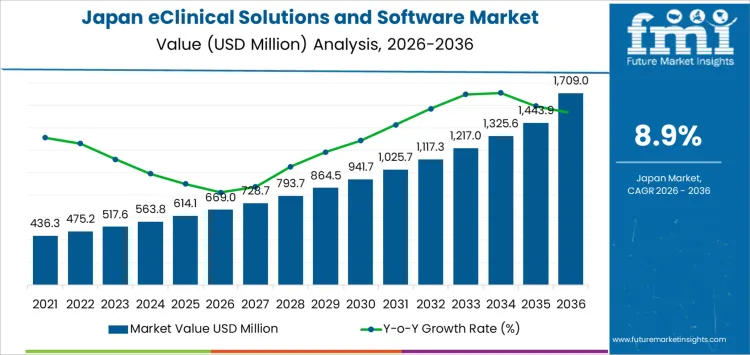
The demand for eClinical solutions and software in Japan is projected to grow significantly, from USD 669.0 million in 2026 to USD 1,709 million by 2036, reflecting a robust CAGR of 8.9%. The pharmaceutical and biotech sectors are key drivers, fueled by the increasing complexity of clinical trials and the need for efficient data management. Cloud-based solutions dominate the industry, accounting for a large portion of the demand, supported by the scalability and flexibility they offer to clinical trial management. The rising need for real-time data collection, enhanced regulatory compliance, and more efficient trial management systems are pushing the growth of eClinical solutions across Japan’s healthcare landscape.
Kyushu & Okinawa leads the growth with a CAGR of 11.2%, driven by their robust pharmaceutical and biotech industries. Kanto, the largest hub for pharmaceutical research, follows with a strong 10.3% CAGR, supported by Tokyo’s expansive clinical trial networks. Kansai and Chubu also show healthy growth at 9.0% and 7.9%, respectively, with increasing adoption of eClinical solutions among healthcare and life sciences companies. As Japan’s focus on digital transformation in healthcare and clinical trials intensifies, demand for advanced eClinical software solutions is expected to grow steadily, facilitating the optimization of trial efficiency, data accuracy, and patient outcomes across various clinical phases and end-user segments such as pharma, biotech, and contract research organizations (CROs).
| Metric | Value |
|---|---|
| Demand for eClinical Solutions and Software in Japan Value (2026) | USD 669.0 million |
| Demand for eClinical Solutions and Software in Japan Forecast Value (2036) | USD 1,709 million |
| Demand for eClinical Solutions and Software in Japan Forecast CAGR (2026-2036) | 8.9% |
The demand for eClinical solutions and software in Japan is growing due to the increasing need for efficient, data-driven approaches in clinical trials, drug development, and healthcare management. eClinical solutions offer a range of software tools that enhance the efficiency and accuracy of clinical trials, from patient recruitment and data management to analysis and reporting. The rise in the number of clinical trials, driven by the growing pharmaceutical, biotech, and medical device industries in Japan, is fueling the demand for such software to streamline operations, ensure regulatory compliance, and accelerate time-to-industry for new therapies and drugs.
Key drivers for this growth include the increasing adoption of digitalization in the healthcare sector, particularly within clinical research organizations (CROs) and pharmaceutical companies. With the growing complexity of clinical trials, eClinical solutions help simplify data management, improve collaboration among stakeholders, and enhance the overall trial process. The focus on precision medicine and the need for more targeted therapies are also driving the demand for advanced data analysis and management tools, which eClinical software can provide.
Japan’s aging population and the increasing prevalence of chronic diseases are pushing healthcare providers and researchers to seek more efficient ways to manage patient data, conduct trials, and deliver treatments. Despite challenges such as regulatory hurdles and the high cost of implementation, the continued shift toward more efficient, data-driven healthcare and clinical trial processes is expected to drive substantial growth in the demand for eClinical solutions and software in Japan through 2036.
