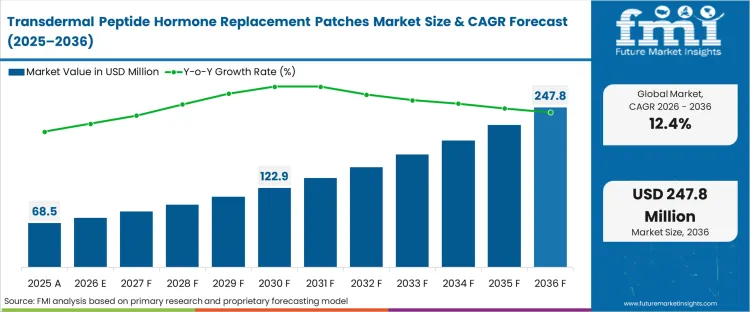
The transdermal peptide hormone replacement patches market was valued at USD 68.5 million in 2025. The hormone microneedle patch adoption is expected to cross USD 77.0 million in 2026 at a CAGR of 12.4% during the forecast period. Steady investment advances the peptide hormone transdermal patch market size to USD 247.8 million through 2036 as the necessity to circumvent gastrointestinal degradation of large-molecule therapies permanently drives developers away from oral reformulations.
Clinical sponsors shaping the non-injectable peptide hormone delivery market are actively transitioning away from traditional subcutaneous injection pens, forced to prioritize patient adherence in chronic endocrine management. This requires substituting familiar liquid vial infrastructure with solid-state microneedle drug delivery systems. Clinical programs that delay integrating transdermal pathways face severe commercial friction, as patients increasingly reject daily injectable regimens. The transition fundamentally redefines the administration lifecycle, moving the burden of compliance from patient behaviour to device design.
The threshold condition unlocking the transdermal peptide patch forecast is the standardization of coating technologies that preserve peptide stability at room temperature. Once contract manufacturers demonstrate scalable, automated coating of high-density microarray structures without degrading the active hormone, the peptide replacement therapy patch industry rapidly unblocks. Formulation stability ceases to be the primary gating factor, allowing subsequent trial phases to move with accelerated velocity.
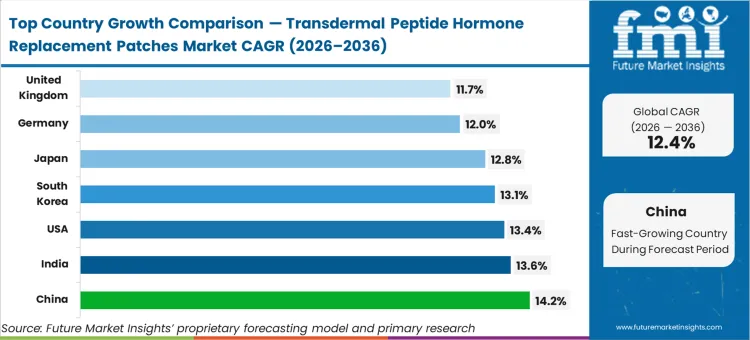
China leads the growth trajectory at a 14.2% compound rate, followed by India at 13.6% and the United States at 13.4%, where deep osteoporosis patient pools intersect with strong patch innovation ecosystems. South Korea tracks at 13.1%, while Japan is estimated to advance at 12.8%. Germany is expected to expand at 12.0%, and the United Kingdom follows at 11.7%. The divergence across this peptide hormone patch CAGR reflects varying technology-assessment thresholds, where markets demanding extensive pivotal success evidence scale more deliberately than those prioritising earlier pilot uptake.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 77.0 million |
| Industry Value (2036) | USD 247.8 million |
| CAGR (2026-2036) | 12.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Answering what are transdermal peptide hormone replacement patches, this sector encompasses microstructured systems engineered to deliver large-molecule endocrine therapies across the stratum corneum. It differs from conventional passive patches by using active mechanical action to deliver peptides into the body. Excluded from this definition are passive estrogen patches and non-therapeutic continuous hormone monitoring devices.
