Fragrance-Free Cleaning Chemicals for Sensitive Environments Market
Market Forecast 2036: USD 5.8 Billion | CAGR 2026–2036: 7.2% | Market Size 2025: USD 2.7 Billion | Incremental Opportunity: USD 2.9 Billion | Market Size 2026: USD 2.89 Billion | Growth Driver: General Surface Cleaners | Key Demand: Commercial Buildings
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Size, Market Forecast and Outlook By FMI
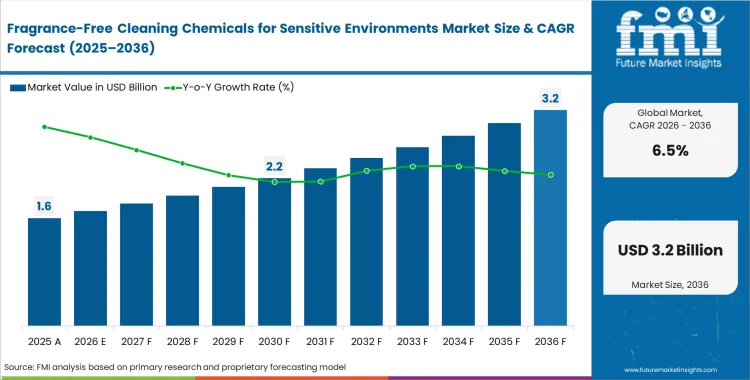
The fragrance-free cleaning chemicals for sensitive environments market valuation stood at USD 1.5 billion in 2025. Valuation is expected to reach USD 1.60 billion in 2026 and expand at a CAGR of 6.5% through 2036, taking total revenue to USD 3.0 billion, as healthcare and institutional buyers shift qualification criteria from basic cleaning performance toward strict indoor air quality and sensory-neutral standards.
Summary of Eco-Label Certified Cleaning Chemicals for Professional Users Market
- The market is estimated at USD 2.7 billion in 2025.
- The market is projected to reach USD 5.8 billion by 2036.
- The market is expected to grow at a CAGR of 7.2% from 2026 to 2036.
- The forecast period represents an incremental opportunity of USD 2.9 billion.
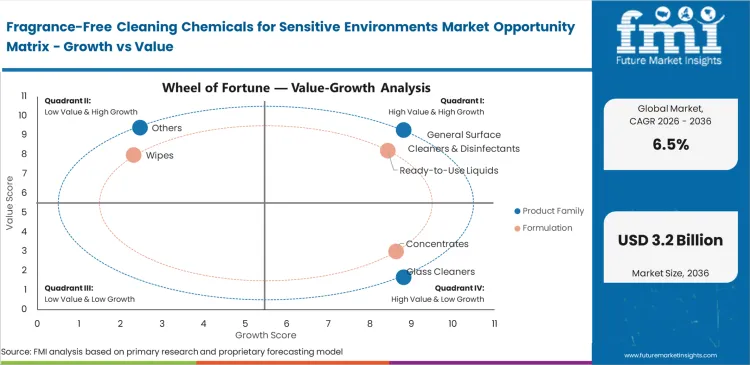
- General surface cleaners lead the product segment with a 28.0% share.
- Commercial buildings dominate the end-use segment with a 33.0% share.
- Concentrated formats lead the format segment with a 37.0% share.
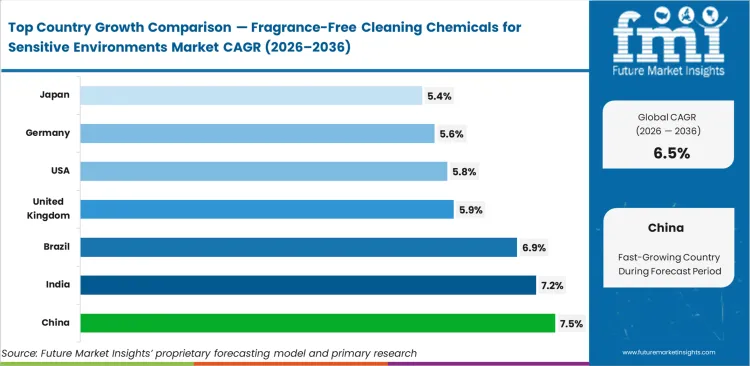
- China (8.2%) and Brazil (8.0%) are among the fastest-growing markets.
- The United States (6.9%) and the United Kingdom (6.9%) show steady growth trends.
- Key companies in the market include Ecolab, Diversey, SC Johnson Professional, 3M, Zep, Spartan Chemical, and Werner & Mertz Professional.
Facility management teams are balancing tighter hygiene protocols with a rising volume of occupant sensitivity complaints, particularly in healthcare, education, and shared public environments. Contract renegotiations are increasingly centered on formulations that deliver broad-spectrum pathogen control without any residual scent, as odor neutrality becomes a measurable requirement rather than a preference. Delays in adopting updated cleaning protocols increase exposure to liability where chemically sensitive or asthmatic occupants report adverse reactions following routine cleaning cycles. Product approvals depend on whether suppliers can demonstrate both indoor air quality improvement and operational efficiency, creating a narrow acceptance range for formulations that meet both conditions. Maintaining complete odor neutrality often requires trade-offs in chemical concentration, which complicates bulk purchasing decisions for cost-sensitive buyers managing large facilities.
