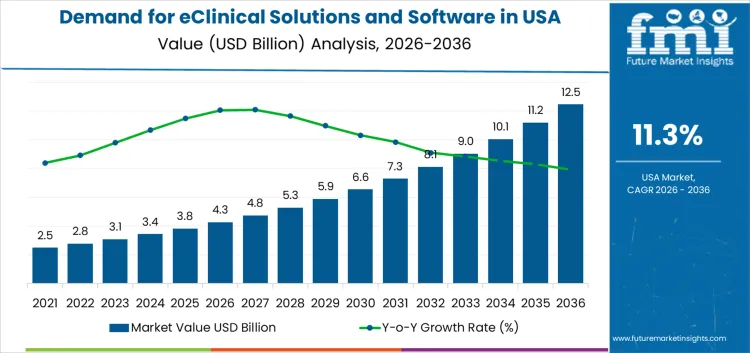
The USA eClinical solutions and software demand is valued at USD 4.3 billion in 2026 and is forecasted to reach USD 12.5 billion by 2036, reflecting a CAGR of 11.3%. Growth is driven by the shift toward decentralized and hybrid clinical trial models, increased reliance on digital data capture, and the need for streamlined trial management across large therapeutic pipelines. Sponsors and CROs prioritize systems that enhance data integrity, reduce cycle times, and support regulatory compliance.
Phase III leads adoption because late-stage trials generate large, complex datasets requiring robust electronic data capture, centralized monitoring, and advanced analytics. Platforms integrate with wearables, ePRO systems, and remote site tools to strengthen real-time oversight. Enhanced interoperability across trial workflows improves patient recruitment efficiency, adherence tracking, and endpoint validation.
The West, South, and Northeast regions show strong demand due to concentrated biopharmaceutical research, extensive CRO presence, and high clinical trial activity. Key suppliers include Oracle, Datatrak International, Inc., Dassault Systèmes, CRF Health, and eClinicalWorks. Their strategies focus on cloud-native architectures, AI-enabled data quality management, and unified trial platforms that support scalable deployment across complex, multi-site clinical studies.
The growth contribution index for eClinical solutions and software in the United States reflects distinct inputs from digital trial operations, data management needs, and regulatory alignment. Core contribution comes from the transition toward electronic data capture environments that support structured clinical workflows across sponsors and contract research units. This factor maintains steady influence because digital consistency improves data integrity and reduces manual processing. Clinical trial volume contributes an additional share, as greater protocol complexity increases reliance on integrated platforms for monitoring, randomization, and safety reporting.
Analytics and real-time visibility tools contribute a rising portion of growth because sponsors require continuous oversight of patient enrollment, deviation trends, and endpoint performance. This segment strengthens its contribution as advanced analytics integrate with operational systems. Remote and decentralized trial models contribute another measurable share because virtual visit structures depend on coordinated digital interfaces, secure communication layers, and device-linked data flows. These requirements elevate demand for interoperable software.
Regulatory expectations for audit readiness and data traceability add a structured contribution by reinforcing investment in validated systems. Integration with legacy infrastructures contributes a smaller but consistent share driven by modernization cycles. The index reflects distributed contributions shaped by operational digitization, regulatory direction, and expanding trial complexity across the domestic research landscape.
| Metric | Value |
|---|---|
| USA eClinical Solutions and Software Sales Value (2026) | USD 4.3 billion |
| USA eClinical Solutions and Software Forecast Value (2036) | USD 12.5 billion |
| USA eClinical Solutions and Software Forecast CAGR (2026-2036) | 11.3% |
Demand for eClinical solutions and software in the United States grows due to expanding clinical research activity, complex trial designs, and increased reliance on digital data workflows. Sponsors and contract research organizations use electronic data capture, clinical trial management systems, and remote monitoring platforms to manage rising trial volumes across oncology, rare disorders, metabolic conditions, and immunology. Decentralized and hybrid study models strengthen adoption of tools that support virtual visits, wearable integration, and real time data review. Regulatory expectations for data integrity and audit readiness increase the use of validated digital systems across study planning, execution, and safety reporting.
Biotech firms depend on cloud based platforms to reduce administrative burden and accelerate decision making during early and late phase development. Integration of genomics, imaging, and patient reported outcomes expands data complexity, which reinforces the need for unified analytics environments. Sites adopt eSource and electronic consent tools to streamline documentation and reduce errors. Investment in real world evidence programs increases demand for platforms capable of linking trial data with observational datasets. Cybersecurity priorities support migration to controlled digital infrastructures across research networks.
