Medical Devices Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The report delivers an in-depth look at the medical devices market, spanning market sizing, share analysis, production volumes, sales forecasting, brand positioning, competitive intelligence, demand dynamics, growth catalysts, market barriers, ai integration trends, precision medicine integration trends, clinical adoption patterns, distribution performance, supply chain mapping, policy and regulatory impact, investment landscape, and forward-looking growth strategy.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Medical Devices Market Size, Market Forecast and Outlook By FMI
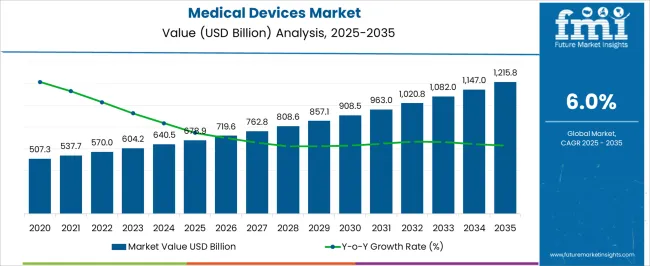
The medical devices market was valued at USD 678.9 Billion in 2025, projected to reach USD 719.63 Billion in 2026, and is forecast to expand to USD 1288.75 Billion by 2036 at a 6% CAGR. Aging populations across developed economies and rising chronic disease prevalence in emerging markets continue to drive demand for diagnostic, therapeutic, and monitoring technologies. Capital expenditure cycles across hospital networks now prioritize acquisition of connected, interoperable platforms that reduce per-procedure costs while improving clinical throughput in both acute and ambulatory care settings.
Summary of Medical Devices Market
- Market valuation is expected to climb to USD 1288.75 billion by 2036.
- The industry is expected to expand at 6% annually from 2026 through 2036.
- The 2025 baseline valuation came in at USD 678.9 billion.
- The ten-year outlook presents a cumulative growth opportunity of USD 569.12. billion.
- In-vitro Diagnostic leads by Type with 21.2% share in 2026, supported by laboratory automation and point-of-care testing demand.
- Hospitals & ASCs leads by End-User with 42.3% share in 2026.
- The market remains a broad, regulation-driven category where clinical interoperability, reimbursement eligibility, and connected device capabilities define competitive positioning.
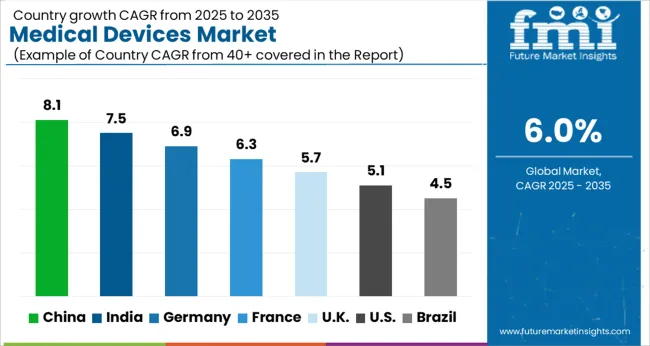
- Among key countries, China leads at 8.1% CAGR, followed by India at 7.5%, Germany at 6.9%, France at 6.3%, the UK at 5.7%, the USA at 5.1%, and Brazil at 4.5%.
Absolute dollar opportunity over the forecast period amounts to USD Billion 569.12, reflecting sustained capital allocation across multiple clinical verticals. As per FMI, regulatory harmonization efforts between the United States FDA and European MDR frameworks are compressing approval timelines for combination products, enabling manufacturers to pursue parallel market entry strategies. Precision medicine integration is reshaping procurement specifications, with health systems demanding devices that generate structured data compatible with electronic health record ecosystems and population health analytics platforms.
