Precision Fermentation Ingredient Supplier Market
The report analyzes the precision fermentation ingredient supplier market by ingredient type, form, application, microbe type, customer type, and region from 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Precision Fermentation Ingredient Supplier Market Size, Market Forecast and Outlook By FMI
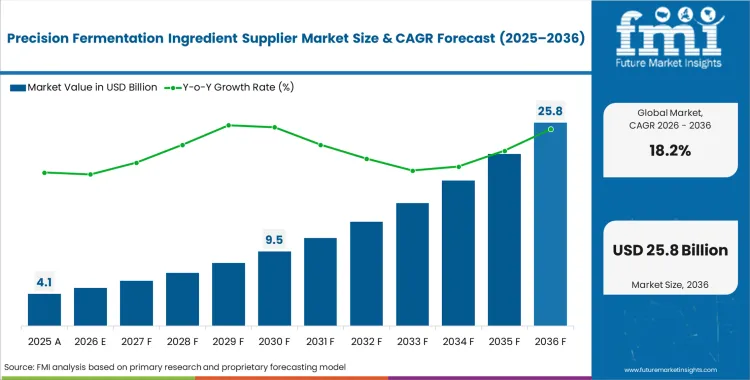
The precision fermentation ingredient supplier market was valued at USD 3.5 billion in 2025. Demand is set to cross USD 4.1 billion in 2026. The market revenue is expected to reach USD 21.8 billion through 2036, at a CAGR of 18.2% during this forecast period, as pilot-scale bio-manufacturing infrastructure finally crosses into commercial volume production for dairy and egg equivalents.
Summary of Precision Fermentation Ingredient Supplier Market
- The market is forecast to reach USD 21.8 billion by 2036.
- The market is expected to grow at a CAGR of 18.2% from 2026 to 2036.
- The market was estimated at USD 3.5 billion in 2025.
- The forecast period represents an incremental opportunity of USD 17.7 billion.
- The market is a B2B ingredient space where functionality, regulatory clearance, and scale-up economics outweigh consumer branding.
- Growth is driven by rising demand for animal-identical proteins offering consistency and reduced dependence on traditional agriculture.
- Precision-fermented whey, casein, and egg proteins are gaining traction due to superior solubility, foaming, and formulation performance.
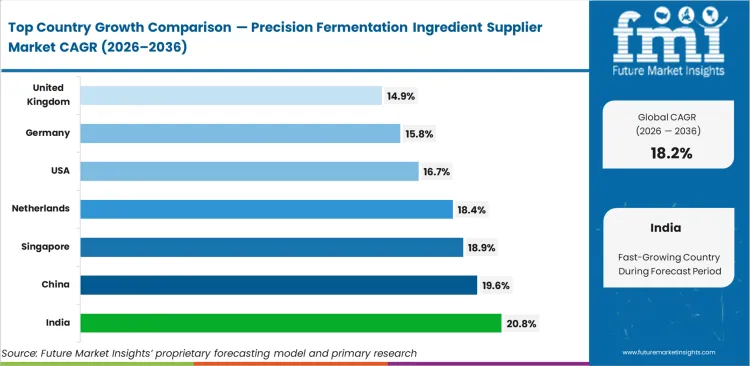
- India, China, and Singapore are among the fastest-growing markets, with India leading at a 20.8% CAGR through 2036.
- Proteins dominate the ingredient segment, while powders remain the leading form due to shelf-stable B2B distribution.
- Food & beverage is the largest application segment, supported by dairy alternatives, sports nutrition, and reformulation demand.
- Key players in the market include Perfect Day, Vivici, Remilk, Imagindairy, The Every Company, Onego Bio, and Verley Food.
Formulation scientists encounter significant constraints with traditional plant-based isolates, notably concerning solubility and flavor mitigation. A pivot toward fermented protein systems directly addresses these functional shortcomings. Research and Development departments currently calculate precise qualification schedules for assessing alternative protein ingredient vendors. This action secures supply before large food organizations consume all available upstream capacity. Procurement teams seldom account for the necessity of restarting full stability timelines. Validating a novel bio-identical whey necessitates this comprehensive examination. Vendor selection delays result in missing retail product refresh periods entirely.
FDA GRAS clearance facilitates immediate, widespread adoption across international supply chains. Federal regulators issue no-questions letters for specific microorganisms, guaranteeing absolute safety. Prudent category managers promptly transition pilot initiatives into complete commercial production stages. This regulatory benchmark rapidly transforms initial experimentation into enduring supply-chain integration. Food processors depend entirely on these federal achievements to proceed safely with introductions of new products. This safety guarantee ensures consistent volume procurement agreements for expanding facilities.
