reach, forcing sponsors to transition to scalable, thermally stable formats.
What is the primary restraint?
The extreme biophysical degradation of capsids during sublimation acts as the primary friction point. Formulating a protective matrix requires lengthy, empirical analytical bridging studies that routinely stall clinical timelines if initiated too late.
Which country grows fastest?
India advances at 20.1%, outpacing China's 19.4%. The difference reflects India's aggressive mandate to upgrade domestic CDMO analytical formulation capabilities specifically to capture outsourced stability contracts from Western sponsors.
Why is AAV formulation so critical to commercialization?
AAV capsids are exceptionally prone to aggregation under freezing stress. Establishing a robust excipient matrix early in clinical development is the only mechanism that prevents catastrophic titer loss during subsequent commercial scale-up.
What role do specialized CDMOs play in this market?
Specialized providers bridge the gap between biological production and solid-state stability. Sponsors rely on these entities not just for capacity, but for their proprietary biophysical assays required to validate the integrity of the dried vector.
How does the choice of primary container impact the therapy?
Maintaining an absolute moisture barrier is paramount for solid-state biologics. Deep-vacuum sealing in glass vials prevents atmospheric moisture from triggering premature vector degradation over a multi-year shelf life.
Why do developers struggle with the liquid-to-solid transition?
The tension lies in the timing. Developers often delay the transition until Phase III to save early-stage costs, only to find that the required analytical comparability studies cannot be completed without delaying the commercial launch window.
What makes European procurement dynamics different?
European HTA bodies aggressively penalize the total lifecycle costs associated with cryogenic handling. This forces regional developers to proactively build 2°C to 8°C thermal stability directly into their product profiles to ensure favorable reimbursement.
How is the US market structured?
The sheer volume of FDA pivotal reviews forces domestic sponsors to finalize their formulations. The focus remains heavily on securing integrated CDMO partnerships capable of executing both complex formulation and flawless sterile fill-finish.
What differentiates premium CDMOs from standard fill-finish operators?
Premium vendors possess proprietary historical datasets mapping the glass transition behaviors of specific serotypes. Standard operators lack this predictive thermal modeling capability, often leading to costly batch failures during qualification.
Why is India's trajectory significant?
India's CDMO sector is elevating its technical baseline from simple small-molecule filling to highly complex biological stabilization, shifting the global outsourcing dynamic for late-stage gene therapies.
What hidden costs emerge if stabilization fails?
Sponsors face the immediate write-off of high-value biological batches, coupled with the systemic cost of restarting multi-year stability trials required to satisfy regulatory comparability demands.
How do dual-chamber systems factor into the market?
These systems simplify the point-of-care reconstitution process, eliminating the risk of handling errors by hospital staff. While technically complex to manufacture, they represent the aspirational standard for decentralized commercial administration.
What limits the push for fully ambient (room-temperature) stability?
While ambient storage is the ultimate goal, the chemical interventions required to achieve it often introduce novel excipient toxicity risks that complicate regulatory reviews, making refrigerated targets a safer commercial strategy.
How does the methodology validate these specialized services?
FMI cross-references public clinical pipeline progressions against the specific capacity expansions of tier-1 biomanufacturing suites, ensuring the forecast tracks actual clinical demand rather than theoretical equipment sales.
What is the 2036 end state for this market?
By 2036, lyophilization will represent the default commercial formulation standard for all systemically administered gene therapies, permanently reserving ultra-cold logistical infrastructure exclusively for patient-specific, autologous treatments.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.