Country-level growth trajectories reflect divergent stages of healthcare infrastructure maturity. China leads at 8.1% CAGR, driven by centralized volume-based procurement reforms and hospital tier expansion programs. India follows at 7.5%, supported by national health protection scheme expansions covering equipment-intensive procedures. Germany advances at 6.9% as MDR compliance investments force device upgrades. France registers 6.3% growth tied to ambulatory surgery center expansion. The United Kingdom grows at 5.7%, with NHS procurement framework renewals directing capital toward next-generation diagnostic platforms. The United States expands at 5.1% on replacement cycles and ambulatory surgical center proliferation. Brazil records 4.5% growth anchored by SUS public health system modernization.
Medical Devices Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 719.63 Billion |
| Industry Value (2036) | USD 1288.75 Billion |
| CAGR (2026 to 2036) | 6% |
Source: Future Market Insights, 2026
Medical Devices Market Definition
Medical devices encompass instruments, apparatus, machines, implants, and in-vitro reagents intended for diagnosis, prevention, monitoring, treatment, or alleviation of disease. The market covers the full spectrum of device classes from low-risk consumables through high-risk implantable systems, spanning cardiovascular, orthopedic, diagnostic imaging, surgical, diabetes care, ophthalmic, dental, nephrology, and wound management categories. Device selection is governed by clinical indication, regulatory classification, facility accreditation requirements, and payer reimbursement eligibility.
Medical Devices Market Inclusions
Market scope includes global and regional revenue sizing for all regulated medical device classes, forecast projections from 2026 to 2036, segmentation by device type and end-user, country-level demand analysis, and competitive landscape mapping across diagnostic, therapeutic, surgical, and monitoring device categories.
Medical Devices Market Exclusions
The scope excludes pharmaceutical products, consumable laboratory reagents sold independently of device platforms, health information technology software-only solutions, veterinary devices, and consumer wellness wearables not classified under medical device regulations.
Medical Devices Market Research Methodology
- Primary Research: Analysts conducted structured interviews with device manufacturers, hospital procurement directors, clinical engineers, and payer reimbursement specialists to map purchasing decision gates and technology adoption cycles.
- Desk Research: Data collection aggregated regulatory filing databases, national health expenditure reports, device approval registries, and published clinical trial outcomes across major markets.
- Market-Sizing and Forecasting: Baseline values derive from a hybrid bottom-up and top-down methodology, cross-referencing company revenue disclosures with regional health expenditure data and device utilization rates.
- Data Validation and Update Cycle: Projections are tested against publicly reported financial guidance from leading companies and verified through quarterly update cycles incorporating the most recent regulatory and clinical evidence.
Why is the Medical Devices Market Growing?
The medical devices market is experiencing steady growth driven by increasing demand for advanced diagnostic and therapeutic equipment across healthcare facilities worldwide. Rising prevalence of chronic diseases, aging populations, and expansion of healthcare infrastructure are significantly supporting market growth. Technological advancements in devices, coupled with increased focus on early disease detection and minimally invasive procedures, are enhancing treatment outcomes and operational efficiency.
Adoption of smart and connected medical devices is being facilitated by integration with electronic health records and telehealth platforms, allowing healthcare providers to monitor patient conditions in real time. Regulatory initiatives promoting high-quality and safe medical equipment are further shaping the market landscape. Growing investments in research and development are driving innovation in device accuracy, durability, and patient safety.
Hospitals and ambulatory surgical centers are increasingly procuring advanced equipment to improve service quality, optimize workflows, and reduce treatment costs As healthcare systems globally continue to modernize, the medical devices market is positioned for long-term expansion with continued opportunities in both diagnostic and therapeutic segments.
