Food Packaging Migration and Barrier Performance Test Instruments Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Food packaging migration and barrier performance test instruments market was is segmented by Test Type (Barrier Testing, Overall Migration, Specific Migration, NIAS Screening, Headspace Testing, Extractables), Analytical Technique (LC-MS, GC-MS, ICP-MS, FTIR, Coulometric, Infrared), Barrier Metric (OTR, WVTR, CO2TR, Aroma, Light, Vacuum), Packaging Material (Plastics, Paperboard, Metal, Glass, Multilayer, Bioplastics), Instrument Format (Benchtop, Floorstanding, Multi-cell, Inline, Portable, Modular), Sample Format (Films, Pouches, Trays, Bottles, Closures, Coatings), End User (Testing Labs, Food Processors, Converters, Resin Makers, Regulators, Universities), and Region. Forecast covers 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Food Packaging Migration and Barrier Performance Test Instruments Market Size, Market Forecast and Outlook By FMI
Food packaging migration and barrier performance test instruments market was was valued at USD 0.82 billion in 2025. Industry valuation is estimated to reach USD 0.88 billion in 2026 at a CAGR of 7.20% during the forecast period. Revenue buildup is expected to take total valuation to USD 1.76 billion by 2036 as food-contact compliance, shelf-life validation, and recycled-content qualification keep migration and barrier testing tied to release decisions rather than optional lab work.
Food Packaging Migration and Barrier Performance Test Instruments Market Summary
- Summary of Paper Cups Market Market valuation is expected to reach USD 19.9 billion by 2036.
- The category is forecast to expand at a 4.5% CAGR from 2026 to 2036. Paper cups generated USD 12.2 billion in 2025.
- Market value is expected to rise to USD 12.8 billion in 2026. Hot beverages are projected to remain the leading end use with 52% share in 2026.
- PE-coated paperboard is set to lead material demand at 58% share in 2026.
- Single-wall cups continue to represent the largest format segment, with 48% share expected in 2026. Among the countries covered, India shows the strongest growth profile through 2036.
Food Packaging Migration and Barrier Performance Test Instruments Market Key Takeaways
| Parameter | Details |
|---|---|
| Market value (2026) | USD 12.8 billion |
| Forecast value (2036) | USD 19.9 billion |
| CAGR (2026 to 2036) | 4.5% |
| Estimated market value (2025) | USD 12.2 billion |
| Leading end use | Hot beverages |
| End-use share (2026) | 52% |
| Leading format | Single-wall cups |
| Format share (2026) | 48% |
| Leading material | PE-coated paperboard |
| Material share (2026) | 58% |
| Leading capacity band | 8 to 12 oz |
| Capacity share (2026) | 46% |
| Leading distribution channel | Distributor-led supply |
| Distribution channel share (2026) | 42% |
| Leading region | East Asia |
| Regional share (2026) | 30% |
| Fastest-growing country | India |
| India CAGR | 6.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
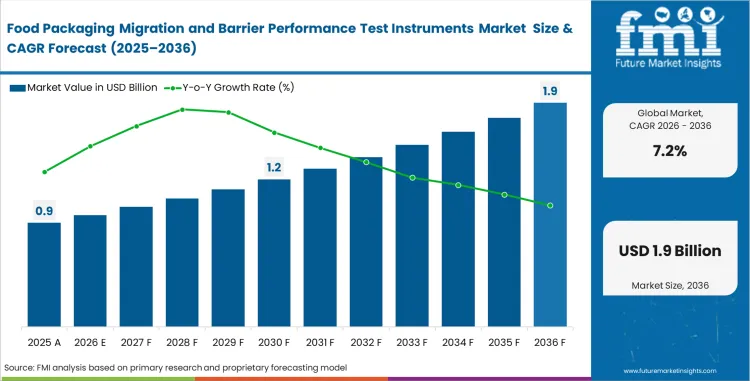
Quality teams are no longer reviewing packaging materials only for basic migration clearance or a routine oxygen transmission result. Current buying decisions increasingly center on whether testing capability should stay scattered across outsourced labs and adapted instruments or move into tighter in-house workflows that support release timing, complaint control, and faster material approval. Multilayer packs, recycled inputs, and low-migration claims add more interpretive burden to package qualification. Buyers with a wider validation agenda are moving closer to combined workflows that connect migration review, permeation measurement, and package performance evidence with adjacent packaging testing equipment decisions.
Installed demand becomes easier to build once converters, food processors, and contract labs begin treating migration and barrier evidence as a routine gate in material sign-off rather than a case-by-case exception. Lab managers and packaging engineers drive that change when they align method selection with release timing instead of audit response alone. Once that habit is in place, replacement demand and method upgrades become easier to justify.
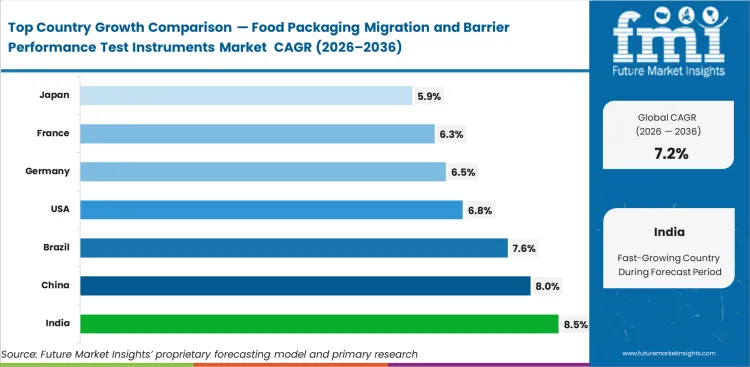
India is projected to record 8.5% CAGR in this market during 2026 to 2036, followed by China at 8%, Brazil at 7.6%, the United States at 6.8%, Germany at 6.5%, France at 6.3%, and Japan at 5.9%. Faster-moving countries are adding test depth while packaged-food output, converter modernization, and compliance burden rise together. Mature markets remain commercially important because accredited lab capacity, installed analytical platforms, and replacement cycles keep valuation anchored even when annual expansion runs lower.
Food Packaging Migration and Barrier Performance Test Instruments Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.88 billion |
| Industry Value (2036) | USD 1.76 billion |
| CAGR (2026-2036) | 7.20% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Raw Material Price Trend Analysis for Packaging Inputs in Food Packaging Migration and Barrier Performance Test Instruments Market
Pricing dynamics for packaging materials used in food packaging migration and barrier performance test instruments are closely aligned with regulatory-grade material requirements, particularly for high-purity polymers, coated paper substrates, aluminum foils, glass containers, and specialty multilayer barrier materials designed to simulate real food-contact conditions. According to the U.S. Bureau of Labor Statistics, Producer Price Index (PPI) data for 2025 indicates continued inflationary pressure across key packaging inputs, with paperboard prices rising by approximately 7-8% year-over-year and aluminum mill shapes recording increases of around 6-8%, while plastic packaging products exhibited moderate monthly fluctuations within the 0.6-1.0% range, reflecting ongoing volatility in petrochemical feedstocks and industrial demand cycles. This pricing environment has resulted in elevated cost sensitivity for high-barrier and migration-testing substrates, where multilayer polymer films and specialty coatings require consistent input quality and stable cost structures to ensure reproducibility in permeability and contamination analysis. Glass and metal-based testing containers continue to reflect energy-driven cost pass-through effects, while paper-based substrates used in absorbent migration testing formats remain exposed to pulp price variability and supply chain adjustments.
