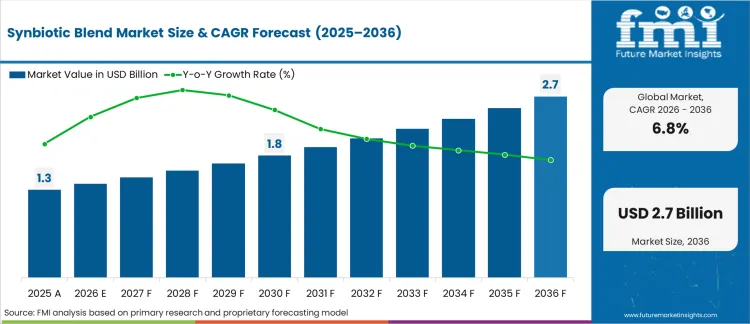
In 2025, the Synbiotic Blend market crossed a valuation of USD 1.3 billion and is projected to reach USD 1.4 billion in 2026 and USD 2.7 billion by 2036, reflecting a CAGR of 6.8%, with momentum tied to clinical substantiation gates. Procurement teams require documented strain survivability and targeted microbiome modulation before authorizing retail listings. Product formulators must specify colony forming units and targeted prebiotic fibers to clear health claim reviews.
Raised compliance thresholds reset minimum capability requirements, forcing manufacturers to invest in strict efficacy trials. A 2025 meta-analysis of 29 randomized controlled trials involving 1,633 older adults confirms that while prebiotics significantly drive Bifidobacterium (SMD: 1.09), synbiotic interventions excel at precision modulation, significantly increasing specific strains like B. longum and B. breve, and boosting beneficial short-chain fatty acids like acetic acid [6]. Manufacturers prioritizing these gut microbiota metrics clear retail review committees faster than those with generic formulations. Adoption of evidence-backed blends reduces consumer skepticism and justifies the premium pricing models essential for maintaining high-margin manufacturing operations.
As stated by Loula Merkel, CEO of COMET, "Prebiotics continue to gain undeniable traction, expanding well beyond supplements and into nearly every aisle of the grocery store." "We developed this annual report to help our customers stay ahead of what's next. I am confident that these findings will help inform their business strategies and lead to the next generation of innovative products." [5]
Country dispersion mirrors health claim exposure, with India, China, Brazil, the United States, the United Kingdom, and Germany posting higher conversion where rising disposable income accelerates preventive health spending. According to FMI's estimates, procurement clears faster for products exhibiting proven synbiotic efficacy for specific conditions. Suppliers align strain sourcing with these regional buying triggers to maximize shelf velocity and limit regulatory pushback.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.4 billion |
| Industry Value (2036) | USD 2.7 billion |
| CAGR (2026-2036) | 6.8% |
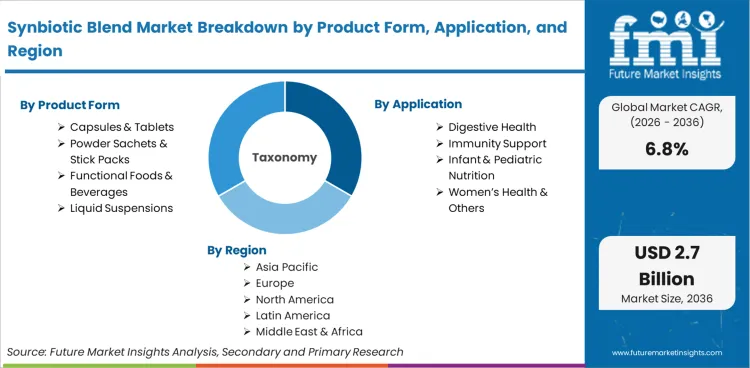
The Synbiotic Blend market represents the global commercialization of finished dietary and functional products that combine live probiotic microorganisms with complementary prebiotic substrates in a single formulation. It measures revenues generated from integrated synbiotic products intended to support probiotic viability and function, with products commonly positioned for digestive and gut-health benefits. Market valuation captures sales across supplement, pediatric nutrition, adult wellness, and fortified food categories distributed through pharmacy, specialty retail, mass-market, and e-commerce channels.
