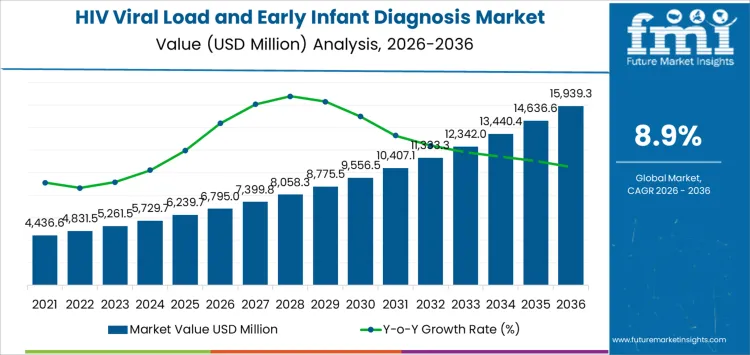
The HIV viral load and early infant diagnosis market is projected to reach USD 6,795.0 million in 2026 and expand to USD 15,939.3 million by 2036, at a CAGR of 8.9%. Value expansion reflects the central role of molecular testing within long-term HIV treatment monitoring and maternal-child health programs rather than episodic diagnostic demand. Testing volumes are structurally linked to antiretroviral therapy coverage, treatment adherence monitoring, and pediatric case identification protocols embedded in national healthcare systems.
Expenditure remains resilient because viral load measurement directly determines clinical decision pathways, regimen switching, and program performance assessment. Assay sensitivity requirements, throughput capacity, instrument placement models, and service obligations, positioning testing platforms as infrastructure-bound healthcare assets rather than discretionary laboratory consumables, influence pricing dynamics.
Momentum across the forecast period is shaped by persistence of treatment-as-prevention strategies and institutional reliance on quantitative viral suppression metrics. Health systems depend on routine viral load testing to replace surrogate markers and reduce delayed detection of treatment failure. Early infant diagnosis demand remains structurally anchored to prevention of mother-to-child transmission frameworks, where turnaround time and diagnostic certainty affect survival outcomes.
Operational complexity related to reagent supply continuity, data connectivity, and quality assurance constrains rapid substitution of platforms once installed. Funding structures, including public budgets and donor-backed procurement, introduce planning discipline but also stabilize long-term volumes. Value growth remains tied to expansion of testing coverage, decentralization of diagnostic networks, and sustained integration of viral load and infant diagnosis within standardized HIV care pathways rather than short-term screening initiatives or prevalence-driven fluctuations.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 6,795.0 million |
| Market Forecast Value (2036) | USD 15,939.3 million |
| Forecast CAGR 2026 to 2036 | 8.9% |
Demand for HIV viral load and early infant diagnosis (EID) testing is increasing as healthcare providers and national programs focus on improving treatment outcomes and reducing transmission among key populations. Viral load assays measure the quantity of HIV RNA in blood to monitor antiretroviral therapy effectiveness, enabling clinicians to adjust regimens when replication persists. Early infant diagnosis identifies HIV infection in newborns born to mothers living with HIV, addressing a window where maternal antibodies can confound conventional antibody tests. Laboratories and hospital systems evaluate platforms based on sensitivity at low viral levels, throughput capacity, and integration with laboratory information systems because reliable results influence clinical decisions, public reporting, and patient care pathways. Procurement teams prioritize platforms with scalable capacity, reagent supply reliability, and service support to ensure uninterrupted testing across reference centers and peripheral health facilities that share treatment responsibilities.
Growth in global HIV treatment targets, expansion of mother-to-child transmission prevention initiatives, and alignment with public health monitoring frameworks reinforce uptake of advanced diagnostic solutions. Health ministries and programs adopt viral load and EID testing to track suppression rates and measure program performance against international goals for epidemic control. Investments in decentralized testing infrastructure support point-of-care and laboratory-based platforms that reduce turnaround time, improve linkage to care, and enable targeted interventions. Manufacturers of diagnostics are enhancing multiplex capabilities and reducing per-test cost to better serve resource-constrained settings while meeting quality standards. These clinical, operational, and public health drivers are contributing to sustained demand growth in the HIV viral load and early infant diagnosis market.
Demand for HIV viral load and early infant diagnosis testing is shaped by treatment monitoring protocols, elimination of mother-to-child transmission programs, and decentralization of diagnostics. Health systems prioritize timely, accurate results to guide therapy decisions and pediatric case identification. Adoption reflects expansion of testing coverage, integration of point-level diagnostics, and sample logistics optimization. Selection criteria emphasize analytical sensitivity, turnaround time, and suitability for resource-variable settings. Segmentation explains how assay format, clinical purpose, and testing setting influence utilization patterns across HIV care pathways.