Transition toward fermented protein systems, supported by FDA GRAS clearances, is accelerating formulation acceptance and commercial rollout across global food markets. India is forecast to register a 20.8% CAGR through 2036, while adoption across China is projected to expand at 19.6% CAGR, supported by state‑led protein self‑sufficiency initiatives. Singapore is expected to post 18.9% CAGR due to agile novel‑food regulation, followed by the Netherlands at 18.4%. The United States is set to advance at 16.7% CAGR, with Germany and the UK projected to grow at 15.8% and 14.9%, respectively through 2036.
Precision Fermentation Ingredient Supplier Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4.1 billion |
| Industry Value (2036) | USD 21.8 billion |
| CAGR (2026-2036) | 18.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
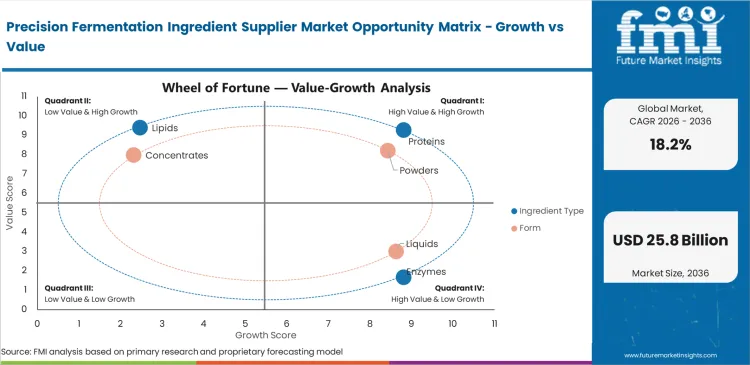
Segmental Analysis
Precision Fermentation Ingredient Supplier Market Analysis by Ingredient Type
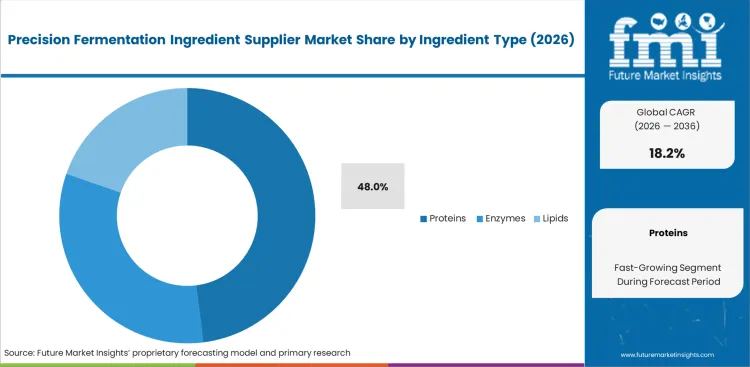
Plant-based ingredients frequently fail to withstand the intense heat processing conditions characteristic of industrial manufacturing. FMI's assessment shows that substituting native dairy products with microbially produced equivalents completely removes the need for off-note masking. Research and development teams at bakery companies consider this clean-label advantage, stemming from precision fermented egg protein suppliers, a premium attribute. The proteins segment is estimated to account for a 48.0% revenue share in 2026, as formulation chemists urgently require heat-stable binding agents offering functionality comparable to animal-derived alternatives. This attribute easily justifies initial price premiums for companies adopting the technology early. Deferring pilot trials forces those same brands to depend on volatile agricultural egg markets. Competitors securing a consistent supply of bio-identical materials rapidly gain market share from hesitant late entrants.
- Initial pilot validation: Bench-scale functionality tests accurately establish successful foaming specifications. Formulation chemists only advance prototypes exhibiting behavioral characteristics identical to native ingredients.
- Commercial scale qualification: Successful pilot runs necessitate stringent full-scale facility stress evaluations. Plant managers thoroughly assess synthetic binders under high-speed industrial extrusion parameters.
- Long-term contract expansion: Reliable commercial performance logically dictates product portfolio growth. Procurement teams consolidate purchasing power with suppliers guaranteeing flawless uninterrupted volume delivery.
