MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market
The MR-Linac And Image-Guided Radiotherapy QA Measurement Systems is Segmented by Product Type (Phantoms, Detector Arrays, Ion Chambers, Electrometers, QA Software), QA Workflow (Periodic QA, Daily QA, Commissioning, Patient QA, End-To-End QA), End User (Cancer Centers, Academic Hospitals, Research Institutes), Modality Focus (Dosimetry QA, Imaging QA, Motion QA, Targeting QA), Platform Compatibility (Unity, MRIdian Legacy, Vendor-Neutral), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Size, Market Forecast and Outlook By FMI
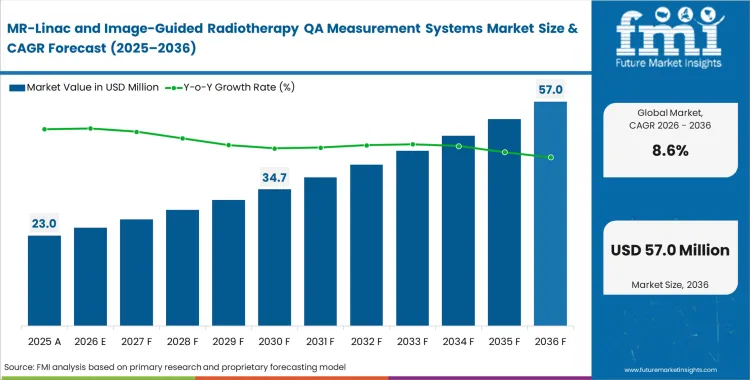
The MR-linac and image-guided radiotherapy QA measurement systems market was valued at USD 21.2 million in 2025. The industry is projected to reach USD 23.0 million in 2026 at a CAGR of 8.6% during the forecast period. Demand outlook carries the market valuation to USD 52.6 million by 2036 as the integration of magnetic resonance imaging with radiotherapy necessitates specialized QA systems capable of precise calibration within high-field environments.
Physics directors face a tough reality when moving to these hybrid systems, as they have to ensure that the radiation beam hits the exact spot the MRI sees in real time. Sticking with older testing tools is a major risk for any oncology department, especially if a patient's internal organs shift during treatment and the equipment does not catch it. A mistake in measuring the radiation dose is much more serious here than on a standard machine because the magnetic field changes to exactly how the beam behaves. Most chief physicists look for dosimetry tools that can check both the picture quality and the beam output at once, so they can avoid spending hours swapping out heavy gear between patient appointments.
Summary of MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market
- Market Snapshot
- The MR-linac and image-guided radiotherapy QA measurement systems market was valued at USD 21.2 million in 2025 and is projected to surpass USD 52.6 million by 2036.
- The industry is expected to advance at an 8.6% CAGR from 2026 to 2036, creating an incremental opportunity of USD 29.6 million over the period.
- This remains a specification-led oncology QA niche where magnetic-field compatibility, geometric distortion control, MR-to-radiation alignment accuracy, and end-to-end workflow validation determine product selection.
- Demand is tied less to treatment volume alone and more to the combination of new MR-linac commissioning, recurring daily and weekly QA, and patient-specific verification in adaptive radiotherapy programs.
- Demand and Growth Drivers
- Demand is rising because MR-linac platforms require QA tools that can operate safely and accurately in a magnetic-field environment, which changes detector behavior, imaging checks, and alignment routines compared with conventional linacs.
- Adoption is being strengthened by the continued rollout of MR-guided radiotherapy, with HIQA citing 90 Unity systems installed internationally by September 2024 and Elekta’s consortium update highlighting 50 centers across 20 countries in early 2025.
- Product demand is also supported by online adaptive workflows because those programs increase the value of distortion phantoms, detector arrays, motion phantoms, and software that can document alignment and dose fidelity across repeated adaptive sessions.
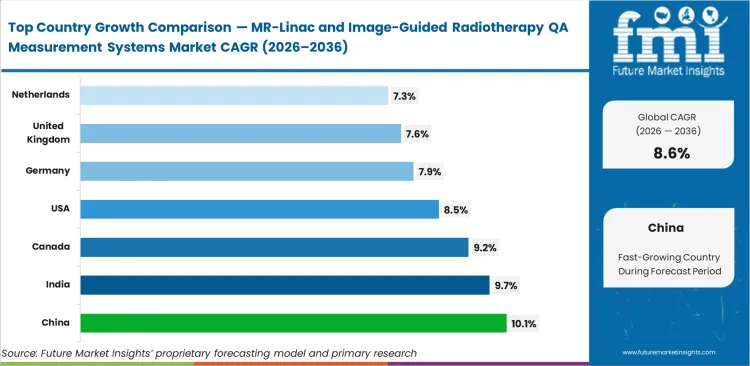
- Among key countries, China is anticipated to lead at 10.1% CAGR, followed by India at 9.7%, Canada at 9.2%, the United States at 8.5%, Germany at 7.9%, the United Kingdom at 7.6%, and the Netherlands at 7.3%.
- Growth is moderated by the still-small installed base, long procurement cycles at cancer centers, and the need for institution-specific validation before centers expand beyond vendor-supplied QA routines.
