The tryptic protein digestion kits market is projected to reach USD 848.0 million in 2026 and expand to USD 1,953.0 million by 2036, at a CAGR of 8.7%. Value expansion is driven by standardized proteomics sample preparation requirements across academic research, biopharmaceutical development, and clinical proteomics workflows. Adoption intensity remains closely linked to mass spectrometry utilization growth, reproducibility mandates, and increasing reliance on kit-based digestion to reduce inter-operator and inter-laboratory variability. Spending concentration favors sequencing-grade and automation-compatible kits that support high-throughput peptide generation with predictable digestion efficiency and minimal autolysis risk.
Demand progression is governed by proteomics standardization, biologics characterization needs, and integration with automated LC-MS pipelines. Kit adoption expands where regulated bioanalytical testing, biosimilar comparability studies, and large cohort biomarker research require consistent peptide maps and validated workflows. Cost sensitivity, enzyme stability constraints, and limited protocol flexibility moderate unrestricted uptake in smaller laboratories. Market behavior remains anchored to repeat consumable demand, laboratory modernization cycles, and method harmonization requirements rather than exploratory proteomics alone. These factors support sustained, structurally stable growth across global life science research environments.
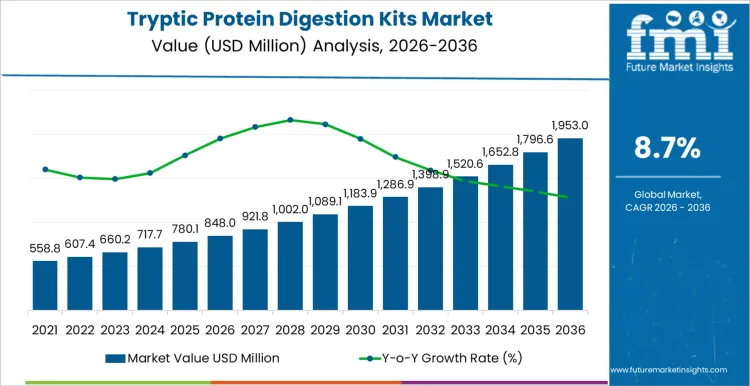
| Metric | Value |
|---|---|
| Market Value (2026) | USD 848.0 million |
| Market Forecast Value (2036) | USD 1,953.0 million |
| Forecast CAGR 2026 to 2036 | 8.7% |
Demand for tryptic protein digestion kits is increasing as life science researchers and proteomics laboratories require reliable, streamlined solutions for preparing complex biological samples prior to mass spectrometry and tandem mass tag analysis. Trypsin-catalyzed digestion of proteins into peptides remains a core step in shotgun proteomics workflows because it generates predictable peptide fragments that improve identification confidence and quantitative reproducibility. Researchers specify kits formulated with optimized trypsin quality, stabilizers, and buffer systems that minimize autolysis and maximize digestion efficiency across varied sample types, including serum, tissue lysates, and cell cultures. Procurement teams evaluate kit performance metrics such as digestion completeness, reproducibility, and ease of workflow integration because consistent peptide generation influences downstream identification, quantitation, and bioinformatics interpretation in high-throughput studies.
Growth in applications such as biomarker discovery, systems biology, and biologic therapeutic characterization reinforces adoption of tryptic digestion kits that support standardized sample preparation across multi-user laboratories. As large-scale proteomic studies expand, laboratories prioritize solutions that reduce sample-to-sample variability and shorten preparation time without sacrificing data quality. Vendor support for protocol optimization, documentation, and troubleshooting enhances confidence among technical staff, particularly when integrating digestion kits with automated liquid handling systems. Advances in formulation stability extend shelf life and support reproducible performance under varied storage conditions, which is especially valuable in decentralized research facilities and contract research settings. These application-specific performance and operational readiness drivers are contributing to sustained demand growth in the tryptic protein digestion kits market.