Demand for eClinical solutions and software in Japan is segmented by clinical trial phase, end-user, and region. By clinical trial phase, Phase III leads with 40% of the demand, followed by Phase II, Phase I, and Phase IV. In terms of end-users, pharma/biotech companies account for 35% of the demand, with contract research organizations, medical device companies, hospitals & clinics, and other sectors contributing to the remainder. Regionally, demand is distributed across Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, and the rest of Japan.
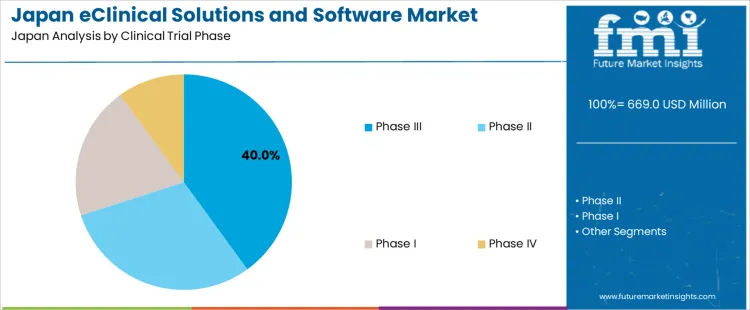
Phase III clinical trials account for 40% of the demand for eClinical solutions and software in Japan due to their critical role in validating the safety and efficacy of new treatments before industry approval. During this phase, large-scale testing is conducted to gather comprehensive data, requiring robust, scalable software solutions to manage complex data, monitor patient outcomes, and ensure regulatory compliance. The demand for eClinical solutions increases as Phase III trials involve extensive coordination between research sites, sponsors, and regulatory bodies. As Japan's pharmaceutical and biotech industries focus on efficient, reliable trial processes, Phase III trials remain a major driver for eClinical software solutions.
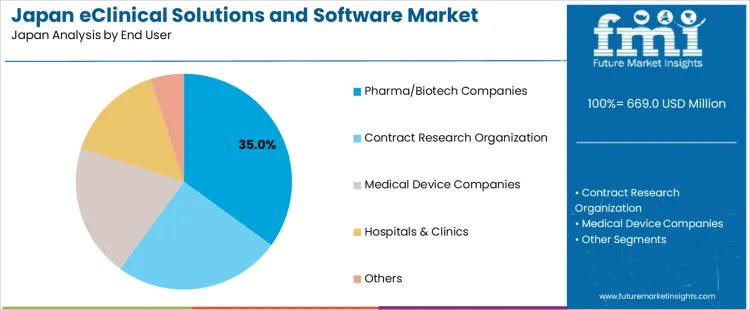
Pharma/biotech companies account for 35% of the demand for eClinical solutions and software in Japan, as these companies heavily rely on advanced software to streamline the clinical trial process. These organizations use eClinical solutions to manage clinical data, ensure compliance with regulations, and enhance collaboration between teams during drug development. As the pharmaceutical and biotech sectors continue to push for faster drug development cycles and more efficient clinical trials, the demand for eClinical solutions grows. The software allows for real-time data analysis, reduces the risk of errors, and speeds up the trial process, making it indispensable for companies looking to bring new therapies to industry quickly and efficiently.
Demand for eClinical solutions and software in Japan is increasing as the life sciences sector embraces digital transformation. Trends include the adoption of electronic data capture, clinical trial management systems, and remote patient data collection. Restraints include legacy systems, challenges with localization, and cautious adoption due to compliance concerns in Japan’s highly regulated clinical environment.
Why is Demand for eClinical Solutions and Software Growing in Japan?
Demand for eClinical solutions and software in Japan is growing as pharmaceutical companies and contract research organizations seek to streamline drug development and improve trial efficiency. Japan’s regulated clinical landscape demands high data integrity and audit readiness, driving digital transformation. The increasing focus on chronic and rare diseases, combined with the need for global clinical trial participation, boosts demand for scalable, integrated software. The need for centralized and accessible data across teams also encourages investment in eClinical solutions, supporting faster decision‑making and better patient outcomes.
What are the Key Challenges and Risks That Could Limit eClinical Solutions Demand in Japan?
