High-throughput food allergen screening test instruments market was valued at USD 192.3 million in 2025. Industry valuation is estimated to reach USD 208.4 million in 2026 at a CAGR of 8.4% during the forecast period. Cumulative category buildup takes total valuation to USD 467 million by 2036 as food manufacturers and contract laboratories treat allergen screening as a routine release, sanitation, and label-verification task rather than an occasional bench check.
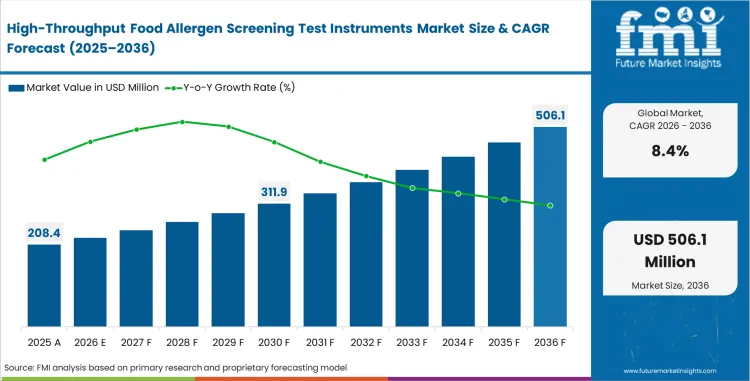
| Metric | Value |
|---|---|
| Market Size (2025) | USD 192.3 million |
| Market Size (2026) | USD 208.4 million |
| Market Size (2036) | USD 467 million |
| CAGR (2026-2036) | 8% |
| Incremental Opportunity | USD 258.6 million |
| Leading Technology | PCR (34%) |
| Leading Instrument Type | PCR Systems (32%) |
| Leading Throughput Format | 96-Well (58%) |
| Leading Sample Matrix | Processed Foods (39%) |
| Leading Application | Finished Foods (34%) |
| Leading End User | Food Manufacturers (43%) |
| Leading Automation Level | Semi-Automated (46%) |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Quality teams in food plants are no longer choosing benches one method at a time. Day-to-day selection now centers on whether a setup can handle extraction, plate work, result review, and follow-up confirmation without slowing batch release or creating another manual checkpoint, and that is pulling this category closer to food allergen testing as a workflow decision instead of a single-device purchase. Faster run speed does not decide the purchase on its own. Laboratories first want repeatable prep, cleaner handoffs between shifts, and a result trail that can stand up during internal review.
Wider bench adoption becomes easier once a laboratory standardizes extraction, plate handling, and result interpretation across sites. Quality teams trigger that condition when allergen checks start sitting beside broader food diagnostics routines instead of staying in a separate corner of the lab. After that point, adding another instrument or another assay places less strain on training, scheduling, and result review.
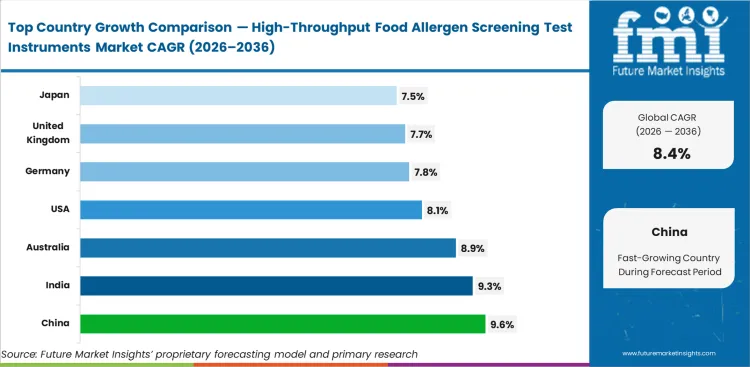
China is expected to record 9.6% CAGR through 2036 as larger packaged-food plants formalize in-house allergen screening. India follows at 9.3%, helped by wider bench adoption in branded food production. Australia is likely to post 8.9% CAGR, while the United States reaches 8.1% as mature laboratories keep refining release workflows. Germany stands at 7.8%, the United Kingdom at 7.7%, and Japan at 7.5%. Countries with closer alignment between routine testing and laboratory information systems usually add capacity with less disruption to daily lab timing.