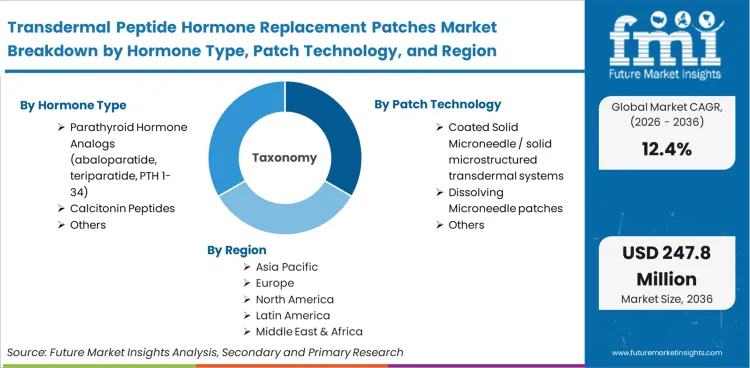
Scope includes solid coated microneedles, dissolving microneedles, and hydrogel-forming systems carrying parathyroid hormone analogs, calcitonin, or growth hormones. Platforms actively in clinical trial phases or specialised commercial supply for endocrine replacement fall within this boundary. The integration of a specialized microarray patch containing proprietary hormone formulations represents the core commercial unit evaluated.
Passive transdermal patches for estrogen or testosterone replacement are explicitly excluded, as small-molecule diffusion relies on entirely different formulation architectures. Similarly, microneedle systems designed for vaccine delivery or continuous glucose monitoring are outside scope. These exclusions maintain analytical focus on the specific manufacturing and stability challenges inherent to peptide hormone delivery.
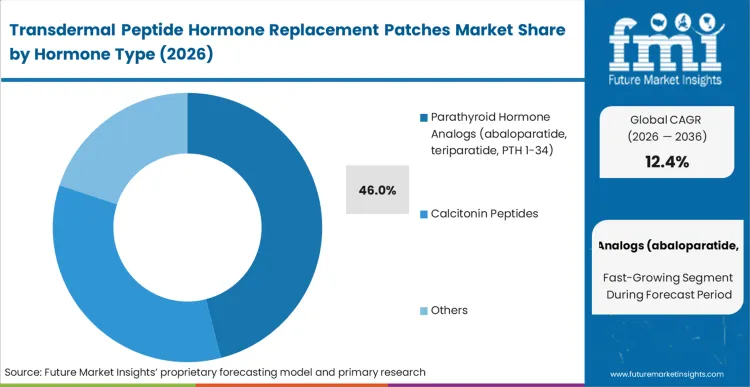
The reason parathyroid hormone analogs (abaloparatide, teriparatide, PTH 1-34) dominate this space is their narrow therapeutic window requiring precise, pulsatile delivery that oral routes cannot achieve and patients resist via injection. Holding a 46.0% share, this sub-segment acts as the proving ground for the osteoporosis drug delivery. Programs operating in the abaloparatide transdermal system and the teriparatide microneedle patch market focus here because the commercial penalty for adherence failure in osteoporosis is immediate. By switching to a solid format, developers eliminate needle anxiety.
FMI's assessment notes that sponsors delaying their low dose hormone alternatives face a hostile reimbursement environment that increasingly penalizes low real-world adherence. Following closely, the emerging salmon calcitonin microneedle patch market demonstrates how alternate bone-health peptides are adopting similar clinical pathways.
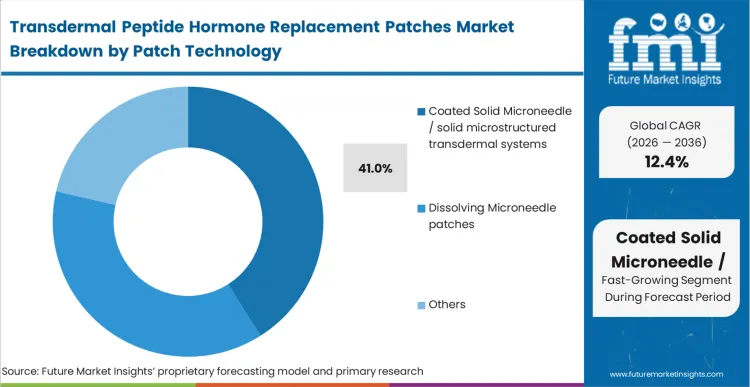
The coated solid microneedle / solid microstructured transdermal systems captures a leading 41.0% share because it decouples the peptide formulation chemistry from the mechanical strength of the needle itself. Unlike the dissolving microneedle hormone patch market where the drug must support the array, coated systems allow developers to utilize proven titanium or polymer bases.
