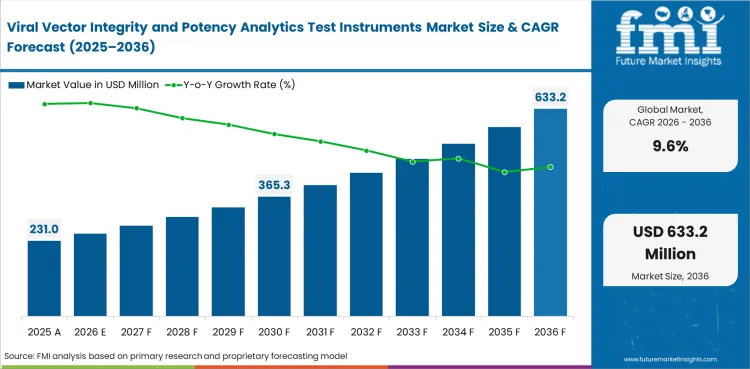
The viral vector integrity and potency analytics test instruments market was valued at USD 211 million in 2025, is projected to reach USD 231 million in 2026, and is forecast to expand to USD 578 million by 2036 at a 9.6% CAGR. Based on Future Market Insights analysis, industry outlook remains positive as later-stage vector programs place more importance on reproducibility, orthogonal confirmation, and assay transfer discipline across development and release-facing work.
| Parameter | Details |
|---|---|
| Market value (2026) | USD 231 million |
| Forecast value (2036) | USD 578 million |
| CAGR (2026 to 2036) | 9.6% |
| Estimated market value (2025) | USD 211 million |
| Leading instrument technology | Digital PCR systems (28.6% of instrument technology segment) |
| Leading assay priority | Genome quantification (24.8% of assay priority segment) |
| Leading vector class | AAV (49% of vector class segment) |
| Leading workflow stage | Release testing (31.4% of workflow stage segment) |
| Leading end user | Biopharma developers (36.7% of end-user segment) |
| Leading throughput | Mid throughput (34.5% of throughput segment) |
| Leading assay mode | Nucleic acid analytics (33.2% of assay mode segment) |
| Leading region | North America (40% regional share) |
| Fastest-growing country | China (11.6% CAGR) |
| Key players | Thermo Fisher Scientific, Bio-Rad Laboratories, Sartorius AG, Agilent Technologies, Waters Corporation, Danaher Corporation, Bruker Corporation |
Source: Future Market Insights, 2026.
Lot readiness depends more on whether one result can be verified by another without long debates on assay reliability or interpretation gaps. Growing activity in adjacent cell and gene therapy clinical trial capabilities is pushing laboratories to build stronger analytical capability instead of relying on a limited set of methods.
China is projected to post 11.6% CAGR through 2036 in this market, followed by South Korea is anticipated to rise at a CAGR of 11.1%, the United Kingdom is poised to expand at a CAGR of 10.2%, Germany is projected to witness a CAGR of 9.4%, the United States is forecast to register at a CAGR of 9.1%, France is projected to grow at a CAGR of 9%, and Japan is expected to reach at a CAGR of 8.8%.
Viral vector integrity and potency analytics test instruments market includes analytical systems used to assess whether viral vectors meet potency, identity, and integrity requirements during development, release, comparability, and stability assessment. Market scope covers instruments used directly for vector characterization and excludes general laboratory equipment that does not serve a defined viral vector analytics purpose.
Scope includes digital PCR, qPCR, chromatography systems, capillary electrophoresis, flow cytometers, bio-layer interferometry systems, analytical ultracentrifuges, and advanced particle readout platforms used in viral vector testing. Configurations used for genome titer and capsid identity are part of the scope. Full-empty evaluation, potency interpretation, and replication competence review are included.
The market excludes upstream bioreactors, vector manufacturing consumables, fill-finish equipment, storage hardware, and routine clinical diagnostics unless they serve a direct viral vector integrity or potency testing role. General laboratory automation is excluded when it has no defined vector analytics function.
Industry outlook is improving as viral vector programs move from exploratory characterization toward tighter control environments. High confidence depends on how well genome, particle, and potency readouts align across the same batch. Instruments that improve repeatability and reduce interpretation gaps are gaining relevance across analytical development and quality workflows. Similar discipline is becoming more visible in cell and gene therapy bioreactor PAT and analytics test platforms, where closer monitoring and decision-grade analytics are becoming more important in platform selection.