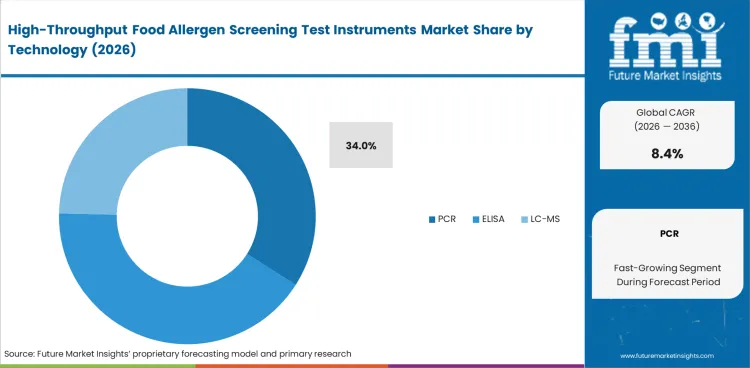
PCR is expected to account for 34% share in 2026 because it fits routine allergen screening across mixed food matrices with less bench disruption than many alternatives. Laboratories working with processed foods need a method that can handle repeated runs, stay manageable during result review, and align with the plate-based logic already familiar from real-time pcr systems. ELISA keeps a firm place in established screening programs, while LC-MS becomes more relevant when deeper follow-up work is required. Digital PCR adds technical precision, yet routine food testing still leans toward methods that match existing extraction, plate handling, and interpretation habits. Laboratories that move toward a technically richer platform without considering daily bench practicality often create more review burden than their teams can comfortably manage.
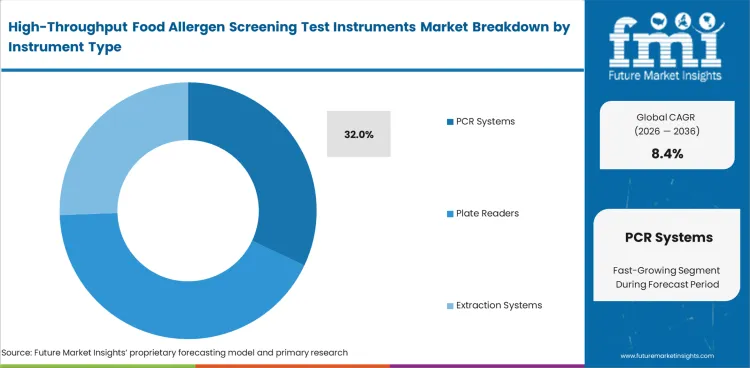
PCR Systems lead this segment because many laboratories build their routine allergen workflow around instruments that can handle repeat screening without adding another layer of bench complexity. By 2026, PCR Systems are projected to contribute 32% of total market share. Position at that level reflects their central role in day-to-day screening, especially in laboratories already comfortable with the workflow logic used in qpcr instruments. Plate Readers remain important where ELISA routines are well established, while Extraction Systems gain attention when prep consistency creates more pressure than detection itself. Mass Spectrometers matter most where broader confirmatory work becomes part of normal laboratory planning. A platform with weak assay compatibility may look workable at first, yet it usually adds slower validation work and more complicated review later.
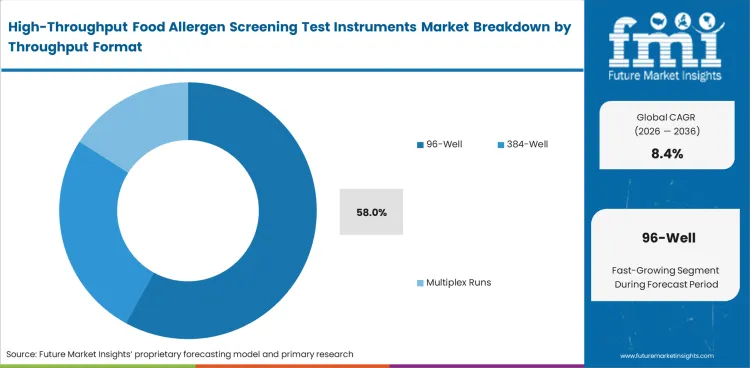
Routine allergen screening depends on more than raw sample volume. Laboratories also need a format that can keep pace with extraction timing, plate setup, result review, and release scheduling through an ordinary working day, which is why 96-Well remains the preferred choice for many sites. It is projected to represent 58% of the market in 2026 because it gives laboratories a workable balance between throughput, validation familiarity, and bench handling control, especially in settings already moving closer to broader lab automation routines. 384-Well options suit larger operations with tighter workflow discipline, while Multiplex Runs appeal where broader analyte coverage matters more than bench familiarity. Batch Benchtop formats still serve smaller operations and lower-volume laboratories. Moving too early toward denser formats often replaces practical bench efficiency with theoretical output that everyday staffing cannot fully capture.