Strict regulatory requirements, such as data protection and GCP standards, make implementation complex. Organizations with legacy systems may face integration challenges, leading to data fragmentation. Localization needs, including Japanese‑language interfaces and support, can delay adoption. High costs for software solutions and training are barriers for smaller research centers and sponsors. Cybersecurity concerns and the need for staff to adapt to new digital workflows create hurdles for full implementation, slowing the uptake of eClinical solutions in some sectors.
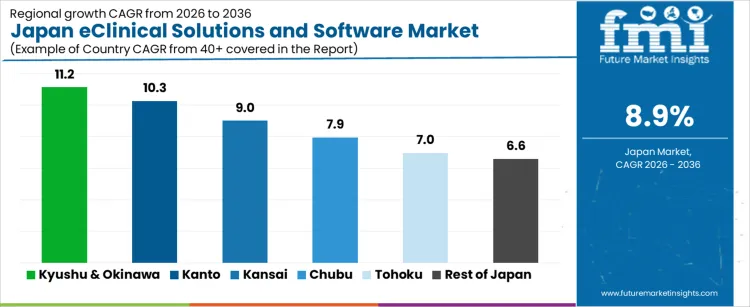
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 11.2% |
| Kanto | 10.3% |
| Kansai | 9.0% |
| Chubu | 7.9% |
| Tohoku | 7.0% |
| Rest of Japan | 6.6% |
Demand for eClinical solutions and software in Japan is growing steadily, with Kyushu & Okinawa leading at an 11.2% CAGR. Kanto follows with a 10.3% CAGR, supported by its large pharmaceutical and biotech industries and concentration of clinical trial organizations. Kansai shows a 9.0% CAGR, fueled by its pharmaceutical sector and growing interest in personalized medicine. Chubu experiences a 7.9% CAGR, driven by its expanding pharmaceutical and medical device sectors. Tohoku and the rest of Japan see moderate growth at 7.0% and 6.6% CAGR, respectively, as digital health adoption spreads across the country.
Kyushu & Okinawa leads the demand for eClinical solutions and software in Japan, growing at an 11.2% CAGR. The region’s strong focus on pharmaceutical and biotech industries, combined with the increasing need for digital transformation in clinical trials and research, is driving the growth of eClinical solutions. Kyushu is home to several pharmaceutical and life sciences companies that are increasingly adopting cloud-based clinical software to improve the efficiency and accuracy of their clinical trials. Okinawa, with its growing focus on health technology, is also seeing an increase in the use of eClinical software solutions. The region’s increasing emphasis on automation and regulatory compliance in clinical research is supporting the adoption of these technologies. As more pharmaceutical companies, contract research organizations (CROs), and healthcare institutions in Kyushu & Okinawa implement eClinical solutions, the demand for software in this sector is expected to continue to rise.
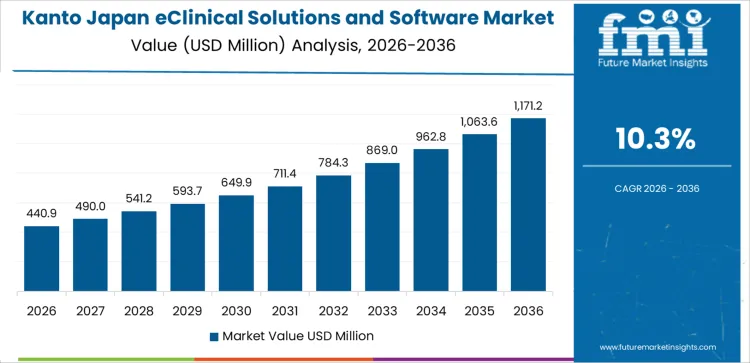
Kanto is experiencing strong demand for eClinical solutions and software, with a 10.3% CAGR. The region, particularly Tokyo, is a major hub for pharmaceutical and biotech industries, which are increasingly adopting eClinical solutions to streamline clinical trial processes, improve data management, and ensure regulatory compliance. Kanto’s large number of contract research organizations (CROs) and clinical trial sites are driving the demand for advanced software tools for trial management, patient recruitment, data analysis, and reporting. The region’s strong healthcare infrastructure and focus on precision medicine and clinical research innovation further contribute to the rising demand for eClinical solutions. With an increasing number of clinical trials and drug development projects in Kanto, the need for efficient and scalable clinical software will continue to grow, making the region a key player in the expansion of eClinical solutions in Japan.