AAV-centered programs are adding further momentum because they often require deeper visibility into capsid composition, full-empty evaluation, and payload packaging consistency. Later-stage programs place more value on platforms that support method transfer and internal review discipline without forcing extensive assay rebuilding. Readout complexity in this market also close to analytical expectations seen in chromatography instrumentation, where system relevance rises when separation quality and reproducibility affect decision confidence.
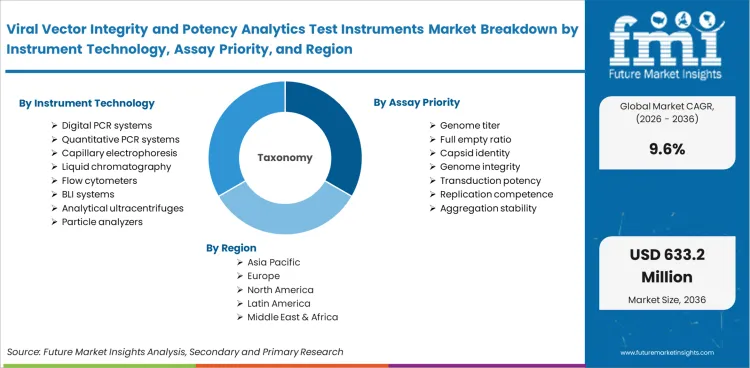
The viral vector integrity and potency analytics test instruments market is segmented by instrument technology, assay priority, vector class, workflow stage, end user, throughput, assay mode, and region. By instrument technology, the market is divided into Digital PCR Systems, Quantitative PCR Systems, Capillary Electrophoresis, Liquid Chromatography, Flow Cytometers, BLI Systems, Analytical Ultracentrifuges, and Particle Analyzers. By assay priority, the market is classified into Genome Titer, Full Empty Ratio, Capsid Identity, Genome Integrity, Transduction Potency, Replication Competence, and Aggregation Stability. Platform relevance across these groups is easier to understand when viewed alongside enabling tool families such as flow cytometry and liquid chromatography systems, both of which support controlled analytical interpretation in adjacent lab environments.
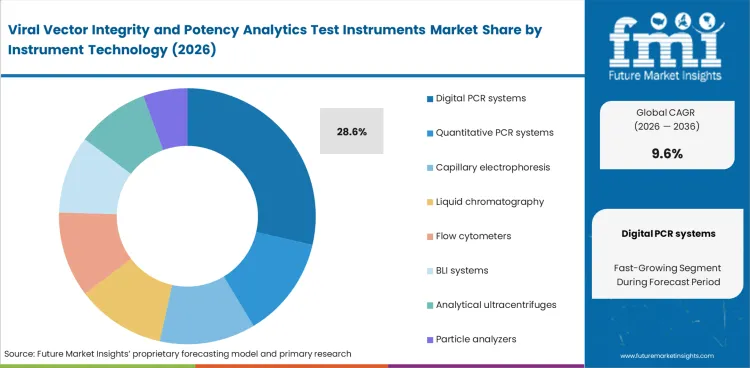
Absolute genome measurement becomes more important as viral vector testing moves into decision-grade analytical work. Digital PCR systems lead this segment because they reduce standard-curve variation and improve repeatability across analytical development and quality workflows. Digital PCR systems are expected to hold 28.6% share in 2026 within instrument technology. Laboratories use them for vector titer, copy number, and residual signal review with more consistent interpretation. Analytical logic in this segment is also close to the operating priorities seen in real-time PCR systems, though viral vector work places stronger emphasis on transfer discipline and orthogonal confirmation.
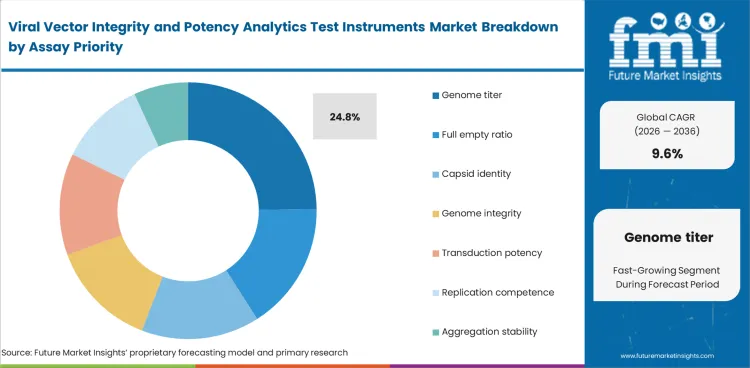
Genome quantification is likely to contribute 24.8% share in 2026 within assay priority because laboratories prefer stable and repeatable setups when early measurement confidence affects downstream interpretation. Testing often starts with measuring genome presence before broader analytical review. This early role also helps explain why molecular tool classes remain commercially relevant in adjacent testing environments such as large molecule bioanalytical testing services.
