Patient-Reported Outcome Data Monetization Market
The patient-reported outcome data monetization market is segmented by Data Type (Clinical PROs, Behavioral PROs, QoL Scores, Symptom Logs), Monetization Model (Data Licensing, Subscription Access, API Access, Insights Sales), End User (Pharma, Payers, Providers, Researchers), Application (Drug Trials, Outcomes Research, Market Access, Post-Market), Deployment (Cloud, On-Premise, Hybrid), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Patient-Reported Outcome Data Monetization Market Size, Market Forecast and Outlook By FMI
The patient-reported outcome data monetization market was valued at USD 1.2 Billion in 2025. Demand is estimated to surpass USD 1.3 Billion in 2026 at a CAGR of 11.9% during the forecast period. The cumulative revenue is expected to be USD 4.0 Billion through 2036 as regulatory agencies increasingly require longitudinal patient feedback for post-market surveillance.
Summary of Patient-Reported Outcome Data Monetization Market
- Patient-Reported Outcome Data Monetization Market Definition
- Commercial infrastructure enabling financial packaging and sale of direct patient health feedback. Brokers tokenize self-reported outcomes and sell access to pharmaceutical developers seeking clinical utility validation.
- Demand Drivers in the Market
- Value-based contracting mandates force formulary managers to buy continuous symptom trackers.
- FDA label expansion requirements compel regulatory affairs directors to purchase legacy patient feedback.
- Specialty drug pricing pressure pushes commercial leads to acquire longitudinal quality-of-life proof.
- Key Segments Analyzed in the FMI Report
- Clinical PROs: This segment is expected to account for 38.5% share in 2026, driven by strict regulatory acceptance frameworks demanding objective baseline symptom metrics.
- Data Licensing: This segment is set to hold 41.2% share in 2026, reflecting buyer preference for raw data manipulation over static dashboard access.
- Pharma: This segment is projected to lead end user demand in 2026, propelled by the necessity to acquire outcomes data to justify premium pricing.
- Drug Trials: This segment is anticipated to capture 36.7% share in 2026, accelerated by sponsors utilizing historical cohorts for early-stage evidence generation.
- Cloud: This segment is estimated to secure 62.4% share in 2026, sustained by the operational reality requiring federated data queries across strict national borders.
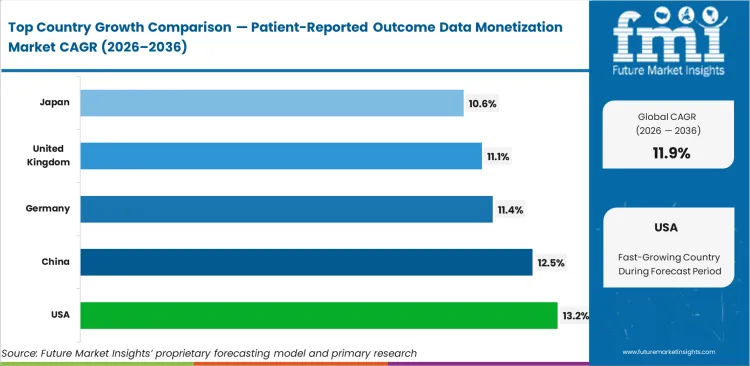
- United States: This segment demonstrates 13.2% compound growth, fueled by rapid greenfield digitization of fragmented clinic networks and value-based care mandates.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Analyst, Healthcare, at FMI, points out, "Pharma procurement assumes PRO datasets act as clean baseline assets. Reality dictates longitudinal decay halves data value every six months unless re-verified. Clinical directors buy legacy symptom logs expecting immediate label expansion utility. Buyers actually receive heavily degraded demographic snapshots requiring massive statistical imputation. Organizations paying premium licensing fees for anything older than eighteen months effectively purchase regulatory noise."
- Strategic Implications / Executive Takeaways
- Chief Data Officers must transition from selling static reports to offering federated API access.
- Market Access VP roles require direct integration of tokenized patient logs to secure formulary wins.
- Clinical Trial Directors risk massive delays without pre-purchasing background symptom data.
- Methodology
- Primary Research: Chief Data Officers at pharma sponsors, VP of Market Access at payers, and registry network administrators.
- Desk Research: FDA Real-World Evidence Program registries, NIH PROMIS adoption datasets, and OECD digital health data governance filings.
- Market-Sizing and Forecasting: Monetizable dataset volume estimated as a percentage of total commercial patient engagement platform interactions.
- Data Validation and Update Cycle: Revenue triangulation cross-checked against publicly disclosed licensing contracts from major health data brokers.