Adoption accelerates when large healthcare networks enforce zero-emission or low-VOC compliance across multiple properties, requiring alignment with approved supplier lists. Integrated dosing systems standardize application, reduce manual dilution errors, and help control overall chemical usage while ensuring adherence to occupational safety requirements in enclosed and high-traffic environments.
China is projected to expand at a CAGR of 7.5% through 2036, followed by India at 7.2% and Brazil at 6.9%, reflecting stronger growth across developing institutional and healthcare infrastructure. More established markets show steadier expansion, with the United Kingdom at 5.9% and the United States at 5.8%. Germany is expected to grow at 5.6%, while Japan records 5.4%, indicating demand driven more by replacement cycles and compliance upgrades than new facility additions.
Segmental Analysis
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by Product Family
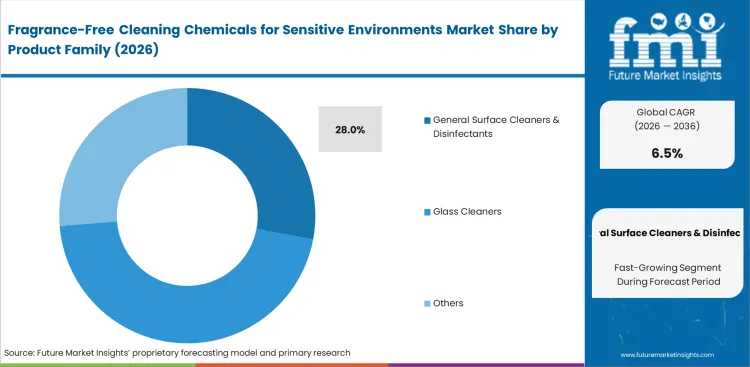
General surface cleaners & disinfectants hold 28.0% share, driven by mandatory infectious disease protocols requiring continuous application across high-touch areas without introducing volatile compounds. Housekeeping supervisors face intense pressure completing room assignments under strict time limits while maintaining absolute olfactory neutrality. FMI's analysis indicates multi surface disinfectant formulations win evaluations only when contact times drop below three minutes without leaving residual chemical scents.
Choosing heavily perfumed products forces custodians to abandon protocol, ventilating rooms extensively before patients or students can safely re-enter. High-frequency chemical application of specialized floor cleaning detergent degrades porous stones, causing directors to quietly seek milder fragrance-free general surface cleaners despite measurable efficacy drops. Delaying formulation upgrades leaves operators vulnerable to cross-contamination events, causing health department interventions.
- Purchasing triggers: Outbreak events force risk management officers to instantly override existing supply contracts. Selecting hospital-grade fragrance-free disinfectant cleaners lacking synthetic fragrances immediately restores brand confidence following localized contamination scares.
- Qualification validation: Infection control audits verify pathogen elimination efficacy on standardized test surfaces. Housekeeping directors demand visual proof confirming no residue remains on reflective glass.
- Renewal drivers: Custodial staff feedback dictates long-term contract survival. Formulations causing respiratory irritation prompt union representatives to block vendor renewals entirely.
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by Formulation
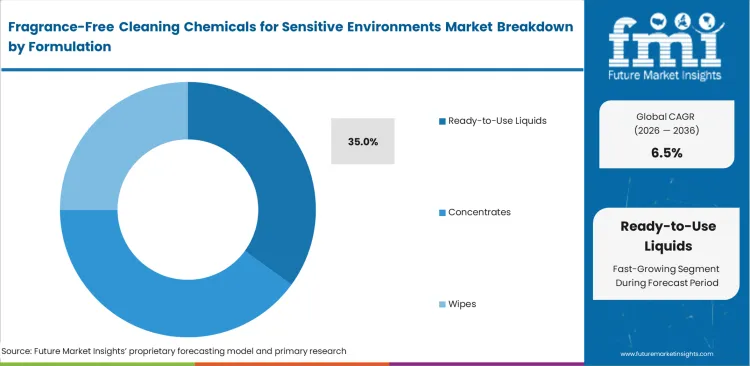
Shipping water across oceans destroys operating margins, yet end-user safety dictates format preferences. Ready-to-use liquids are anticipated to capture 35.0% share due to healthcare and educational facilities prioritize immediate, error-free application by rotating custodial staff. Facility managers avoid complex mixing stations that risk exposing workers to concentrated raw chemical odors. Automated dosing of super concentrate cleaning systems guarantees exact chemical ratios for larger operations, standardizing sanitation quality regardless of staff experience levels.
Rising adoption of sealed concentrate pods eliminates manual pouring hazards entirely, preventing accidental splashes that could release intense base-chemical fumes into sensitive corridors. Directors miscalculating localized water hardness find concentrated formulas failing to lather properly, creating streaky residue on bathroom mirrors. Operators evaluating ready-to-use vs concentrate fragrance-free cleaners must balance shipping logistics against the liability of manual dilution errors.
- Procurement savings: Eliminating mixing errors cuts liability expenses. Supply chain directors calculate exact ROI timelines based on reduced worker compensation claims regarding chemical exposure.