From an industry perspective, SGS expanded its food safety and packaging testing capabilities during 2024-2025, including advanced migration and barrier performance testing services aligned with evolving regulatory frameworks such as EU and FDA compliance standards. This development reinforces the growing demand for high-integrity, chemically stable, and contamination-resistant packaging materials capable of supporting precise analytical testing. The convergence of persistent input cost variability and increasing regulatory scrutiny is accelerating the shift toward standardized, high-performance packaging substrates, supporting cost optimization while maintaining analytical accuracy across food packaging testing ecosystems.
Segmental Analysis
Key Facts of Segments
- Test Type: Barrier Testing leads due to its direct linkage with shelf-life validation and package performance under real storage and distribution conditions.
- Analytical Technique: LC-MS dominates as migration studies increasingly require deeper compound identification and low-level detection across complex materials.
- Barrier Metric: OTR remains the most critical metric since oxygen ingress directly impacts product freshness, spoilage risk, and packaging redesign decisions.
- Packaging Material: Plastics account for the largest share due to widespread use across films, trays, pouches, and multilayer packaging formats requiring repeated validation.
- Instrument Format: Benchtop systems hold the highest share, supported by routine lab workflows that prioritize compact setup, repeatability, and operational efficiency.
- Sample Format: Films lead as they represent the first stage of barrier and migration evaluation before conversion into finished packaging formats.
- End User: Testing labs dominate owing to their role in accredited testing, overflow capacity, and documentation-heavy compliance requirements.
- Application: Compliance release remains the primary application, as final approval decisions depend on validated migration and barrier performance evidence.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Test Type
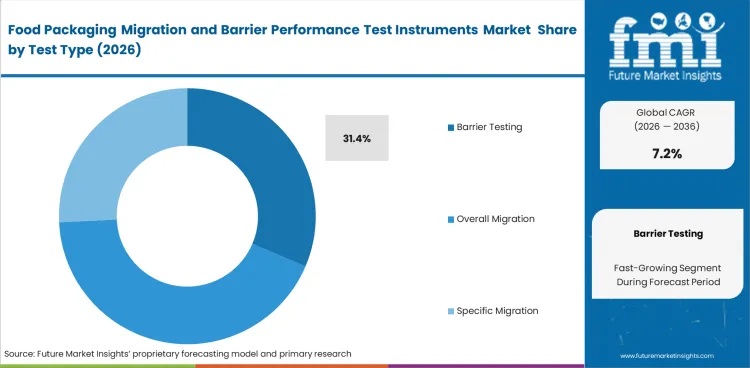
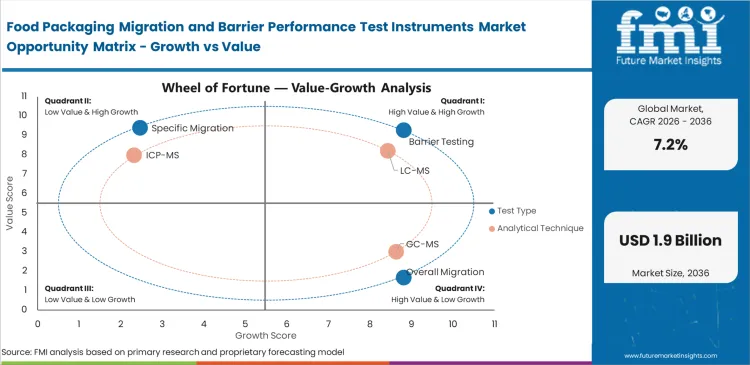
Release discipline stays closest to Barrier Testing because food-contact approval rarely stops at a chemical transfer result alone. Barrier performance determines how long a product remains commercially viable once the pack enters distribution, storage, and retail handling. In 2026, Barrier Testing is expected to account for 31.4% share of this market, supported by its direct relevance to oxygen control, moisture control, and package-gas stability. Quality teams rely on this evidence when a material change could alter spoilage risk even if migration limits remain acceptable. Overall migration and specific migration still matter for compliance, but barrier work often carries clearer day-to-day commercial consequences because shelf-life erosion turns quickly into waste, complaint volume, and line-side revalidation. Buying decisions stay strongest where one instrument can support repeat comparison across films, laminates, trays, and finished packs without slowing routine lab output through adjacent barrier packaging evaluation.
- Shelf-life control: Barrier testing links pack approval with real product life in storage and distribution. Weak evidence here leaves quality teams exposed to faster freshness loss and repeat review cycles.
- Ingress discipline: Oxygen and moisture checks help laboratories distinguish acceptable materials from formats that only appear compliant on paper. Poor screening raises the chance of avoidable pack complaints.
- Release confidence: Routine barrier evidence gives technical teams a firmer basis for final approval when material substitutions enter a live packaging program. Delayed review can push validation burdens later into the commercial schedule.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Analytical Technique
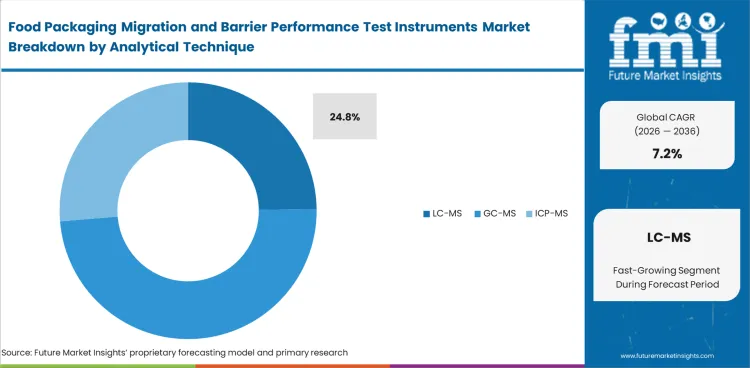
Deeper interpretive burden has kept LC-MS ahead where migration review moves beyond simple known-target checks. Food-contact testing increasingly needs a method that can handle complex mixtures, lower-level compounds, and difficult matrices without leaving too much uncertainty in the result. LC-MS is likely to represent 24.8% of total market share in 2026. Analytical teams favor it where package chemistry, recycled inputs, inks, adhesives, or coatings introduce compounds that require stronger separation and more dependable confirmation. GC-MS and ICP-MS retain clear roles, but LC-MS stays commercially relevant when a lab needs broader coverage without relying on repeated external confirmation. Buying interest becomes firmer once migration work moves from occasional troubleshooting to recurring qualification across multiple pack types. Wrong platform selection rarely fails in one obvious moment; it usually shows up as longer interpretation cycles, repeat sample work, and slower release decisions under related packaging testing services pressure.
- Resolution depth: LC-MS gives laboratories stronger interpretive support when migrants are difficult to isolate or confirm. Limited depth can leave quality teams arguing over borderline results instead of closing a release file.
- Method flexibility: Broader method utility matters when multiple materials and coating systems pass through one testing queue. Narrower fit increases retest burden and slows sample throughput.