- Product and Segment View
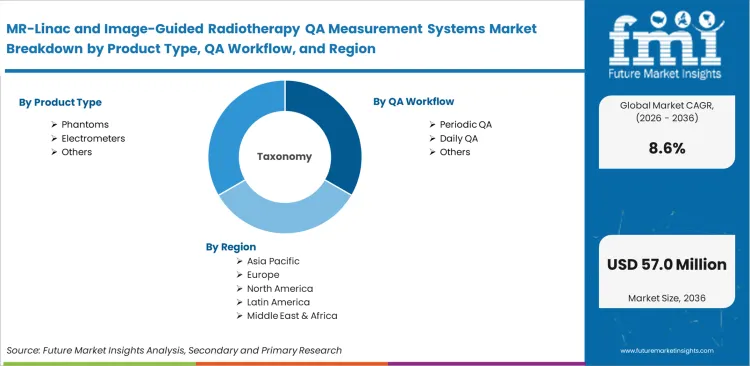
- The market covers MR-compatible phantoms, detector arrays, ion chambers, electrometers, and QA software used for commissioning, routine dosimetry, image-quality checks, targeting validation, and motion-management verification on MR-guided radiotherapy platforms.
- These systems are used across daily QA, periodic QA, patient-specific QA, end-to-end testing, and commissioning in cancer centers running adaptive or MR-guided radiation therapy workflows.
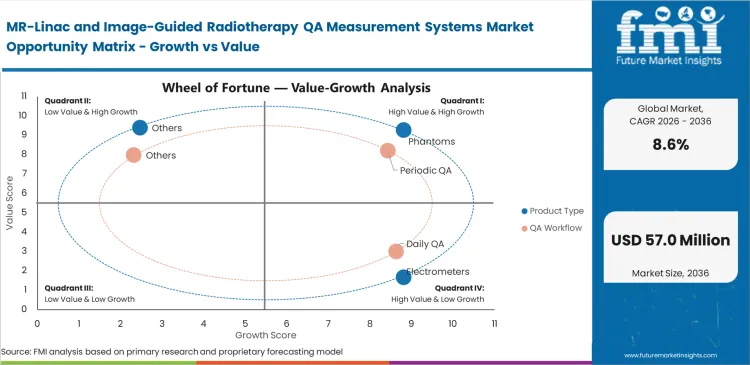
- Phantoms are expected to lead the Product Type segment with 34% share, because MRgRT programs need repeated checks for distortion, image quality, targeting, and workflow validation rather than single-point beam tests alone.
- Anticipating periodic QA to lead the QA Workflow segment with 41% share, reflecting the scheduled imaging, alignment, and dosimetric checks that recur throughout system life after commissioning is complete.
- As the buying decision usually sits with tertiary oncology providers rather than stand-alone research sites, Cancer centers are predicted to lead the End User segment with a 61% share.
- Dosimetry QA is poised to lead the Modality Focus segment with 38% share, as dose verification remains the minimum acceptance layer before imaging and adaptive workflows are trusted clinically.
- Unity is set to represent the Platform Compatibility segment with 68% share, supported by the larger documented active installed base for Elekta’s high-field MR-linac platform.
- The scope includes measurement systems used directly for MR-linac and MR-guided RT QA, but excludes the MR-linac treatment machine itself, treatment-planning systems sold as primary therapy platforms, and broad conventional linac QA products with no MR-specific positioning.
- Geography and Competitive Outlook
- China, India, and Canada are the fastest-rising demand pockets, while the United States remains the largest stable high-value procurement base for advanced oncology QA tools.
- Competition is shaped by vendors that can supply a fuller QA stack, especially those covering phantoms, arrays, chambers, electrometers, and workflow software instead of single-purpose accessories.
- Key participants include PTW Freiburg, IBA Dosimetry, Standard Imaging, Mirion Medical, ScandiDos, and The Phantom Laboratory.
- The market is moderately concentrated, with PTW Freiburg at an estimated 18% share, because it offers one of the most complete current MR-linac QA portfolios across water phantoms, arrays, and detectors.
As soon as a hospital decides to buy one of these image guided radiotherapy machines, getting the right non-magnetic sensors and testing kits becomes the top priority. Purchasing teams generally avoid looking at a vendor unless their tools can fit directly to the machine's main computer. Clinics cannot start treating people until these specialized sensors map out how the magnet pulls on the images and set the ground rules for every future treatment.
China is poised to project a CAGR at 10.1%, as its rapid expansion of specialized cancer infrastructure involves leapfrogging traditional setups straight to hybrid platforms. India is likely to track closely at 9.7% driven by private hospital network investments, while Canada anticipated to advance at 9.2%. The United States is ready to expand at 8.5%, supported by a mature installed base requiring dedicated MR-linac dosimetry systems. Germany will likely see 7.9% growth as strict European radiation safety mandates compel continuous equipment modernization. The United Kingdom expectedly follows at 7.6% and the Netherlands sets the pace at an anticipated 7.3% growth. This divergence highlights a structural split where emerging economies are outfitting entirely new bunkers while established Western networks focus on workflow optimization for existing fleets.