- Hidden costs: Automated dispenser maintenance requires specialized technician visits. Franchise owners face unexpected repair bills whenever hard water calcifies delicate internal mixing valves.
- Lifecycle comparison: Initial equipment installation capital looks high, yet chemical waste reduction pays off within eight months. Financial controllers strongly prefer predictable monthly refill expenditures.
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by End-use Environment
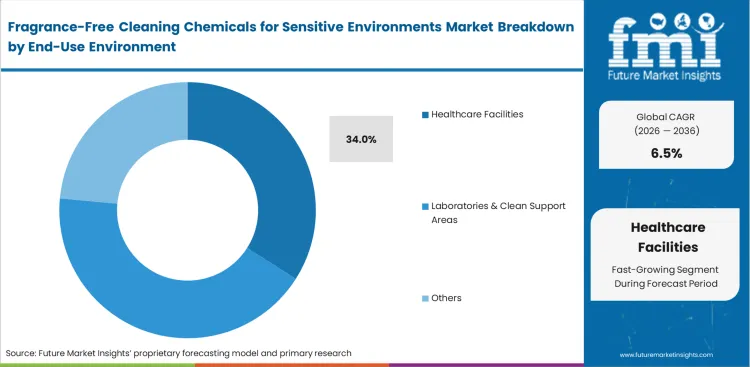
Healthcare facilities are anticipated to contribute 34.0% share in 2026, supported by strict hygiene protocols and the need to maintain respiratory-safe indoor environments. Cleaning practices in these settings prioritize scent-free formulations to prevent adverse reactions among patients, particularly in sensitive areas such as pediatric and elderly care units. Operational requirements extend beyond hygiene to include patient comfort and safety. Cleaning staff must manage high turnover rates while ensuring that chemical application does not interfere with indoor air quality standards. Product selection is therefore influenced by both efficacy and the absence of residual odor. Improper chemical usage introduces both financial and operational risks, including damage to medical equipment and increased maintenance costs. Facilities that fail to standardize fragrance-free protocols often face declining satisfaction metrics, which can directly impact institutional performance indicators.
- Efficacy failures: Using generic degreasers on premium clinical equipment causes irreversible etching. Restoration expenses quickly exceed any perceived chemical purchasing discounts.
- Residual risks: Lingering chemical odors generate immediate occupant complaints. Facility managers must issue formal apologies when synthetic fragrances trigger asthmatic reactions in pediatric wings.
- Mitigation mandates: Standardized training programs ensure custodians match specific unfragranced chemistries to distinct surface materials. Executive housekeepers must conduct random UV-light inspections verifying actual pathogen removal.
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by Chemistry Platform
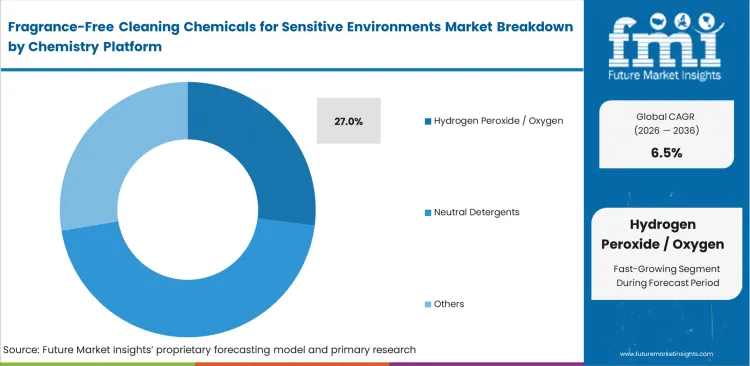
Efficacy requirements collide continually with occupant safety concerns. Hydrogen peroxide / oxygen-based systems are predicted to capture 27.0% share, offering stable broad-spectrum performance without corrosive bleaching side effects or harsh fumes. Contract cleaning managers rely on these compounds for routine sanitation, valuing long shelf life and compatibility with diverse architectural finishes. FMI analysts note that staff respiratory complaints quietly force unpublicized brand switching away from disinfectant chemicals lacking proper odor mitigation. Substituting standard industrial degreasers with green solvents neutralizes active pathogen threats while maintaining safe indoor air quality metrics. Teams ignoring staff feedback experience surging employee turnover, multiplying recruitment costs far beyond chemical budget limits.
- Performance peaks: Oxygen-based formulas maintain active pathogen-killing properties even when contaminated by organic soil. Custodial supervisors trust these solutions for heavily trafficked public restrooms.
- Edge failures: Cold water environments severely restrict activation speed. Operations managers must install localized water heaters to ensure proper chemical performance during winter months.
- Acceptability thresholds: Safety data sheets dictate exact maximum exposure limits. Environmental compliance officers reject any formulation lacking comprehensive dermal toxicity testing documentation.