Demand for tryptic protein digestion kits is driven by reproducibility requirements, increasing sample throughput, and standardization across proteomics workflows. Laboratories prioritize consistent peptide generation, minimized variability, and compatibility with high-resolution mass spectrometry. Adoption reflects wider use of automation, expansion of regulated bioanalytical testing, and growth in large-cohort studies. Selection decisions focus on digestion efficiency, workflow integration, and data comparability across runs. Segmentation clarifies how kit configuration, analytical purpose, and organizational setting influence utilization patterns across discovery, development, and quality environments.
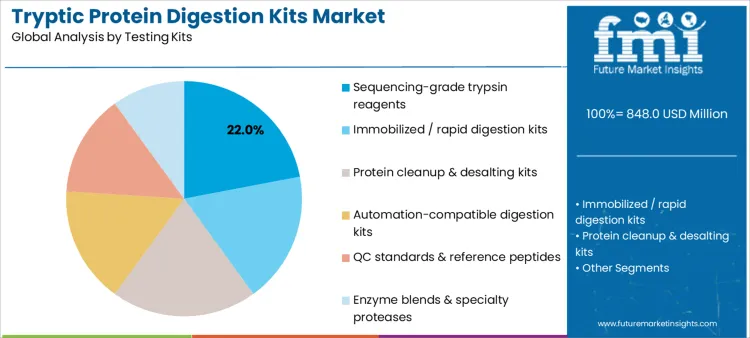
Sequencing-grade trypsin reagents hold 22.0%, reflecting their central role in achieving specific and reproducible protein cleavage. These reagents support consistent peptide maps required for comparative analysis across experiments. Protein cleanup and desalting kits account for 20.0%, addressing matrix interference and improving downstream signal quality. Immobilized or rapid digestion kits at 18.0% shorten processing timelines while maintaining digestion control. Automation-compatible digestion kits represent 16.0%, supporting standardized high-throughput operations. QC standards and reference peptides at 14.0% enable method verification, while enzyme blends at 10.0% serve specialized proteolysis needs. Kit-type distribution reflects emphasis on digestion fidelity and operational efficiency.
Key Points
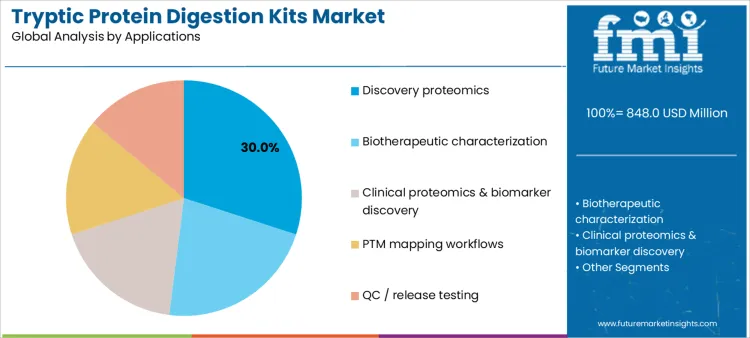
Discovery proteomics represents 30.0%, driven by exploratory studies requiring broad protein coverage and reliable digestion across diverse samples. Biotherapeutic characterization at 22.0% relies on precise peptide mapping to assess structure, purity, and degradation pathways. Clinical proteomics and biomarker discovery at 18.0% emphasize consistency across large patient cohorts. PTM mapping workflows at 16.0% require controlled digestion to preserve modification information. QC and release testing at 14.0% depend on validated, repeatable protocols supporting compliance. Application segmentation highlights stronger utilization where data reliability, comparability, and regulatory confidence are critical.