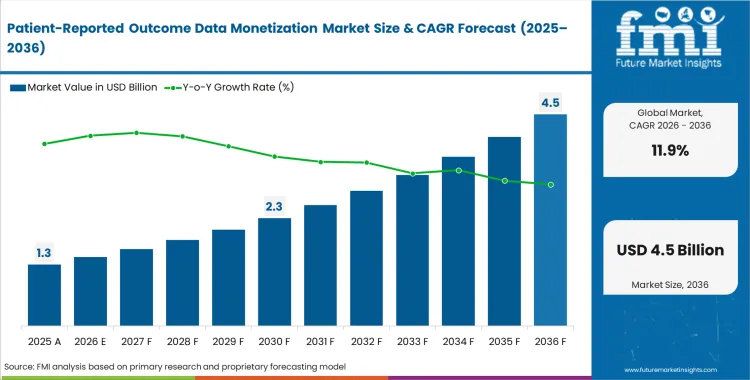
Patient-Reported Outcome Data Monetization Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.3 Billion |
| Industry Value (2036) | USD 4.0 Billion |
| CAGR (2026 to 2036) | 11.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Clinical directors in pharmaceutical companies immediately face a ceiling on drug pricing because they currently lack access to real world evidence solutions that can conclusively prove improvements in patients' quality of life. In the past, procurement teams typically regarded patient diaries as merely secondary documentation for clinical trials. Now, however, buyers are actively purchasing tokenized clinical datasets in order to justify a drug's placement on a formulary for commercial payers. If companies delay this procurement, they will be forced to negotiate reimbursement terms using clinical trial data that is quickly becoming outdated. This process of data brokering effectively constitutes a shadow market for gaining pricing leverage.
The widespread viability of scalable data monetization platforms will be achieved once federated privacy networks are established to tokenize patient identities across various, previously separated hospital systems. Data brokers are the catalysts for this change by offering a zero-copy architecture. The process of aggregating symptom logs, which are often disjointed, transforms the raw textual information into structured, usable assets.
The United States leads at a 13.2% CAGR, driven by aggressive medicare value-based care contracting and an increasingly competitive landscape among health tech startups vying for market dominance. China market revenue expands at a 12.5% CAGR, supported by state-sponsored clinical data consolidation initiatives and significant investment in domestic AI healthcare infrastructure. Germany market grows at an 11.4% CAGR, reflecting stringent GDPR consent frameworks structuring the regional data economy, which encourages secure and transparent data exchange. The United Kingdom reaches an 11.1% CAGR, based on localized NHS data trusts scaling regional access and fostering research collaborations. Japan scales at a 10.6% CAGR, facing slow legacy hospital system integration, though recent government policies aim to accelerate digital transformation efforts. Asian providers build monetization directly into primary care workflows.
Patient-Reported Outcome Data Monetization Market Definition
The market for monetizing patient-reported outcome (PRO) data focuses on commercializing health feedback provided directly by patients through structured licensing. This process involves organizations collecting self-reported data—such as symptom logs, quality-of-life scores, and behavioral data—from clinical settings or consumer devices. Brokers then de-identify this raw feedback and bundle it for sale to external buyers. The core value of this data is its ability to link subjective patient experiences with objective medical claims to demonstrate and validate the efficacy of drugs and treatments.
Patient-Reported Outcome Data Monetization Market Inclusions
Core scope captures revenue generated from selling, licensing, or providing subscription access to structured patient-generated health data. Commercialization of clinical trials support software data falls squarely within parameters. API access fees for continuous patient symptom streams constitute primary inclusion criteria. Federated learning network access fees also qualify, enabling researchers to analyze remote symptom registries directly.
Patient-Reported Outcome Data Monetization Market Exclusions
Hardware sales for patient monitoring wearables fall completely outside analytical boundaries. Direct patient care revenue generated from utilizing these tools remains excluded. Software licensing fees strictly for internal hospital operational use do not qualify. Selling purely clinical electronic health records lacking subjective patient-reported components falls entirely beyond this specific financial boundary.
Patient-Reported Outcome Data Monetization Market Research Methodology
- Primary Research: Chief Data Officers at pharma sponsors, VP of Market Access at payers, and registry network administrators.
- Desk Research: FDA Real-World Evidence Program registries, NIH PROMIS adoption datasets, and OECD digital health data governance filings.
- Market-Sizing and Forecasting: Monetizable dataset volume estimated as a percentage of total commercial patient engagement platform interactions.
- Data Validation and Update Cycle: Revenue triangulation cross-checked against publicly disclosed licensing contracts from major health data brokers.