Segmental Analysis
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis by Product Type
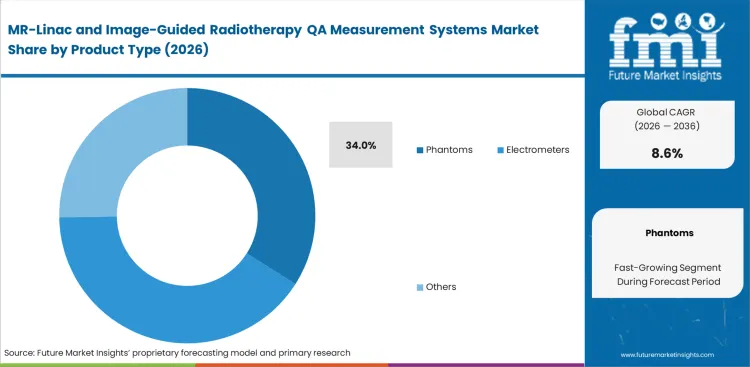
Strict MR environment compatibility dictates why physical testing units maintain their prominent position in oncology physics budgets. Phantoms are estimated to account for 34% share in 2026. FMI's analysis indicates this segment remains critical because clinical physicists require a physical baseline to measure spatial distortion caused by the magnetic field interacting with radiotherapy device components. Facilities relying entirely on virtual mapping expose themselves to severe targeting errors during complex treatments. What procurement officers evaluating the best QA phantom for MR-guided radiotherapy rarely realize is that phantom geometry establishes the absolute coordinate system upon which every subsequent software calculation depends. R&D directors at major academic hospitals specify MR-linac phantom systems first because passing a physical end-to-end test is the only universally accepted proof of system integrity for regulatory sign-off.
- Spatial Validation Testing: Non-magnetic phantoms simulate patient tissue density to confirm radiation tracks accurately through the MR bore. Clinical physicists secure precise geometrical coordinates to avoid mistreatment liabilities.
- Detector Array Deployment: Specialized grids measure beam profiles without introducing signal artifacts into the imaging stream. Oncology department heads gain faster daily equipment sign-offs that reduce early morning staffing requirements.
- Electrometer Precision: High-fidelity electronic readers capture minute charge variations inside shielded bunkers. Radiation safety officers confirm absolute dose output to ensure compliance with strict national medical exposure regulations.
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis by QA Workflow
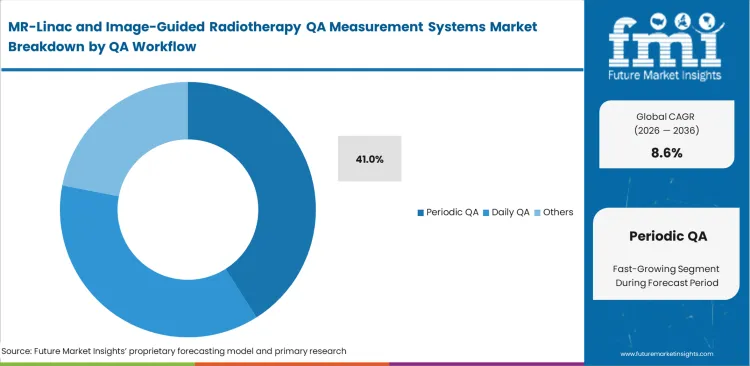
Comprehensive system auditing forms the backbone of regulatory compliance for hybrid radiation platforms. Based on FMI's assessment, senior medical physicists dedicate entire weekends to execute these deep diagnostic sweeps. While daily QA for MR-linac confirms basic functionality, periodic routines involve cross-validating the primary radiation therapy software against independent absolute dosimetry measurements. The non-obvious reality is that periodic workflow suites are highly profitable for vendors because they bundle multiple distinct software licenses under a single mandatory maintenance contract. The periodic QA segment leads the category due to the exhaustive nature of monthly and annual equipment checks, thereby anticipated to capturing 41% share in 2026. Department administrators navigating MR-linac commissioning QA budgets who attempt to piecemeal their periodic testing tools inevitably face scheduling gridlocks when trying to reconcile mismatched data formats across different vendor platforms.
- Routine Daily Verification: Automated routines check basic beam output and imaging isocenter alignment before the first patient arrives. Operations managers avoid schedule delays to keep tightly booked MR-Linac machines running on time.
- System Commissioning Phase: Extensive initial testing establishes the baseline performance parameters for a newly installed machine. Commissioning physicists rely on specialized multi-tool packages to prevent delayed clinical go-live dates.
- Patient-Specific Execution: Custom treatment plans are verified against simulated delivery logs prior to actual therapy. Dosimetrists executing patient-specific QA for MR-linac confirm highly complex dose gradients to guarantee the intended radiation matches the prescribed anatomical target.
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis by End User
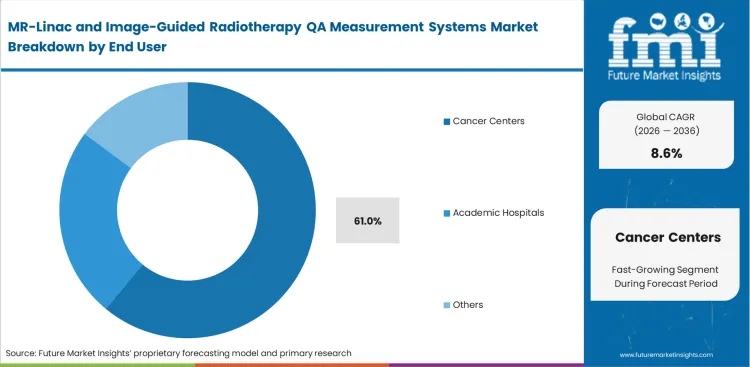
Cancer centers are poised to garner 61% share in 2026 as massive capital requirements restrict hybrid radiotherapy systems primarily to large, well-funded institutions. In FMI's view, these facilities drive hardware volume because they process the highest daily patient throughput, necessitating highly durable cancer center QA equipment for MR-linac workflows. Smaller clinics simply lack the bunker space and specialized physics personnel required to operate MR-guided platforms. Academic hospitals though pioneer the complex radiotherapy positioning devices protocols, high-volume cancer centers establish the commercial baseline by demanding ruggedized QA tools that withstand constant use. A facility director who under-equips a high-traffic center with entry-level calibration tools will face immediate pushback from clinical staff suffering through extended setup times.