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by Supply Channel
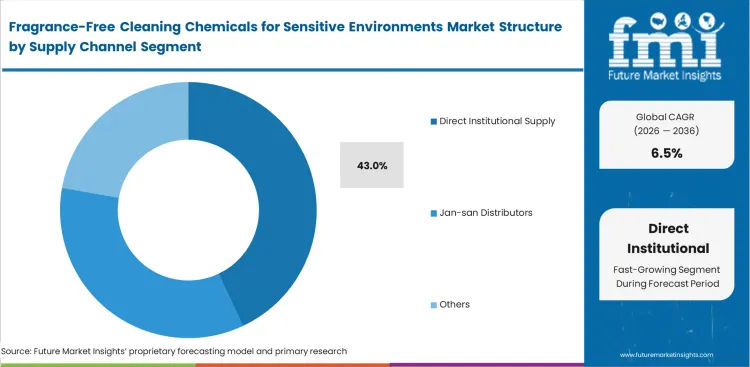
Direct institutional supply is estimated to represent a 43.0% share, enabling global healthcare brands to apply strict zero-fragrance hygiene standards across thousands of united properties. Supply chain vice presidents negotiate bulk pricing, implementing standardized institutional fragrance-free cleaning suppliers programs worldwide. Localized distributor networks secretly fulfill emergency shortages at huge markups when direct supply chains inevitably falter. Relying strictly on centralized hubs leaves remote senior care facilities vulnerable during logistical strikes. Lacking backup regional suppliers leaves remote senior care facilities vulnerable to operational disruptions, forcing unacceptable compromises utilizing unapproved scented retail chemicals.
- First adopters: Flagship clinical properties pioneer direct supply agreements to guarantee absolute sensory neutrality. Corporate compliance officers monitor consumption metrics via cloud-connected dispensers.
- Subsequent followers: Mid-scale academic institutions adopt centralized purchasing to match premium hygiene standards. Regional managers realize standardized unscented chemistry simplifies staff training across multiple locations.
- Late converts: Independent daycares finally abandon local hardware stores when parents demand hospital-grade scent-free disinfection proofs. Owner-operators surrender chemical autonomy to secure recognizable branded sanitation certificates.
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Drivers, Restraints, and Opportunities
Indoor air quality requirements are pushing executive housekeeping teams toward zero-emission and unscented formulations, particularly in facilities operating sealed HVAC systems where even low levels of volatile compounds can trigger building-wide complaints. Faster room turnover without residual odor directly improves patient intake flow, making single-pass, fragrance-free disinfection a practical requirement rather than a preference. Delays in upgrading cleaning protocols can restrict room availability during peak admission periods, creating operational bottlenecks. Facilities that adopt hypoallergenic floor care and surface cleaning chemistries, even at higher per-liter costs, often achieve higher room completion rates while reducing occupant irritation linked to routine cleaning.
Implementation remains constrained by infrastructure limitations within older institutional buildings. Dilution system rollout is frequently delayed where plumbing systems cannot support the pressure requirements of modern venturi-based equipment. Retrofitting janitorial closets across large facilities requires capital approval and coordinated installation, which slows adoption timelines. Portable, battery-operated dosing systems offer an interim solution, though limited reservoir capacity reduces suitability for high-volume environments where continuous supply is required.
Opportunities in the Fragrance-Free Cleaning Chemicals for Sensitive Environments Market
- Probiotic formulations: Living bacteria strains consume organic waste long after initial application. Facilities directors eliminate chronic drain odors organically without introducing synthetic masking agents.
- Concentrated soluble pods: Water-soluble packaging eliminates pouring hazards entirely. Managers slash shipping weights while guaranteeing perfect unscented chemical dosing every single time.
- IoT-enabled dispensing: Cloud-connected equipment tracks exact chemical consumption per zone. Operations managers predict supply shortages weeks before empty bottles impact custodial workflows utilizing specialized surface disinfectant products.
Regional Analysis
Based on regional analysis, fragrance-free cleaning chemicals for sensitive environments market is segmented into Asia Pacific, North America, Europe, and Latin America across numerous key countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.5% |
| India | 7.2% |
| Brazil | 6.9% |
| United Kingdom | 5.9% |
| United States | 5.8% |
| Germany | 5.6% |
| Japan | 5.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis
Centralized environmental mandates are shaping sanitation standards across newly developed commercial zones and clinical facilities, requiring property developers to secure green building approvals before occupancy. This requirement forces early adoption of sensory-neutral cleaning chemistries across large mixed-use projects. Facility operators managing high-density assets are negotiating bulk supply agreements across entire building portfolios, favoring concentrated formulations that reduce storage constraints in urban environments. Food service areas within institutional settings are also shifting toward unscented treatments to comply with stricter exhaust and air handling regulations without introducing masking agents into preparation zones. Cross-border facility management firms are consolidating supplier bases to ensure consistent compliance across regions, selecting manufacturers capable of delivering uniformly certified formulations across multiple jurisdictions.
- China: Urban development policies are linking commercial occupancy approvals to strict green building credentials, pushing early adoption of certified unscented cleaning formulations across large metropolitan projects. Building service contractors that transition ahead of these requirements gain an advantage in municipal bidding, particularly in high-density coastal cities where compliance standards are tightly enforced. Adoption within newly constructed healthcare facilities is accelerating as operators align sanitation protocols with international accreditation expectations before commissioning. China is projected to grow at a CAGR of 7.5%, as regulatory approval processes increasingly treat compliance as a baseline requirement rather than a differentiating factor for property developers.