Precision Fermentation Ingredient Supplier Market Analysis by Form
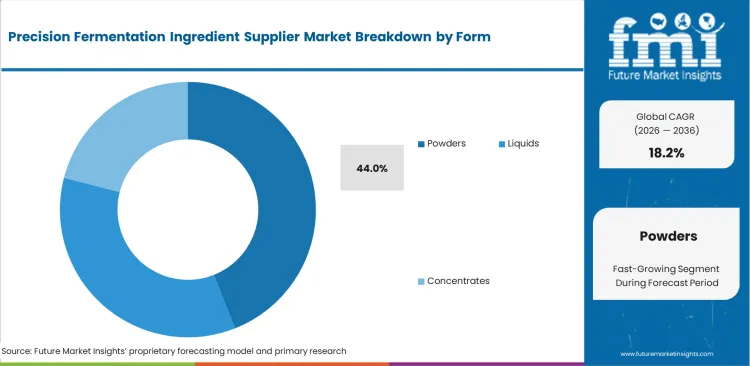
Standardized shelf-life requirements necessitate particular formatting decisions throughout global supply chains. The powders segment is likely to account for a 44.0% revenue share in 2026 as the unit economics of shipping water internationally are compromised, pushing buyers toward the concentrated powder alternative. Shipping water internationally compromises baseline unit economics, leading buyers toward this concentrated format. Converting fermentation broths into stable dry formats enables seamless integration, based on FMI's estimates. This perfectly aligns with existing material handling infrastructure for large-scale distributors. Logistics managers at multinational distribution companies reject liquid systems needing specialized cold-chain transport. Generalized market forecasts frequently overlook a critical processing vulnerability. Excessive drying temperatures often denature fragile fermented casein molecules prior to reaching a mixing vat. Drying engineers at precision fermented casein suppliers devote months to optimizing spray-drying parameters. This action preserves delicate protein functionality during the vital moisture removal phase. Brands selecting sub-optimal powder suppliers face the risk of catastrophic product failures during final consumer preparation. Careful vendor selection remains essential for ultimate product.
- Spray-drying economics: Moisture removal employing high-efficiency spray dryers represents a significant capital expenditure. Facility engineers meticulously balance energy consumption against ultimate powder solubility parameters.
- Hidden rehydration costs: Poorly milled particles clump severely during downstream processing operations. Mixing personnel spend extensive amounts of time attempting to fully dissolve inferior dry formats.
- Lifecycle transport savings: Concentrated dry shipments significantly reduce global freight expenditures. Supply chain departments maximize margin capture through the elimination of liquid weight from international cargo manifests.
Precision Fermentation Ingredient Supplier Market Analysis by Application
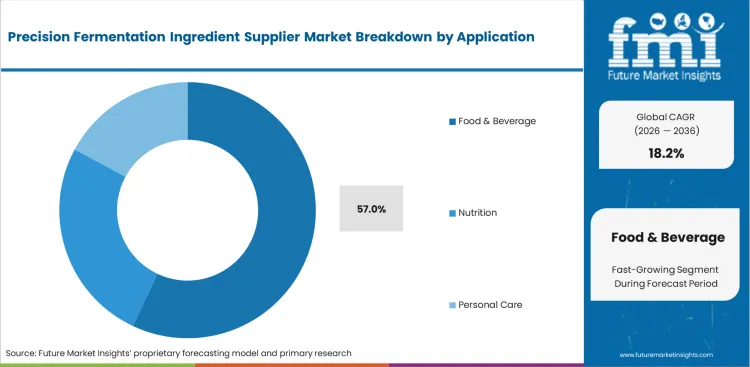
Consumer demand for sustainable nutrition clashes directly with stubborn taste preferences frequently. Brands attempt to bridge this exact taste divide, supporting this massive application footprint. Based on FMI's assessment, introducing bio-identical whey into performance drinks satisfies vegan positioning. The food & beverage segment is likely to hold 57.0% revenue share in 2026. Utilizing fermented flavors often triggers unpredictable foaming during high-speed bottling runs. Packaging line supervisors must recalibrate filling equipment to accommodate these novel behavioral characteristics. Failing to adjust line speeds results in unacceptable retail defect rates directly. Production delays escalate costs for unprepared manufacturing facilities significantly. Anticipating these behavioral quirks saves vast amounts of operational capital.
- Early sports adoption: Performance nutrition labels prioritize functional whey replacements first. Target demographics readily accept novel biotechnology delivering superior muscle-recovery metrics reliably.
- Mainstream dairy transition: Everyday yogurt and cheese brands adopt microbial inputs eventually. Category managers focus on seamless taste substitution without alienating conservative shoppers.
- Late bakery conversion: Commercial bakeries resist transition until egg-replacement economics become overwhelmingly favorable. Cost-obsessed procurement departments require absolute price parity before modifying legacy recipes.
Precision Fermentation Ingredient Supplier Market Analysis by Microbe Type
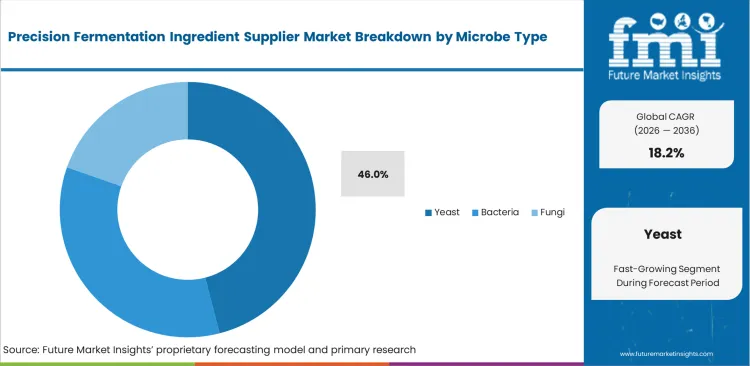
Scaling upstream production requires chassis organisms with proven industrial suitability, a need currently dominated by the yeast segment, which is estimated to account for a 46.0% share in 2026. This market leadership is firmly secured by yeast's inherent genetic flexibility and the existence of decades of established brewery-scale infrastructure. In sharp contrast, alternative systems pose considerable vulnerabilities, such as hyper-productive mycoprotein strains suffering oxygen deprivation in standard commercial tanks, which necessitates expensive retrofitting to prevent batch loss.