FMI analysts opine that this architectural separation provides the fastest regulatory bridge for existing molecules. The buyer, the clinical trial sponsor, validates the choice by demonstrating that the active pharmaceutical ingredient remains unchanged during the coating process. CDMOs failing to master uniform coating techniques on solid arrays face immediate exclusion from tier-1 dissolving microneedles and coated-array clinical supply contracts.
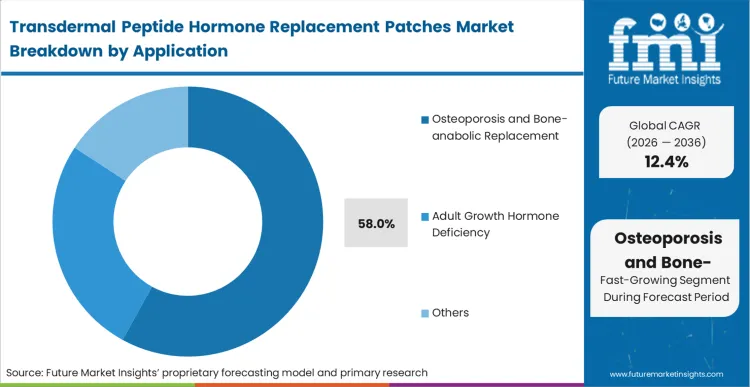
The demand for osteoporosis treatment patches commands a 58.0% share by representing the most urgent intersection of chronic treatment duration and high molecular complexity. The choice facing strategists in the osteoporosis peptide patch therapy market is whether to continue fighting attrition rates of daily injectables or fundamentally change the user experience.
According to FMI's estimates, while human growth hormone treatment and drugs and growth hormone deficiency patch therapy offer longer-term pediatric potential, bone-anabolic therapies provide the immediate volume required to justify microarray manufacturing scale-up. Formulators also leverage niche hypoparathyroidism patch delivery applications to justify premium patch development costs, leaving legacy injectable sponsors disadvantaged.
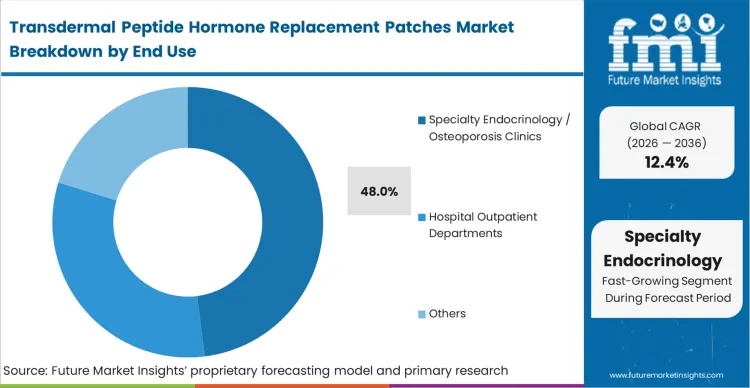
Specialty endocrinology clinics operate as the critical qualification gatekeepers for the endocrine microneedle patch market, commanding a 48.0% share of current trial and pilot volume. These facilities establish the real-world training protocols that determine whether peptide patch self-administration is viable for broader anti osteoporosis therapy and fracture healing. As per FMI's projection, the decision centers on workflow efficiency—specifically, whether teaching peptide endocrine therapy self-administration requires less nursing time than injection training. When the transdermal format proves it reduces clinical overhead, adoption accelerates. Manufacturers who assume general practitioners will drive early adoption misread the market.
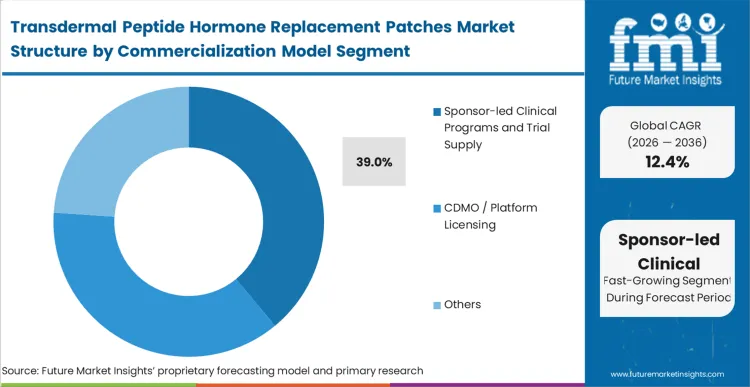
The peptide hormone patch clinical pipeline currently dictates the ecosystem, with sponsor-led trial supply holding 39.0% share because fully scaled commercialization remains gated by pivotal trial completions. This sub-segment reflects a market in active transition, where postmenopausal osteoporosis treatment developers must fund customised manufacturing lines to secure phase III material. In FMI's view, the decision to internalise supply versus partnering with a peptide microneedle patch CDMO defines the company's risk profile. Internalizing ensures IP control but risks profound delays if coating automation fails, directly impacting the broader microneedle patch contract manufacturing for peptides sector.