Demand for eClinical solutions and software in the USA is shaped by the expansion of clinical research programs, regulatory expectations for digital data integrity, and the need for workflow automation across trial phases. Buyers prioritize data capture accuracy, monitoring efficiency, and interoperability with existing research systems. Adoption patterns reflect trial complexity, patient recruitment challenges, and requirements for real-time analytics across sponsors and research partners.
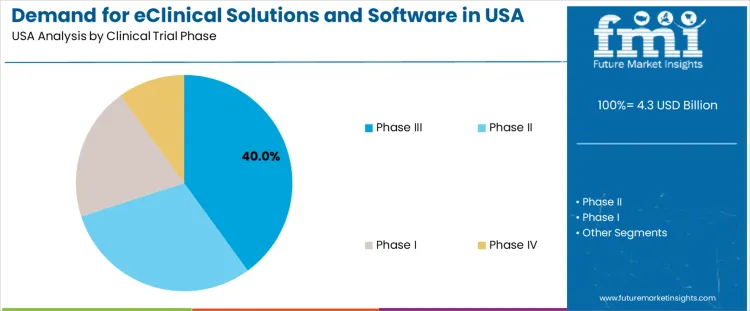
Phase III holds 40.0%, making it the leading phase segment in USA demand for eClinical platforms. Large patient populations and multicenter coordination require advanced electronic data capture, remote monitoring, and centralized analytics to manage safety signals and efficacy outcomes. Phase II holds 30.0%, driven by mid-stage trials that require structured protocol management and detailed pharmacodynamic assessments. Phase I holds 20.0%, supporting early safety evaluations and tightly controlled data environments. Phase IV holds 10.0%, reflecting post-marketing surveillance needs and long-term data collection. Phase distribution aligns with workload intensity, data volume, and operational complexity.
Key Points:
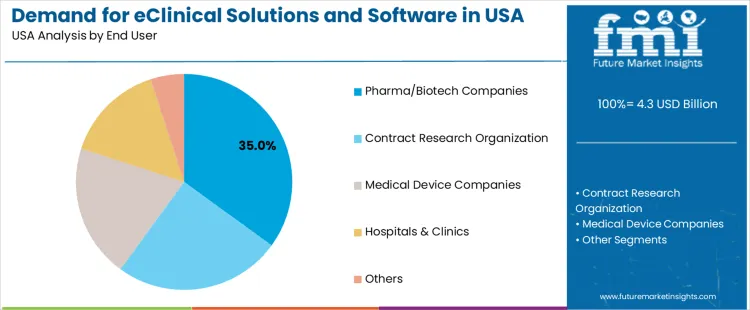
Pharma and biotech companies hold 35.0%, making them the leading end-user segment for eClinical systems in the USA. These organizations run diverse pipelines that depend on integrated digital platforms for trial oversight, regulatory documentation, and data governance. Contract research organizations hold 25.0%, using eClinical tools to manage outsourced trial operations and streamline coordination with sponsors. Medical device companies hold 20.0%, supporting device-specific protocols, usability studies, and post-approval data requirements. Hospitals and clinics hold 15.0%, contributing patient enrollment data and site-level monitoring. Others hold 5.0%, representing limited but specialized research activities. Distribution reflects trial ownership, operational scale, and regulatory compliance needs.
Key Points:
Demand increases as US pharmaceutical sponsors manage growing trial volumes that require integrated electronic data capture, clinical trial management, and remote monitoring systems. Contract research organizations expand use of unified platforms to coordinate multi-site studies with standardized documentation. Decentralized trial models introduce electronic patient-reported outcomes and telehealth-based visit scheduling. Real-time data access supports faster safety review and protocol adjustments. Federal initiatives promoting interoperability strengthen interest in validated digital platforms. Academic medical centres adopt electronic consent systems to streamline recruitment. Biotech firms with limited internal infrastructure rely on cloud-based eClinical tools to manage early-phase research efficiently.
Sponsors evaluate software licensing and implementation fees that increase total operational expenditure for long-duration trials. Several organizations encounter data integration difficulties when linking legacy systems with modern eClinical platforms. Smaller research sites report limited staff capacity for training on complex digital tools. Variability in connectivity across rural trial locations affects reliability of remote data capture. Compliance requirements for audit trails, user access controls, and validation testing raise administrative workload. Some sponsors delay upgrades when technology transitions risk interrupting ongoing trials. Sites with limited hardware resources experience performance issues during high-volume data entry periods.