- High-Volume Processing: Dedicated cancer facilities require fast-setup QA arrays to handle dense patient schedules. Clinic directors achieve higher daily throughput to directly improve the return on their capital equipment investment.
- Protocol Development Focus: Academic institutions utilize highly granular measurement tools to research novel adaptive therapy techniques. Chief researchers secure grant funding by publishing validated data on new MR-guided treatment methodologies.
- Clinical Trial Standardization: Research institutes demand strict, reproducible data formats to pool multi-center study results. Trial coordinators ensure uniform dose delivery validation to prevent data rejection by national regulatory authorities.
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis by Modality Focus
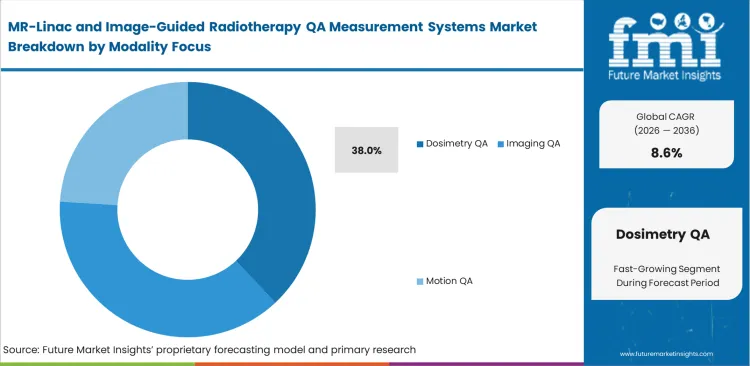
Confirming the exact amount of radiation delivered remains the non-negotiable legal threshold for oncology clinics. FMI observes that this foundational requirement forces every facility to maintain specialized MR-safe ion chamber devices and electrometers specifically calibrated for magnetic environments, resulting with dosimetry QA projected to secure a 38% share in 2026. While clear anatomical visualization is the main selling point of hybrid machines, precise dose verification in MR-guided radiotherapy dictates actual patient survival rates. The hidden complexity here is that the magnetic field physically alters the trajectory of secondary electrons, requiring complex correction factors that standard fiducial markers software cannot calculate natively. Medical physics directors managing MR-to-MV alignment QA who ignore these field-induced perturbations risk systemic under-dosing, a technical failure that undermines the entire clinical rationale for purchasing an MR-Linac.
- Absolute Dose Calibration: Specialized chambers capture exact radiation output under specific reference conditions. Chief physicists establish the legal baseline for treatment safety to avoid overexposure incidents.
- Dynamic Motion Tracking: Dedicated tools verify that the radiation beam pauses accurately when target anatomy shifts outside established margins. Therapy radiographers confidently treat tumors near moving organs to minimize damage to healthy tissue.
- Targeting Alignment Verification: Geometric phantoms ensure the imaging center perfectly matches the radiation focal point. Radiation oncologists deliver highly conformal doses to maximize tumor destruction while preserving critical adjacent structures.
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis by Platform Compatibility
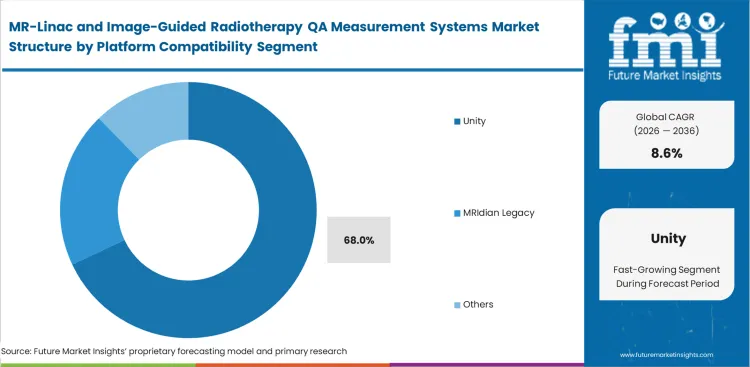
Installed base scale heavily influences which quality assurance ecosystems receive prioritized development from third-party vendors. Unity compatibility is set to represent 68% of market share in 2026. As per FMI's projection, Elekta's dominant global footprint with its 1.5T system creates a massive captive audience for QA developers. Vendors invest heavily in validating their products specifically for this platform to access the largest pool of ready buyers. Interestingly, a QA vendor's technical superiority matters less than their ability to cleanly handshake with the Unity control interface; a clunky software bridge will kill a sale faster than a slight dip in measurement resolution. Procurement managers sourcing radiotherapy QA software for MR-guided treatment who select vendor-neutral intensity modulated radiation therapy tools without verifying deep API integration often burden their physics teams with permanent manual data entry workflows.
- Native Interface Integration: QA software communicates seamlessly with the dominant 1.5T treatment planning architecture. Physics teams eliminate manual data transfers to dramatically reduce the risk of transcription errors during calibration.
- Legacy System Support: Specialized tools maintain validation capabilities for earlier generation low-field MR radiotherapy systems. Hospital administrators extend the functional lifespan of existing capital equipment to delay massive replacement expenditures.
- Cross-Platform Validation: Agnostic measurement systems allow clinics operating mixed equipment fleets to standardize their testing protocols. Regional health directors simplify staff training requirements to allow physicists to float across different clinic locations seamlessly.