Key Points
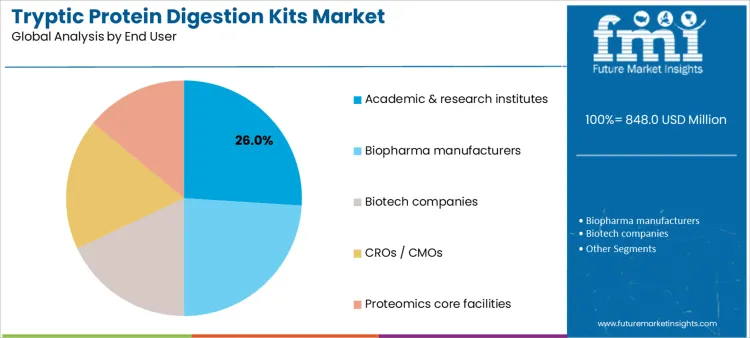
Academic and research institutes account for 26.0%, reflecting extensive use in method development and hypothesis-driven proteomics. Biopharma manufacturers at 24.0% integrate digestion kits into development and quality programs supporting biologics pipelines. Biotech companies at 18.0% apply kits in translational research and early-stage characterization. CROs and CMOs at 18.0% require standardized kits adaptable to multiple client protocols. Proteomics core facilities at 14.0% support shared instrumentation and varied project needs. End-user segmentation indicates concentration where analytical depth, throughput, and protocol standardization guide kit selection.
Key Points
Demand for tryptic protein digestion kits reflects standardized sample preparation requirements in proteomics, biopharmaceutical analysis, and clinical research. Adoption concentrates in academic research laboratories, contract research organizations, biopharmaceutical manufacturers, and proteomics core facilities. Global scope aligns with growth in mass spectrometry-based protein analysis. Usage centers on ready-to-use kits containing sequencing-grade trypsin and buffers optimized for reproducible enzymatic digestion prior to peptide-based analysis.
How do analytical reproducibility need and workflow efficiency shape adoption?
Proteomic workflows rely on consistent and complete protein digestion to generate reliable peptide maps. Demand increases where variability in in-house digestion protocols affects mass spectrometry data quality and comparability. Tryptic digestion kits provide controlled enzyme activity, defined reaction conditions, and reduced preparation steps, supporting inter-laboratory consistency. Biopharmaceutical development uses these kits for protein characterization, impurity profiling, and comparability studies under regulated environments. High-throughput research settings value reduced hands-on time and predictable digestion kinetics. Compatibility with automation platforms and downstream LC-MS workflows enhances utility. Adoption reflects analytical priority for reproducibility, time efficiency, and data integrity in complex protein analysis pipelines.
How do enzyme stability limits, cost exposure, and method flexibility influence market scalability?
Trypsin activity is sensitive to storage conditions, autolysis, and contamination, affecting shelf life and performance reliability. Demand sensitivity rises where cold chain handling increases logistical complexity. Kit cost exceeds bulk enzyme procurement, influencing adoption in cost-constrained academic laboratories. Fixed protocols limit flexibility for atypical proteins requiring alternative digestion conditions. Batch-to-batch enzyme variability necessitates quality control oversight. Regulatory environments require extensive validation when switching suppliers or formats. Dependence on animal-derived or recombinant enzyme sourcing affects supply stability. Scalability remains constrained by enzyme stability considerations, budget sensitivity, and need for method adaptability across diverse proteomic applications.
Demand for tryptic protein digestion kits is expanding globally as proteomics workflows become integral to biopharmaceutical development, clinical research, and academic life sciences. Standardized sample preparation supports reproducibility across mass spectrometry platforms. Growth in biologics characterization, biomarker discovery, and quality control increases routine kit usage. Automation compatibility and reduced preparation variability influence purchasing decisions. Growth rates in India at 11.2%, Brazil at 10.7%, China at 10.2%, the USA at 7.2%, and Germany at 7.1% indicate sustained expansion driven by proteomics adoption, laboratory throughput requirements, and method standardization rather than exploratory research alone.