US research programmes expand use of remote patient monitoring devices that feed data directly into eClinical systems. Sponsors adopt analytics tools that support risk-based monitoring and predictive assessment of site performance. AI-assisted data cleaning improves query resolution efficiency. Unified platforms that combine EDC, CTMS, eCOA, and safety reporting gain traction as organizations seek reduced operational fragmentation. Cloud-native architectures support scalable trial deployment across dispersed sites. Patient-centric interfaces enhance engagement in decentralized protocols. Regulatory interest in real-time oversight encourages uptake of systems capable of continuous data traceability and structured reporting across therapeutic portfolios.
Demand for eClinical solutions and software in the USA is rising due to broader digital transformation in clinical research, increased trial complexity, and stronger adoption of data-integration systems. West USA records a CAGR of 13.0% supported by its concentration of technology-enabled research operations and biotechnology firms. South USA shows an 11.6% CAGR driven by expanding clinical-trial networks and growth in CRO activity. Northeast USA posts a 10.4% CAGR due to dense pharmaceutical and academic research environments. Midwest USA holds a 9.0% CAGR supported by hospital-linked research programs and steady adoption of digital trial-management platforms.
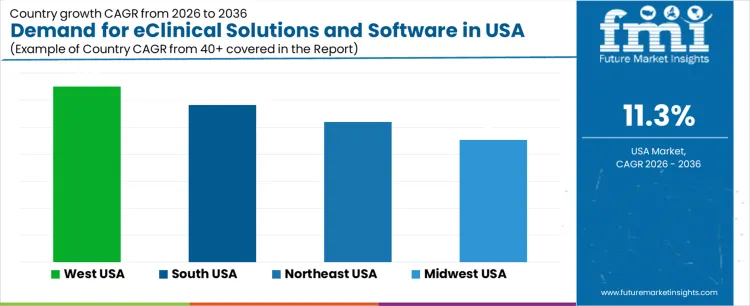
| Region | CAGR (2026-2036) |
|---|---|
| West USA | 13.0% |
| South USA | 11.6% |
| Northeast USA | 10.4% |
| Midwest USA | 9.0% |
West USA drives demand through its strong presence of biotechnology firms, digital health developers, and research institutions that depend on integrated clinical-data systems. The region’s CAGR of 13.0% reflects high utilization of platforms supporting electronic data capture, clinical-trial management, and real-time analytics. Organizations conducting early and mid-stage trials rely on digital workflows to manage complex datasets and improve operational oversight. Technology firms in the region contribute to continuous advancement of cloud-based and automation-enabled eClinical architectures. The region’s active research ecosystem encourages adoption of structured platforms that support regulatory compliance and high-frequency data monitoring.
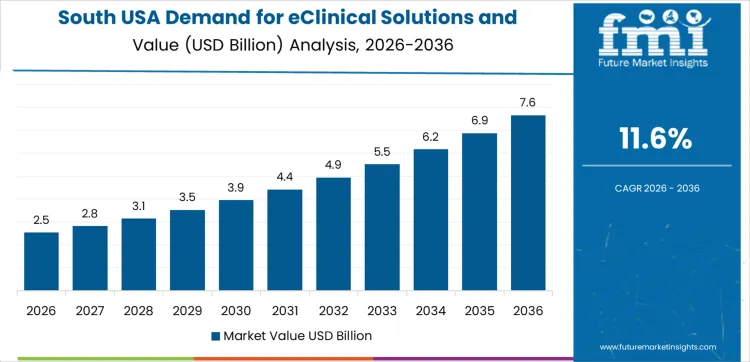
South USA supports rising demand due to expanding clinical-research networks, growing CRO activity, and increasing numbers of multi-site trials. The region’s CAGR of 11.6% reflects steady usage of eClinical platforms for workflow standardization, data consolidation, and trial oversight. Hospitals and academic centers adopt digital tools to support protocol compliance and patient-data integrity. CROs rely on eClinical systems to manage large trial portfolios involving diverse therapeutic areas. Regional life-science development zones contribute to wider adoption of electronic data capture, remote-monitoring platforms, and site-management systems.
Northeast USA drives demand due to its dense pharmaceutical, academic, and clinical-research infrastructure that requires structured data-management systems. The region’s CAGR of 10.4% reflects consistent utilization of eClinical tools for trial planning, regulatory documentation, and real-time evaluation of patient outcomes. Established pharmaceutical firms rely on integrated platforms to coordinate complex global studies. Academic medical centers generate high trial volumes requiring secure data pathways. Diagnostic and translational research initiatives maintain strong engagement with analytics-enabled software for continuous assessment.