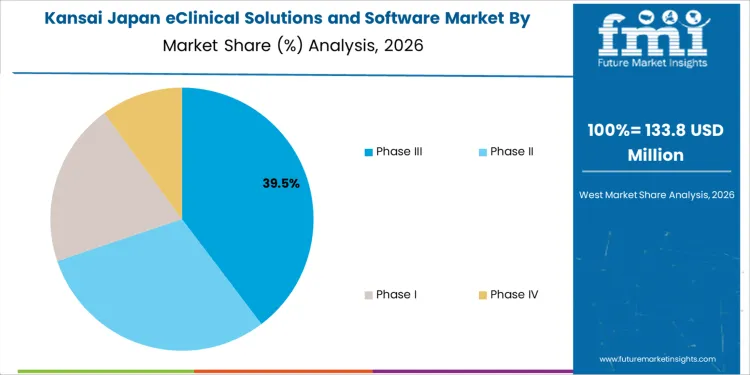
Kansai is seeing steady demand for eClinical solutions and software, with a 9.0% CAGR. The region’s focus on pharmaceutical research, biotechnology, and medical devices is fueling the growth in clinical software adoption. Kansai, with major cities like Osaka and Kyoto, is home to numerous academic institutions and pharmaceutical companies that are adopting eClinical solutions for clinical trial management, regulatory compliance, and data analytics. The growing emphasis on personalized medicine and the increasing need for real-time data collection and monitoring during clinical trials are driving the demand for software solutions. Kansai’s commitment to advancing healthcare and life sciences innovation is further supporting the growth of eClinical solutions in the region. As the demand for clinical trials and digital health solutions continues to rise, Kansai is expected to see continued growth in the adoption of eClinical software.
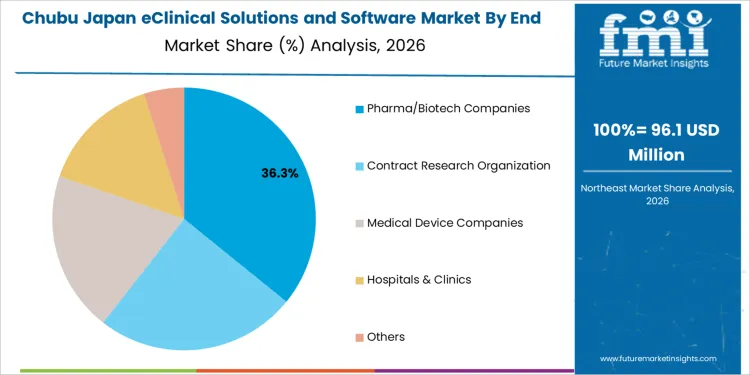
Chubu is experiencing moderate demand for eClinical solutions and software, with a 7.9% CAGR. The region’s strong manufacturing sector, particularly in pharmaceuticals and medical devices, is increasingly integrating eClinical software into clinical trials and research processes to improve efficiency and reduce costs. Cities like Nagoya, which are home to large pharmaceutical companies, are seeing a growing need for software solutions that support clinical trial management, data collection, and regulatory compliance. Chubu’s increasing focus on health technology and innovation in clinical research is further boosting the demand for eClinical solutions. As Chubu’s life sciences sector continues to grow and expand, the need for digital solutions in clinical trials, patient management, and data analysis will continue to rise, supporting the region’s steady growth in eClinical software demand.
Tohoku is seeing moderate demand for eClinical solutions and software, with a 7.0% CAGR. While the region is not as heavily concentrated in pharmaceutical activity as other regions, Tohoku is increasingly adopting eClinical software solutions to support clinical trials, research, and patient care. The region’s growing focus on healthcare innovation and digital health, combined with government support for biotech and pharmaceutical research, is contributing to the rise in demand for eClinical solutions. Tohoku’s emphasis on improving clinical trial efficiency and ensuring regulatory compliance in healthcare practices is driving the adoption of these technologies. As more pharmaceutical and healthcare companies in Tohoku implement digital solutions for clinical trials and patient management, the demand for eClinical solutions and software is expected to continue growing.