Segmental Analysis
Patient-Reported Outcome Data Monetization Market Analysis by Data Type
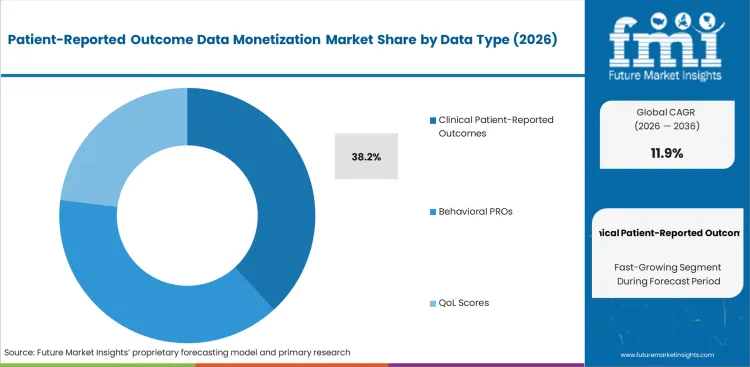
Stringent regulatory compliance frameworks fundamentally drive procurement decisions in this sector. Clinical Patient-Reported Outcomes (PROs) currently account for a substantial 38.5% market share, primarily because explicit recognition of these specific validated instruments, rather than generic patient feedback, is mandated by FDA label reviewers. Regulatory affairs executives acquire these comprehensive datasets specifically to circumvent the substantial time and financial investment associated with mandatory Phase IV post-market trials. The strategic utilization of validated clinical surveys significantly alters compliance officers' negotiating leverage with health authorities. Conversely, selecting eclinical solutions that do not incorporate linked, validated clinical outcome data effectively renders those technology investments entirely unproductive.
- Regulatory validation: FDA approval pathways explicitly require the submission of validated clinical scores for labeling indications related to specific chronic diseases. This mandatory validation provides regulatory affairs directors with an immediate and powerful advantage during critical label expansion negotiations.
- Imputation burden: Raw patient symptom data invariably contains significant longitudinal gaps that necessitate specialized statistical imputation and patching. This forces biostatisticians to confront unexpected and substantial compute costs solely for the purpose of making the licensed datasets analytically usable.
- Phase IV replacement: The strategic purchase of established historical clinical registries serves as a direct replacement for conducting entirely new post-market surveillance studies. This enables clinical trial directors to avoid millions of dollars typically associated with site activation and operational fees.
Patient-Reported Outcome Data Monetization Market Analysis by Monetization Model
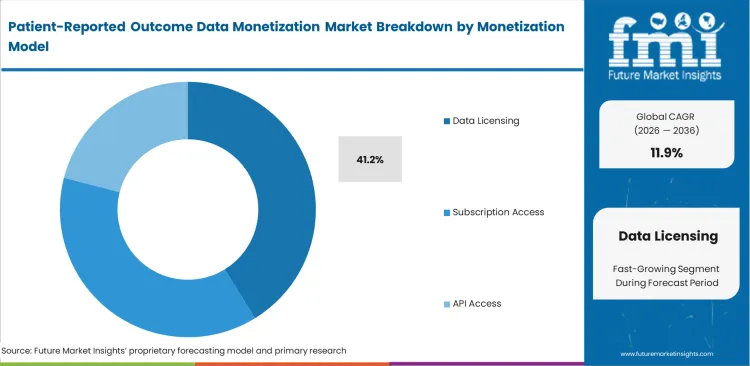
Buyers require direct manipulation capabilities for raw data, moving beyond reliance on pre-packaged dashboard views. Data licensing segment commands a 41.2% market share in 2026, primarily driven by mid-cap pharmaceutical companies acquiring raw patient registry software extracts to fuel their proprietary machine learning models. Obtaining data licenses enables biostatisticians to seamlessly integrate patient feedback with existing medical claims databases. Exclusive dependence on subscription-based insights severely constrains internal data science teams. Although data licensing appears highly lucrative in theory, the continuous compliance overhead associated with de-identification significantly erodes margins for data brokers operating under stringent privacy regulations. Organizations that neglect direct data licensing are unable to construct proprietary AI training sets, forcing total reliance on costly third-party consulting services.
- Internal model training: Raw licensing directly supports proprietary algorithmic development, allowing Chief Data Officers to build unique predictive assets instead of leasing generic industry analyses.
- Compliance cost: Continuous data tokenization necessitates ongoing legal oversight and software updates. Privacy officers allocate substantial operational budgets to maintain the legal liquidity of data.
- Claims integration: Licensed patient feedback integrates smoothly with acquired pharmacy fill data, enabling health economists to accurately calculate actual drug efficacy curves across diverse populations.