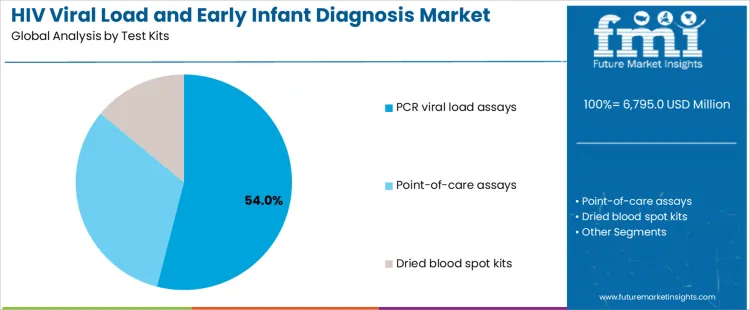
PCR viral load assays account for 54.0%, reflecting their role as reference methods for quantitative monitoring and confirmatory diagnosis. These assays provide high sensitivity and reproducibility essential for longitudinal patient management. Point-of-care assays at 32.0% support decentralized testing with faster results, improving linkage to care in remote settings. Dried blood spot kits at 14.0% facilitate sample transport and storage where cold-chain access is limited. Test-kit segmentation highlights preference for laboratory-grade accuracy complemented by formats improving access and operational feasibility.
Key Points
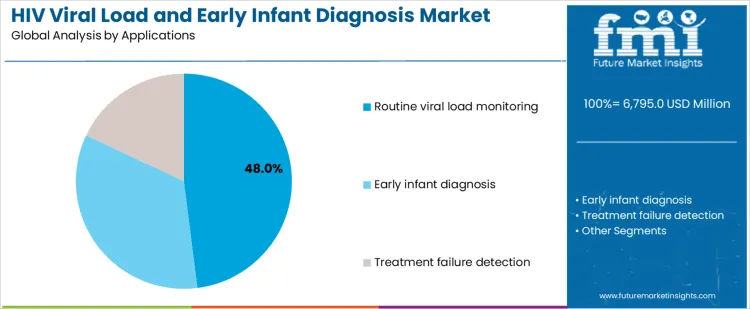
Routine viral load monitoring represents 48.0%, driven by treatment guidelines requiring regular assessment of therapy effectiveness. Early infant diagnosis at 34.0% addresses timely identification of perinatal infections to enable rapid intervention. Treatment failure detection at 18.0% supports regimen adjustments based on virologic response. Application-based segmentation reflects concentration where testing directly informs clinical decision-making, program performance tracking, and pediatric outcomes within HIV care frameworks.
Key Points
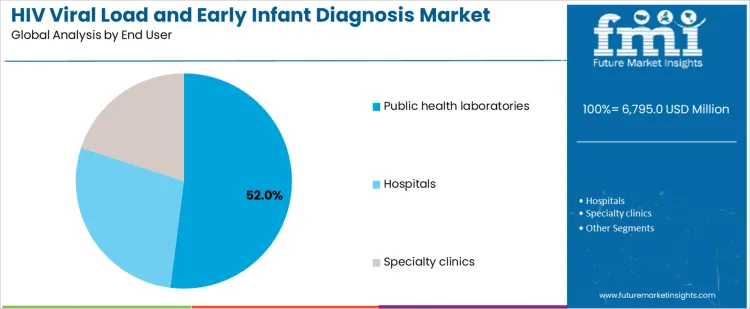
Public health laboratories account for 52.0%, reflecting centralized testing for national programs and surveillance initiatives. Hospitals at 28.0% conduct testing for inpatient and outpatient HIV management. Specialty clinics at 20.0% focus on targeted populations and integrated care models. End-user segmentation indicates concentration where testing volume, programmatic oversight, and diagnostic infrastructure support sustained utilization across HIV services.
Key Points
Demand for HIV viral load and early infant diagnosis testing reflects clinical necessity to monitor treatment effectiveness and identify infection in newborns with maternal exposure. Adoption concentrates in public health laboratories, hospital networks, and decentralized testing sites supporting HIV care programs. Global scope aligns with treatment-as-prevention strategies and maternal-child health initiatives. Usage centers on nucleic acid amplification assays, point-of-care platforms, and centralized laboratory systems delivering quantitative viral load results and definitive infant diagnosis.