- India: India’s fragrance-free cleaning chemicals sector is expected to grow at a CAGR of 7.2% through 2036. Private hospital networks and diagnostic chains are standardizing sanitation protocols across multiple locations, replacing traditional scented disinfectants with low-emission alternatives. Regional climate conditions, particularly humidity-driven mold risks, influence formulation demand toward products that combine antimicrobial performance with minimal airborne irritation. Local supply networks remain dominant due to cost sensitivity and distribution reach.
- Japan: Japan integrates workforce and industrial requirements into chemical selection, where the fragrance-free cleaning chemicals market advances at a CAGR of 5.4% from 2026 to 2036 as aging labor conditions restrict exposure to volatile compounds. Demand is concentrated in controlled environments such as precision manufacturing and healthcare facilities, where even minor chemical emissions can disrupt operations. Domestic production covers most needs, with limited reliance on imports for specialized applications.
North America Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis
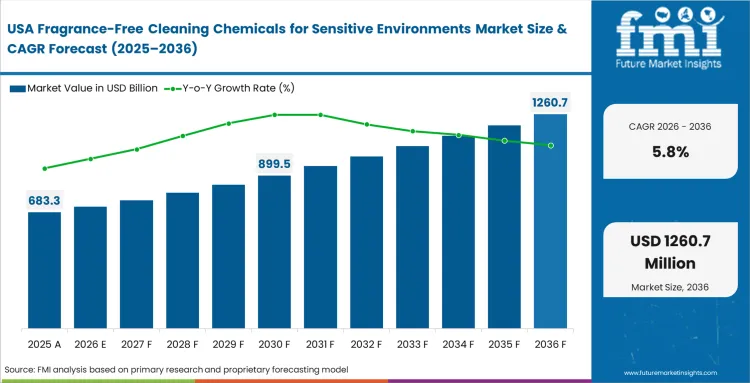
Institutional demand across North America is structured around occupational health compliance, where fragrance-free formulations are embedded into supply eligibility for large contracts. Government facilities, schools, and healthcare systems enforce restrictions on volatile compounds and masking agents, which removes non-compliant products before they enter distribution networks. Private commercial real estate follows similar standards to meet tenant expectations around indoor air quality and liability reduction. Equipment preferences are shifting toward flexible dispensing systems that support multiple suppliers, reducing dependence on closed chemical ecosystems.
- United States: The demand for fragrance-free cleaning chemicals for sensitive environments in US is poised to rise at a CAGR of 5.8% through 2036. Corporate real estate trusts mandate certified protocols to satisfy escalating environmental reporting demands from demanding institutional investors. Premium rental rates are secured by marketing buildings as fully compliant green environments as fully compliant green environments lacking harsh chemical odors. Fragrance-free cleaning chemicals USA adoption outpaces historical averages as these strict compliance mandates cascade from federal facilities down to independent regional clinics. Mastering complex safety documentation allows distributors to deeply entrench themselves within regional commercial supply chains, preventing agile startups from gaining market share. Resolving indoor air quality complaints immediately reduces legal liabilities tied to occupational asthma claims.
Europe Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis
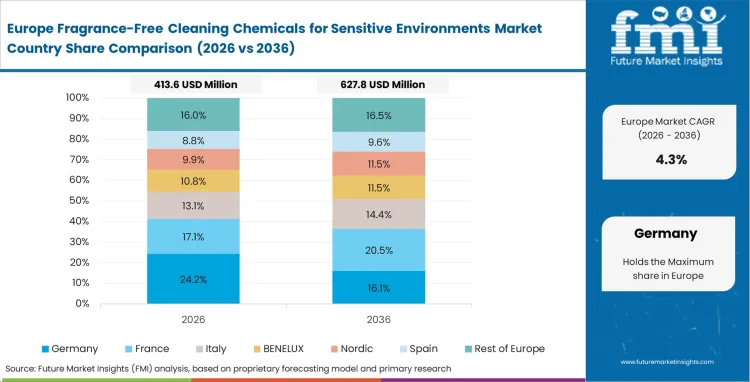
Regulatory enforcement across Europe defines both product composition and supplier access, with strict limits on fragranced and high-emission chemicals across institutional settings. Wastewater controls, occupational exposure limits, and indoor air quality standards operate simultaneously, requiring formulations that meet multiple compliance layers.
- United Kingdom: The United Kingdom aligns cost control with compliance, where the fragrance-free cleaning chemicals market is expected to grow at a CAGR of 5.9% from 2026 to 2036 as local authorities shift toward concentrated unscented products to reduce transport and packaging overhead while maintaining environmental standards. Cross-border service providers reduce supplier fragmentation by standardizing fragrance-free chemistries that can be deployed consistently across different regulatory jurisdictions.
- Germany: Germany integrates environmental implementation directly into facility operations, with the fragrance-free cleaning chemicals market advancing at a CAGR of 5.6% through 2036 as wastewater penalties and workplace safety regulations eliminate conventional scented formulations from institutional use. Early compliance reduces exposure to regulatory action and supports long-term operational stability.