- Established genetic libraries: Suppliers utilize extensively documented yeast strains to accelerate initial target molecule development. Molecular biologists avoid regulatory hurdles by selecting organisms with proven safety histories.
- Bioreactor oxygen constraints: High-density fermentations demand massive aeration infrastructure continuously. Facility managers struggle to maintain dissolved oxygen levels during peak exponential growth phases.
- Next-generation algae shifts: Phototrophic hosts offer theoretical cost advantages regarding feedstock inputs. R&D departments monitor these early-stage platforms for future decade-level capacity planning.
Precision Fermentation Ingredient Supplier Market Analysis by Customer Type
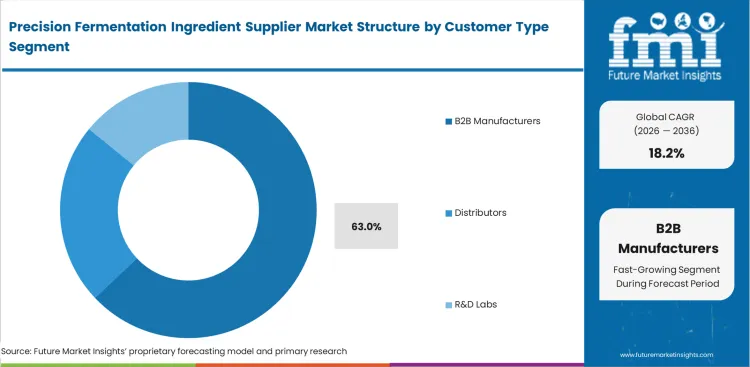
Achieving substantial volume necessitates direct sales into significant industrial production schedules. The B2B manufacturers segment is expected to account for a 63.0% share, factors driving this dominance are established procurement infrastructure, demand for consistent high-volume supply, and consumer brand reliance on industrial partners. Upstream bio-facilities establish direct alignment with global food processors, sustaining this principal customer channel. Establishing these direct pipelines, in FMI's perspective, completely bypasses fragmented retail distribution constraints. Corporate sourcing departments mandate stringent audits before issuing a precision fermentation ingredient Request for Quotation (RFQ). These departments integrate new alternative protein ingredients into core brands with prudence. Outside observers frequently overlook a demanding administrative reality.
- Premium niche performance: Small luxury brands accept higher pricing for initial bespoke ingredient deliveries. Sourcing departments at these agile companies validate novel concepts faster than massive conglomerates.
- Mid-market formulation gaps: Regional processors struggle to secure allocations from overbooked upstream bioreactors. Operations departments face severe supply risks relying on single-source fermentation partners.
- Global conglomerate standards: Massive multinational corporations impose draconian purity and volume requirements strictly. Quality assurance departments enforce zero-tolerance defect policies reliably met only by mature suppliers.
Precision Fermentation Ingredient Supplier Market Drivers, Restraints, and Opportunities
Frequent supply chain disruptions involving avian flu and volatile dairy pricing force massive food conglomerates into action. They must fundamentally rethink their primary ingredient sourcing strategies immediately. Procurement departments face escalating pressure to stabilize input costs regardless of regional agricultural shocks. Locking in bio-identical proteins via precision fermentation protein suppliers offers exact pricing predictability. This financial stability extends over multi-year horizons securely. Transitioning away from animal agriculture reduces corporate carbon footprints drastically. This strategic pivot satisfies aggressive internal sustainability targets mandated by executive boards. Delaying this supply chain evolution leaves brands dangerously exposed to future commodity spikes. Brands also face increasingly stringent environmental regulations globally. This urgency pushes pilot programs into full commercial execution rapidly.
Downstream processing bottlenecks create severe friction slowing widespread commercial adoption. Extracting, purifying, and drying target molecules from complex fermentation broths requires highly specialized filtration infrastructure. Plant managers consistently discover a frustrating operational reality during initial scaling attempts. Downstream purification costs easily exceed the actual upstream fermentation expenses continuously. This physical limitation persists because legacy dairy processing equipment rarely handles microbial separation efficiently. Upgrading this equipment demands extensive retrofitting budgets. Category managers learn a harsh financial truth quickly. Capital requirements for continuous-filtration technologies remain prohibitively high for early-stage start-ups.