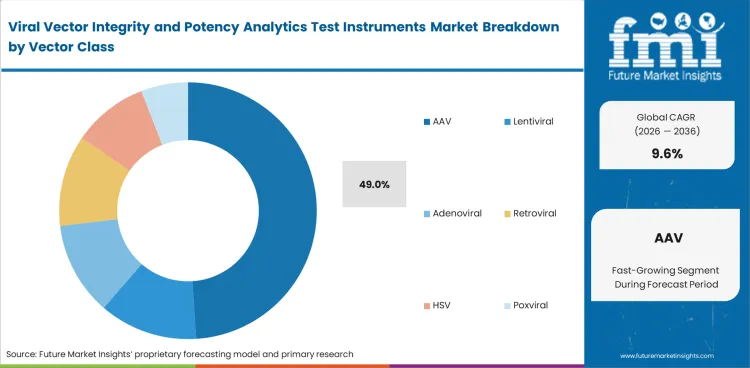
AAV leads vector class demand because active programs often require deeper testing across capsid composition, full-empty analysis, and payload packaging consistency. The segment is projected to hold 49% share in 2026 within vector class. Many laboratories use more than one method before accepting a result, which keeps instrument demand strong in AAV-related work.
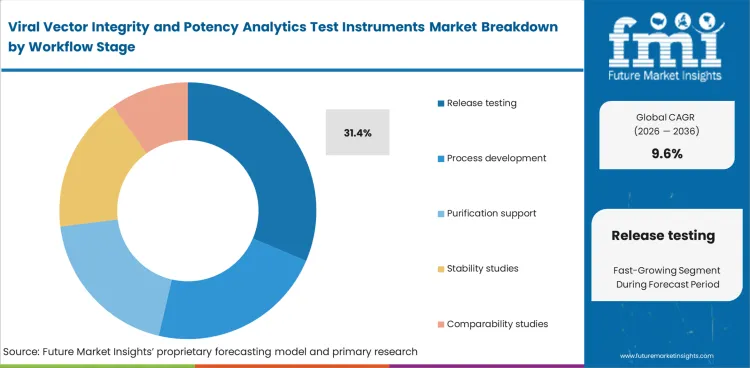
Release testing is expected to account for 31.4% share in 2026 within workflow stage because final lot decisions depend on reliable and repeatable results. Instruments used at this stage become more valuable when they help teams review data clearly and reduce uncertainty during product assessment.
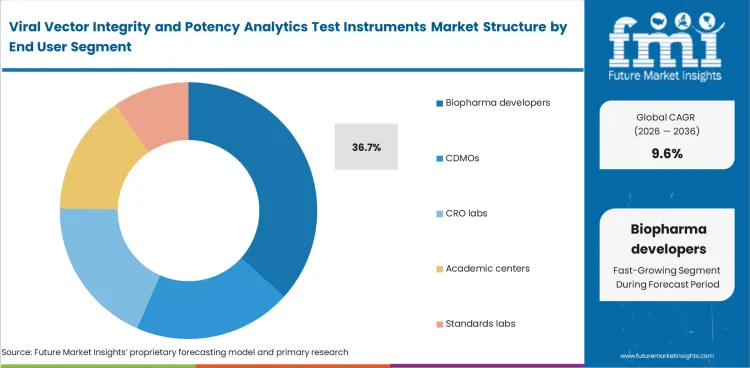
Biopharma developers are expected to represent 36.7% share in 2026 within end-user demand because key analytical work usually stays with sponsors as programs move closer to pivotal and commercial stages. Method selection, data review, and result interpretation often remain under direct sponsor control because product understanding is strongest at that level.