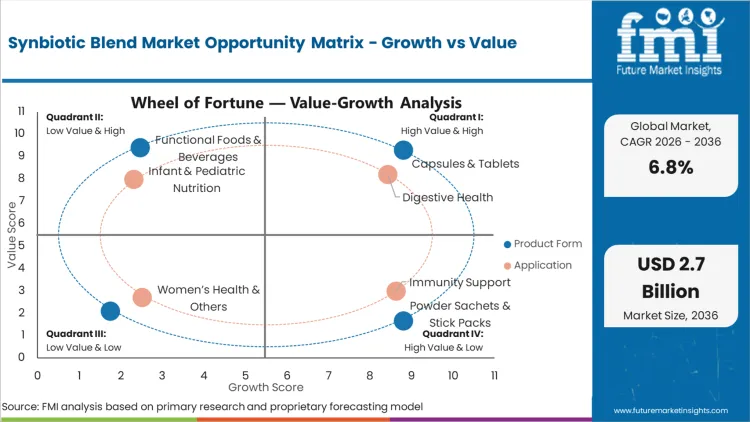
The scope includes co-formulated capsules and tablets containing clinically validated probiotic strains paired with specific prebiotic fibers. Powder sachets, stick packs, and liquid suspensions positioned for digestive and immune support are included. Functional foods and beverages fortified with defined synbiotic combinations, where both components are declared and quantified, form part of the measured revenue base.
Standalone probiotic supplements without a prebiotic substrate and isolated prebiotic fibers without live cultures are excluded. Postbiotic preparations, heat-killed bacterial lysates, and non-viable formulations are not considered. Traditional fermented foods lacking standardized strain identification and quantified synbiotic composition fall outside the defined market scope.
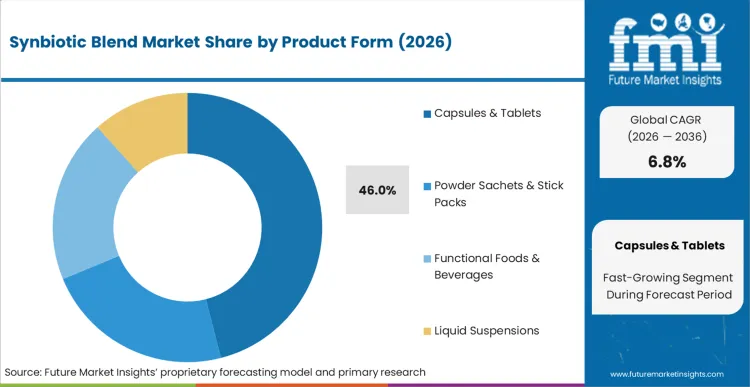
Capsules & Tablets command 46% of the volume in 2026, driven by stability requirements that dictate formulation preferences. Manufacturers gravitate toward encapsulated formats to protect live cultures from harsh gastric acid during transit. Liquid and powder variations serve pediatric and functional beverage segments but face strict shelf-life constraints. According to FMI's estimates, formulators balance survivability with consumer pill fatigue by adapting encapsulation techniques to house specialized pre- and probiotic combinations safely.