- Qualification burden: Better alignment with advanced migration workflows helps labs defend findings during customer review and internal approval. Gaps in method confidence often return as added sign-off delays.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Barrier Metric
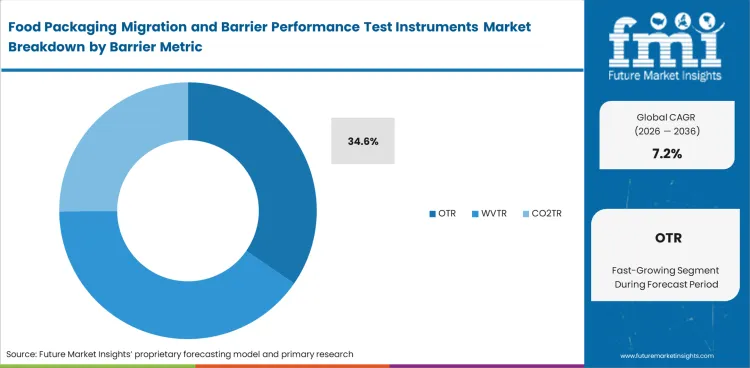
Oxygen control remains one of the clearest commercial questions in food packaging, which is why OTR stays at the center of barrier instrument demand. Spoilage risk, sensory decline, and shorter usable life all become harder to manage when oxygen movement is not measured with enough consistency. Market estimates place OTR at 34.6% share in 2026. Packaging engineers use it as a first screening layer when comparing films, laminates, or coated structures intended to preserve freshness and product stability. WVTR, CO2TR, aroma, and light metrics remain important, but oxygen transmission often enters the decision earliest because its package consequences are widely understood across foods with different moisture and respiration profiles. OTR demand stays strong where test results can be linked quickly to pack redesign, line approval, and shelf-life argument. Poor oxygen data does more than weaken a lab report; it leaves technical teams exposed to avoidable pack-performance disputes inside adjacent high barrier films work.
- Freshness retention: OTR testing gives buyers a clearer read on how well a package protects sensitive products. Weak control can shorten usable life and raise complaint risk.
- Comparison speed: Reliable oxygen data helps technical teams compare candidate materials without extended debate. Slow comparison makes redesign work harder to close.
- Pack-performance linkage: OTR results translate directly into commercial decisions around format approval and material replacement. Unclear results can keep package choices open longer than operations teams can afford.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Packaging Material
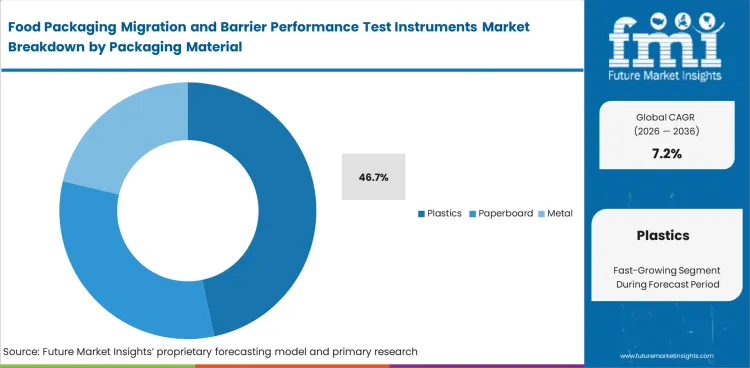
Plastic food-contact formats generate the largest testing load because they dominate the everyday mix of films, pouches, trays, bottles, closures, and multilayer packs used across packaged foods. Material variety within this group also increases the need for repeat migration and barrier review rather than one-time approval logic. Plastics are expected to account for 46.7% share in 2026. Teams working across plastic formats often need both chemical-transfer evidence and pack-performance data before moving a material into routine production. Paperboard, metal, glass, multilayer, and bioplastics remain relevant, yet plastics carry the broadest mix of barrier expectations, additive concerns, coating variation, and package redesign activity. This material class remains commercially central because even small composition or thickness changes can reopen validation questions that were once considered settled. Misreading that burden usually surfaces later as repeat testing, supplier disputes, or release hesitation around linked barrier coated carton decisions.
- Material diversity: Plastic formats cover a wide mix of resins, additives, coatings, and layer combinations. Each change can reopen testing work that quality teams thought was already closed.
- Validation frequency: Repeat review stays high because plastic packs are often the first place where reformulation and downgauging efforts appear. Light oversight can lead to approval gaps later in the cycle.
- Commercial exposure: Plastics sit close to high-volume food applications, so mistakes carry wider consequences than a niche substrate error. One weak approval can create avoidable pressure across multiple SKUs.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Instrument Format
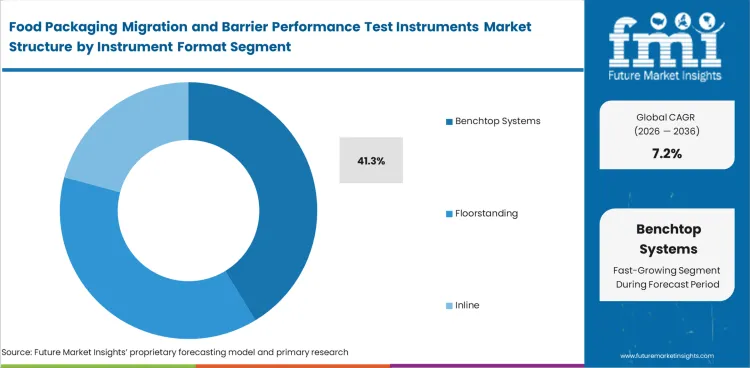
Bench space, method repeatability, and routine throughput keep Benchtop systems ahead in this category. Most migration and barrier workflows still happen in controlled lab settings where compact installation, method discipline, and service accessibility matter more than large-scale integrated layouts. Benchtop systems are projected to emerge with 41.3% market share in 2026. Laboratories prefer them when daily testing volume is steady enough to justify dedicated equipment but not so large that full inline or multi-cell investment becomes necessary across every task. Floorstanding and modular systems remain useful in heavier-duty environments, while portable options serve narrower needs. Benchtop demand stays strongest where labs want dependable operation without rebuilding the room around a single test method. Poor fit at this stage rarely looks like immediate equipment failure; it appears as underused capacity, awkward sample handling, and added service interruptions linked to nearby modified atmosphere packaging equipment workflows.
- Installation fit: Benchtop systems suit laboratories where space, workflow sequence, and sample access must remain manageable. Oversized equipment can burden room layout and slow routine work.
- Method consistency: Stable bench-based operation helps teams maintain repeatable conditions across recurring tests. Inconsistent handling raises the chance of rework and disputed results.