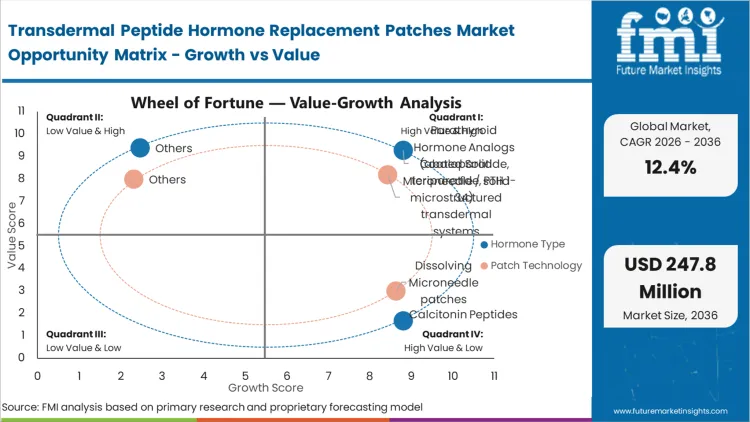
Explaining why are peptide hormones difficult to deliver through the skin, formulation architects face the absolute failure of oral formulations to protect large peptide molecules from gastrointestinal degradation. This compels the pursuit of mechanical transdermal solutions. Clinical sponsors developing next-generation osteoporosis therapies confront the reality that an effective drug is useless if patients refuse daily injections. This pressure makes the transition to solid-state microarray formats an operational necessity, removing capital away from traditional subcutaneous drug delivery devices.
Assessing what are the main adoption barriers for peptide hormone transdermal patches, the primary friction is the severe complexity of automated microarray coating at scale. While pilot-scale manual coating yields highly precise dosing, moving to continuous roll-to-roll or high-throughput batch processing frequently results in unacceptable active pharmaceutical ingredient variance across the patch surface. This scale-up bottleneck forces sponsors into extended manufacturing validation cycles, delaying clinical timelines until CDMOs can prove batch-to-batch uniformity that satisfies stringent regulatory requirements.
Opportunities in the Transdermal Peptide Hormone Replacement Patches Market
Based on the regional analysis, the Transdermal Peptide Hormone Replacement Patches Market is segmented into North America, East Asia, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14.2% |
| India | 13.6% |
| United States | 13.4% |
| South Korea | 13.1% |
| Japan | 12.8% |
| Germany | 12.0% |
| United Kingdom | 11.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Infrastructure maturity shapes the rapid advancement across the East Asian ecosystem, where established high-precision manufacturing bases readily adapt to microarray production requirements. Rather than building novel supply chains, developers leverage existing advanced materials engineering hubs. In FMI's view, this infrastructure advantage allows rapid iteration of patch designs, compressing the time between pilot formulation and clinical trial readiness. The region's dense aging populations provide the necessary volume to justify these specialized investments, driving the hormone replacement therapy market delivery trends.
FMI's report includes extensive analysis of emerging biomanufacturing centers across the broader Asia-Pacific rim, functioning primarily as specialized testing grounds.
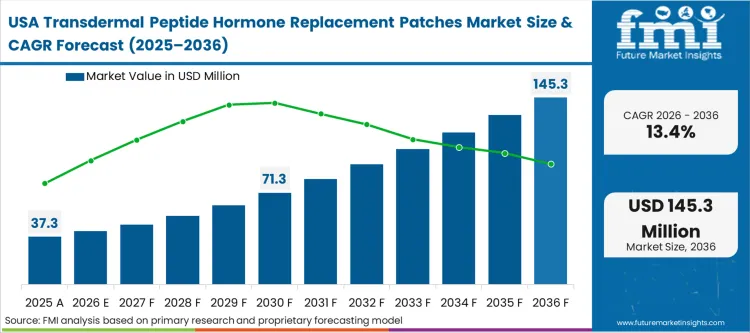
Policy-led dynamics, specifically the rigorous FDA requirements for combination product approvals, dictate the development cadence across North American markets. Developers must simultaneously prove the efficacy of the peptide and the flawless mechanical reliability of the device, effectively doubling the regulatory burden compared to traditional formulations. According to FMI's estimates, this environment forces extreme consolidation in the endocrine drug-device combination market, as only well-capitalized clinical sponsors possess the resources to navigate the dual-track approval process.