Patient-Reported Outcome Data Monetization Market Analysis by End User
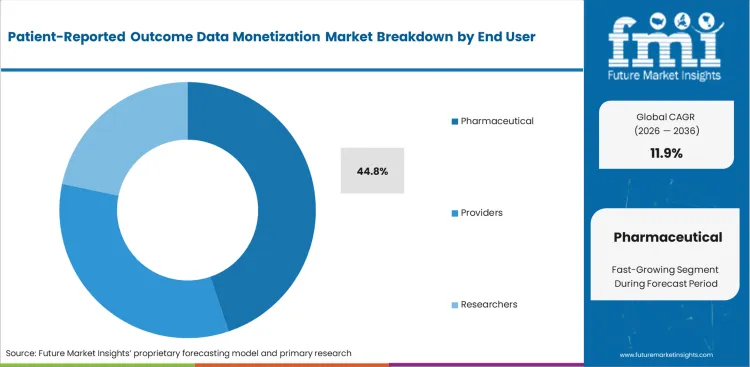
Justifying premium pricing necessitates extensive data acquisition campaigns. The pharmaceutical sector segment accounts for 44.8% share in 2026, driven by the requirement to substantiate improved patient quality-of-life for specialty drug reimbursement. Market access Vice Presidents routinely authorize substantial purchases of patient engagement platforms datasets to counteract payer resistance; leveraging these patient narratives transforms formulary negotiations from theoretical cost-benefit analyses to discussions centered on actual human impact. While competitors primarily perceive pharmaceutical companies as the key buyers, payers are rapidly emerging as 'shadow-purchasers,' surreptitiously procuring identical data through actuarial intermediaries to ultimately deny the very claims submitted by those pharma companies. Drug developers who forgo this data acquisition are critically disadvantaged in pricing negotiations, and pricing teams lacking this evidential support face immediate formulary rejection.
- Formulary negotiation: Specialty drug pricing requires verifiable quality-of-life improvement documentation. Market access VPs secure Tier 1 placement utilizing purchased patient feedback.
- Actuarial defense: Health insurers purchase outcomes data to challenge expensive drug claims. Medical directors deny coverage based on real-world failure rates.
- Targeted marketing: Commercial leads identify specific demographic clusters reporting poor competitor drug experiences. Marketing teams deploy highly targeted switching campaigns based on symptom prevalence.
Patient-Reported Outcome Data Monetization Market Analysis by Application
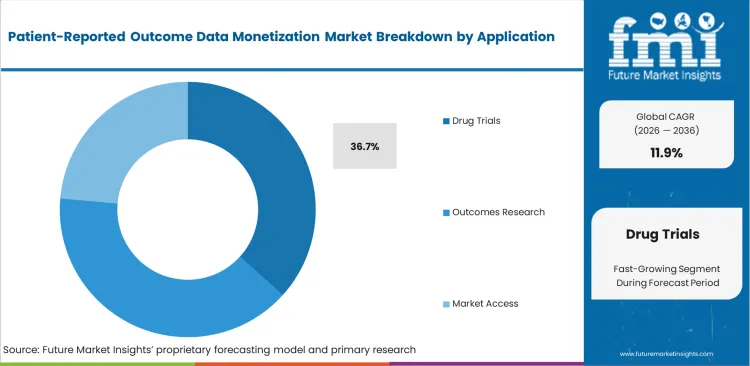
Generating evidence in the early stages of drug development requires extensive historical baselines. The drug trials segment holds 36.7% share in 2026, driven by sponsors acquiring historical cohort data to optimize inclusion criteria. Clinical operations directors purchase clinical data provenance logs to pinpoint the precise geographical locations of targeted patient populations, a practice where the acquisition of historical data fundamentally transforms site selection strategy. While most trial planners view this data solely as a recruitment resource, historical PRO datasets can effectively serve as synthetic control arms, potentially eliminating the requirement for placebo groups altogether. Sponsors initiating trials without this historical intelligence often encounter significant enrollment delays, as the failure to accurately map historical patient clusters compels clinical operations directors to activate unproductive clinical sites.
- Site selection: Historical symptom density maps identify optimal trial locations. Clinical operations directors avoid activating sites in patient deserts.
- Synthetic controls: Dense historical patient logs serve as baseline comparison groups. Biostatisticians eliminate actual placebo administration.
- Endpoint design: Reviewing past patient complaints highlights exactly the symptoms mattering to target populations. Trial designers write highly relevant primary endpoints.