- Service access: Easier maintenance and operator familiarity matter when labs cannot absorb long downtime. Poor equipment fit often turns into avoidable interruptions during release-sensitive periods.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Sample Format
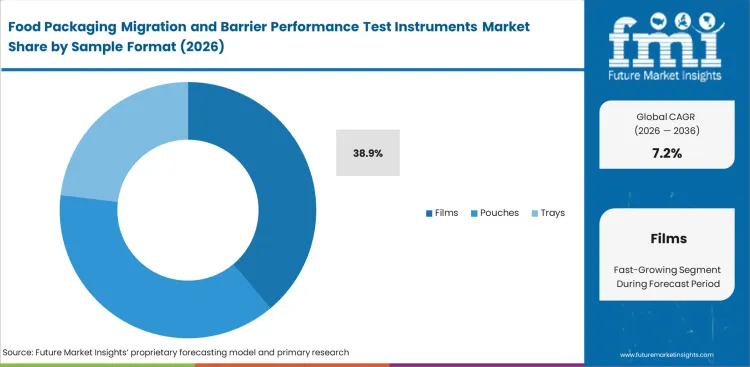
Barrier films remain the most common sample class because much of food packaging performance still starts with web materials before the final pack is formed, sealed, or filled. Technical teams often need film-level evidence early, especially when comparing coatings, laminates, downgauged structures, or recycled-content blends. Films are forecast to represent 38.9% share in 2026. Pouches, trays, bottles, closures, and coatings all carry testing demand, but films sit closest to the first decision point in many package programs. Position gives them a wider role in screening, supplier comparison, and material qualification. Laboratories prioritize film testing because it can reveal whether a candidate material deserves further pack-level work before additional time and conversion cost are committed. When film evidence is weak or delayed, downstream pack evaluation becomes harder to interpret and more expensive to repeat in related barrier packaging in Europe work.
- Early screening: Film samples let laboratories compare barrier behavior before full package conversion begins. Weak early insight can carry avoidable cost into later validation stages.
- Material comparison: Side-by-side film evaluation gives technical teams a cleaner basis for judging downgauged, coated, or recycled-content options. Poor comparison quality slows material selection.
- Conversion discipline: Film data helps determine whether a candidate deserves more pack-level work. Skipping this layer can lead to wasted converter time and repeat approval effort.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by End User
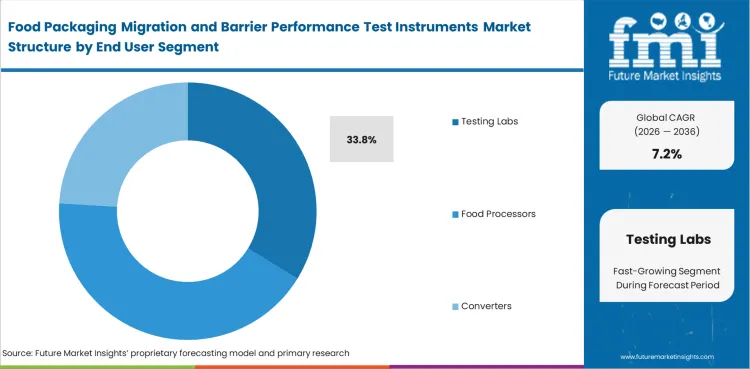
Testing Labs stay ahead because accredited capability, method discipline, and overflow handling remain central to how food-contact evidence is generated and accepted. Many converters and food processors still rely on external lab capacity when test complexity rises or internal queues become too tight. In 2026, Testing Labs are expected to contribute 33.8% of total market share. Their role extends beyond simple execution because they often support method validation, comparability review, and documentation needs tied to release decisions. Food processors, converters, resin makers, regulators, and universities all contribute demand, yet contract and accredited laboratories remain more exposed to recurring multi-client testing loads. This end-user lead persists because many organizations prefer to build selective in-house capability while leaving the heaviest interpretive work with specialist facilities. Underinvestment here does more than create longer lead times; it can weaken confidence in the evidence package carried into adjacent food packaging approval cycles.
- Accredited execution: Testing labs provide a level of method discipline many end users do not maintain across every test type. Gaps in execution quality can weaken acceptance of final results.
- Overflow support: External lab capacity helps organizations manage peaks in validation work without overbuilding internal capability. Limited support can create queue pressure at release-sensitive moments.
- Documentation depth: Testing labs often carry stronger reporting discipline for customer review and conformity checks. Thin documentation can reopen closed questions during approval review.
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Application
Release timing sits at the center of Compliance Release because instrument demand becomes easier to justify when a result directly determines whether a material can move into commercial use. Compliance Release is expected to account for 30.6% share in 2026. Shelf-life work, R&D, failure analysis, recycled-content evaluation, and benchmarking all matter, yet final sign-off remains the most commercially immediate application because it connects testing cost with approval risk, supply continuity, and pack-change accountability. Quality heads and packaging engineers lean toward instruments that can support defensible release evidence rather than exploratory insight alone. This application leads because a package that cannot clear internal or customer review still carries cost even if its technical concept appears promising. Delayed release evidence usually returns as inventory strain, supplier disagreement, or extended reliance on parallel materials around adjacent packaging testing services activity.
- Approval linkage: Compliance release ties instrument output directly to go or no-go decisions on material use. Weak evidence can stall commercial introduction even after other work is complete.
- Audit readiness: Clear release records help technical teams defend package choices during customer and internal review. Poor preparation often reopens decisions that seemed settled.
- Supply continuity: Timely sign-off matters when alternative materials or revised formulations are waiting for clearance. Delay can force temporary workarounds that add handling burden and cost.
Food Packaging Migration and Barrier Performance Test Instruments Market Drivers, Restraints, and Opportunities
Food-contact approval is becoming harder to defend with partial evidence, and that keeps instrument demand tied to release work rather than discretionary laboratory spending. Packaging engineers and quality heads now face a tighter requirement to show that a material can remain suitable for contact and preserve product stability through storage and distribution. Migration review, barrier validation, and package-gas checks increasingly move together when recycled inputs, downgauged films, or low-migration claims enter a program. Industry outlook benefits from that wider burden because buyers need tools that shorten the path from material screening to final approval without sending every complex question outside the business.
Internal coordination remains the biggest brake on faster adoption. Labs may want newer capability, yet budget release, method ownership, sample responsibility, and final sign-off often sit with different teams. That slows equipment decisions even when current workflows are visibly stretched. Outsourced testing can reduce immediate capital pressure, but it also leaves some end users dependent on external queues and less able to compare materials at the pace packaging changes now require. Method validation burden adds another layer because a new instrument is only useful once its output is trusted across the people who approve the pack.
Opportunities in the Food Packaging Migration and Barrier Performance Test Instruments Market
- Recycled-content qualification: Wider use of recycled and reformulated food-contact materials creates room for instruments that help laboratories compare material behavior with less ambiguity. End users that can shorten that review cycle gain a clearer path to package approval.
- Method consolidation: Buyers increasingly value equipment that supports barrier, migration, and package-evidence workflows in a more connected manner. Laboratories that reduce handoff gaps can defend release timing with less back-and-forth.
- Regional lab depth: Expanding packaged-food activity in Asia and Latin America supports stronger demand for local testing capability tied to packaging testing equipment. Suppliers with dependable service coverage and application support stand to benefit where method discipline is being built alongside installed capacity.
Regional Analysis
Key Facts of Country
- India fastest growth at 8.5% CAGR, driven by expanding packaged food consumption, converter upgrades, and increasing compliance requirements tied to food-contact materials.
- China growth at 8% CAGR supported by large-scale manufacturing, export-oriented packaging standards, and rising need for consistent material validation.
- Brazil records 7.6% CAGR with demand shaped by food export requirements and increasing reliance on validated shelf-life and packaging integrity data.