Latin America Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis
Multinational corporate mandates are establishing uniform hygiene standards across regional operations, reducing reliance on slower municipal regulatory processes and requiring immediate alignment with internal compliance frameworks. International hospital networks entering new markets maintain strict zero-fragrance clinical protocols, which compels local distributors to realign institutional chemical portfolios to meet eligibility requirements for supply contracts. Facility managers operating within premium commercial real estate environments must balance stringent pathogen control with the preservation of sensitive interior finishes, leading to increased adoption of oxygen-based systems that leave no residual odor after application.
- Brazil: Brazil reflects this transition, where the fragrance-free cleaning chemicals market is expected to grow at a CAGR of 6.9% between 2026 and 2036 as multinational tenants impose uniform indoor air quality standards across commercial properties. Early adoption of fragrance-free formulations enables property owners to secure long-term leases with global firms, strengthening occupancy stability while reducing health-related operational disruptions.
Competitive Aligners for Market Players
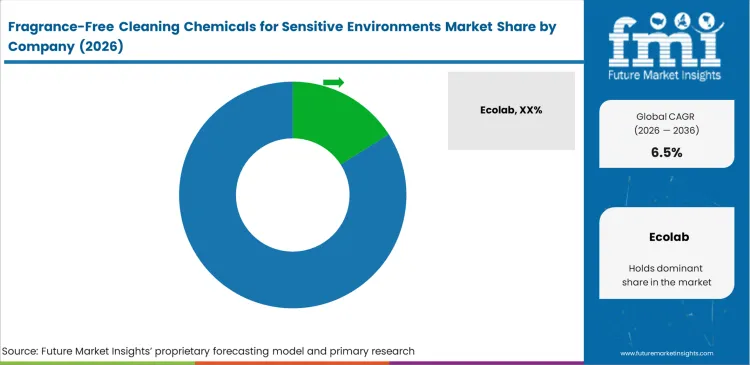
Chemical manufacturers in this segment operate closely within facility workflows rather than acting as standalone suppliers. Companies such as Ecolab and Solenis secure long-term agreements by installing proprietary dilution systems within hospital plumbing networks. This setup raises switching barriers, as operators must evaluate equipment removal, retraining requirements, and service disruption before changing suppliers. These transition burdens extend contract durations for unfragranced chemical programs.
Established suppliers maintain service coverage that allows rapid technician deployment across dispersed institutional sites. Smaller entrants offering niche formulations often face rejection when they cannot support centralized dispensing systems in remote locations. Large manufacturers also provide multilingual compliance documentation, allowing facility teams to meet zero-VOC and safety reporting requirements without delay. Competing suppliers must build comparable service and regulatory support before gaining entry into multinational contracts.
Facility operators are countering supplier dependency by specifying open-format dispensing systems compatible with multiple chemical inputs. Hardware decoupling allows regional sourcing flexibility during disruptions, while internal audits of dilution ratios ensure consistency and prevent excess consumption. Supplier selection increasingly depends on maintaining both operational flexibility and consistent sensory neutrality.
Key Players in Fragrance-Free Cleaning Chemicals for Sensitive Environments Market
- Ecolab
- Solenis (Diversey)
- SC Johnson Professional
- CloroxPro
- Reckitt Pro Solutions
- Betco
- Zep
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.6 billion to USD 3.0 billion, at a CAGR of 6.5% |
| Market Definition | Commercial-grade sanitation agents formulated specifically without masking scents, volatile organic perfumes, or synthetic odorants. |
| Segmentation | Product Family, Formulation, End-use Environment, Chemistry Platform, Supply Channel |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Spain, Italy, Russia, China, India, Japan, South Korea, Australia, Saudi Arabia, United Arab Emirates, South Africa |
| Key Companies Profiled | Ecolab, Solenis (Diversey), SC Johnson Professional, CloroxPro, Reckitt Pro Solutions, Betco, Zep |
| Forecast Period | 2026 to 2036 |
| Approach | Total institutional square footage mapped against average chemical consumption volume per occupied room-night. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Fragrance-Free Cleaning Chemicals for Sensitive Environments Market Analysis by Segments
Product Family
- General surface cleaners & disinfectants
- Restroom cleaners
- Glass cleaners
- Floor care chemicals
- Laundry care chemicals
Formulation
- Ready-to-use liquids
- Concentrates
- Wipes
- Sprays
- Pods / cartridges
End-use Environment
- Healthcare facilities
- Laboratories & clean support areas
- Schools & childcare facilities
- Senior care & assisted living
- Corporate / public buildings
Chemistry Platform
- Hydrogen peroxide / oxygen-based systems
- Neutral detergents
- Quat-based disinfectants
- Citric / organic acid cleaners
- Enzyme-based cleaners
Supply Channel
- Direct institutional supply
- Jan-san distributors
- Group purchasing contracts
- Facility service providers
- Online / catalog procurement
Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- Asia Pacific
- Middle East and Africa
Bibliography
- Carson, A., Henneberger, P. K., Park, J.-H., Curwin, B., Piacentino, J., Bailey, R. L., & LeBouf, R. (2025). A qualitative assessment of changes in occupational exposure to cleaning and disinfection agents among healthcare workers. Workplace Health & Safety.
- Salonen, H., et al. (2024). Cleaning products: Their chemistry, effects on indoor air quality, and links to health. Indoor Air.