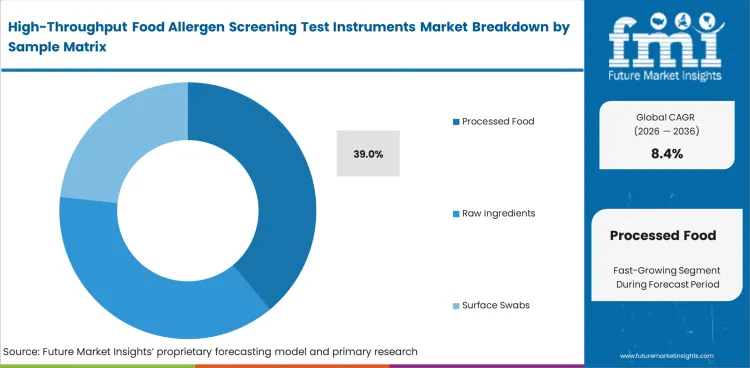
Processed foods keep laboratories under the greatest analytical pressure because ingredients, heat treatment, texture variation, and cross-contact exposure make screening more difficult than it is for a cleaner raw-material sample. Raw Ingredients remain important for supplier control, while Surface Swabs and Rinse Water support sanitation and changeover verification. Processed Foods are expected to make up 39% of the market in 2026, reflecting how often finished formulations create the hardest bench work and the highest label exposure, particularly in settings that also rely on a service laboratory for overflow or confirmation. Sample complexity matters more here than sample count alone. Laboratories that underestimate matrix difficulty often end up with extra prep steps, repeat runs, or outside confirmation they could have avoided with a better bench setup.
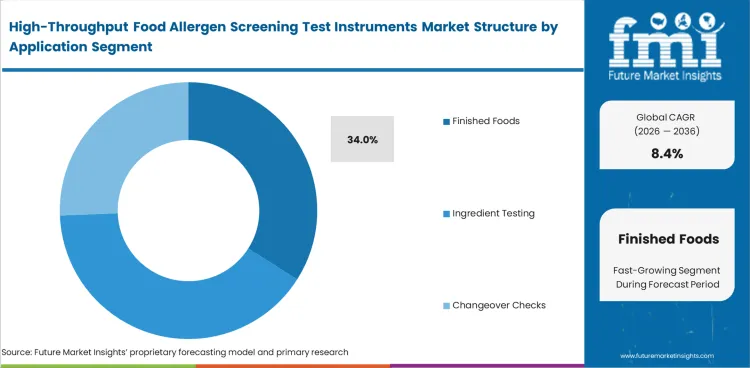
Release decisions carry more commercial weight than most other allergen checks, which keeps Finished Foods at the center of instrument use. Ingredient Testing and Changeover Checks remain necessary, yet final product screening sits closest to the point where a labeling error becomes a shipment hold, rework event, or recall exposure. Label Claims also stay relevant, especially where clean allergen communication supports premium positioning and where analytical laboratory service support is used for overflow or secondary review. Finished Foods are anticipated to represent 34% of the market in 2026. Priority at that level reflects the importance of final-release confidence rather than a simple preference for end-product sampling. Leaving final-release screening too late in the production cycle can turn a small analytical delay into a broader warehouse and scheduling problem.