FMI's report includes analysis of the Canadian clinical ecosystem, where facilities increasingly serve as critical specialized overflow sites for North American pivotal trials.
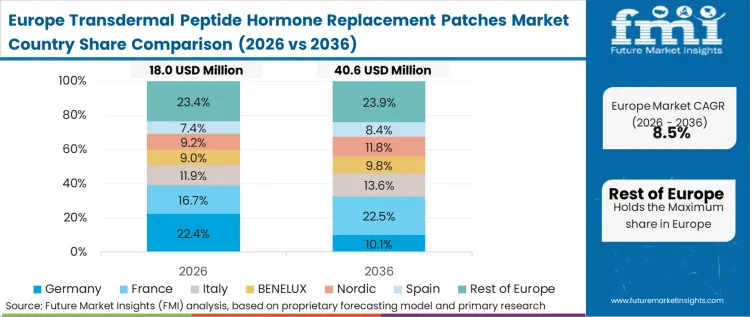
Economics-led pressures define the European trajectory, where nationalized healthcare systems strictly evaluate the cost-per-outcome of transitioning from inexpensive legacy injectables to premium transdermal patches. Clinical sponsors must demonstrate that the upfront cost of the microarray technology is entirely offset by reduced downstream fracture interventions resulting from improved patient adherence. As per FMI's projection, this dynamic forces formulators in the biologics transdermal delivery market to prioritize data that directly correlates delivery mechanics with long-term systemic health savings.
FMI's report includes extensive analysis of the Nordic and Western European healthcare networks, which serve as early indicators for how rigid reimbursement models will ultimately value the operational efficiencies of anti osteoporosis fracture healing.
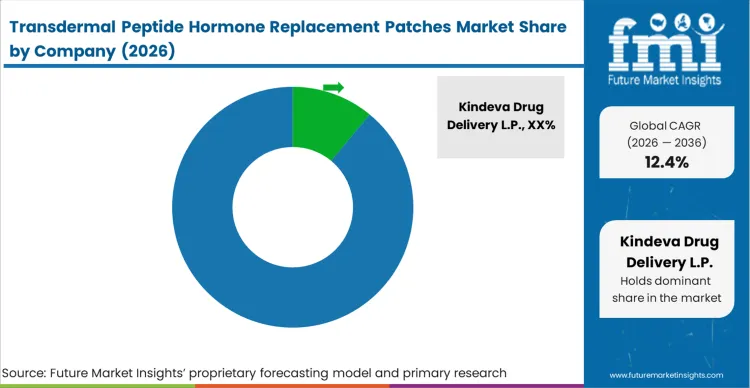
The specialized nature of solid-state peptide delivery creates a highly concentrated competitive situation dominated by peptide hormone patch manufacturers and dedicated delivery platform developers rather than traditional pharmaceutical giants. Companies like Kindeva Drug Delivery L.P. and LTS Lohmann Therapie-Systeme AG hold outsized influence because they possess the rare combination of polymer engineering expertise and pharmaceutical drug delivery coating capabilities. Finding a reliable hormone patch development partner or transdermal peptide patch supplier capable of manufacturing reproducible, high-density microarrays at a commercial scale is the absolute dividing line between qualified programs and theoretical research.
Incumbents in this space maintain their advantage through heavily guarded trade secrets related to active pharmaceutical ingredient stabilisation during the automated coating process. Entities like Corium, LLC and Raphas Co., Ltd. have spent years optimising the specific fluid dynamics required to deposit peptides onto microscopic tips without degrading the molecule. Challengers attempting to replicate this must build entirely novel manufacturing lines, as standard transdermal or sterile fill-finish equipment for controlled release drug delivery technology cannot be retrofitted for microarray production.