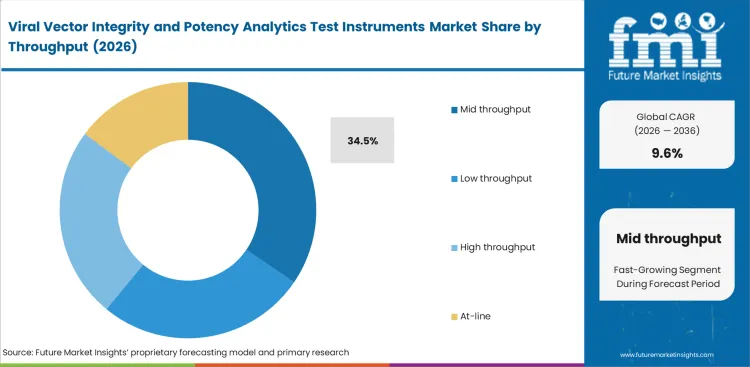
Mid throughput is projected to hold 34.5% share in 2026 because many laboratories need enough capacity to support study work and release-related testing without carrying the utilization risk of oversized systems. This leads comes from balanced bench use rather than maximum installed capacity.
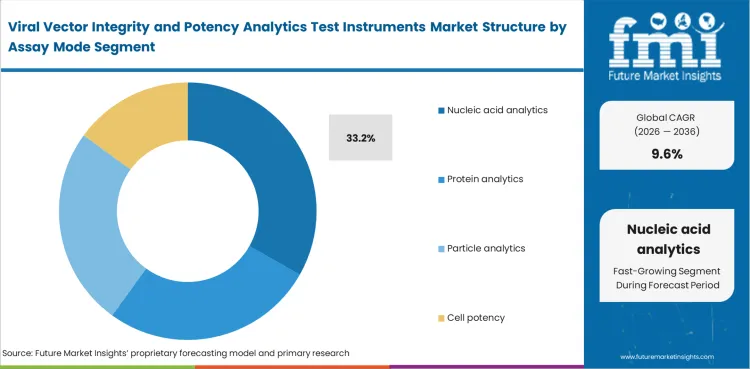
Nucleic acid analytics is expected to account 33.2% share in 2026 within assay mode because genome-linked questions usually come first before laboratories move into particle-focused or cell-based review. Titer and payload interpretation continue to shape assay flow across many viral vector evaluation settings.
The market is expanding steadily as viral vector programs require stronger analytical discipline across release testing, comparability studies, and stability work. Laboratories place more value on platforms that generate results they can defend during internal review. Opportunity continues to widen where instruments support reproducibility, method transfer, and orthogonal confirmation. A similar pattern is visible in cell and gene therapy clinical trial activity, where program advancement increases the need for stronger control logic around data and development decisions.
Process changes often bring instrument purchases forward. Comparability work requires methods strong enough to support a definitive conclusion, which raises the value of platforms that remain dependable through scale-up, site transfer, automation changes, and raw-material adjustments. This level of analytical scrutiny also supports growing interest in advanced characterization tools such as next generation mass spectrometer where large and complex biomolecular analysis is becoming more useful in specialized settings.
.webp)
| Country | CAGR |
|---|---|
| China | 11.6% |
| South Korea | 11.1% |
| United Kingdom | 10.2% |
| Germany | 9.4% |
| United States | 9.1% |
| France | 9% |
| Japan | 8.8% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research.
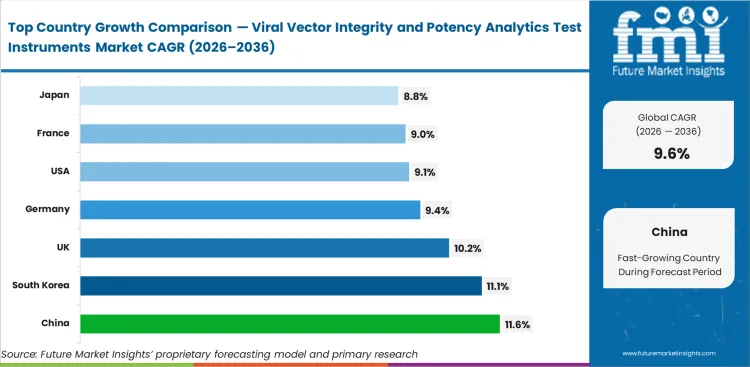
The United States is projected to grow at a 9.1% CAGR through 2036. An established development and manufacturing base, later-stage program activity, and continuing bench refinement support demand in this market. Platform choice often depends on repeatability, method transfer, and how well one readout supports another during product review.
The United Kingdom is expected to rise at a 10.2% CAGR through 2036. Dense gene therapy research activity and a capable analytical services base keep the country ahead of much of Europe. Laboratories often favor platforms that can support development flexibility without forcing a major assay rebuild later.