- United States expands at 6.8% CAGR with demand anchored in replacement cycles, advanced lab infrastructure, and strong emphasis on defensible testing outcomes.
- High-quality standards, and structured packaging validation workflows, Germany growth at 6.5% CAGR supported by established testing discipline.
- France registers 6.3% CAGR with steady demand linked to food safety validation and consistent migration and barrier testing practices.
- Reflecting a mature market where demand is sustained by precision testing, method reliability, and equipment replacement cycles, Japan moderates at 5.9% CAGR.
Based on the regional analysis, the Food Packaging Migration and Barrier Performance Test Instruments Market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.5% |
| China | 8% |
| Brazil | 7.6% |
| United States | 6.8% |
| Germany | 6.5% |
| France | 6.3% |
| Japan | 5.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Food Packaging Migration and Barrier Performance Test Instruments Market Analysis
Food packaging test demand in Asia Pacific is being shaped by a mix of expanding packaged-food output, heavier converter modernization, and a wider need to validate materials before they move into high-volume use. Laboratory capacity in this region is becoming more important because packaging programs are handling more reformulation work, more substrate comparison, and more compliance review than they did a few years ago. India and China carry the clearest pull because testing burden rises alongside scale, while Japan contributes a steadier installed base with more mature analytical discipline. Asia Pacific remains commercially important not only for faster annual expansion but also for the way local testing capacity changes approval timing and supplier confidence. Related active and modified atmospheric packaging activity adds another reason barrier and package-gas validation stay relevant.
- India: Converter upgrades, broader packaged-food consumption, and a tighter food-contact review burden are keeping India at the top of the country range. Industry outlook in India points to 8.5% CAGR through 2036, with valuation supported by a rising need to compare materials more rigorously before release. Local laboratories and food processors are under pressure to build cleaner evidence packages as packaging portfolios widen. Faster movement here reflects more than volume; it reflects a stronger need for repeat validation across changing material mixes.
- China: Manufacturing depth and scale keep China central to this category, especially where films, multilayer packs, and export-oriented quality expectations raise the testing load. China is likely to post 8% CAGR in the sector by 2036. Analytical teams benefit from a wide local supply base, but that also raises the burden of comparing substrate quality and consistency across suppliers. Stronger installed capability helps, yet method discipline still matters because a weak approval call can travel across large packaging volumes quickly.
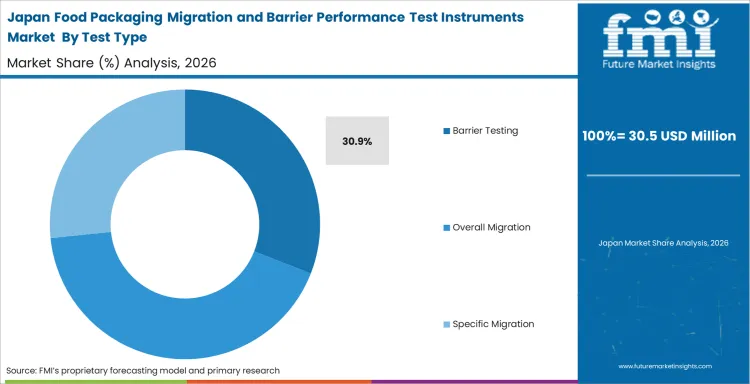
- Japan: Mature analytical practice and deeper lab discipline make Japan a steadier market rather than the fastest one. Industry outlook in Japan is expected to record 5.9% CAGR during 2026 to 2036, supported by repeat replacement cycles, controlled validation routines, and continued emphasis on defensible package evidence. Buyers here tend to favor methods that reduce interpretive uncertainty rather than capacity alone. That keeps valuation anchored even with a lower annual pace.
FMI's report includes South Korea, ASEAN, and Oceania. Regional progression outside the highlighted countries is supported by food-processing expansion, rising compliance burden, and a wider need for local package validation once imported materials and local conversion formats begin to mix in the same supply chain.
North America and Latin America Food Packaging Migration and Barrier Performance Test Instruments Market Analysis
North America and Latin America present two different commercial patterns, yet both remain important for instrument demand. North America carries a deeper installed analytical base and stronger replacement logic, while Latin America offers more room for first-time capability build-out as food exporters, converters, and packers tighten validation discipline. United States valuation stays tied to repeat qualification, shelf-life accountability, and customer-facing evidence needs. Brazil stands out because food-packaging expansion and exporter requirements make test discipline harder to postpone. This region pair is best read through operating urgency: one side values speed and defensibility in established lab systems, while the other is building more local capacity to reduce dependence on thin testing coverage. Adjacent smart food packaging activity keeps package-performance data visible in broader packaging decisions.
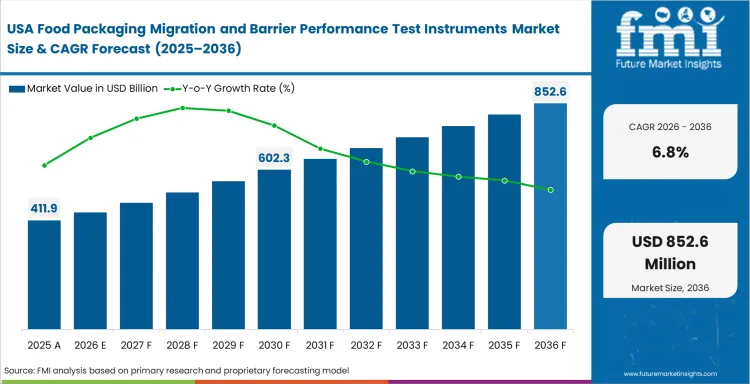
- United States: Established food packaging operations and a dense installed analytical base give the United States a more replacement-led profile. Quality teams still face a sizable burden because material substitutions, recycled-content trials, and shelf-life commitments all need defensible package evidence. United States is forecast to register 6.8% CAGR in this market over the study period. Commercial importance remains high because buyers place strong value on method credibility, service access, and turnaround discipline when release timing is tight.
- Brazil: Food exports, local packaging expansion, and a wider need for shelf-life control make Brazil one of the stronger country opportunities in this study. A CAGR of 7.6% is expected for this market in Brazil through 2036. Testing capability matters here because a weak evidence package can complicate both domestic pack approval and export-facing quality assurance. Suppliers that support method consistency and dependable service timing are better placed to win recurring demand.
FMI's report includes Mexico, Argentina, and the rest of Latin America. Additional country demand in this region is influenced by packaged-food output, exporter requirements, and the practical need to reduce delays caused by limited local test depth.