The trajectory through 2036 points toward increasing integration, as large pharmaceutical sponsors seek to internalize these delivery capabilities to protect their next-generation biological pipelines. The tension lies between developers wanting broad peptide patch technology licensing and pharmaceutical buyers demanding exclusive rights to specific microarray architectures. Answering which companies are developing hormone microneedle patches, these dynamic forces have specialised patch manufacturers to carefully maintain their early clinical partnerships in the drug delivery technology sector to avoid inadvertently locking themselves out of broader therapeutic categories.
| Metric | Value |
|---|---|
| Quantitative Units | USD 77.0 million to USD 247.8 million, at a CAGR of 12.4% |
| Market Definition | The sector encompasses microarray and solid microstructured systems engineered specifically to deliver large-molecule endocrine therapies across the stratum corneum, requiring active mechanical disruption to transport peptides systemically. |
| Hormone type Segmentation | Parathyroid hormone analogs (abaloparatide, teriparatide, PTH 1-34), Calcitonin peptides, Growth hormone patches, Other peptide endocrine hormones |
| Patch technology Segmentation | Coated solid microneedle / solid microstructured transdermal systems, Dissolving microneedle patches, Hydrogel-forming microneedle patches, Reservoir / osmotic wearable microneedle patches, Other microarray patch formats |
| Application Segmentation | Osteoporosis and bone-anabolic replacement, Adult growth hormone deficiency, Hypoparathyroidism / calcium-bone disorders, Other endocrine replacement uses |
| End use Segmentation | Specialty endocrinology / osteoporosis clinics, Hospital outpatient departments, Home-care self-administration, Clinical research centers |
| Commercialization model Segmentation | Sponsor-led clinical programs and trial supply, CDMO / platform licensing, Specialty pharmacy commercialization, Hospital pharmacy commercialization |
| Regions Covered | North America, East Asia, Europe |
| Countries Covered | United States, China, India, Japan, South Korea, Germany, United Kingdom, and 40 plus countries |
| Key Companies Profiled | Kindeva Drug Delivery L.P., LTS Lohmann Therapie-Systeme AG, Corium, LLC, Hisamitsu Pharmaceutical Co., Inc., Raphas Co., Ltd., Micron Biomedical, Inc., PharmaTher Holdings Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted clinical trial directors and formulation leads managing solid-state delivery. Baselines were anchored to active clinical program funding and CDMO licensing milestones. Forecasts were cross validated against reported patient adherence dropout rates for legacy subcutaneous injectables. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What are transdermal peptide hormone replacement patches?
The industry represents USD 77.0 million in 2026, comprising microstructured systems engineered to actively transport large-molecule endocrine therapies across the stratum corneum. These patches utilise solid-coated or dissolving arrays to bypass the gastrointestinal tract without requiring traditional needles.
Build a forecast for peptide hormone replacement patch demand by 2036.
Ongoing investment fuels the valuation to USD 247.8 million through 2036. The expansion signals the anticipated transition from restricted clinical phase manufacturing to scaled commercial distribution as room-temperature stable peptide formats clear rigid combination-product regulatory hurdles.
What is the growth outlook for peptide hormone patches?
The industry is expected to grow at a CAGR of 12.4% during the forecast period. This rate is strictly constrained by the extended timelines of combination-product regulatory reviews, preventing the immediate spike in revenue that simpler oral drug reformulations typically experience.
Which hormone therapies are most suitable for microneedle patches?
Parathyroid hormone analogues secure the dominant position primarily because they target the severe adherence failures present in osteoporosis treatment. Clinical sponsors prioritize this chemistry because the commercial and biological penalties for patients abandoning legacy daily injections are immediate and catastrophic.
Explain the market potential for transdermal peptide hormone patches.
Coated solid microstructured transdermal systems capture the majority share by decoupling the active peptide from the mechanical needle. This architecture holds massive potential because it allows developers to utilize proven rigid materials for stratum corneum penetration while maintaining formulation stability.
Why is osteoporosis the leading application for peptide hormone patches?
Osteoporosis and bone-anabolic replacement drives the highest volume demand due to massive patient cohorts suffering from injection fatigue. Providers prescribe long-acting hormone microneedle patch options to definitively close the adherence gap and prevent subsequent severe fractures associated with regimen abandonment.
Why are peptide hormones difficult to deliver through the skin?
The absolute failure of passive formulations to transport large peptide molecules across the intact stratum corneum forces the shift to mechanical microarrays. When formulators successfully stabilise a peptide on a solid microstructure, they eliminate refrigerated distribution, altering unit economics for the sponsor.