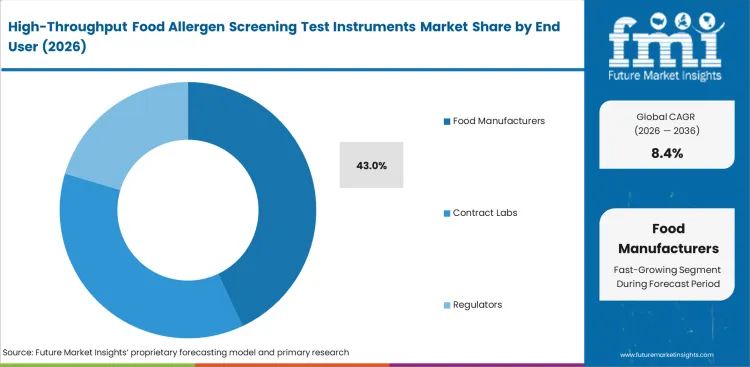
Food Manufacturers are forecast to account for 43% share in 2026 because routine sample ownership remains concentrated inside plant-linked laboratories. In-house teams handle recurring checks tied to supplier control, line clearance, final release, and internal verification, which gives manufacturers a steadier need for dedicated screening capacity than most other user groups. Contract Laboratories still play an important role where smaller processors outsource work or need extra confirmation capacity, while Regulators and Research Labs contribute more selectively. Many manufacturer-linked laboratories also look at adjacent benchtop automation options when trying to reduce manual handling without rebuilding the whole room. Test frequency and workflow immediacy explain why manufacturers stay ahead. User groups without direct exposure to production timing can tolerate longer turnarounds. Plant-linked laboratories usually cannot.
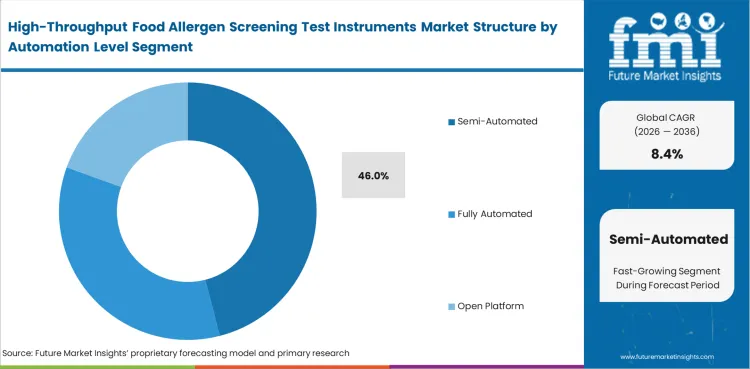
Bench automation creates the most value when it removes repetitive handling without forcing the laboratory into a working model it cannot maintain. Many food laboratories want faster extraction, cleaner result capture, and steadier run timing, yet they still want direct operator control over loading, review, and exception handling. Semi-Automated systems lead because they answer that need better than either a fully manual or fully integrated setup, and they also sit comfortably beside related molecular diagnostics workflows where targeted automation is already familiar. Market estimates place Semi-Automated systems at 46% share in 2026. Fully Automated options remain attractive for larger and more standardized environments, while Open Platform and Standalone setups hold value where method flexibility or budget discipline outweighs full workflow integration. A laboratory can usually absorb targeted automation faster than a full-room redesign, especially where staffing patterns and validation routines are already set.
Based on the regional analysis, the High-Throughput Food Allergen Screening Test Instruments Market is segmented into North America, Latin America, Europe, East Asia, South Asia and Pacific, and Middle East and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 9.6% |
| India | 9.3% |
| Australia | 8.9% |
| United States | 8.1% |
| Germany | 7.8% |
| United Kingdom | 7.7% |
| Japan | 7.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America remains the reference point for routine allergen bench discipline because release timing, internal documentation, and contract-laboratory expectations are already tied closely to food quality practice. Laboratories here rarely need to be convinced that allergen screening belongs inside daily operations. Buying discussions usually center on how far automation can go without adding review burden or creating a bench format that site teams cannot support consistently. Method familiarity, software clarity, service response, and fit with existing result-control routines carry more weight than headline technical claims alone. Closer alignment with digital quality handling also makes this region more receptive to setups that borrow logic from molecular assay environments, provided bench use remains practical for ordinary food testing conditions.