Europe Food Packaging Migration and Barrier Performance Test Instruments Market Analysis
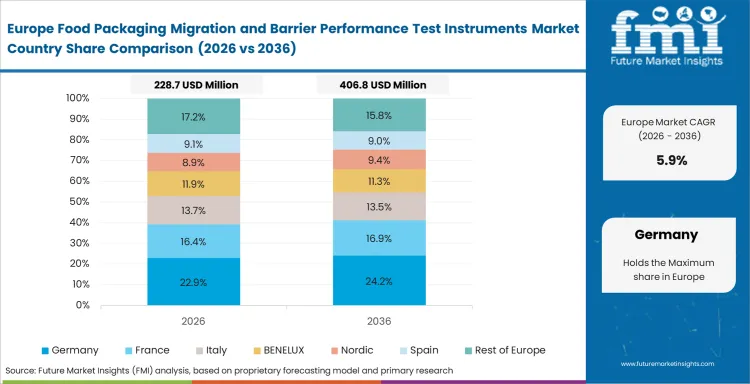
Europe remains commercially important because food-contact review and package-performance discipline are deeply embedded in packaging approval routines. Germany and France do not sit at the top of the CAGR range, yet both carry value because converters, food manufacturers, and contract laboratories already work within more established testing habits. Barrier, migration, and conformity evidence often travel together in package discussions here, which keeps instrument demand closely tied to method reliability and documentation quality. Buyers in Europe care less about adding equipment for its own sake and more about whether it reduces approval uncertainty across films, coatings, laminates, and recycled-content applications. Related high barrier metallized paper snack packaging and barrier packaging activity keeps barrier evaluation visible across the region.
- Germany: Manufacturing discipline, converter depth, and strong technical review culture keep Germany important in this category. Germany is expected to register 6.5% CAGR in the market during the forecast period. Buyers tend to prioritize method repeatability and documentation quality because package approval often travels through multiple technical checkpoints. That makes the country attractive for suppliers that can support dependable performance rather than one-time sales activity.
- France: Food-packaging validation in France stays closely tied to release quality and package suitability across a broad mix of food applications. Industry outlook points to 6.3% CAGR through 2036, supported by continued need for migration review and barrier confirmation where material claims must be defended with cleaner evidence. Laboratories and converters both benefit when methods reduce ambiguity in comparison work. Lower pace than India or China does not reduce importance here; it simply reflects a more mature installed base with steadier replacement demand.
FMI's report includes Benelux and Nordic regions. Food-contact review in these areas supports regional demand because accredited testing, imported material assessment, and package-evidence discipline remain closely tied to converter and food-manufacturer approval routines.
Competitive Aligners for Market Players
Competitive standing in this market depends less on broad catalog size and more on whether suppliers can support dependable package evidence. Buyers compare vendors on method credibility, service responsiveness, application support, and the ease with which results can be defended in internal and customer review. Agilent Technologies, AMETEK MOCON, Labthink, Shimadzu Corporation, Waters Corporation, SCIEX, and Industrial Physics all hold relevant positions because they cover either migration-focused analytical work or barrier and package-performance measurement. Preference tends to favor suppliers that reduce interpretive burden and keep validation work moving when release timing is under pressure.
Incumbents retain an advantage when they combine trusted analytical depth with service coverage and stronger familiarity inside laboratory routines. Barrier specialists benefit from a clearer fit in oxygen, moisture, and package-gas work, while broader analytical suppliers hold ground where migration review requires deeper interpretation. Challengers can still win, but they need more than a competitive instrument. Buyers usually need confidence that installation, training, method setup, and support will hold once testing moves from trial use into recurring package approval under adjacent next generation packaging technology decisions.
Large end users often resist lock-in by keeping migration and barrier work split across different vendor relationships. That limits how far one supplier can dominate the full workflow, even when its installed base is strong. Competitive concentration is therefore likely to remain moderate through 2036 rather than tightening into a narrow-vendor pattern. Reason is straightforward: package validation needs differ across labs, sample types, and approval routines, so buyers keep room for more than one technical path when the cost of a wrong method choice is delayed release and weaker evidence.
Key Players in Food Packaging Migration and Barrier Performance Test Instruments Market
- Agilent Technologies
- AMETEK MOCON
- Labthink
- Shimadzu Corporation
- Waters Corporation
- SCIEX
- Industrial Physics
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.88 billion to USD 1.76 billion, at a CAGR of 7.20% |
| Market Definition | Food Packaging Migration and Barrier Performance Test Instruments Market covers equipment used to evaluate chemical transfer from food-contact materials and to measure package resistance to oxygen, moisture, and related barrier challenges. Scope centers on instruments that support release, qualification, and shelf-life evaluation in food packaging applications. |
| Test Type Segmentation | Barrier Testing, Overall Migration, Specific Migration, NIAS Screening, Headspace Testing, Extractables |
| Analytical Technique Segmentation | LC-MS, GC-MS, ICP-MS, FTIR, Coulometric, Infrared |
| Barrier Metric Segmentation | OTR, WVTR, CO2TR, Aroma, Light, Vacuum |
| Packaging Material Segmentation | Plastics, Paperboard, Metal, Glass, Multilayer, Bioplastics |
| Instrument Format Segmentation | Benchtop, Floorstanding, Multi-cell, Inline, Portable, Modular |
| Sample Format Segmentation | Films, Pouches, Trays, Bottles, Closures, Coatings |
| End User Segmentation | Testing Labs, Food Processors, Converters, Resin Makers, Regulators, Universities |
| Application Segmentation | Compliance Release, Shelf-life, R&D, Failure Analysis, Recycled-content, Benchmarking |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, Brazil, United States, Germany, France, Japan, and 40 plus countries |
| Key Companies Profiled | Agilent Technologies, AMETEK MOCON, Labthink, Shimadzu Corporation, Waters Corporation, SCIEX, Industrial Physics |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews covered laboratory managers, packaging engineers, quality heads, and food-contact specialists involved in package approval. Baseline demand was anchored to attributable instrument exposure across barrier analyzers and migration-focused analytical systems used in food packaging. Forecasts were validated against end-use adoption depth, replacement logic, and regional packaging activity. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Food Packaging Migration and Barrier Performance Test Instruments Market Analysis by Segments
Test Type
- Barrier Testing
- Overall Migration
- Specific Migration
- NIAS Screening
- Headspace Testing
- Extractables
Analytical Technique
- LC-MS
- GC-MS
- ICP-MS
- FTIR
- Coulometric
- Infrared
Barrier Metric
- OTR
- WVTR
- CO2TR
- Aroma
- Light
- Vacuum
Packaging Material
- Plastics
- Paperboard
- Metal
- Glass
- Multilayer
- Bioplastics
Instrument Format
- Benchtop
- Floorstanding
- Multi-cell
- Inline
- Portable
Modular
- Sample Format
- Films
- Pouches
- Trays
- Bottles
- Closures
- Coatings
End User
- Testing Labs
- Food Processors
- Converters
- Resin Makers
- Regulators
- Universities
Application
- Compliance Release
- Shelf-life
- R&D
- Failure Analysis
- Recycled-content
- Benchmarking
Region
- North America
- United States
- Latin America
- Brazil
- Europe
- Germany
- France
- East Asia
- China
- Japan
- South Asia
- India
- Oceania
- Middle East and Africa
Bibliography
- U.S. Bureau of Labor Statistics. (2025, July 16). Producer Price Indexes-June 2025.
https://www.bls.gov/news.release/archives/ppi_07162025.htm - U.S. Food and Drug Administration. (2024, July 24). Food Packaging & Other Substances that Come into Contact with Food. U.S. Food and Drug Administration. https://www.fda.gov/food/food-ingredients-packaging/food-packaging-other-substances-come-contact-food-information-consumers
- Consumer Affairs Agency. (2025, June 20). Questions & Answers of the Positive List System. Consumer Affairs Agency. https://www.caa.go.jp/policies/policy/standards_evaluation/appliance/notice/assets/standards_cms101_250808_01.pdf
- Gupta, R. K., Sharma, S., Kumar, V., & Dhawan, V. (2024, September 30). Migration of Chemical Compounds from Packaging Materials: A Comprehensive Review on the Impact on Food Safety. Foods. https://www.mdpi.com/2304-8158/13/19/3125
- Phelps, D. W., De Atley, S., Nguyen, N. T., & Peaslee, G. F. (2024, March 19). Per- and Polyfluoroalkyl Substances in Food Packaging. Environmental Science & Technology. https://doi.org/10.1021/acs.est.3c03702
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary source documentation.