| Country | CAGR (%) |
|---|---|
| India | 11.2% |
| Brazil | 10.7% |
| China | 10.2% |
| USA | 7.2% |
| Germany | 7.1% |
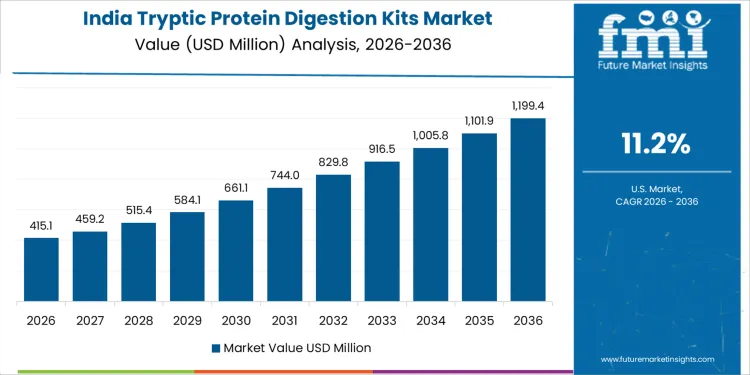
Demand for tryptic protein digestion kits in India is growing at a CAGR of 11.2%, supported by expansion of proteomics research within pharmaceutical and academic laboratories. Contract research organizations increase digestion throughput to support biologics characterization and biosimilar comparability studies. Universities and research institutes adopt standardized kits to improve reproducibility in mass spectrometry workflows. Growing availability of LC-MS platforms increases routine sample preparation volumes. Local distributors improve kit availability and technical support. Growth reflects laboratory modernization, research funding expansion, and standardization needs rather than niche exploratory proteomics activity.
Tryptic protein digestion demand in Brazil is expanding at a CAGR of 10.7%, driven by growth in academic proteomics and public research initiatives. Universities and national research centers increase use of standardized digestion protocols for biomarker and disease research. Biopharmaceutical laboratories apply digestion kits for formulation and stability studies. Government-funded projects support instrumentation and consumables procurement. Regional core facilities provide shared proteomics services. Growth reflects institutional research investment and method harmonization rather than high-volume commercial testing.
Demand for tryptic protein digestion kits in China is growing at a CAGR of 10.2%, aligned with large-scale proteomics research and biopharmaceutical development. Domestic biologics developers integrate standardized digestion within quality and characterization workflows. National research programs support proteome mapping and disease biomarker studies. High-throughput laboratories favor ready-to-use kits to reduce variability. Domestic and international suppliers ensure broad availability. Growth reflects scale-driven adoption and research prioritization rather than small-lab experimentation.
Tryptic protein digestion demand in the United States is expanding at a CAGR of 7.2%, supported by mature proteomics infrastructure and standardized workflows. Pharmaceutical and biotech companies use digestion kits for routine characterization and comparability studies. Clinical research laboratories apply kits within translational proteomics programs. Automation and reproducibility requirements favor kit-based preparation. Replacement demand arises from steady research activity rather than rapid lab expansion. Growth reflects workflow optimization and consistency requirements rather than increased proteomics adoption rates.
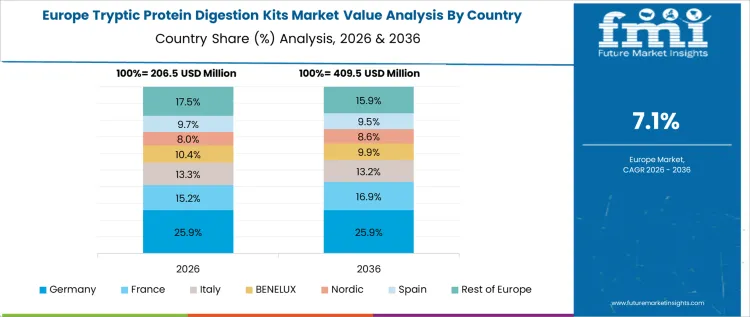
Tryptic protein digestion demand in Germany is growing at a CAGR of 7.1%, shaped by strong academic research and industrial biotechnology presence. Research institutes emphasize method validation and reproducibility in proteomics experiments. Pharmaceutical laboratories use digestion kits to support analytical consistency. Funding stability supports continuous consumables procurement. Emphasis on quality standards moderates rapid volume increases. Growth remains steady, reflecting disciplined research activity and standardized laboratory practices rather than aggressive scale-up.