Patient-Reported Outcome Data Monetization Market Analysis by Deployment
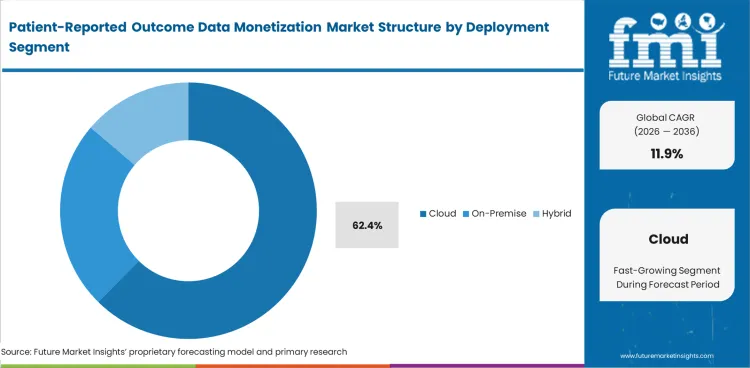
Effective cross-border data mobility necessitates zero-copy architectures. The cloud segment commands a 62.4% share in 2026, sustained by the operational reality that federated learning networks cannot function efficiently on isolated servers. IT infrastructure directors are implementing cloud healthcare API solutions to securely query data, thus circumventing the requirement to physically transfer patient records across international boundaries. The utilization of cloud infrastructure immediately resolves data residency legal mandates, and while the market generally perceives cloud adoption as an IT cost reduction measure, its primary function is actually to serve as a legal liability shield, ensuring that data custodianship remains firmly with the originating hospitals. Conversely, facilities that insist on localized, on-premise monetization strategies consequently isolate themselves from global pharmaceutical procurement networks and entirely forfeit highly profitable international research grants.
- Federated queries: Remote algorithms analyze patient logs without extracting raw text. Privacy officers approve data commercialization without violating local residency laws.
- Scale economics: Massive datasets require elastic compute resources during tokenization processing. IT infrastructure directors avoid massive capital hardware expenditures.
- Network effects: Cloud platforms instantly connect localized registries to global buyers. Hospital administrators gain immediate access to international pharma budgets.
Patient-Reported Outcome Data Monetization Market Drivers, Restraints, and Opportunities
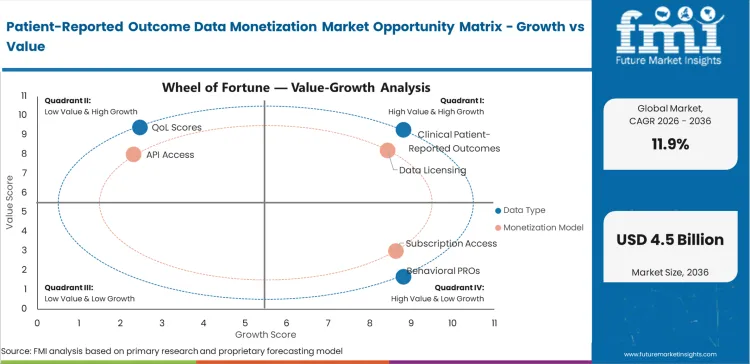
Losing Tier 1 formulary placement jeopardizes billions in specialty drug revenue. Market access vice presidents are acquiring tokenized patient datasets, because commercial payers now refuse to cover costly therapeutics without real-world documentation of quality-of-life benefits. Delaying this purchase completely shifts negotiating leverage to health insurers. Buyers currently contract with digital healthcare brokers to link specific symptom relief to pharmacy fill rates. Waiting for phase four trial results is a multi-year process. Buying existing patient logs offers instant statistical support for pricing negotiations today.
Severe long-term data gaps compromise the accuracy of predictive models. Biostatisticians struggle to derive value from licensed datasets because patients rarely maintain consistent symptom diaries. Hardware manufacturers prioritize initial user engagement over multi-year retention, embedding this core challenge. New telehealth software integrates passive background tracking to combat abandonment, yet passive systems still lack the granular, subjective emotional scores required by regulatory bodies.
Opportunities in the Patient-Reported Outcome Data Monetization Market
- Tokenized linkage pipelines: Connecting isolated symptom apps to national claims databases. Chief Data Officers sell highly enriched longitudinal profiles.
- Synthetic control generation: Packaging historical disease progression logs for trial sponsors. Clinical directors bypass massive placebo-group recruitment costs entirely.
- Automated consent architectures: Deploying smart contracts for patient data monetization platform agreements. Compliance officers drastically reduce legal overhead per transaction.
Regional Analysis
.webp)
Based on regional analysis, the patient-reported outcome data monetization market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 13.2% |
| China | 12.5% |
| Germany | 11.4% |
| United Kingdom | 11.1% |
| Japan | 10.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Patient-Reported Outcome Data Monetization Market Analysis
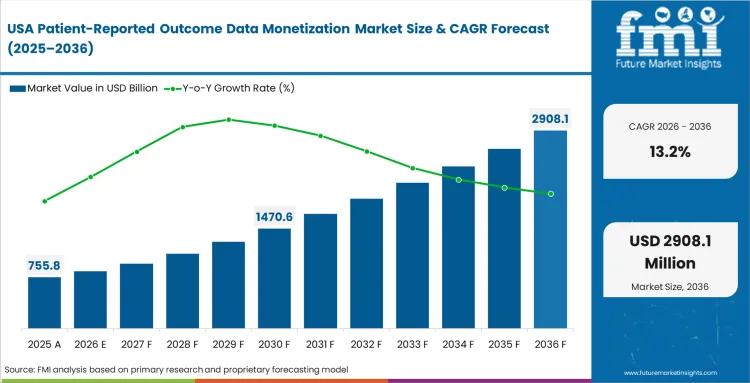
The increasing pressure from aggressive medicare value-based care mandates make it necessary for significant data consumption, imposing severe financial penalties on health systems that fail to prove actual patient improvement. Commercial payers echo this requirement, demanding similar proof for specialty drug coverage. According to FMI's estimates, these dual pressures are driving substantial investment in tokenized health records. This shift allows hospitals to convert compliance reporting costs into new profit centers through direct data sales.