Effective HIV management requires precise measurement of viral suppression to guide antiretroviral therapy adjustments. Demand increases where routine viral load testing replaces CD4 monitoring to assess treatment success and transmission risk. Early infant diagnosis addresses diagnostic gaps created by maternal antibody interference in serological testing. Timely identification enables rapid initiation of therapy during critical developmental windows. Program design emphasizes turnaround time, sample stability, and access in remote settings. Dried blood spot compatibility expands reach beyond centralized laboratories. Integration with prevention of mother-to-child transmission programs strengthens testing continuity. Adoption reflects clinical reliance on accurate, early detection and longitudinal monitoring to improve survival and reduce onward transmission.
Testing platforms require stable power, trained personnel, and quality assurance systems, limiting deployment in resource-constrained areas. Demand sensitivity rises where reagent cost and instrument maintenance exceed program budgets. Sample transport delays affect result timeliness, particularly for centralized testing models. Point-of-care systems reduce delays yet face throughput and cartridge supply limitations. Data connectivity challenges hinder result reporting and patient follow-up. Regulatory approval and procurement cycles vary across regions, affecting standardization. Sustained funding dependence introduces volatility in testing volumes. Scalability remains constrained by infrastructure readiness, operational cost exposure, and need for reliable end-to-end diagnostic networks.
Demand for HIV viral load and early infant diagnosis testing is expanding globally as treatment monitoring and prevention of mother-to-child transmission programs scale. Health systems emphasize routine viral suppression assessment to guide therapy optimization and resistance management. Point-of-care platforms and centralized laboratory expansion improve access and turnaround times. International funding, national guidelines, and procurement frameworks influence adoption consistency. Growth rates in India at 10.3%, China at 10.1%, Brazil at 9.7%, the USA at 8.5%, and Germany at 8.4% indicate sustained expansion driven by programmatic testing requirements, laboratory capacity scaling, and outcome-focused HIV management rather than screening-led volume growth.
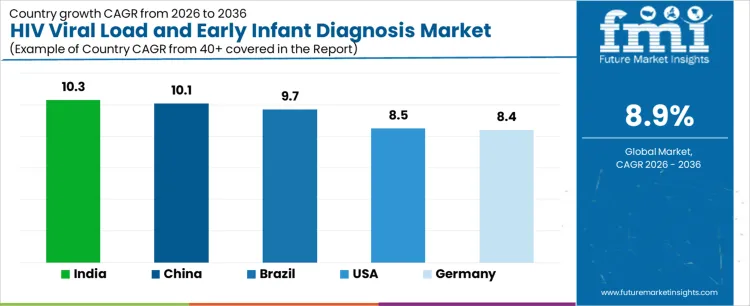
| Country | CAGR (%) |
|---|---|
| India | 10.3% |
| China | 10.1% |
| Brazil | 9.7% |
| USA | 8.5% |
| Germany | 8.4% |
HIV viral load and early infant diagnosis demand in India is growing at a CAGR of 10.3%, supported by expansion of national treatment monitoring programs. Public health facilities increase routine viral load testing to assess therapy adherence and suppression. Prevention of mother-to-child transmission initiatives expand early infant testing coverage. Decentralized laboratories reduce turnaround times in high-burden states. International donor support complements domestic procurement. Growth reflects program scale-up, protocol adherence, and capacity expansion rather than increases in HIV incidence.
HIV viral load and early infant diagnosis demand in China is expanding at a CAGR of 10.1%, aligned with standardized infectious disease management frameworks. Centralized reference laboratories process high testing volumes with consistent quality control. Routine viral load monitoring supports long-term therapy management. Maternal and child health programs integrate early infant diagnosis within broader screening pathways. Domestic assay production improves supply security. Growth reflects systematized monitoring and centralized capacity rather than outreach-led testing expansion.
HIV viral load and early infant diagnosis demand in Brazil is growing at a CAGR of 9.7%, driven by universal treatment access and monitoring mandates. Public health networks require regular viral load assessment to guide regimen adjustments. Early infant diagnosis supports timely initiation of pediatric therapy. Regional laboratories extend testing reach beyond major cities. Legal frameworks ensure access to diagnostic services. Growth reflects institutional commitment to comprehensive HIV care rather than episodic testing initiatives.