- Centers for Disease Control and Prevention. (2024, June). Guideline for disinfection and sterilization in healthcare facilities (updated version).
- Environmental Protection Agency. (2025, September 11). Learn about the Safer Choice label.
- NHS England. (2025). National standards of healthcare cleanliness 2025: Health and safety.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Labor cost reduction metrics linked to single-pass unfragranced chemical adoption rates.
- Transition friction delaying automated dilution dispenser installations across legacy clinical properties.
- Qualification requirements for hospital-grade virucidal formulations lacking volatile organic compounds.
- Hidden shipping constraints negating intended concentrate cost savings for remote educational districts.
- Material degradation risks associated with high-frequency scent-neutral surface cleaner application.
- Supply chain vulnerabilities exposing senior care facilities to localized unscented chemical shortages.
- Respiratory compliance factors driving chemistry shifts away from aggressive quaternary blends.
- Vendor lock-in mechanics tied directly to proprietary dosing hardware installation inside commercial plumbing.
Frequently Asked Questions
How big is the fragrance-free cleaning chemicals sector?
The industry valuation stood at USD 1.50 billion in 2025. Projections indicate total sales will cross USD 1.6 billion in 2026, steadily advancing to reach USD 3.0 billion by 2036.
What is the CAGR for fragrance-free cleaning chemicals?
The sector is expected to expand at a 6.50% compound annual growth rate through 2036. Regulatory constraints regarding indoor air quality force institutional operators toward sensory-neutral formulations.
Why are fragrance-free cleaning chemicals growing?
Eliminating volatile organic compounds directly prevents building-wide respiratory complaints. Executive housekeepers authorize premium chemical expenditures when products demonstrably reduce asthma-related operational disruptions and legal liabilities.
What is the difference between fragrance-free vs unscented cleaning products?
Fragrance-free indicates the complete absence of any scenting ingredients, including masking agents. Unscented products frequently contain chemical maskers designed to neutralize the harsh chemical smell of raw base ingredients, which can still trigger severe respiratory distress in sensitive populations.
How do buyers evaluate fragrance-free vs low-voc cleaning chemicals?
Low-VOC products minimize chemical emissions but may still contain natural or synthetic fragrances that irritate occupants. Fragrance-free products completely eliminate olfactory triggers. Procurement teams targeting absolute indoor air quality often require formulations that satisfy both criteria simultaneously.
Why do ready-to-use liquids capture 35.0% share?
Healthcare and educational facilities prioritize immediate application by rotating custodial staff. Facility managers avoid complex mixing stations that risk exposing workers to concentrated raw chemical fumes. Convenience directly overrides the bulk cost savings associated with traditional dilution systems.
How does direct institutional supply maintain 43.0% channel dominance?
Enterprise operations demand absolute zero-fragrance hygiene standardization worldwide. Corporate teams negotiate bulk contracts, forcing franchised operators to utilize approved vendor lists. Direct channels guarantee formula consistency across thousands of diverse clinical locations.
What creates hidden costs for dilution systems?
Hard water calcifies delicate internal mixing valves rapidly. Operations managers face unexpected repair bills from specialized maintenance technicians. Uncalibrated equipment wastes expensive hypoallergenic chemicals, inflating quarterly custodial budgets unexpectedly.
Why do healthcare facilities lead application environments?
Clinical wards represent massive square footage demanding daily attention without compromising patient respiratory stability. Room attendants require versatile unscented formulations capable of tackling multiple surfaces simultaneously. High turnover speed dictates chemical selection entirely to ensure rapid room readmission.
How do premium medical fixtures complicate chemical purchasing?
Aggressive hospital-grade disinfectants etch expensive imaging equipment and tarnish clinical fittings. Facility directors quietly seek milder agents to preserve specialized diagnostic hardware. Balancing pathogen elimination against material preservation creates constant tension.
Why do hydrogen peroxide systems capture 27.0% share?
Stable broad-spectrum performance anchors routine sanitation workflows without releasing harsh fumes. These compounds offer extended shelf life and broad compatibility across diverse clinical surfaces. Custodial supervisors trust oxygen-based formulations for heavily trafficked pediatric wards.
What restricts aggressive chemical adoption?
Staff respiratory complaints force unpublicized brand switching. Union representatives block vendor renewals if formulations cause persistent asthmatic reactions among custodial teams. Environmental compliance officers strictly enforce indoor air quality exposure limits.
How do corporate mandates shape enterprise demand?
Real estate investment trusts install advanced closed-loop dosing architecture during initial construction phases to achieve LEED certification. Directors source massive quantities of verified unfragranced chemicals. Corporate brands enforce global hygiene standards strictly, fearing negative exposure regarding sick building syndrome.
Why do academic institutions struggle with automated dilution?
Tight operational budgets prohibit expensive dispenser installations across aging campus infrastructure. Regional supervisors rely on basic unfragranced blends supplied by domestic manufacturers. Localized chemical storage depots on multiple floors manage high-volume turnover demands between classes.
What drives zero-fragrance adoption across mature economies?
Occupational safety frameworks require strict environmental impact documentation to protect workers. Managers demand verifiable reductions in volatile organic compound emissions per facility. Green certification attainment unlocks lucrative corporate leasing contracts for compliant property managers.