- United States: The market is projected to expand at a 13.2% CAGR, driven by the need to comply with evolving Medicare reimbursement and value-based care models. Private commercial insurers frequently deny specialty drug claims lacking real-world quality-of-life documentation, prompting market access directors to purchase extensive tokenized datasets to secure coverage approvals. Hospitals are establishing proprietary patient registries, securing highly lucrative exclusive licensing deals with major drug developers. These direct data sales are transforming traditional hospital IT compliance centers into major revenue streams, consequently altering the balance of power during regional payer negotiations.
- Canada: Market growth is steady, supported by centralized public health data repositories seeking to monetize anonymized clinical registries. Provincial health authorities are increasingly mandating structured outcomes tracking for novel therapeutics, deploying standardized symptom collection tools across public networks. This allows researchers to access deep demographic pools for longitudinal efficacy studies, circumventing the high overhead associated with independent cohort recruitment.
Europe Patient-Reported Outcome Data Monetization Market Analysis
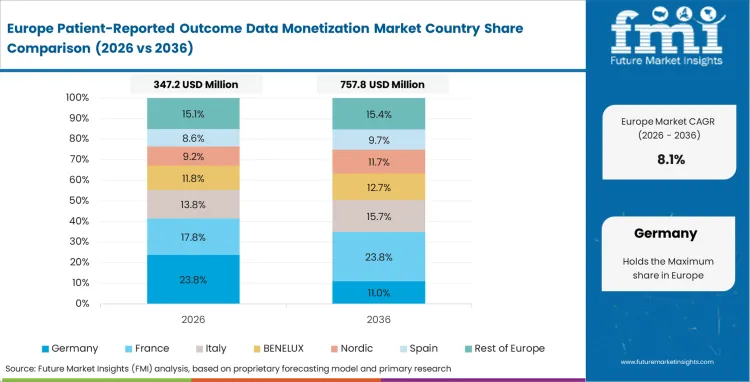
Strict GDPR consent frameworks intentionally impede data velocity. Health data exchanges necessitate explicit opt-in mechanisms for every secondary commercial application. Data brokers allocate substantial operational budgets to maintaining legal compliance instead of acquiring new datasets. FMI's analysis suggests Europe countries prioritize privacy maintenance over commercial AI development.
- Germany: Stringent local data privacy regulations prohibit the cross-border export of raw patient logs. Compliance officers mandate federated query architectures for all external pharmaceutical research. Germany registers 11.4% CAGR, supported by the rapid deployment of localized federated learning networks specifically engineered to circumvent international data transfer restrictions. Organizations proficient in localized federated learning establish a significant competitive advantage against foreign data aggregators attempting to penetrate the domestic health data market.
- United Kingdom: Localized NHS data trusts maintain rigorous control over citizen health records. Regional health administrators offer restricted analytical access rather than raw data exports. The United Kingdom is projected to register 11.1% CAGR through 2036, driven by government-supported initiatives aimed at commercializing public health data while rigorously protecting patient anonymity. Global pharmaceutical purchasers are adjusting their expectations to accept aggregated insights instead of the preferred raw patient logs, fundamentally reshaping clinical research strategies throughout the region.
Asia Pacific Patient-Reported Outcome Data Monetization Market Analysis
- State‑sponsored clinical data consolidation removes interoperability barriers, with centralized health authorities enforcing strict formats across public hospitals. Researchers access massive longitudinal cohorts without negotiating multiple institutional review boards. FMI’s assessment indicates centralized architecture enables unprecedented AI model training speeds.
- China: Centralized hospital networks deploy standardized symptom tracking apps across millions of chronic disease patients. Provincial health directors license structured datasets to domestic biotech firms. China grows at 12.5% CAGR, fueled by state investment in unified health data systems and a push to accelerate pharmaceutical innovation. Domestic datasets shorten drug development timelines, giving China a competitive edge in global specialty drug markets.
- Japan: Aging hospital infrastructure creates bottlenecks for real‑time symptom data extraction. IT directors struggle to export patient logs from legacy servers. Japan expands at 10.6% CAGR, supported by government subsidies for healthcare IT upgrades. Slower integration sustains domestic market share to some extent for specialized local data integration vendors operating exclusively within national hospital networks.