Midwest USA maintains steady demand due to its broad network of hospital research programs, academic institutions, and mid-scale biopharmaceutical firms. The region’s CAGR of 9.0% reflects routine reliance on eClinical systems for structured trial execution, data validation, and compliance management. Healthcare systems conduct numerous therapeutic studies requiring digital oversight tools. Academic investigators use electronic platforms to streamline trial operations and manage multisite collaborations. Mid-scale pharmaceutical and device firms adopt eClinical software to enhance data accuracy and meet documentation requirements.
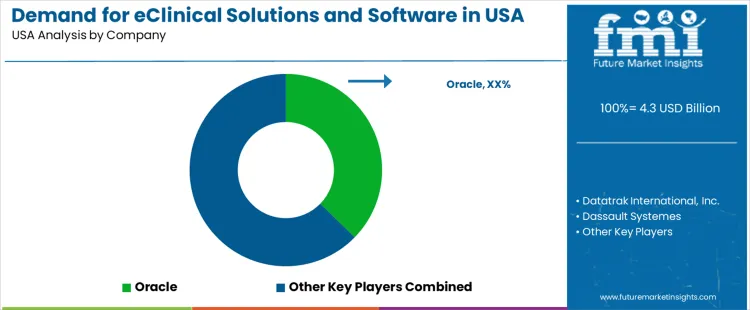
Demand for eClinical solutions and software in the USA is shaped by requirements for centralized data management, secure clinical workflow integration, and validated digital tools that support clinical trial planning, execution, and monitoring. Buyers evaluate platform interoperability, audit readiness, user interface stability, and compatibility with electronic data capture, randomization, and clinical trial management systems. Procurement teams prioritize regulatory conformity, cybersecurity safeguards, and scalable architectures supporting studies with varying protocol complexity. Adoption patterns reflect the expansion of decentralized trials, higher data volume from digital endpoints, and increased reliance on cloud-based clinical technologies.
Oracle holds an estimated 37.3% share. Its position reflects widespread use of clinical data platforms, integrated trial management modules, and strong adoption among contract research organizations and large pharmaceutical sponsors. Datatrak International, Inc. participates through configurable electronic data capture systems and workflow tools supporting mid-sized trial portfolios. Dassault Systèmes maintains visibility through modeling, simulation, and data frameworks used in research environments requiring structured data integration.
CRF Health contributes with electronic clinical outcome assessment solutions supporting patient-reported data collection in both site-based and remote settings. EClinicalWorks serves healthcare networks conducting observational research and registry-based studies using practice management and electronic health record systems. Competitive positioning in the USA reflects data integrity, deployment flexibility, validation rigor, and long-term support capabilities across clinical research operations.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Clinical Trial Phase | Phase III, Phase II, Phase I, Phase IV |
| End User | Pharma/Biotech Companies, Contract Research Organization, Medical Device Companies, Hospitals & Clinics, Others |
| Solution | Randomization & Trial Management (RTSM), Clinical Data Management System (CDSM), Clinical Trial Management System (CLMS), Electronic Clinical Outcome Assessment (eCOA), Electronic Trial Master File (eTMF) |
| Delivery Mode | Cloud-based (SAAS) Solution, Licensed Enterprise (On-premise), Web-hosted (On-demand) Solution |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Oracle, Datatrak International, Inc., Dassault Systèmes, CRF Health, eClinicalWorks |
| Additional Attributes | Dollar sales by clinical trial phase, end-user segment, solution type, and delivery mode; regional adoption patterns across West, South, Northeast, and Midwest USA; digitization trends in clinical research workflows; increasing integration of eCOA, eTMF, and cloud-based CTMS platforms; competitive positioning of SaaS vendors and enterprise eClinical providers; regulatory alignment with FDA and GCP requirements; advancements in remote trial conduct, decentralized trial solutions, and real-time trial analytics. |
How big is the demand for eclinical solutions and software in USA in 2026?
The demand for eclinical solutions and software in USA is estimated to be valued at USD 4.3 billion in 2026.
What will be the size of eclinical solutions and software in USA in 2036?
The market size for the eclinical solutions and software in USA is projected to reach USD 12.5 billion by 2036.
How much will be the demand for eclinical solutions and software in USA growth between 2026 and 2036?
The demand for eclinical solutions and software in USA is expected to grow at a 11.3% CAGR between 2026 and 2036.
What are the key product types in the eclinical solutions and software in USA?
The key product types in eclinical solutions and software in USA are phase iii, phase ii, phase i and phase iv.
Which end user segment is expected to contribute significant share in the eclinical solutions and software in USA in 2026?
In terms of end user, pharma/biotech companies segment is expected to command 35.0% share in the eclinical solutions and software in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.