How do incumbents lock in enterprise buyers?
Proprietary dilution equipment integrates directly into hospital plumbing infrastructure. Directors evaluating alternative unfragranced suppliers must calculate complete equipment removal costs. Hidden transition expenses effectively secure decade-long chemical supply relationships.
Why do facility owners demand open-source dispensing hardware?
Decoupled equipment accepts generic unscented chemical cartridges seamlessly. Regional managers source locally during acute supply chain disruptions. Independent hardware prevents chemical vendors from quietly decreasing concentration levels to inflate overall consumption volumes.
What role do probiotic formulations play?
Living bacteria strains consume organic waste long after initial application. Facilities directors eliminate chronic drain odors effectively without masking them with synthetic perfumes. Biological action maintains sensory neutrality while reducing toxic chemical runoff.
How do localized distributor networks survive?
Centralized supply chains inevitably falter during logistical strikes. Local dealers secretly fulfill emergency shortages of unfragranced chemicals at huge markups. Independent clinics abandon local hardware stores only when patients demand hospital-grade sensory-neutral disinfection proofs.
What degrades chemical performance in winter?
Cold water environments severely restrict activation speed. Operations managers must install localized water heaters to ensure proper chemical performance. Dropping temperatures delay required wet-contact times, slowing hospital room turnover rates.
Why do cross-contamination events trigger immediate brand switching?
Localized outbreaks expose institutional operators to severe liability. Risk management officers instantly override existing supply contracts. Selecting recognized unscented virucidal formulas immediately restores occupant confidence following health department interventions.
How do seasonal influxes affect school district procurement?
Massive student volumes prioritize high-efficiency sensory-neutral chemicals. Operations directors demand formulas capable of operating effectively using cold water to reduce energy costs. Mastering seasonal inventory fluctuation determines annual operational profitability for academic facilities.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Family
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Family , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Family , 2026 to 2036
- General Surface Cleaners & Disinfectants
- Glass Cleaners
- Others
- General Surface Cleaners & Disinfectants
- Y to o to Y Growth Trend Analysis By Product Family , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Family , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Formulation
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Formulation, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Formulation, 2026 to 2036
- Ready-to-Use Liquids
- Concentrates
- Wipes
- Ready-to-Use Liquids
- Y to o to Y Growth Trend Analysis By Formulation, 2021 to 2025
- Absolute $ Opportunity Analysis By Formulation, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-Use Environment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-Use Environment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-Use Environment, 2026 to 2036
- Healthcare Facilities
- Laboratories & Clean Support Areas
- Others
- Healthcare Facilities
- Y to o to Y Growth Trend Analysis By End-Use Environment, 2021 to 2025
- Absolute $ Opportunity Analysis By End-Use Environment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Chemistry Platform
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Chemistry Platform, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Chemistry Platform, 2026 to 2036
- Hydrogen Peroxide / Oxygen
- Neutral Detergents
- Others
- Hydrogen Peroxide / Oxygen
- Y to o to Y Growth Trend Analysis By Chemistry Platform, 2021 to 2025
- Absolute $ Opportunity Analysis By Chemistry Platform, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Supply Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Supply Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Supply Channel, 2026 to 2036
- Direct Institutional Supply
- Jan-san Distributors
- Others
- Direct Institutional Supply
- Y to o to Y Growth Trend Analysis By Supply Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Supply Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Family
- By Formulation
- By End-Use Environment
- By Chemistry Platform
- By Supply Channel
- Competition Analysis
- Competition Deep Dive
- Ecolab
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Solenis (Diversey)
- SC Johnson Professional
- CloroxPro
- Reckitt Pro Solutions
- Betco
- Ecolab
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Family , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Formulation, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End-Use Environment, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Chemistry Platform, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Supply Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Family
- Figure 6: Global Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Formulation
- Figure 9: Global Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End-Use Environment
- Figure 12: Global Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Chemistry Platform
- Figure 15: Global Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Supply Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Family
- Figure 32: North America Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Formulation
- Figure 35: North America Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End-Use Environment
- Figure 38: North America Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Chemistry Platform
- Figure 41: North America Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Supply Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Family
- Figure 48: Latin America Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Formulation
- Figure 51: Latin America Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by End-Use Environment
- Figure 54: Latin America Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Chemistry Platform
- Figure 57: Latin America Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Supply Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Family
- Figure 64: Western Europe Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Formulation
- Figure 67: Western Europe Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by End-Use Environment
- Figure 70: Western Europe Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Chemistry Platform
- Figure 73: Western Europe Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Supply Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Family
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Formulation
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End-Use Environment
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Chemistry Platform
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Supply Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Family
- Figure 96: East Asia Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Formulation
- Figure 99: East Asia Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by End-Use Environment
- Figure 102: East Asia Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Chemistry Platform
- Figure 105: East Asia Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Supply Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Family
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Formulation
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End-Use Environment
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Chemistry Platform
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Supply Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Family , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Family , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Family
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Formulation, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Formulation, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Formulation
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End-Use Environment, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End-Use Environment, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End-Use Environment
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Chemistry Platform, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Chemistry Platform, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Chemistry Platform
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Supply Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Supply Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Supply Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