Competitive Aligners for Market Players
Control over the interoperability layer dictates competitive power entirely. IQVIA and Optum maintain massive influence not by generating most patient feedback, but by owning tokenization engines linking isolated symptom logs to actual medical claims. Smaller app developers generate highly engaged symptom data. Niche vendors cannot prove clinical value until they pay toll fees to connect data with pharmacy fill records held by dominant life science analytics players.
Incumbents possess deep libraries of pre-cleared institutional review board approvals. Navigating hospital legal departments takes years. Established data brokers hold master agreements covering hundreds of clinical sites. Startups building slick patient tracking apps hit a massive wall attempting to license that data externally. Hospitals refuse to share liability with unproven vendors. TriNetX leverages its massive established academic medical center network to bypass this exact friction.
Large pharmaceutical buyers actively resist exclusive data broker dependencies. Procurement directors split contracts across multiple data vendors to prevent pricing extortion during critical Phase III trials. Relying purely on Flatiron Health for oncology data limits negotiation leverage. Sponsors increasingly fund open-source tokenization standards to commoditize linkage layers. Proprietary data silos face severe pricing pressure as interoperability protocols force brokers to compete on analytical quality rather than mere data access.
Key Players in Patient-Reported Outcome Data Monetization Market
- IQVIA
- Oracle Health
- Optum (UnitedHealth Group)
- Medidata Solutions (Dassault Systèmes)
- Flatiron Health (Roche)
- TriNetX
- HealthVerity
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.3 Billion in 2026 to USD 4.0 Billion by 2036, at a CAGR of 11.9% |
| Market Definition | The patient-reported outcome data monetization market commercializes direct-from-patient health feedback through structured licensing architectures. Organizations aggregate self-reported symptom logs, quality-of-life scores, and behavioral trackers from clinical or consumer devices. Brokers de-identify this raw feedback and package it for external buyers. Value stems entirely from linking subjective patient narratives with verifiable medical claims to prove drug efficacy. |
| Segmentation | Data Type, Monetization Model, End User, Application, Deployment, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, Japan, South Korea, Taiwan, Singapore, Brazil, Mexico, Argentina, GCC Countries, South Africa, Israel, Rest of Middle East & Africa |
| Key Companies Profiled | IQVIA, Oracle Health, Optum, Medidata Solutions, Flatiron Health, TriNetX, HealthVerity |
| Forecast Period | 2026 to 2036 |
| Approach | Monetizable dataset volume estimated as a percentage of total commercial patient engagement platform interactions. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Patient-Reported Outcome Data Monetization Market Analysis by Segments
- Data Type
- Clinical PROs
- Behavioral PROs
- QoL Scores
- Symptom Logs
- Monetization Model
- Data Licensing
- Subscription Access
- API Access
- Insights Sales
- End User
- Pharma
- Payers
- Providers
- Researchers
- Application
- Drug Trials
- Outcomes Research
- Market Access
- Post-Market
- Deployment
- Cloud
- On-Premise
- Hybrid
- Region
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Rest of Middle East & Africa
- North America
Bibliography
- USA Food and Drug Administration. (2024, January). Focus area: Patient-reported outcomes and clinical outcome assessments.
- Brown, B. R., Samuel, J., Teeters, M., Vyas, A., & Bossaer, J. (2025, November). Characterizing patient-reported outcome data availability for oral anticancer therapies.
- Paravathaneni, M., et al. (2024, January). Patient-reported outcomes in clinical trials leading to cancer drug approvals: A systematic review.
- Zhang, X., et al. (2026, March). Patient-reported outcome measures in clinical trials: Trends from 2008 to 2023.
- European Medicines Agency. (2025). Use of patient experience data and real-world evidence in regulatory decision-making.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Tokenization strategies linking symptom logs to medical claims.
- Pricing leverage generated by clinical outcomes datasets during formulary negotiations.
- Interoperability constraints blocking raw data exports from legacy hospital systems.
- Federated query architectures solving cross-border privacy mandates.
- Actuarial reliance on real-world evidence for specialty drug claim denials.
- Synthetic control group creation utilizing historical patient feedback cohorts.
- Statistical imputation costs associated with longitudinal data gaps.
- Regional infrastructure disparities dictating monetization protocol deployment speed.
Frequently Asked Questions
What is the valuation of the patient-reported outcome data monetization market?
Demand scales to USD 1.3 Billion in 2026. This signals massive pharma reliance on real-world evidence.
What value does the patient-reported outcome data monetization market reach by 2036?
Revenue crosses USD 4.0 Billion by 2036. Regulatory acceptance of synthetic control arms drives this expansion.