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Drivers, Restraints, and Opportunities
Hospital directors face intense pressure to keep their expensive hybrid machines running at full capacity. This financial reality makes calibration speed a daily operational priority rather than a simple technical concern. Clinical physicists must run spatial safety checks before the first patient arrives to meet strict legal regulations. Doctors who adjust treatment plans while a patient lies inside the scanner also need instant validation of their new dose calculations. Relying on older manual testing methods causes severe scheduling delays that back up the entire oncology department. Clinics are forced to purchase fast, automated measurement arrays simply to prevent their stereotactic radiation therapy platforms from sitting idle during peak working hours. A slow quality assurance routine directly reduces the number of patients a facility can treat each week.
Software communication blocks represent the biggest operational headache for clinical teams. Third-party testing tools often fail to connect directly with the main intraoperative radiation therapy systems used by the hospital. Physicists end up typing daily test results into central compliance databases by hand when these platforms refuse to share data. Major equipment manufacturers deliberately restrict access to their internal software code to protect their own ecosystems from outside developers. This defensive strategy forces mixed-vendor clinics to deal with permanent administrative delays. Real plug-and-play functionality remains rare, leaving hospital physics teams to manage frustrating manual data transfers instead of focusing on actual patient care.
- Instant Dose Verification: Writing software programs that confirm fresh dose calculations in under a minute give doctors the confidence to change treatment plans safely while the patient is actively on the table.
- Early Warning Alerts: Feeding daily test results into smart algorithms allows service teams to spot tiny hardware issues early. Clinics avoid unexpected machine breakdowns that cancel entire days of scheduled therapies.
- Measuring Moving Anatomy: Building sensitive detector grids that record how radiation hits a shifting target allows physics teams to prove their oncology imaging software accurately tracks a breathing lung or moving organ during active treatment.
Regional Analysis
Based on regional analysis, MR-Linac and Image-Guided Radiotherapy QA Measurement Systems market is segmented into North America, Latin America, Europe, Asia-Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
| China | 10.1% |
| India | 9.7% |
| Canada | 9.2% |
| United States | 8.5% |
| Germany | 7.9% |
| United Kingdom | 7.6% |
| Netherlands | 7.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia-Pacific MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis
National funding and private corporate hospital investments are driving a massive wave of new oncology center construction across Asia-Pacific. Health ministries want to process high patient volumes quickly. They skip older equipment and install hybrid machines directly into new concrete bunkers. Clinic directors need automated QA tools that warm up quickly so they can start treating patients early in the morning and maximize clinical hours.
- China: Centralized healthcare programs fund the direct deployment of hybrid accelerators in provincial capital hospitals. China is predicted to record a CAGR of 10.1% in the QA measurement systems market through 2036. Clinic directors demand comprehensive suites to ensure these new installations meet strict national safety standards immediately upon commissioning. Equipment vendors securing early validation contracts in these state-funded centers lock in long-term service agreements and recurring upgrade revenue.
- India: Corporate hospital conglomerates use MR-Linac technology to attract premium medical tourism and secure higher billing rates. Demand for MR-linac QA systems in India is anticipated to rise at a CAGR of 9.7% from 2026 to 2036. These private institutes require top-tier arrays to prove their clinical accuracy to private insurers, creating a lucrative opportunity for vendors providing localized technical support and fast hardware replacement services.
FMI analyses, emerging hubs across the broader Asia-Pacific region are starting to pool their healthcare resources. They aim to fund centralized national cancer institutes equipped with similar hybrid capabilities and high-speed QA infrastructure.
North America MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis
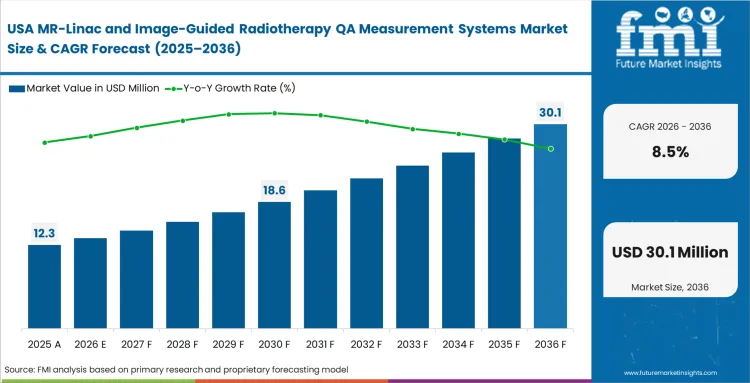
A vast installed base of aging radiation therapy equipment forces widespread modernization across North American clinical networks. Hospitals upgrading to hybrid modalities must throw out their old quality assurance inventories because traditional ferromagnetic tools cannot safely enter the MR environment. Large academic consortiums publish strict new guidelines for hadron therapy and precision cancer calibration. Department heads face constant pressure from domestic regulatory bodies to document every aspect of spatial accuracy, pushing demand toward software that handles massive daily data logging.
- Canada: Canada is forecast to record steady growth in QA measurement systems adoption at a CAGR of 9.2% through 2036. Provincial health authorities govern centralized procurement for major cancer treatment facilities and prioritize standardized workflows across regions. Regional physics directors seek vendor-neutral platforms to harmonize testing procedures, meaning once a QA system secures approval from a major health board, it becomes the default standard for subsequent installations.
- United States: Highly competitive private healthcare markets drive hospitals to adopt rapid adaptive therapy techniques to maintain regional prestige. Radiation safety officers require highly sophisticated 4D measurement tools to validate complex moving-target treatments before patient exposure. The United States is projected to witness 8.5% CAGR in the MR-guided radiotherapy QA market through 2036. This mature landscape demands continuous software upgrades rather than just initial hardware sales.