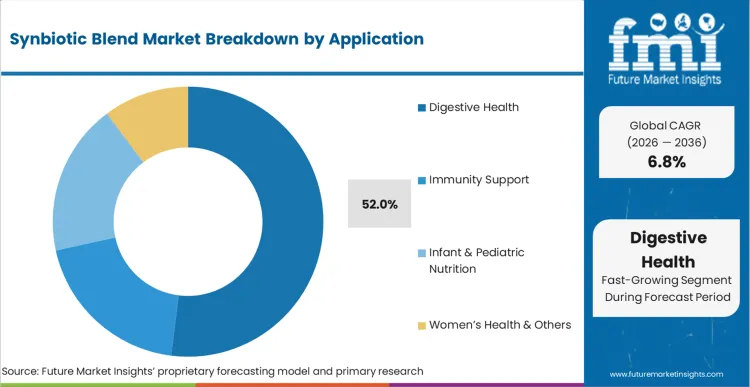
Digestive Health accounts for 52% of the sector in 2026, supported by high incidence rates of chronic gastrointestinal distress. Growing awareness of microbiome balance funnels consumer spending into targeted therapeutic blends rather than generic vitamins. FMI analysts opine that emerging applications in cognitive support and metabolic health broaden the addressable base. Brands link specific microbial strains to measurable health outcomes, moving the category from general wellness to symptom-specific intervention.
Clinical validation mandates act as the primary catalyst for category expansion across mature health markets. Regulatory bodies and major retail chains demand rigorous scientific backing for functional health claims, pushing formulators away from generic blends toward proprietary, evidence-based synbiotic products. This shift forces heavy investment in human clinical trials to demonstrate specific microbiome modulation effects. The focus on therapeutic efficacy accelerates consumer adoption and justifies premium pricing models.
Strain survivability and manufacturing constraints limit rapid scaling for emerging independent brands. Combining live bacterial cultures with prebiotic substrates introduces complex moisture and stability challenges during production and storage. Poor formulation leads to rapid CFU degradation, resulting in product recalls and damaged brand equity. Companies without specialized infrastructure face high contract manufacturing costs, squeezing margins and delaying product launches. Consistent viability remains the critical hurdle for mass-market expansion as brands fight for premium placement.
Based on the regional analysis, the Synbiotic Blend market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
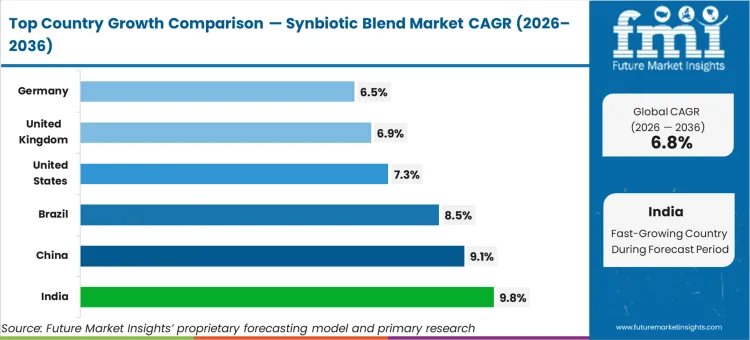
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.8% |
| China | 9.1% |
| Brazil | 8.5% |
| United States | 7.3% |
| United Kingdom | 6.9% |
| Germany | 6.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific exhibits rapid adoption fueled by rising disposable income and a cultural affinity for digestive wellness. Formulators adapt products to local dietary habits, developing powder and liquid formats suited for varied climates. Regulatory frameworks evolve to establish clearer guidelines for functional claims, filtering out low-quality entrants from retail shelves. According to FMI's estimates, regional manufacturers scale production rapidly to meet surging demand for immunity-boosting supplements.
FMI’s report includes an extensive evaluation of the Asia Pacific landscape and its emerging regulatory frameworks. Japan and South Korea represent critical markets for advanced functional foods and geriatric nutrition. Buyers monitor evolving health claim regulations in these nations to dictate future formulation strategies regarding heat-stable probiotics and specialized delivery mechanisms.
North America features a mature, highly competitive landscape driven by clinical validation and premium retail expansion. The region commands a massive share of global biotic sales, with consumers demanding transparency in strain selection and CFU counts. Companies invest heavily in retail partnerships and direct-to-consumer platforms to capture market share. Formulation advancements push the category beyond traditional capsules into functional foods.
FMI’s report includes detailed tracking of North American channel dynamics and consumer purchasing behaviors. Canada and Mexico offer secondary expansion targets through pharmacy and specialized retail networks. Suppliers must navigate stringent labeling standards across these borders to place multistrain probiotics successfully and maintain cross-border compliance.