What is the CAGR for the patient-reported outcome data monetization market?
Growth registers at 11.9% through 2036. Value-based contracting mandates force continuous data procurement.
Why do Clinical PROs lead by Data Type?
FDA pathways explicitly recognize validated clinical instruments. Regulatory affairs directors buy these specific datasets to accelerate label expansion without launching costly Phase IV studies.
Why does Data Licensing dominate the Monetization Model?
Biostatisticians require raw manipulation capabilities. Renting dashboard views restricts internal machine learning development. Licensing allows direct merging with existing medical claims databases.
Why does Pharma represent the top End User?
Formulary placement requires verifiable quality-of-life documentation. Market access VPs spend millions on patient data to justify premium pricing for specialty therapeutics.
Why do Drug Trials capture significant Application share?
Sponsors use historical symptom datasets as synthetic control arms. Clinical trial planners bypass massive placebo-group recruitment costs using existing demographic intelligence.
Why does Cloud deployment secure maximum share?
Federated query architecture requires elastic compute resources. IT directors query cross-border patient logs without violating strict localized data residency laws.
How does India compare to the United States ?
India grows faster due to greenfield digital rollouts lacking legacy technical debt. The United States scales on aggressive Medicare value-based contracting and private payer requirements.
Why do Europe markets trail Asia growth rates?
Strict GDPR consent frameworks force explicit opt-in mechanics. Data brokers spend massive operational budgets maintaining legal compliance rather than accelerating dataset acquisition.
What prevents niche app developers from monetizing data directly?
Standalone symptom data lacks clinical utility. Apps must pay tokenization fees to major brokers to link logs with verifiable pharmacy claims.
How do large buyers resist data vendor lock-in?
Procurement directors split contracts across multiple data aggregators. Sponsoring open-source tokenization standards commoditizes linkage layers entirely.
What operational friction destroys data licensing margins?
Continuous tokenization requires constant legal oversight. Privacy officers consume massive operational budgets maintaining compliance under constantly shifting regional data residency laws.
How do payers utilize this data against drug sponsors?
Medical directors buy outcomes data through third-party actuaries. Identifying high real-world failure rates justifies denying expensive specialty drug claims.
What role do academic medical centers play in supply?
Large research hospitals hold massive deep-phenotype registries. Exclusive master agreements with these facilities grant data brokers massive leverage over pharma buyers.
How does site selection change with historical PRO data?
Clinical operations directors map symptom density geographically. Activating trial sites in verified high-density patient zones prevents massive enrollment delays.
What dictates cloud adoption beyond simple storage costs?
Cloud acts primarily as a liability shield. Federated learning keeps actual custodianship firmly with originating hospitals while permitting remote commercial algorithms.
How do patient registries function as competitive weapons?
Exclusive licensing deals lock competitors out of specific disease states. Holding validated outcomes datasets forces rivals into longer traditional trial formats.
What commercial consequence faces sponsors delaying data procurement?
Entering pricing negotiations without real-world quality-of-life proof strips sponsors of leverage. Insurers immediately demand massive discounts on targeted specialty drugs.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Data Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Data Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Data Type , 2026 to 2036
- Clinical Patient-Reported Outcomes
- Behavioral PROs
- QoL Scores
- Clinical Patient-Reported Outcomes
- Y to o to Y Growth Trend Analysis By Data Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Data Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Monetization Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Monetization Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Monetization Model, 2026 to 2036
- Data Licensing
- Subscription Access
- API Access
- Data Licensing
- Y to o to Y Growth Trend Analysis By Monetization Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Monetization Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharmaceutical
- Providers
- Researchers
- Pharmaceutical
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Drug Trials
- Outcomes Research
- Market Access
- Drug Trials
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud
- On-Premise
- Hybrid
- Cloud
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Data Type
- By Monetization Model
- By End User
- By Application
- By Deployment
- Competition Analysis
- Competition Deep Dive
- IQVIA
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Oracle Health
- Optum (Value (USD Million)edHealth Group)
- Medidata Solutions (Dassault Systèmes)
- Flatiron Health (Roche)
- TriNetX
- IQVIA
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Monetization Model, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Data Type
- Figure 6: Global Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Monetization Model
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Application
- Figure 15: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Deployment
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Data Type
- Figure 32: North America Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Monetization Model
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Application
- Figure 41: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Deployment
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Data Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Monetization Model
- Figure 51: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by End User
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Deployment
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Data Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Monetization Model
- Figure 67: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Application
- Figure 73: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Deployment
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Data Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Monetization Model
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End User
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Application
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Data Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Monetization Model
- Figure 99: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by End User
- Figure 102: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Application
- Figure 105: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Deployment
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Data Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Monetization Model
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Data Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Monetization Model, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Monetization Model, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Monetization Model
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