FMI reports, hardware replacement cycles in North America will eventually stabilize. The commercial focus will then shift almost entirely to recurring software licenses and specialized maintenance contracts.
Europe MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Analysis
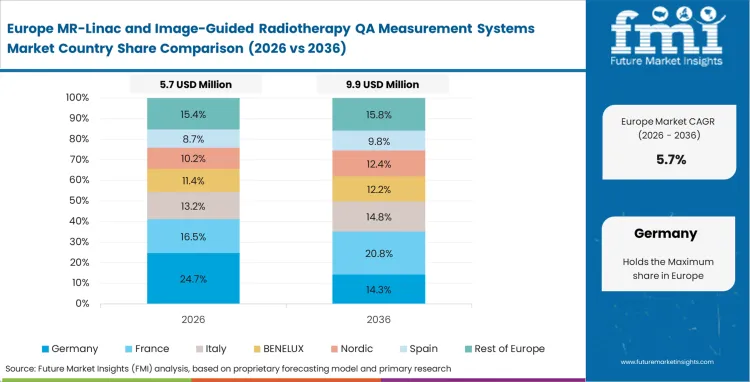
Strict radiation protection directives mandate meticulous equipment verification across all European member states. Public health systems prioritize rigorous testing protocols over raw patient throughput speed. European clinics pioneered early MR-Linac adoption, resulting in a highly experienced base of clinical physicists who demand advanced, customizable measurement tools rather than basic entry-level arrays. Facility administrators allocate significant budgets to maintain compliance with these uncompromising safety frameworks and ensure the initial targeting data matches the radiation isocenter.
- Germany: Germany is anticipated to witness MR-compatible QA devices grow at a CAGR of 7.9% over the forecast period. Strict national radiation protection laws require extensive auditing of all clinical radiation-emitting devices. Senior medical physicists rely on high-precision ion chambers to provide indisputable legal documentation of dose output, allowing domestic manufacturers to leverage their deep understanding of local regulations and maintain strong positions.
- United Kingdom: The National Health Service utilizes centralized technology assessment programs to evaluate the cost-effectiveness of new clinical tools. Oncology department heads require QA software that proves its value by reducing labor hours spent on routine calibration. UK adoption is likely to advance at a CAGR of 7.6% by 2036. Vendors must demonstrate clear workflow efficiencies to pass rigorous NHS procurement reviews and secure long-term contracts.
- Netherlands: The Netherlands sector is expected to register a CAGR of 7.3% from 2026 to 2036. Leading academic research hospitals drive the development of highly advanced, real-time adaptive radiotherapy protocols. Research directors demand ultra-sensitive detector arrays to validate complex, small-field treatments safely, turning these pioneering centers into global beta-testing sites for next-generation QA software modules.
FMI assesses, European clinical networks will continue to set the global standard for rigorous QA testing. Independent verification processes established here frequently shape the regulatory frameworks adopted by other global regions.
Competitive Aligners for Market Players
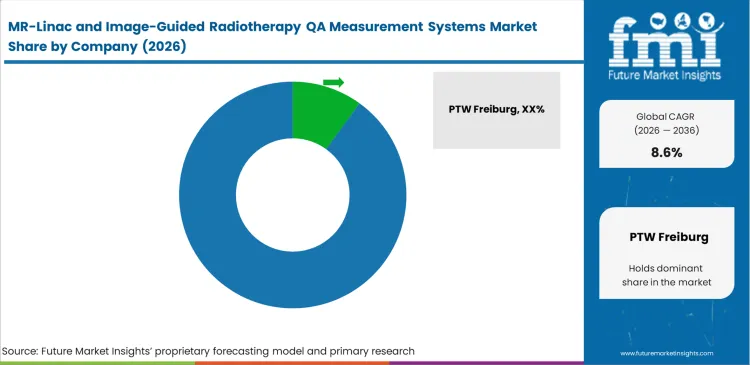
Building measurement tools that survive inside a 1.5T magnetic field requires specific materials engineering, limiting the number of capable suppliers. Companies such as PTW Freiburg and IBA Dosimetry maintain their lead because they have already solved these physical challenges. Hospital buyers evaluate any MR-linac QA system supplier based strictly on past performance. A clinical physicist will rarely risk a highly expensive hybrid machine on unproven calibration devices. Hardware durability matters, yet the primary deciding factor is how smoothly the testing software connects to the main treatment console without causing system delays.
Early collaboration with major machine builders gives established brands a strong commercial edge. When procurement committees compare PTW vs IBA dosimetry MR-linac QA setups, they look for the system that contains the most complete set of built-in correction codes. These codes are necessary to get accurate readings from standard dose monitoring devices under heavy magnetic interference. New competitors face a steep climb because they must write and test these validation algorithms from zero, requiring years of regulatory review. The older brands also benefit from daily habit. Thousands of medical physicists already know exactly how to use their specific software, making hospitals reluctant to switch and retrain their staff.
Hospital administrators actively try to avoid getting trapped by a single supplier. They evaluate vendor-supplied vs independent MR-linac QA tools to find flexible software that can read data from several different hardware brands. Clinic directors reject closed data systems and demand testing programs that send standardized compliance reports straight into the central oncology information systems. This demand for open software forces equipment makers to adapt their product designs. The most successful developers will automate the math behind the tests. They will build systems that read the raw physical data and immediately tell the therapy team whether it is safe to proceed with the patient treatment.