Germany is projected to rise at a 9.4% CAGR from 2026 to 2036. Reproducibility, operational fit, and controlled workflow compatibility carry strong importance in platform evaluation. Adoption tends to be deliberate, though long-run bench use remains strong once methods are accepted internally.
France is anticipated to record 9% CAGR through 2036. Translational research capability and continued interest in advanced therapy development support the country’s position in this market. Laboratories often favor instruments that help explain why payload, capsid, and potency signals do not align smoothly across batches.
China is anticipated to rise at a CAGR of 11.6% by 2036. Fresh analytical capacity and a broader local advanced therapy base are helping laboratories install wider assay capability from the outset. Current practices can be embedded earlier before legacy bench constraints become harder to manage.
South Korea is projected to grow at 11.1% CAGR through 2036. Manufacturing ambition and disciplined quality practice support a strong position near the top of the country ranking. Laboratories often prefer platforms that deliver dependable results in controlled environments without adding unnecessary assay burden.
Japan is expected to grow at an 8.8% CAGR through 2036. Instrument selection emphasizes repeatable laboratory performance, long-use suitability, and compatibility with controlled workflows. The lower pace than China or South Korea reflects deliberate adoption rather than weak market conditions.
The viral vector integrity and potency analytics test instruments market includes suppliers active across molecular analysis, chromatography, particle characterization, and label-free interaction workflows used in viral vector testing. Thermo Fisher Scientific and Bio-Rad Laboratories hold strong positions because genome quantification remains a central analytical priority, and both companies have broad relevance in decision-grade molecular workflows.
Agilent Technologies, Waters Corporation, and Danaher Corporation compete through established analytical platforms that fit characterization, separation, and control needs across vector development and quality workflows. Sartorius AG remains relevant where assay packages require broader analytical support, while Bruker Corporation serves more specialized environments where method quality and interpretation depth matter more.
Barriers to entry are moderate at the instrument level, though they rise in later-stage analytical use, where laboratories need repeatable performance, transfer-friendly workflows, application support, and confidence across orthogonal readouts before a platform gains routine acceptance.
Key global companies leading the viral vector integrity and potency analytics test instruments market include:
| Company | Molecular Analytics Relevance | Characterization Breadth | Application Support | Method Transfer Fit |
|---|---|---|---|---|
| Thermo Fisher Scientific | High | High | Strong | High |
| Bio-Rad Laboratories | High | Medium | Strong | High |
| Sartorius AG | Medium | Medium | Moderate | Medium |
| Agilent Technologies | Medium | High | Moderate | Medium |
| Waters Corporation | Medium | High | Moderate | Medium |
| Danaher Corporation | Medium | High | Strong | Medium |
| Bruker Corporation | Medium | Medium | Moderate | Medium |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Viral Vector Integrity and Potency Analytics Test Instruments Market
Major Global Players
Emerging / Specialized Participants
| Metric | Value |
|---|---|
| Quantitative Units | USD 231 million to USD 578 million, at a 9.6% CAGR |
| Market Definition | Viral Vector Integrity and Potency Analytics Test Instruments Market covers analytical platforms used to measure genome titer, payload integrity, capsid composition, potency-linked response, and related quality attributes across viral vector development and control activities. Coverage is limited to instruments with a direct viral vector analytics role. |
| Segmentation |
|
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, United Kingdom, Germany, France, Japan, China, South Korea, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Bio-Rad Laboratories, Sartorius AG, Agilent Technologies, Waters Corporation, Danaher Corporation, Bruker Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews and analytical review of public guidance pages, association publications, peer-reviewed viral vector characterization literature, and company technical material were used. Baseline valuation reflects the installed and expanding bench used for integrity, content, potency, and comparability work. Forecasts were validated against later-stage assay intensity, site maturity, and vendor participation in cell and gene therapy analytics. |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the demand for viral vector integrity and potency analytics test instruments in the global market in 2026?
Demand for viral vector integrity and potency analytics test instruments in the global market is estimated to be valued at USD 231 million in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 578 million by 2036.
What is the expected demand outlook between 2026 and 2036?
The market is expected to expand at a 9.6% CAGR between 2026 and 2036.
Which instrument technology is poised to lead by 2026?
Digital PCR systems are expected to account for 28.6% share in 2026.
Which assay priority leads the market?
Genome quantification is expected to hold 24.8% share in 2026 because many workflows begin there.
Which vector class remains ahead?
AAV is projected to hold 49% share in 2026 due to broader analytical depth requirements.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.