Latin America represents an emerging frontier characterized by growing awareness of metabolic health. Economic factors push consumers toward cost-effective preventive nutrition rather than expensive pharmaceutical interventions. Brands navigate varied import regulations to establish local distribution networks and secure early market share. Formulators prioritize shelf-stable powders to overcome regional cold-chain logistics challenges.
FMI’s report includes an analysis of Latin American market entry barriers and distribution bottlenecks. Argentina and Chile present opportunities for affordable powder formulations tailored to public health initiatives. Manufacturers observe regional clinical trials to align product claims with local infant gut health priorities and secure institutional purchasing contracts.
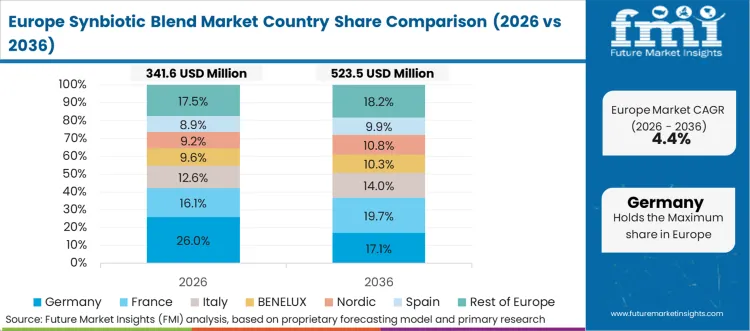
Europe operates under strict health claim regulations, demanding robust clinical evidence for synbiotic products. Established pharmaceutical and nutraceutical companies dominate the landscape, utilizing extensive pharmacy distribution networks. Strategic partnerships accelerate cross-border expansion and product innovation in a highly fragmented regulatory environment. Regional authorities heavily scrutinize functional claims before allowing market entry.
FMI’s report includes comprehensive mapping of European regulatory environments and pharmacy distribution structures. France and Italy remain pivotal for digestive health supplements driven by aging demographics. Companies track European Food Safety Authority guidelines to ensure formulation compliance for microbiome therapeutics and avoid costly product recalls.
Strategic acquisitions consolidate proprietary technology and expand clinical portfolios across the nutritional landscape. Large corporations absorb specialized biotechnology firms to acquire validated strains and advanced delivery mechanisms. This consolidation pressures mid-tier brands to differentiate through niche applications or specialized formats, forcing them to secure exclusive licensing agreements to remain viable.
Manufacturing control determines margin viability and product quality for biotic formulators. Contract manufacturing bottlenecks force leading brands to internalize production or forge long-term partnerships with dedicated facilities. Internalized production limits exposure to supply chain volatility and ensures consistent CFU viability, allowing brands to scale operations without compromising the delicate microbial balance required for health claims.
Retail channel dominance dictates brand survival in a saturated wellness sector. Companies secure national listings by demonstrating clinical efficacy and strong unit economics to category managers. Securing premium shelf space in mass-market retail requires substantial marketing investment and proven consumer retention, pushing brands to adopt omnichannel strategies that combine physical grocery presence with recurring digital subscriptions.
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.4 billion (2026) to USD 2.7 billion (2036), at a CAGR of 6.8% |
| Market Definition | The synbiotic blend sector comprises the commercialization of dietary products containing both live microorganisms and selective substrates utilized by host microorganisms conferring a health benefit. |
| Product Form Segmentation | Capsules & Tablets, Powder Sachets & Stick Packs, Functional Foods & Beverages, Liquid Suspensions |
| Application Segmentation | Digestive Health, Immunity Support, Infant & Pediatric Nutrition, Women’s Health & Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, United Kingdom, Germany, India, China, Brazil and 40 plus countries |
| Key Companies Profiled | Danone S.A., Yakult Honsha Co., Ltd., DSM-Firmenich, Lallemand Inc., Probi AB, Nestlé S.A., Sabinsa Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up modeling validated through primary interviews with synbiotic blend manufacturers, probiotic strain developers, contract formulators, and dietary supplement brands, supported by retail sales benchmarking, trade flow analysis, and production capacity verification across encapsulation and powder blending facilities |
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
How large is the demand for Synbiotic Blend in the global market in 2026?