Opportunities in the Precision Fermentation Ingredient Supplier Market
- High-margin specialty enzymes: Developing bespoke biocatalysts offers superior margins compared to bulk commodity proteins. R&D departments target these niche applications to generate early revenue streams rapidly. This strategic focus ensures immediate cash flow for expanding upstream operations.
- Hybrid formulation platforms: Blending synthetic whey with traditional plant isolates creates optimized texture profiles. Formulation chemists use fermented functional lipids to mask lingering off-notes in hybrid dairy alternatives. This approach accelerates consumer acceptance of novel product lines significantly.
- Decentralized facility partnerships: Co-locating bioreactors near major agricultural feedstock sources reduces transportation overhead. Operations departments establish these strategic hubs to secure cheap carbon inputs. Securing localized feedstocks insulates production from volatile global shipping costs.
Regional Analysis
Based on regional analysis, the precision fermentation ingredient supplier market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 20.8% |
| China | 19.6% |
| Singapore | 18.9% |
| Netherlands | 18.4% |
| United States | 16.7% |
| Germany | 15.8% |
| United Kingdom | 14.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Precision Fermentation Ingredient Supplier Market Analysis
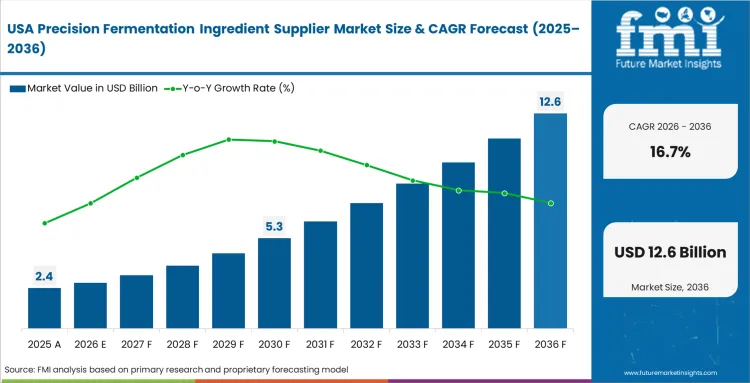
Venture capital density sustains aggressive early-stage platform development across North America hubs. Massive funding rounds allow domestic start-ups to build proprietary pilot facilities independently. This financial independence frees them from relying entirely on contract manufacturers abroad. FMI observes this capital advantage accelerates strain optimization cycles significantly. Procurement teams at major consumer brands readily test these rapid iterations. This testing secures early mover advantages in retail dairy-alternative aisles. Deep integration between software analytics and biological engineering defines this regional approach perfectly. The North America region is expected to dominate early commercialization volumes aggressively. Massive production runs guarantee strong B2B supply chain integrations over the forecast period.
- United States: United States market is expected to register a 16.7% Compound Annual Growth Rate (CAGR) during the forecast period, key factors driving this trajectory include robust Food and Drug Administration (FDA) Generally Recognized As Safe (GRAS) framework establishing a relatively straightforward commercialization path for new microbial proteins, and the substantial early commercialization presence that gives US-based fermented ingredients suppliers a significant global competitive advantage. The clarity provided by the GRAS framework allows category managers confidence in incorporating approved ingredients into prominent beverage product lines. Furthermore, operations departments efficiently scale production capacity to meet the substantial domestic demand for ingredient formulations.
Europe Precision Fermentation Ingredient Supplier Market Analysis
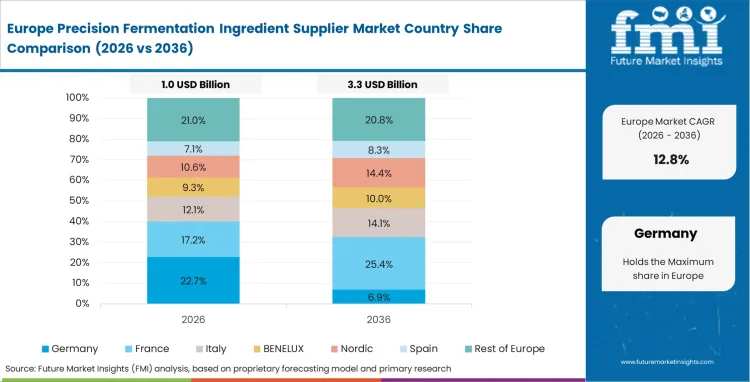
Strict precision fermentation ingredient regulatory challenges dictate adoption speeds across European jurisdictions. Companies face extended safety dossier reviews before commercializing any genetically engineered outputs. These rigorous standards inadvertently create massive protective moats for early movers. Successful navigators of the bureaucratic gauntlet secure uncontested market shares easily. Sourcing departments at Europe dairy conglomerates increasingly invest directly in compliant upstream start-ups. This proactive investment strategy guarantees future supply access securely. A highly consolidated supplier landscape emerges, dominated by well-funded consortiums. The Europe region is expected to lead in specialized dairy-protein replacements eventually. Long-term Europe commitments promise immense profitability for fully authorized biotechnology firms.