The rest of Japan is experiencing moderate demand for eClinical solutions and software, growing at a 6.6% CAGR. While the demand for eClinical software is more concentrated in major regions like Kanto and Kansai, other areas of Japan are increasingly adopting these solutions in the healthcare and pharmaceutical sectors. Government and regional support for biotech innovation and clinical research is fueling the adoption of digital solutions for clinical trial management, regulatory compliance, and data analysis. As the focus on digital health and healthcare modernization continues across Japan, the demand for eClinical solutions will gradually rise in these regions. The continued expansion of clinical trials and drug development projects in the rest of Japan, supported by both private and public investments, will contribute to steady growth in the adoption of eClinical software solutions.
Demand for eClinical solutions and software in Japan is growing as the pharmaceutical and healthcare industries look to streamline clinical trials, improve data management, and comply with increasingly complex regulations. eClinical software enables more efficient data collection, monitoring, and analysis, reducing the time and costs associated with bringing new therapies to industry. With Japan's advanced healthcare infrastructure and regulatory focus on data accuracy and patient safety, the need for specialized eClinical solutions continues to rise.
Oracle is a leader in the eClinical solutions industry, offering comprehensive software platforms that integrate clinical trial management, data analytics, and regulatory compliance tools. Oracle’s strategy focuses on providing scalable and customizable solutions for the pharmaceutical, biotechnology, and healthcare sectors, allowing organizations to optimize clinical trial operations and ensure better patient outcomes.
Dassault Systemes provides end-to-end solutions, including simulation and 3D modeling tools, for clinical trials and healthcare. CRF Health specializes in eSource solutions that improve the accuracy and efficiency of data collection in clinical trials. EClinicalWorks focuses on electronic health record (EHR) and practice management software, providing clinical solutions that improve patient care and streamline healthcare operations. These companies are competing by innovating their software platforms, offering more integrated, efficient, and user-friendly solutions to meet Japan's evolving clinical research needs.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Key Segments | Phase III, Phase II, Phase I, Phase IV |
| End User | Pharma/Biotech Companies, Contract Research Organization, Medical Device Companies, Hospitals & Clinics, Others |
| Solution | Randomization & Trial Management (RTSM), Clinical Data Management System (CDSM), Clinical Trial Management System (CLMS), Electronic Clinical Outcome Assessment (eCOA), Electronic Trial Master File (eTMF) |
| Delivery Mode | Cloud-based (SAAS) Solution, Licensed Enterprise (On-premise), Web-hosted (On-demand) Solution |
| Key Country | Japan |
| Region | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Key Players Profiled | Oracle, Datatrak International, Inc., Dassault Systemes, CRF Health, EClinicalWorks |
| Additional Attributes | Dollar sales by clinical trial phase, end user, solution, and delivery mode; regional CAGR and growth trends in eClinical Solutions and Software demand in Japan |
How big is the demand for eclinical solutions and software in Japan in 2026?
The demand for eclinical solutions and software in Japan is estimated to be valued at USD 669.0 million in 2026.
What will be the size of eclinical solutions and software in Japan in 2036?
The market size for the eclinical solutions and software in Japan is projected to reach USD 1,709.0 million by 2036.
How much will be the demand for eclinical solutions and software in Japan growth between 2026 and 2036?
The demand for eclinical solutions and software in Japan is expected to grow at a 8.9% CAGR between 2026 and 2036.
What are the key product types in the eclinical solutions and software in Japan?
The key product types in eclinical solutions and software in Japan are phase iii, phase ii, phase i and phase iv.
Which end user segment is expected to contribute significant share in the eclinical solutions and software in Japan in 2026?
In terms of end user, pharma/biotech companies segment is expected to command 35.0% share in the eclinical solutions and software in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.