This Report Addresses
- Market intelligence to support strategic decision making across overall migration testing, specific migration testing, NIAS screening, extractables review, oxygen transmission testing, water vapor transmission testing, headspace gas analysis, and related food-contact package validation workflows
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by food-contact approval logic, instrument exposure review, replacement-cycle assessment, and application mapping across release, shelf-life, and material qualification use cases
- Growth opportunity mapping across Test Type, Analytical Technique, Barrier Metric, Packaging Material, Instrument Format, Sample Format, End User, and Application with emphasis on repeat validation needs at food packaging operating sites
- Segment and regional revenue forecasts covering Barrier Testing, LC-MS, OTR, Plastics, Benchtop Systems, Films, Testing Labs, and Compliance Release across key food-packaging geographies
- Competition assessment including method credibility, service reliability, application support, installation fit, documentation quality, turnaround discipline, and result defensibility in live food-contact testing environments
- Product development tracking across chromatography-linked migration review, mass spectrometry workflows, permeation analyzers, package-gas analysis systems, bench-based validation equipment, and related food packaging test platforms
- Market access analysis covering food processors, packaging converters, resin suppliers, contract testing labs, regulatory bodies, and research institutions where equipment utility depends on dependable lab operation and repeatable package evidence
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive strategy, packaging category planning, technical evaluation, and operating benchmark use
Frequently Asked Questions
What is Food Packaging Migration and Barrier Performance Test Instruments Market?What CAGR is expected from 2026 to 2036?
Food Packaging Migration and Barrier Performance Test Instruments Market covers equipment used to test whether food packaging releases unwanted chemicals and whether it can block oxygen, moisture, and other outside elements that affect product quality.
How much was the market worth in 2025?
Food Packaging Migration and Barrier Performance Test Instruments Market was valued at USD 0.82 billion in 2025.
What is the estimated market size in 2026?
Industry valuation is estimated at USD 0.88 billion in 2026.
What is the forecast value by 2036?
Food Packaging Migration and Barrier Performance Test Instruments Market is projected to reach USD 1.76 billion by 2036.
What CAGR is expected from 2026 to 2036?
Food Packaging Migration and Barrier Performance Test Instruments Market is expected to expand at a CAGR of 7.20% during the forecast period.
Which Test Type leads the market?
Barrier Testing is expected to lead the Test Type segment with 31.4% share in 2026.
Why does Barrier Testing lead the market?
Barrier Testing stays important because it helps packaging teams judge how well a pack protects food from oxygen, moisture, and gas exposure during storage and distribution.
Which Analytical Technique leads the market?
LC-MS is projected to lead the Analytical Technique segment with 24.8% share in 2026.
Why is LC-MS widely used in this market?
LC-MS is widely used because it helps laboratories examine complex migration questions where packaging materials contain compounds that are harder to identify or confirm.
Which Barrier Metric leads the market?
OTR is expected to lead the Barrier Metric segment with 34.6% share in 2026.
What does OTR mean in food packaging testing?
OTR means oxygen transmission rate. It shows how much oxygen can pass through a packaging material and helps explain how well the pack can protect product freshness.
Which Packaging Material leads the market?
Plastics are expected to lead the Packaging Material segment with 46.7% share in 2026.
Why do plastics create the largest testing load?
Plastics are used across films, pouches, trays, bottles, and multilayer packs, so they generate a wider mix of migration checks and barrier studies than most other materials.
Which Instrument Format leads the market?
Benchtop systems are projected to lead the Instrument Format segment with 41.3% share in 2026.
Why are benchtop systems preferred?
Benchtop systems fit routine lab work well because they support repeat testing in controlled conditions without requiring large installation space.
Which Sample Format leads the market?
Films are expected to lead the Sample Format segment with 38.9% share in 2026.
Why are films tested so often?
Films are often checked early in package development because they help teams compare barrier performance before a full pack is produced.
Which End User leads the market?
Testing Labs are projected to lead the End User segment with 33.8% share in 2026.
Why do Testing Labs lead the market?
Testing Labs lead because they handle accredited studies, overflow testing, and documentation-heavy work that many food processors and converters still send outside.
Which Application leads the market?
Compliance Release is expected to lead the Application segment with 30.6% share in 2026.
Why is Compliance Release the leading application?
Compliance Release leads because many testing decisions are tied directly to whether a material can move into approved commercial use.
Which country shows the fastest outlook in this study?
India shows the fastest outlook, with 8.5% CAGR expected through 2036.
How do the other key countries compare?
China is projected at 8%, Brazil at 7.6%, the United States at 6.8%, Germany at 6.5%, France at 6.3%, and Japan at 5.9% for 2026 to 2036.
What is the main restraint in this market?
A major restraint is slow internal coordination, because budget release, method ownership, sample handling, and final approval often sit with different teams.
What do companies usually compare when selecting suppliers?