FMI's report includes Canada and Mexico. Food laboratories across those countries add weight to North America through export-linked testing, wider private-label participation, and a stronger preference for methods that can move between internal and outsourced bench environments without lengthy retraining.
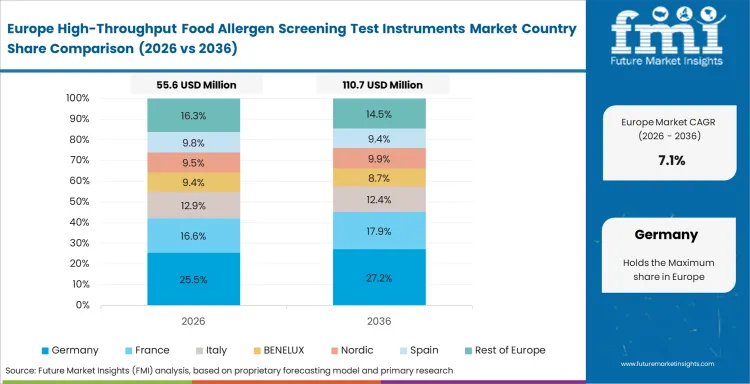
Europe keeps category expansion steady because food labeling discipline, audit culture, and cross-border supply exposure leave little room for weak result handling. Laboratories across the region usually want bench setups that produce dependable records, stay manageable under mixed sample loads, and support repeatable interpretation across shifts. Speed matters, but it does not settle the decision on its own. Review quality, method comfort, and operator consistency carry equal weight in most laboratory settings. Categories such as molecular spectroscopy remain relevant here when laboratories consider how far to widen analytical capability without making routine bench work harder to run.
FMI's report includes France, Italy, Spain, Benelux, and Nordic countries. Food processing and contract-laboratory activity across those areas keep Europe commercially important, especially where mixed product portfolios make method reliability more valuable than headline throughput alone.
Asia Pacific carries the strongest upside because bench formalization is widening across large branded-food operations, export-oriented processing, and independent contract laboratories handling more complex sample volumes. Installed-base maturity still varies sharply from one country to another, which creates very different buying conversations inside the same region. Some laboratories are selecting their first high-throughput setup, while others are refining extraction consistency, bench flow, and result handling after earlier adoption. Adjacent analytical categories such as process spectroscopy matter here less as direct substitutes and more as signs that laboratories are becoming more comfortable with instrument-led food analysis.
FMI's report includes South Korea, ASEAN countries, and New Zealand. Wider adoption across those countries depends less on one single trigger and more on how quickly laboratories can add capacity without stretching staffing, validation effort, or service support beyond what local operations can comfortably carry.
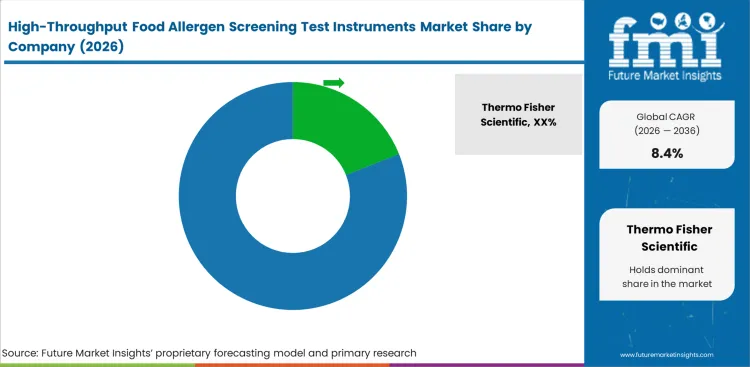
Competition in this category sits in the middle ground between concentration and fragmentation. Thermo Fisher Scientific, Hygiena, Bio-Rad Laboratories, QIAGEN, Bruker Corporation, Revvity, and Waters Corporation all have room to compete, yet laboratories do not choose among them on brand visibility alone. Bench fit, assay compatibility, reporting comfort, service response, and ease of validation decide more deals than headline technical claims. Food laboratories want instruments that reduce repeat work and review burden while still fitting existing SOPs. That is why adjacent categories such as automated molecular diagnostics matter more as workflow reference points than as direct substitutes.