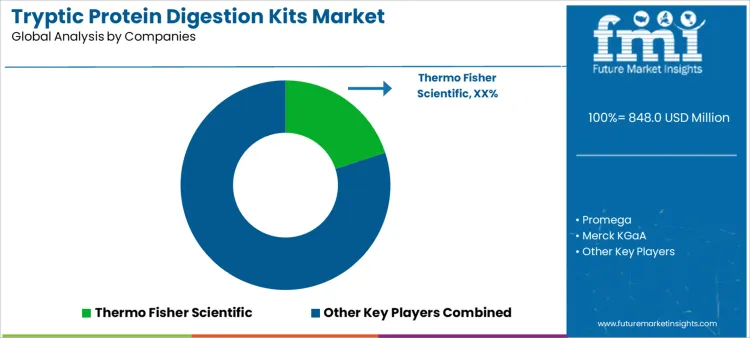
Demand for tryptic protein digestion kits is shaped by proteomics standardization, biomarker validation workflows, and regulated bioanalytical testing requirements. Laboratories assess digestion reproducibility, enzyme specificity, autolysis suppression, batch consistency, and compatibility with mass spectrometry platforms. Buyer evaluation includes protocol simplicity, sample throughput suitability, storage stability, and robustness across complex biological matrices. Procurement behavior emphasizes lot traceability, validated performance documentation, and supplier capability to support method transfer across sites. Trend in the tryptic protein digestion kits market reflects increasing use in clinical proteomics, translational research, and high-throughput discovery pipelines requiring reliable digestion performance.
Thermo Fisher Scientific leads competitive positioning through high-purity trypsin kits integrated within comprehensive proteomics and mass spectrometry ecosystems. Promega competes by supplying sequencing-grade digestion kits valued for consistency, ease of use, and adoption in regulated laboratories. Merck KGaA supports demand through branded proteomics reagents aligned with quality management systems and global distribution reach. Waters maintains relevance by positioning digestion kits alongside chromatography and mass spectrometry workflows used in analytical laboratories. Competitive differentiation centers on enzyme activity consistency, documentation quality, workflow compatibility, and dependable supply supporting reproducible proteomic analysis.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Testing Kits | Sequencing-grade trypsin reagents; Immobilized / rapid digestion kits; Protein cleanup & desalting kits; Automation-compatible digestion kits; QC standards & reference peptides; Enzyme blends & specialty proteases |
| Applications | Discovery proteomics; Biotherapeutic characterization; Clinical proteomics & biomarker discovery; PTM mapping workflows; QC / release testing |
| End User | Academic & research institutes; Biopharma manufacturers; Biotech companies; CROs / CMOs; Proteomics core facilities |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Thermo Fisher Scientific; Promega; Merck KGaA; Waters; Agilent Technologies; Others |
| Additional Attributes | Dollar sales by testing kit type, application, and end user; digestion efficiency, specificity, and reproducibility across sequencing-grade and immobilized trypsin systems; compatibility with automated and high-throughput proteomics workflows; impact on peptide coverage, PTM preservation, and data quality; quality control requirements for regulated biopharma analysis; procurement patterns driven by research funding cycles, biologics development pipelines, and centralized proteomics facilities. |
How big is the tryptic protein digestion kits market in 2026?
The global tryptic protein digestion kits market is estimated to be valued at USD 848.0 million in 2026.
What will be the size of tryptic protein digestion kits market in 2036?
The market size for the tryptic protein digestion kits market is projected to reach USD 1,953.0 million by 2036.
How much will be the tryptic protein digestion kits market growth between 2026 and 2036?
The tryptic protein digestion kits market is expected to grow at a 8.7% CAGR between 2026 and 2036.
What are the key product types in the tryptic protein digestion kits market?
The key product types in tryptic protein digestion kits market are sequencing-grade trypsin reagents, immobilized / rapid digestion kits, protein cleanup & desalting kits, automation-compatible digestion kits, qc standards & reference peptides and enzyme blends & specialty proteases.
Which applications segment to contribute significant share in the tryptic protein digestion kits market in 2026?
In terms of applications, discovery proteomics segment to command 30.0% share in the tryptic protein digestion kits market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.