Key Players in MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market
- PTW Freiburg
- IBA Dosimetry
- Standard Imaging
- Mirion Medical
- ScandiDos
- The Phantom Laboratory
Scope of the Report
| Metric | Value |
| Quantitative Units | USD 23.0 million to USD 52.6 million, at a CAGR of 8.6% |
| Market Definition | MR-Linac and Image-Guided Radiotherapy QA Measurement Systems encompass the specialized non-ferromagnetic physical measurement instruments and dedicated validation software designed explicitly to operate safely and accurately within strong magnetic fields to calibrate and verify hybrid radiation therapy platforms. |
| Segmentation | Product Type, QA Workflow, End User, Modality Focus, Platform Compatibility, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, Netherlands, China, India |
| Key Companies Profiled | PTW Freiburg, IBA Dosimetry, Standard Imaging, Mirion Medical, ScandiDos, The Phantom Laboratory |
| Forecast Period | 2026 to 2036 |
| Approach | Global capital equipment installation tracking correlated with oncology department operating budgets and compliance upgrade cycles. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
MR-Linac and Image-Guided Radiotherapy QA Measurement Systems Market Segments
Product Type
- Phantoms
- Detector arrays
- Ion chambers
- Electrometers
- QA software
QA Workflow
- Periodic QA
- Daily QA
- Commissioning
- Patient QA
- End-to-end QA
End User
- Cancer centers
- Academic hospitals
- Research institutes
Modality Focus
- Dosimetry QA
- Imaging QA
- Motion QA
- Targeting QA
Platform Compatibility
- Unity
- MRIdian legacy
- Vendor-neutral
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Netherlands
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Riis, H. L., Chick, J., Dunlop, A., & Tilly, D. (2024). The quality assurance of a 1.5 T MR-Linac. Seminars in Radiation Oncology, 34(1), 120-128.
- Hilgers, G. C., Ikink, M., Potters, I., Schuring, D., & Minken, A. W. (2024). Characterization of the on-board megavoltage imager in a magnetic resonance-guided radiotherapy machine for beam output checks. Physics and Imaging in Radiation Oncology, 29, 100558.
- Crosby, J., et al. (2024). Technical note: Characterization of a multi-point scintillation dosimetry research platform for a low-field MR-Linac. Medical Physics, 51(9), 6475-6484.
- Kaza, E., & Williams, C. L. (2024). Receive coil quality assurance procedure and automated analysis for ViewRay MRIdian MR-Linac. Journal of Applied Clinical Medical Physics, 25(3), e14275.
- Frick, S., Schneider, M., Kapsch, R.-P., & Thorwarth, D. (2024). Experimental characterization of four ionization chamber types in magnetic fields including intra-type variation. Physics and Imaging in Radiation Oncology, 29, 100561.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Workflows utilized by senior medical physicists to validate hybrid MR-Linac dose delivery accuracy without bottlenecking daily schedules.
- Magnetic field interference challenges requiring specialized non-ferromagnetic detector array materials to prevent severe imaging artifacts.
- Adoption variations between high-volume cancer centers managing dense patient loads and specialized academic research hospitals pioneering adaptive protocols.
- Software integration gaps between third-party quality assurance tools and proprietary oncology information systems that create administrative burdens.
- Regulatory compliance mandates driving regular periodic audits and rigorous geometric validation for clinical radiation-emitting devices.
- Spatial distortion mapping techniques necessary for safe online adaptive radiotherapy protocols during live patient treatments.
- Strategic positioning of major vendors producing Unity-compatible validation software suites with extensive proprietary correction factor libraries.
- Geographic divergence in capital equipment purchasing cycles across established Western networks and rapidly expanding East Asian oncology hubs.
Frequently Asked Questions
What QA tools are required for an MR-linac?
Medical physicists require non-ferromagnetic physical phantoms, MR-compatible detector arrays, specialized ion chambers, and dedicated validation software to map spatial distortion and verify absolute dose output safely within a high-strength magnetic field.
How is MR-linac QA different from conventional linac QA?
Unlike standard linear accelerators, hybrid MR-Linac systems require calibration tools built entirely without ferromagnetic components to prevent dangerous projectile hazards and severe magnetic susceptibility artifacts during the measurement process.
What is the market size of MR-linac QA systems?
The sector was valued at USD 21.2 million in 2025 and is expected to hit USD 52.6 million by 2036, anchored by initial capital outlays for specialized non-magnetic measurement equipment.
Which companies sell QA measurement systems for MR-linac centers?
Dominant suppliers include PTW Freiburg, IBA Dosimetry, Standard Imaging, and Mirion Medical, all of which maintain deep materials engineering expertise and extensive libraries of proprietary magnetic field correction factors.
What product segments dominate MR-guided radiotherapy QA?
Phantoms capture the highest spend at 34% share because they provide the mandatory physical baseline for spatial distortion mapping, which establishes the absolute coordinate system for all subsequent software calculations.
Which QA workflow drives the most spend in MRgRT?
Periodic QA leads with 41% share because comprehensive monthly and annual compliance audits demand expensive, multi-tool software suites to satisfy strict national radiation protection mandates.
Why do MR-linac centers need dedicated detectors?
Standard detectors contain metallic parts that interfere with the magnetic resonance imaging stream, forcing facilities to acquire specialized arrays that capture radiation profiles without distorting the anatomical visualization.
What is geometric distortion in MR-guided radiotherapy?
It is the physical alteration of the imaging coordinate system caused by the magnetic field interacting with internal machine components, requiring specialized phantoms to map and correct these spatial errors before treatment begins.
Can conventional linac QA tools be used on MR-linac systems?