Most supplier evaluations focus on method credibility, service responsiveness, application support, and how easily test results can be defended during review.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Barrier Testing
- Overall Migration
- Specific Migration
- Barrier Testing
- Y-o-Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Analytical Technique
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Analytical Technique, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Analytical Technique, 2026 to 2036
- LC-MS
- GC-MS
- ICP-MS
- LC-MS
- Y-o-Y Growth Trend Analysis By Analytical Technique, 2021 to 2025
- Absolute $ Opportunity Analysis By Analytical Technique, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Barrier Metric
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Barrier Metric, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Barrier Metric, 2026 to 2036
- OTR
- WVTR
- CO2TR
- OTR
- Y-o-Y Growth Trend Analysis By Barrier Metric, 2021 to 2025
- Absolute $ Opportunity Analysis By Barrier Metric, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging Material, 2026 to 2036
- Plastics
- Paperboard
- Metal
- Plastics
- Y-o-Y Growth Trend Analysis By Packaging Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging Material, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Instrument Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Instrument Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Instrument Format, 2026 to 2036
- Benchtop Systems
- Floorstanding
- Inline
- Benchtop Systems
- Y-o-Y Growth Trend Analysis By Instrument Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Instrument Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sample Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sample Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sample Format, 2026 to 2036
- Films
- Pouches
- Trays
- Films
- Y-o-Y Growth Trend Analysis By Sample Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Sample Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Testing Labs
- Food Processors
- Converters
- Testing Labs
- Y-o-Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Analytical Technique
- By Barrier Metric
- By Packaging Material
- By Instrument Format
- By Sample Format
- By End User
- Competition Analysis
- Competition Deep Dive
- Agilent Technologies
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AMETEK MOCON
- Labthink
- Shimadzu Corporation
- Waters Corporation
- SCIEX
- Industrial Physics
- Agilent Technologies
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 8: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 15: North America Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 16: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 22: Latin America Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 23: Latin America Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 24: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 29: Western Europe Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 30: Western Europe Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 31: Western Europe Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 32: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 36: Eastern Europe Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 37: Eastern Europe Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 38: Eastern Europe Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 39: Eastern Europe Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 40: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 43: East Asia Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 44: East Asia Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 45: East Asia Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 46: East Asia Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 47: East Asia Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 48: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 50: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 51: South Asia and Pacific Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 52: South Asia and Pacific Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 53: South Asia and Pacific Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 54: South Asia and Pacific Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 55: South Asia and Pacific Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 56: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 57: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 58: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 59: Middle East & Africa Market Value (USD Million) Forecast by Analytical Technique, 2021 to 2036
- Table 60: Middle East & Africa Market Value (USD Million) Forecast by Barrier Metric, 2021 to 2036
- Table 61: Middle East & Africa Market Value (USD Million) Forecast by Packaging Material, 2021 to 2036
- Table 62: Middle East & Africa Market Value (USD Million) Forecast by Instrument Format, 2021 to 2036
- Table 63: Middle East & Africa Market Value (USD Million) Forecast by Sample Format, 2021 to 2036
- Table 64: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Analytical Technique
- Figure 9: Global Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Barrier Metric
- Figure 12: Global Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Packaging Material
- Figure 15: Global Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Instrument Format
- Figure 18: Global Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Sample Format
- Figure 21: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by End User
- Figure 24: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 25: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 26: Global Market Attractiveness Analysis by Region
- Figure 27: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 32: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 33: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 34: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 35: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Test Type
- Figure 38: North America Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Analytical Technique
- Figure 41: North America Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Barrier Metric
- Figure 44: North America Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Packaging Material
- Figure 47: North America Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Instrument Format
- Figure 50: North America Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 51: North America Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 52: North America Market Attractiveness Analysis by Sample Format
- Figure 53: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 54: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 55: North America Market Attractiveness Analysis by End User
- Figure 56: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 57: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Test Type
- Figure 60: Latin America Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Analytical Technique
- Figure 63: Latin America Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Barrier Metric
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging Material
- Figure 69: Latin America Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 70: Latin America Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 71: Latin America Market Attractiveness Analysis by Instrument Format
- Figure 72: Latin America Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 73: Latin America Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 74: Latin America Market Attractiveness Analysis by Sample Format
- Figure 75: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 76: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 77: Latin America Market Attractiveness Analysis by End User
- Figure 78: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 79: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Test Type
- Figure 82: Western Europe Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Analytical Technique
- Figure 85: Western Europe Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Barrier Metric
- Figure 88: Western Europe Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 89: Western Europe Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 90: Western Europe Market Attractiveness Analysis by Packaging Material
- Figure 91: Western Europe Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 92: Western Europe Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 93: Western Europe Market Attractiveness Analysis by Instrument Format
- Figure 94: Western Europe Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 95: Western Europe Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 96: Western Europe Market Attractiveness Analysis by Sample Format
- Figure 97: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 98: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 99: Western Europe Market Attractiveness Analysis by End User
- Figure 100: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Analytical Technique
- Figure 107: Eastern Europe Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 108: Eastern Europe Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 109: Eastern Europe Market Attractiveness Analysis by Barrier Metric
- Figure 110: Eastern Europe Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 111: Eastern Europe Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 112: Eastern Europe Market Attractiveness Analysis by Packaging Material
- Figure 113: Eastern Europe Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 114: Eastern Europe Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 115: Eastern Europe Market Attractiveness Analysis by Instrument Format
- Figure 116: Eastern Europe Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 117: Eastern Europe Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 118: Eastern Europe Market Attractiveness Analysis by Sample Format
- Figure 119: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 120: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 121: Eastern Europe Market Attractiveness Analysis by End User
- Figure 122: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 123: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Test Type
- Figure 126: East Asia Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 127: East Asia Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 128: East Asia Market Attractiveness Analysis by Analytical Technique
- Figure 129: East Asia Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 130: East Asia Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 131: East Asia Market Attractiveness Analysis by Barrier Metric
- Figure 132: East Asia Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 133: East Asia Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 134: East Asia Market Attractiveness Analysis by Packaging Material
- Figure 135: East Asia Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 136: East Asia Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 137: East Asia Market Attractiveness Analysis by Instrument Format
- Figure 138: East Asia Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 139: East Asia Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 140: East Asia Market Attractiveness Analysis by Sample Format
- Figure 141: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 142: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 143: East Asia Market Attractiveness Analysis by End User
- Figure 144: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 145: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 146: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 147: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 148: South Asia and Pacific Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 149: South Asia and Pacific Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 150: South Asia and Pacific Market Attractiveness Analysis by Analytical Technique
- Figure 151: South Asia and Pacific Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 152: South Asia and Pacific Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 153: South Asia and Pacific Market Attractiveness Analysis by Barrier Metric
- Figure 154: South Asia and Pacific Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 155: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 156: South Asia and Pacific Market Attractiveness Analysis by Packaging Material
- Figure 157: South Asia and Pacific Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 158: South Asia and Pacific Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 159: South Asia and Pacific Market Attractiveness Analysis by Instrument Format
- Figure 160: South Asia and Pacific Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 161: South Asia and Pacific Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 162: South Asia and Pacific Market Attractiveness Analysis by Sample Format
- Figure 163: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 164: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 165: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 166: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 167: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 168: Middle East & Africa Market Y-o-Y Growth Comparison by Test Type , 2026-2036
- Figure 169: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 170: Middle East & Africa Market Value Share and BPS Analysis by Analytical Technique, 2026 and 2036
- Figure 171: Middle East & Africa Market Y-o-Y Growth Comparison by Analytical Technique, 2026-2036
- Figure 172: Middle East & Africa Market Attractiveness Analysis by Analytical Technique
- Figure 173: Middle East & Africa Market Value Share and BPS Analysis by Barrier Metric, 2026 and 2036
- Figure 174: Middle East & Africa Market Y-o-Y Growth Comparison by Barrier Metric, 2026-2036
- Figure 175: Middle East & Africa Market Attractiveness Analysis by Barrier Metric
- Figure 176: Middle East & Africa Market Value Share and BPS Analysis by Packaging Material, 2026 and 2036
- Figure 177: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging Material, 2026-2036
- Figure 178: Middle East & Africa Market Attractiveness Analysis by Packaging Material
- Figure 179: Middle East & Africa Market Value Share and BPS Analysis by Instrument Format, 2026 and 2036
- Figure 180: Middle East & Africa Market Y-o-Y Growth Comparison by Instrument Format, 2026-2036
- Figure 181: Middle East & Africa Market Attractiveness Analysis by Instrument Format
- Figure 182: Middle East & Africa Market Value Share and BPS Analysis by Sample Format, 2026 and 2036
- Figure 183: Middle East & Africa Market Y-o-Y Growth Comparison by Sample Format, 2026-2036
- Figure 184: Middle East & Africa Market Attractiveness Analysis by Sample Format
- Figure 185: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 186: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 187: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 188: Global Market - Tier Structure Analysis
- Figure 189: Global Market - Company Share Analysis