- Netherlands: Dense food-tech ecosystems drive rapid collaborative innovation between universities and commercial processors. Innovation heads utilize this concentrated expertise to solve stubborn downstream purification bottlenecks. Market revenue is estimated to grow at a rate of 18.4% during the forecast period, this synergistic environment continues to strengthen the Netherlands position as a leading hub for advanced dairy protein alternatives and specialized fermentation derived ingredients.
- Germany: Plant managers optimize energy consumption metrics to meet stringent domestic sustainability mandates. This operational excellence reduces unit costs significantly for domestic manufacturers over time. Germany is estimated to register 15.8% CAGR during the forecast period, owing to deep precision engineering heritage accelerates the deployment of hyper-efficient automated bioreactor systems, supporting this strong expansion.
- United Kingdom: United Kingdom is set to expand at a 14.9% CAGR during the forecast period, as Post-Brexit regulatory divergence presents unique opportunities for accelerated novel food approvals, sustaining this steady adoption rate. Strategy departments monitor these framework shifts to bypass slower continental authorization timelines. This environment encourages localized pilot investments by ambitious international biotechnology firms.
Asia Pacific Precision Fermentation Ingredient Supplier Market Analysis
State-backed bioeconomy priorities accelerate domestic capacity building across Asian manufacturing hubs. Aggressive government funding transforms pilot concepts into massive industrial reality swiftly. This localized infrastructure boom directly targets severe regional protein deficits. It eliminates reliance on imported agricultural commodities effectively. Regional brand owners pivot swiftly toward these novel inputs. They satisfy expanding middle-class nutritional demands through advanced bio-manufacturing. This apparent food security initiative simultaneously establishes formidable export capabilities. The Asia Pacific is expected to scale production volumes massively. Dominating the global contract manufacturing sector remains the primary regional objective.
- India: Massive contract manufacturing infrastructure pivots toward synthetic biology to capture global B2B supply contracts. Operations directors leverage deep pharmaceutical fermentation expertise to scale food-grade ingredients rapidly. India is expected to register 20.8% CAGR during the forecast period, as this aggressive capacity expansion creates immense commercial opportunities for Western brands seeking outsourced bio-manufacturing partners.
China: Centralized five-year planning explicitly targets alternative protein self-sufficiency, resulting in long-term growth trajectory and the market expecting to register a 19.6% CAGR over the forecast period. Supply chain executives deploy immense state capital to build world-class bioreactor facilities. This coordinated industrial strategy alters competitive positioning globally as suppliers enter international markets. - Singapore: Agile regulation and novel food clearances are significant factors driving revenue expansion at approximately 18.9% Compound Annual Growth Rate during the forecast period. Agile regulatory frameworks transform this city-state into a premier global testing ground, driving rapid localized commercialization. Regulatory affairs departments secure novel food clearances here months before attempting European approvals. This dynamic forces multinational companies to establish their primary Asia Pacific R&D headquarters locally.
Competitive Aligners for Market Players
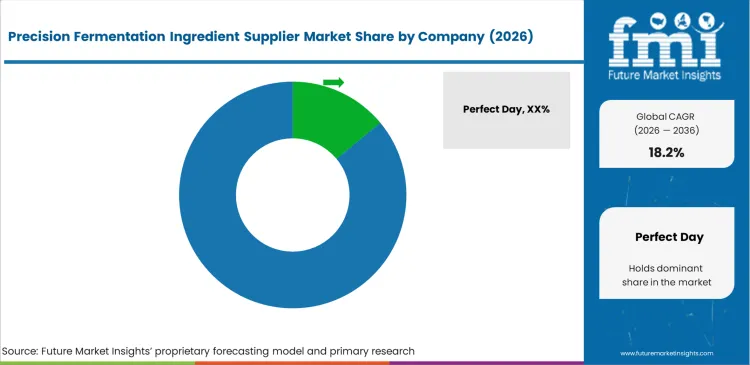
Navigating regulatory clearance creates the ultimate competitive moat shaping the precision fermentation supplier landscape. Start-ups with superior microbial strains mean nothing commercially without FDA or EFSA validation. Incumbents hold a distinct advantage through their proprietary downstream purification libraries. Extracting pure target molecules from messy fermentation broth requires highly specific bio-fermented actives protocols. Optimizing these parameters determines final unit profitability directly. Challengers attempting a favorable precision fermentation ingredient supplier comparison must build separation capabilities independently.