No, utilizing traditional radiotherapy QA tools in a hybrid bunker introduces lethal projectile risks and corrupts the imaging data, making dedicated MR-safe calibration instrumentation an absolute operational necessity.
Which countries are adopting MR-linac QA tools fastest?
China leads global adoption with a 10.1% CAGR as its national healthcare modernization programs fund the rapid, direct deployment of advanced hybrid accelerators in newly constructed provincial capital hospitals.
Which phantom is used for MR-guided radiotherapy QA?
Clinical physicists utilize specialized MR-safe 3D and 4D geometric testing units that simulate patient tissue density to confirm that radiation tracks perfectly through the magnetic bore without spatial deviation.
What is MR-to-radiation isocenter alignment?
This critical verification process uses physical phantoms to ensure that the exact center of the magnetic resonance image perfectly matches the focal point of the radiation beam, preventing catastrophic mistreatment of adjacent healthy tissue.
What is the expected compound annual growth rate for this equipment?
Demand for these specialized measurement suites is poised to advance steadily at an 8.6% CAGR through the assessment period.
Which end-user facility type accounts for the highest procurement volume?
Cancer centers hold 61% share due to their ability to fund massive capital hybrid installations and their need for highly durable tools to maintain dense daily patient throughput.
What modality focus holds the most critical position in physics budgets?
Dosimetry QA captures 38% share because quantifying the exact radiation output under magnetic influence remains the fundamental legal requirement for patient safety.
Which platform compatibility standard leads the sector?
Unity compatibility accounts for 68% share, reflecting Elekta's dominant installed base of 1.5T hybrid systems and the resulting vendor focus on native software integration.
What is the primary operational friction slowing adoption in mixed clinics?
A lack of native API integration between third-party QA software and proprietary oncology information systems creates major administrative bottlenecks by forcing manual data entry.
How does adaptive radiotherapy influence QA purchasing decisions?
Clinicians redrawing treatment contours on the fly require ultra-fast, automated validation tools to confirm the safety of dose recalculations within minutes without delaying the patient schedule.
Why do academic hospitals play a pivotal role in shaping this sector?
Research institutes develop the highly granular MR-guided measurement protocols that eventually become standard regulatory mandates for all routine clinical environments.
What drives private hospital investment in the Indian sector?
Corporate networks utilize MR-Linac capabilities to attract premium medical tourism, requiring top-tier QA arrays to definitively prove their advertised clinical excellence to private insurers.
Why do provincial health authorities dictate Canadian adoption patterns?
Centralized procurement boards prioritize vendor-neutral platforms to harmonize testing procedures and simplify physics staff training across geographically dispersed regional clinic locations.
What forces continuous modernization within the United States market?
Intense regional competition pushes private networks to adopt complex 4D tracking techniques, which inherently require continuous measurement software upgrades rather than just initial hardware sales.
How do European radiation laws impact institutional equipment choices?
Strict directives compel German and UK facilities to prioritize independent, highly documented verification over raw operational speed, favoring premium multi-tool validation suites.
What future technological shift will define workflow efficiency?
Developers are moving toward AI-driven predictive software that automates raw data analysis, turning physical measurements into immediate clinical go/no-go decisions while flagging subtle machine degradation early.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Phantoms
- Electrometers
- Others
- Phantoms
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By QA Workflow
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By QA Workflow, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By QA Workflow, 2026 to 2036
- Periodic QA
- Daily QA
- Others
- Periodic QA
- Y to o to Y Growth Trend Analysis By QA Workflow, 2021 to 2025
- Absolute $ Opportunity Analysis By QA Workflow, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Cancer Centers
- Academic Hospitals
- Others
- Cancer Centers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Modality Focus
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Modality Focus, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Modality Focus, 2026 to 2036
- Dosimetry QA
- Imaging QA
- Motion QA
- Dosimetry QA
- Y to o to Y Growth Trend Analysis By Modality Focus, 2021 to 2025
- Absolute $ Opportunity Analysis By Modality Focus, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platform Compatibility
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platform Compatibility, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platform Compatibility, 2026 to 2036
- Value (USD Million)y
- MRIdian Legacy
- Others
- Value (USD Million)y
- Y to o to Y Growth Trend Analysis By Platform Compatibility, 2021 to 2025
- Absolute $ Opportunity Analysis By Platform Compatibility, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By QA Workflow
- By End User
- By Modality Focus
- By Platform Compatibility
- Competition Analysis
- Competition Deep Dive
- PTW Freiburg
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- IBA Dosimetry
- Standard Imaging
- Mirion Medical
- ScandiDos
- PTW Freiburg
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by QA Workflow, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Modality Focus, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Platform Compatibility, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by QA Workflow
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Modality Focus
- Figure 15: Global Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Platform Compatibility
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by QA Workflow
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Modality Focus
- Figure 41: North America Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Platform Compatibility
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by QA Workflow
- Figure 51: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by End User
- Figure 54: Latin America Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Modality Focus
- Figure 57: Latin America Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Platform Compatibility
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by QA Workflow
- Figure 67: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Modality Focus
- Figure 73: Western Europe Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Platform Compatibility
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by QA Workflow
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End User
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Modality Focus
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Platform Compatibility
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by QA Workflow
- Figure 99: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by End User
- Figure 102: East Asia Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Modality Focus
- Figure 105: East Asia Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Platform Compatibility
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by QA Workflow
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Modality Focus
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Platform Compatibility
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by QA Workflow, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by QA Workflow, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by QA Workflow
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Modality Focus, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Modality Focus, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Modality Focus
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Platform Compatibility, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Compatibility, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Platform Compatibility
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