Incumbents hold an edge when they can support both screening and follow-up work without forcing the laboratory to piece together too many disconnected steps. Thermo Fisher Scientific, QIAGEN, and Bio-Rad Laboratories benefit from broad method familiarity, while Hygiena is well placed where routine food safety bench practice matters more than wider laboratory portfolio breadth. Bruker Corporation and Waters Corporation gain attention when confirmation depth becomes part of the selection discussion, and Revvity remains relevant where microplate-based routines stay central. Challengers can still win, especially when they improve service coverage, shorten implementation time, or give laboratories a cleaner path between daily screening and adjacent biosensors or analytical workflows.
Laboratory buying power will keep suppliers from locking in easy advantage through 2036. Larger food producers and multi-site contract laboratories want method portability, manageable validation effort, and vendor support that does not weaken after installation. Preference like that limits how far any supplier can push a closed bench logic if it creates extra retraining or slow review habits. Category direction points toward more connected screening setups, broader follow-up capability, and steadier software-led result control. Suppliers that improve bench usability without overcomplicating daily work are better placed than those trying to sell technical breadth that the laboratory cannot absorb.

| Metric | Value |
|---|---|
| Quantitative Units | USD 208.4 million to USD 467 million, at a CAGR of 8.4% |
| Market Definition | High-throughput food allergen screening test instruments include bench systems used to detect allergen presence across ingredients, finished foods, swabs, and rinse-related samples inside food testing operations. Scope covers instrument-led screening and follow-up confirmation workflows rather than outsourced testing service fees or clinical diagnostics. |
| Technology Segmentation | PCR, ELISA, LC-MS, Digital PCR |
| Instrument Type Segmentation | PCR Systems, Plate Readers, Extraction Systems, Mass Spectrometers |
| Throughput Format Segmentation | 96-Well, 384-Well, Multiplex Runs, Batch Benchtop |
| Sample Matrix Segmentation | Processed Foods, Raw Ingredients, Surface Swabs, Rinse Water |
| Application Segmentation | Finished Foods, Ingredient Testing, Changeover Checks, Label Claims |
| End User Segmentation | Food Manufacturers, Contract Labs, Regulators, Research Labs |
| Automation Level Segmentation | Semi-Automated, Fully Automated, Open Platform, Standalone |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, Japan, Australia, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Hygiena, Bio-Rad Laboratories, QIAGEN, Bruker Corporation, Revvity, Waters Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | FMI combined interviews with quality assurance heads, laboratory managers, and food safety leads. Baseline sizing anchored to installed benches, replacement timing, and workflow fit across PCR, ELISA, extraction, and LC-MS setups. Forecasts were checked against vendor participation, laboratory format mix, and adoption patterns across food manufacturers, contract laboratories, and public testing settings. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive planning, quality planning, and operational benchmarking use
How large is the High-Throughput Food Allergen Screening Test Instruments Market in 2026?
The market is expected to reach USD 208.4 million in 2026. This covers instruments used to test food for allergen traces at higher sample volumes in food plants and laboratories.
What will the High-Throughput Food Allergen Screening Test Instruments Market be worth by 2036?
The market is projected to reach USD 467 million by 2036. This points to wider use of allergen testing instruments in routine food quality checks over the forecast period.
What CAGR is projected for the market from 2026 to 2036?
FMI projects a CAGR of 8.4% for 2026 to 2036. This suggests steady expansion rather than a short burst of buying.
Which Technology segment leads the market?
PCR leads the Technology segment. PCR is expected to account for 34.0% share in 2026 because many laboratories use it for repeatable, high-volume allergen screening across different food types.
Which Instrument Type segment leads the market?
PCR Systems lead Instrument Type. They are widely used because they fit routine allergen testing workflows in food laboratories.
Which Throughput Format segment leads the market?
96-Well leads Throughput Format. FMI expects it to account for 58.0% share in 2026 because it offers a practical balance between sample volume, lab handling, and workflow control.
What is driving the market forward?