Large food processors fiercely resist vendor lock-in by designing product formulations around generic functional specifications. They avoid relying entirely on proprietary branded ingredients constantly. R&D departments intentionally qualify multiple microbial protein suppliers simultaneously to maintain negotiating leverage. This aggressive multi-sourcing strategy prevents any single biotechnology firm from dictating price terms. The industry is expected to see massive dairy conglomerates simply acquire successful fermentation platforms outright. Internalizing production margins entirely guarantees long-term supply chain security for these global giants.
Key Players in Precision Fermentation Ingredient Supplier Market
- Perfect Day
- Vivici
- Remilk
- Imagindairy
- The Every Company
- Onego Bio
- Verley Food
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 4.1 billion to USD 21.8 billion, at a CAGR of 18.2% |
| Market Definition | Operations center on cultivating genetically optimized microbes to secrete specific functional molecules for downstream commercial food and industrial applications. |
| Segmentation | Ingredient Type, Form, Application, Microbe Type, Customer Type, Region |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, Japan, South Korea, Taiwan, Singapore, Brazil, Mexico, Argentina, GCC Countries, South Africa, Israel, Rest of Middle East & Africa |
| Key Companies Profiled | Perfect Day, Vivici, Remilk, Imagindairy, The Every Company, Onego Bio, Verley Food |
| Forecast Period | 2026 to 2036 |
| Approach | Pilot-plant yield assessments quantify realistic commercial volumes. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Precision Fermentation Ingredient Supplier Market Analysis by Segments
Ingredient Type
- Proteins
- Enzymes
- Lipids
- Sweeteners
Form
- Powders
- Liquids
- Concentrates
- Blends
Application
- Food & Beverage
- Nutrition
- Personal Care
- Industrial Biotech
Microbe Type
- Yeast
- Bacteria
- Fungi
- Algae
Customer Type
- B2B Manufacturers
- Distributors
- R&D Labs
- Brand Owners
Region
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Rest of Middle East & Africa
Bibliography
- Health Canada. (2024, January). Novel food information: β-lactoglobulin protein from yeast strain Komagataella phaffii YRMK-66. Government of Canada
- Health Canada. (2024, July). Cellular agriculture. Government of Canada.
- USA Food and Drug Administration. (2025, January). GRAS Notice No. GRN 001200: β-lactoglobulin produced by Komagataella phaffii “VIPLA”.
- Vivici. (2025, May). A revolutionary protein for nutrition and functionality.
- Eastham, J. L., & Leman, P. (2024, August). Precision fermentation for food proteins: Ingredient innovation through synthetic biology. Current Opinion in Food Science, 59, 101245.
- Knychala, M. M., Augustin, M. A., Williams, T. C., Devenish, R. J., & Abuine, R. (2024, June). Precision fermentation as an alternative to animal protein, a review of current animal protein production and its environmental impacts. Fermentation, 10(6), 315.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
Frequently Asked Questions
Who are the top precision fermentation ingredient suppliers?
Leading suppliers executing B2B commercial scale-up include Perfect Day, Vivici, Remilk, Imagindairy, The Every Company, Onego Bio, and Verley Food. Corporate sourcing departments monitor these firms heavily due to their advanced regulatory dossiers and pilot-scale throughput.
How big is the precision fermentation ingredients market?
Demand is projected to cross USD 4.1 billion in 2026. Consistent downstream investments propel total valuation toward USD 21.8 billion by 2036 as pilot-scale bio-manufacturing infrastructure transitions into permanent commercial volume production.
What ingredients are made with precision fermentation?
Suppliers predominantly produce bio-identical dairy proteins, egg albumen, customized enzyme complexes, and specialized functional lipids. Operations center on target molecules rather than full biomass extraction.
When is precision fermentation expected to reach price parity?
Achieving cost parity relies entirely on solving downstream processing bottlenecks and scaling bioreactor volumes. Procurement teams expect commodity-level pricing only after continuous filtration technologies reduce purification overhead significantly.
What regulatory approvals matter for precision-fermented proteins?
Securing an FDA no-questions letter instantly validates safety profiles for nervous category managers. This regulatory milestone transforms tentative pilot experimentation into immediate commercial supply contracts.
How does precision fermentation compare with plant protein ingredients?
Beverage formulators urgently need heat-stable proteins that dissolve cleanly without chalky textures. Transitioning toward microbial equivalents eliminates secondary masking agents entirely, simplifying total ingredient labels compared to gritty plant isolates.
Why are precision fermentation ingredients expensive?
Extracting target molecules from complex broths requires highly specialized filtration equipment. Downstream purification costs frequently obliterate profit margins before facilities achieve their optimal operational efficiencies.
Which countries are scaling precision fermentation fastest?
Aggressive state-backed bio-manufacturing investments supercharge domestic capacity building across Asia. China leads with a 19.6% compound growth rate, as pharmaceutical contract manufacturers repurpose existing infrastructure rapidly to capture massive new alternative protein export volumes.