HIV viral load and early infant diagnosis demand in the United States is expanding at a CAGR of 8.5%, supported by guideline-driven care and payer coverage. Routine viral load testing is embedded within long-term HIV management protocols. Early infant diagnosis supports prevention of perinatal transmission. Advanced laboratory automation ensures high throughput and accuracy. Public health surveillance programs reinforce consistent testing intervals. Growth reflects protocol adherence and care continuity rather than changes in disease prevalence.
HIV viral load and early infant diagnosis demand in Germany is growing at a CAGR of 8.4%, shaped by evidence-based infectious disease management. Certified laboratories provide standardized viral load assessment. Health insurance coverage supports routine monitoring without access barriers. Early infant diagnosis remains integrated within maternal care pathways. Emphasis on quality assurance ensures testing consistency. Growth remains steady, reflecting structured care models and compliance-driven testing rather than rapid volume escalation.
Demand for HIV viral load and early infant diagnosis testing is shaped by treatment monitoring mandates, prevention programs, and elimination targets. National programs and reference laboratories evaluate assay sensitivity at low copy thresholds, turnaround time, sample type flexibility, and throughput reliability. Buyer assessment includes compatibility with dried blood spots, decentralized testing suitability, reagent stability, and data connectivity for surveillance reporting. Procurement behavior reflects donor funding cycles, tender-based purchasing, instrument placement models, and service coverage in low resource settings. Trend in the HIV viral load and early infant diagnosis market shows continued decentralization, faster result delivery, and integration with national treatment monitoring systems.
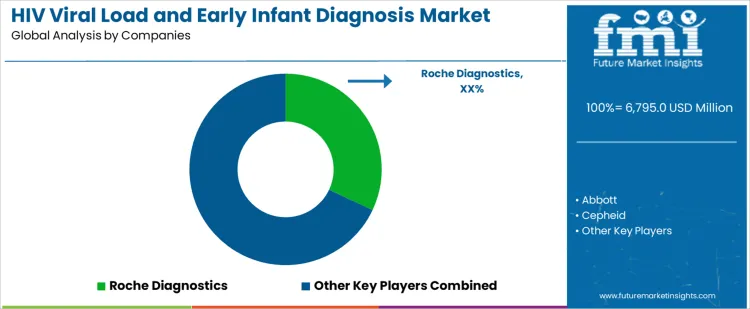
Roche Diagnostics leads competitive positioning through high throughput platforms widely adopted for centralized viral load and infant testing programs. Abbott competes with scalable systems supporting both laboratory and near patient deployment across national networks. Cepheid supports demand through cartridge-based molecular platforms enabling rapid turnaround in decentralized and rural settings. Hologic maintains relevance by supplying high sensitivity molecular assays aligned with reference laboratory workflows. bioMérieux participates with molecular solutions supporting confirmatory testing and program expansion initiatives. Competitive differentiation centers on detection sensitivity, throughput scalability, service infrastructure strength, and alignment with public health procurement frameworks.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Test Kits | PCR viral load assays; Point-of-care assays; Dried blood spot kits |
| Applications | Routine viral load monitoring; Early infant diagnosis; Treatment failure detection |
| End User | Public health laboratories; Hospitals; Specialty clinics |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Roche Diagnostics; Abbott; Cepheid; Hologic; bioMérieux; Others |
| Additional Attributes | Dollar sales by test kit type, application, end user, and sales channel; analytical sensitivity and lower limit of detection for PCR and point-of-care viral load assays; turnaround time advantages supporting early infant diagnosis programs; compatibility of dried blood spot sampling with decentralized testing and transport logistics; role in treatment monitoring and detection of virologic failure; procurement dynamics driven by public health tenders, donor-funded programs, and hospital laboratory testing volumes. |
How big is the HIV viral load and early infant diagnosis market in 2026?
The global HIV viral load and early infant diagnosis market is estimated to be valued at USD 6,795.0 million in 2026.
What will be the size of HIV viral load and early infant diagnosis market in 2036?
The market size for the HIV viral load and early infant diagnosis market is projected to reach USD 15,939.3 million by 2036.
How much will be the HIV viral load and early infant diagnosis market growth between 2026 and 2036?
The HIV viral load and early infant diagnosis market is expected to grow at a 8.9% CAGR between 2026 and 2036.
What are the key product types in the HIV viral load and early infant diagnosis market?
The key product types in HIV viral load and early infant diagnosis market are pcr viral load assays, point-of-care assays and dried blood spot kits.
Which applications segment to contribute significant share in the HIV viral load and early infant diagnosis market in 2026?
In terms of applications, routine viral load monitoring segment to command 48.0% share in the HIV viral load and early infant diagnosis market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.