Food companies are checking allergens more often during routine production, final product release, and sanitation review. Laboratories need benches that can process more samples without slowing down daily work.
What is the main restraint in this market?
Validation and qualification take time. Laboratories cannot replace a core testing bench quickly because new systems must match existing methods, staff routines, and result records.
Which country is expected to grow the fastest?
China is expected to post the fastest pace, with a CAGR of 9.6% through 2036. Wider in-house testing across packaged food production supports that outlook.
What does this market include?
This market includes high-throughput instruments used to detect food allergens in ingredients, finished foods, swabs, and rinse samples. It mainly covers PCR systems, ELISA readers and washers, extraction platforms, and LC-MS benches used in food testing laboratories.
What is not included in this market?
This market does not include lateral-flow strips, consumables sold on their own, outsourced testing service fees, or clinical allergy diagnostics. Those belong to different product groups and follow different buying patterns.
Why is sample preparation so important in allergen testing?
Fast instruments are useful only when sample prep is consistent. Poor prep can lead to repeat runs, slower review, and less confidence in the final result.
Why do Semi-Automated systems still lead Automation Level?
Semi-Automated systems improve bench speed without forcing a laboratory to rebuild the whole workflow. Many laboratories prefer that step because it is easier to manage and validate.
Why does PCR lead the Technology segment?
PCR works well across many food matrices and fits repeatable plate-based routines. Laboratories also value it because it supports higher sample volumes with clear result interpretation.
Why does 96-Well lead Throughput Format?
96-Well formats are familiar to many laboratories and are easier to fit into daily bench work than denser formats. They support higher output without making routine handling too difficult.
Why do Processed Foods lead the Sample Matrix segment?
Processed foods are harder to test because ingredients, heat treatment, and cross-contact can make allergen screening more complex. That keeps them at the center of instrument use.
Why do Finished Foods lead the Application segment?
Finished foods sit closest to label claims and shipment release. A testing error at that stage can create a hold, rework, or recall risk.
Why do Food Manufacturers lead the End User segment?
Food manufacturers run frequent allergen checks inside their own quality programs. They need regular testing for supplier control, line clearance, sanitation checks, and final release.
Why is China ahead of the other countries in the study?
China has a large packaged food base and more plants are building in-house testing capability. That combination supports faster adoption of high-throughput allergen screening instruments.
Why is India also growing at a strong pace?
India is seeing wider branded food production and more formal laboratory routines. Many facilities are still moving from narrower testing practice toward more regular allergen screening.
Why is Australia growing faster than the United States?
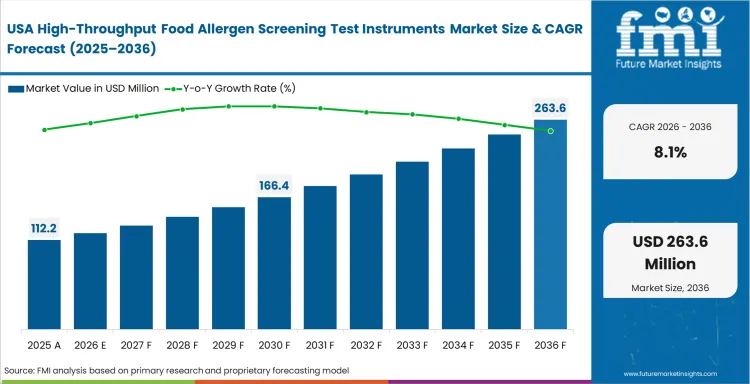
Australia still has room to add more bench capacity, while many larger laboratories in the United States already have an installed base. United States demand remains important, but more of it comes from replacement and workflow refinement.
Why is the United States still a key market?
United States laboratories already run mature food testing programs, which makes it a strong replacement and upgrade market. Suppliers that fit existing methods and reduce review burden are well placed there.
What role does Germany play in this market?
Germany remains important because food manufacturing depth keeps allergen testing close to day-to-day production support. Laboratories there often place high value on method control and steady bench performance.
Why is Japan growing more slowly than some other countries?
Japan usually adopts new laboratory systems in a careful and measured way. Buyers there often want low-disruption integration and dependable result handling before moving ahead.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.