What are the main adoption barriers for peptide hormone transdermal patches?
The severe complexity of automated microarray coating at scale creates a massive scale-up bottleneck. Moving from manual pilot batches to continuous high-throughput processing frequently results in unacceptable active pharmaceutical ingredient variance, extending manufacturing validation cycles by years.
Which countries will adopt hormone microneedle patches fastest?
China expands at a 14.2% compound rate, outpacing India's 13.6% trajectory due to its rapid scaling of domestic biomanufacturing hubs. Formulation architects in China leverage this specialized infrastructure to compress the time between laboratory design and automated commercial production lines faster than competing regions.
Are any peptide hormone patches approved yet?
While multiple programs are in late-stage clinical trials, full commercial approval remains evidence-gated by stringent FDA requirements. Sponsors must simultaneously prove peptide bioequivalence and mechanical device reliability, which heavily consolidates the vendor pool to highly capitalized entities capable of funding extended safety bridging studies.
How do microneedle hormone patches work?
Removing the necessity for refrigerated clinical supply chains drastically reduces distribution costs and logistical failures. Microarrays physically disrupt the top skin layer just enough to deposit the peptide into the interstitial fluid, where it is rapidly absorbed systemically without triggering pain receptors.
Which companies are developing hormone microneedle patches?
Engineering-heavy CDMOs like Kindeva Drug Delivery and LTS Lohmann possess heavily guarded trade secrets related to fluid dynamics and automated peptide deposition. Challengers attempting to replicate these precise coating techniques must build entirely novel manufacturing lines from scratch, securing the incumbents' position as gatekeepers.
Compare peptide hormone patches with injectable hormone therapies.
Transitioning to microarray patches significantly reduces the nursing time previously dedicated to patient injection training. Clinics that integrate these solid-state formats increase their daily patient throughput, altering the baseline efficiency metrics used by healthcare administrators compared to legacy vials and pens.
What are non-injectable hormone delivery alternatives?
Beyond traditional nasal sprays which suffer from highly variable absorption, the industry focuses heavily on microarrays for consistent dosing. Peptide hormones are large molecules that require the active mechanical disruption provided by microneedles to achieve systemic absorption comparable to subcutaneous injections.
When evaluating a dissolving microneedle patch vs coated microneedle patch, how do they differ?
Peptides that cannot maintain integrity when integrated directly into dissolving polymer matrices are pushed toward coated solid arrays. This decoupling ensures the delicate hormone is deposited only on the outer layer, avoiding the thermal or chemical stresses of the base manufacturing process.
Analyzing the hormone microneedle patch vs injection, what drives the transition?
Patient resistance to chronic daily injections creates unsustainable adherence dropout rates, rendering highly effective peptides useless in real-world applications. Sponsors are forced to prioritise patch formats because the commercial penalty for adherence failure in diseases like osteoporosis is too severe to ignore.
Looking at the abaloparatide patch vs abaloparatide injection, what did recent trials reveal?
Recent phase 3 data showed that an investigational abaloparatide transdermal system failed to meet the non-inferiority margin for bone mineral density increases compared to subcutaneous injection. This forced the sponsor to halt the program after regulatory feedback indicated another pivotal trial was required, underscoring the severe clinical barriers in this space.
Comparing the teriparatide patch vs teriparatide injection, why is delivery transition difficult?
Unlike traditional liquid vials, solid-state hormone patches demand specific handling to prevent physical damage to the microarray prior to application. Pharmacies must validate new dispensing workflows that account for combination-product tracking and specialized packaging integrity, extending the commercialization timeline.
How does a peptide patch vs wearable injector compete in pediatric settings?
Needle phobia heavily restricts adherence in juvenile treatments like chronic endocrine replacement patch therapies. While wearable injectors hide the needle, they still cause puncture anxiety; dissolving microneedle patches leave zero sharp residue post-application, offering a distinct competitive advantage in pediatric endocrinology.
How does a peptide patch vs wearable injector compete in pediatric settings?
Needle phobia heavily restricts adherence in juvenile treatments like chronic endocrine replacement patch therapies. While wearable injectors hide the needle, they still cause puncture anxiety; dissolving microneedle patches leave zero sharp residue post-application, offering a distinct competitive advantage in pediatric endocrinology.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.