How do powders maintain segment leadership?
Global shipping economics punish companies transporting liquid weight across oceans. Logistics departments demand concentrated dry formats to maximize container efficiency and prevent cold-chain spoilage.
What advantage do incumbents hold over startups?
Established suppliers possess highly optimized downstream separation protocols. Perfecting these proprietary filtration parameters takes years, preventing new entrants from matching established commercial unit costs.
Why do brands utilize hybrid formulation platforms?
Blending synthetic whey with plant isolates balances superior texture with acceptable cost profiles. Formulation chemists use functional lipids to perfect mouthfeel in these transitional dairy alternatives.
What role do B2B manufacturers play?
Direct supply agreements guarantee consistent volume absorption for massive bioreactor facilities. Sourcing departments dictate strict purity specifications that upstream platforms must meet before commercializing.
How does Europe regulation impact supply chains?
Rigorous novel food frameworks impose multi-year delays on product commercialization. Sourcing executives invest directly in compliant suppliers to guarantee future access behind these protective bureaucratic moats.
Why is spray-drying a critical operational hurdle?
Excessive temperatures easily denature fragile protein structures during moisture removal. Plant engineers spend months fine-tuning thermal parameters to preserve functionality without spiking energy consumption.
What triggers late mainstream bakery conversion?
Commercial bakeries operate on razor-thin margins requiring absolute cost parity. Procurement officers refuse to modify legacy recipes until synthetic egg replacements match conventional commodity pricing exactly.
How do conglomerates resist vendor lock-in?
R&D teams intentionally design product specifications around generic functional requirements. Approving multiple microbial suppliers simultaneously ensures continuous price competition and secures supply chain redundancy.
What advantage does Singapore offer biotech firms?
Agile novel food frameworks permit rapid consumer testing and iteration. Regulatory affairs departments utilize this dynamic city-state to generate vital safety data before expanding globally.
Why are phototrophic algae strains monitored closely?
Algae platforms offer theoretical advantages regarding cheap carbon feedstock requirements. R&D departments evaluate these early-stage chassis organisms for potential deployment in next-generation capacity planning.
How does ingredient purity impact B2B contracts?
Multinational quality assurance departments enforce strict zero-defect policies. Failing to remove microscopic fermentation byproducts triggers immediate batch rejections and jeopardizes long-term supplier relationships.
What hidden costs affect liquid ingredient systems?
Specialized refrigerated transport drastically inflates total lifecycle shipping expenses. Logistics managers avoid liquid formulations whenever possible to protect delicate profit margins across extended distribution networks.
Why do premium niche brands adopt early?
Small agile companies readily accept higher unit costs to secure novel functional claims. Innovation departments at these brands validate concepts faster than bureaucratic corporate sourcing committees.
How do oxygen constraints limit fungal platforms?
High-density fermentations suffocate rapidly without massive aeration infrastructure. Facility managers must retrofit standard tanks extensively to support hyper-productive strains during exponential growth.
What strategic role do decentralized facilities play?
Co-locating bioreactors near major agricultural hubs secures cheap, reliable carbon inputs. Operations departments establish these strategic nodes to slash ongoing transportation overhead for basic feedstocks.
How does US venture capital impact commercialization?
Massive funding density allows startups to construct proprietary pilot facilities quickly. This financial independence accelerates strain optimization cycles, granting domestic suppliers a global head start.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Opportunity
- Trends
- Desk Research Programme (Secondary Evidence)
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type , 2026 to 2036
- Proteins
- Enzymes
- Lipids
- Proteins
- Y to o to Y Growth Trend Analysis By Ingredient Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powders
- Liquids
- Concentrates
- Powders
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Food & Beverage
- Nutrition
- Personal Care
- Food & Beverage
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Microbe Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Microbe Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Microbe Type, 2026 to 2036
- Yeast
- Bacteria
- Fungi
- Yeast
- Y to o to Y Growth Trend Analysis By Microbe Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Microbe Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- B2B Manufacturers
- Distributors
- R&D Labs
- B2B Manufacturers
- Y to o to Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Ingredient Type
- By Form
- By Application
- By Microbe Type
- By Customer Type
- Competition Analysis
- Competition Deep Dive
- Perfect Day
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Vivici
- Remilk
- Imagindairy
- The Every Company
- Onego Bio
- Verley Food
- Perfect Day
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Microbe Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Ingredient Type
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Microbe Type
- Figure 15: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Customer Type
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Ingredient Type
- Figure 32: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Form
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Microbe Type
- Figure 41: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Customer Type
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Form
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Microbe Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Customer Type
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Form
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Microbe Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Form
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Microbe Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Form
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Microbe Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Customer Type
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Microbe Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Microbe Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Microbe Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Microbe Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