Demand for Synbiotic Blend in the global market is estimated to be valued at USD 1.4 billion in 2026.
What will be the market size of Synbiotic Blend in the global market by 2036?
Market size for Synbiotic Blend is projected to reach USD 2.7 billion by 2036.
What is the expected demand growth for Synbiotic Blend in the global market between 2026 and 2036?
Demand for Synbiotic Blend in the global market is expected to grow at a CAGR of 6.8% between 2026 and 2036.
Which Product Form is poised to lead global sales by 2026?
Capsules & Tablets format commands 46% share, given its ability to protect live cultures from gastric degradation.
How significant is the role of Digestive Health in driving Synbiotic Blend adoption in 2026?
Digestive Health captures 52% share as chronic gastrointestinal distress funnels consumer spending into targeted therapeutic blends.
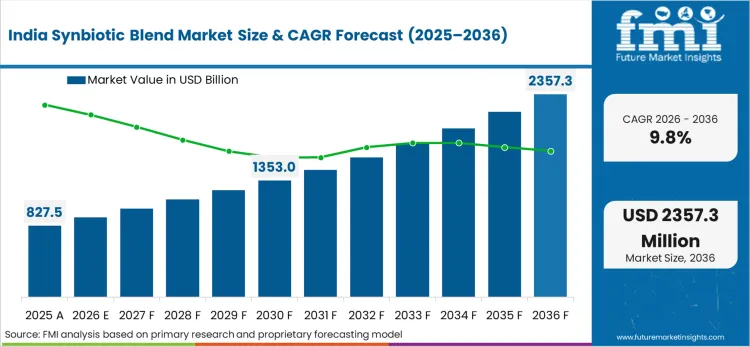
What is driving demand in India?
Growing awareness of preventive digestive care among an expanding middle class drives adoption and retail velocity.
What compliance standards or regulations are referenced for India?
National health survey associations provide baseline tracking for biotic consumption trends.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 9.8% during 2026 to 2036.
Why is North America described as a priority region in this report?
High consumer awareness and massive retail penetration for premium, clinically validated formulations anchor long-term demand.
What type of demand dominates in North America?
Clinically substantiated product replacement demand drives value over generic supplement formats.
What is China growth outlook in this report?
China is projected to expand at a CAGR of 9.1% during 2026 to 2036.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America under the regional scope of analysis.
What are the sources referred to for analyzing Brazil?
Analysis relies on clinical studies tracking gut microbiota profiles in local overweight populations.
What is the main demand theme linked to Brazil in its region coverage?
Obesity rates and metabolic intervention interest steer targeted product development and regional marketing.
Does the report cover the United States in its regional analysis?
Yes, the United States is included within North America under the regional coverage framework.
What is the main United States related demand theme in its region coverage?
Stringent retail category management demands clinically backed formulations for mass-market placement.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Powder sachets and liquid suspensions hold importance for climate adaptability and localized consumer preferences.
What is Synbiotic Blend and what is it mainly used for?
Synbiotic blends combine live probiotic microorganisms with selective prebiotic substrates to enhance gastrointestinal health and immunity.
What does Synbiotic Blend mean in this report?
The market tracks commercial dietary products formulating targeted bacterial strains with compatible prebiotic fibers.
What is included in the scope of this Synbiotic Blend report?
Co-formulated capsules, tablets, powder stick packs, liquid suspensions, and fortified functional foods are included.
What is excluded from the scope of this report?
Standalone probiotics, isolated prebiotic fibers, and general fermented foods lacking quantified strains are excluded.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the Synbiotic Blend forecast?
Forecasts use hybrid top-down and bottom-up modeling validated against corporate capacity data and clinical trial benchmarks.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public clinical datasets are used instead of unverified syndicated market estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.