Segmental Analysis
The medical devices market is segmented by type, end-user, and geographic regions. By type, medical devices market is divided into In-vitro Diagnostic (IVD), Orthopedic Devices, Cardiovascular Devices, Diagnostic Imaging Devices, Minimally Invasive Surgery Devices, Wound Management, Diabetes Care Devices, Ophthalmic Devices, Dental Devices, Nephrology Devices, General Surgery, and Others. In terms of end-user, medical devices market is classified into Hospitals & ASCs, Clinics, and Others. Regionally, the medical devices industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the In-vitro Diagnostic (IVD) Type Segment
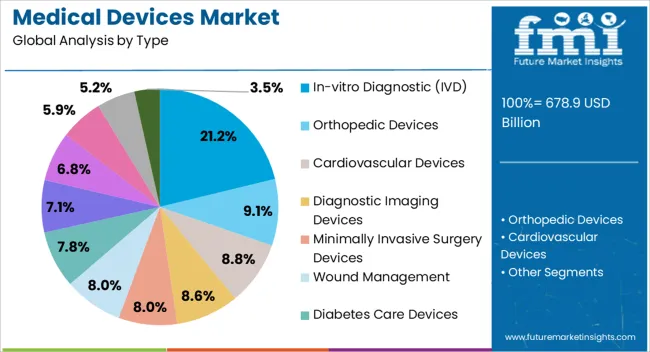
The in-vitro diagnostic type segment is projected to hold 21.2% of the medical devices market revenue share in 2026, establishing it as a leading product type. This leadership is being driven by the growing need for early disease detection and preventive healthcare across hospitals and clinical laboratories. IVD devices provide accurate, rapid, and cost-effective testing for a wide range of conditions, including infectious diseases, metabolic disorders, and cancers.
Their adoption is being facilitated by advancements in automation, molecular diagnostics, and point-of-care testing platforms, which improve throughput and diagnostic reliability. Hospitals and laboratories are increasingly integrating IVD systems into routine workflows to enhance patient outcomes and reduce diagnostic turnaround times. The segment benefits from continuous innovation that expands the range of detectable conditions, improves sensitivity and specificity, and reduces operational complexity.
Increasing awareness about preventive healthcare and government initiatives promoting laboratory diagnostics are further supporting adoption As healthcare providers continue to prioritize efficiency, accuracy, and compliance with international standards, the in-vitro diagnostic segment is expected to maintain strong growth and remain a key revenue contributor.
Insights into the Hospitals and ASCs End-User Segment
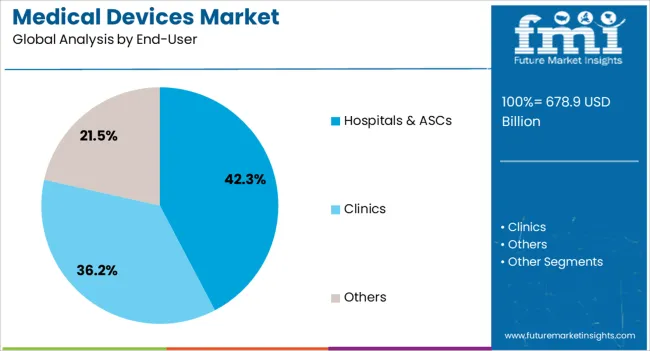
The hospitals and ambulatory surgical centers end-user segment is anticipated to account for 42.3% of the medical devices market revenue share in 2026, making it the largest end-user category. This dominance is being driven by the continuous expansion of hospital networks, increasing surgical volumes, and the need for comprehensive patient care across outpatient and inpatient settings. Hospitals and ASCs are increasingly procuring advanced medical devices that enable high-precision diagnostics, minimally invasive surgeries, and real-time patient monitoring.
Adoption is further supported by operational efficiency requirements, regulatory mandates, and the growing emphasis on improving patient outcomes while controlling costs. The segment benefits from the integration of medical devices with hospital information systems, enhancing workflow automation and enabling data-driven decision-making.
Rising investments in modernizing healthcare facilities and expanding capacity, particularly in emerging economies, are sustaining demand for high-quality devices As hospitals and ASCs continue to evolve as central hubs for diagnostic and therapeutic services, the reliance on sophisticated medical devices is expected to drive sustained growth and reinforce their leading position in the market.
Market Overview
Medical Devices Market: Global Industry Analysis and Opportunity Assessment 2026 - 2026 Overview
Medical devices market is the fastest increasing market worldwide due to its extensive use in hospitals, research and development centers, educational institutes and clinical laboratories.
Medical devices market is majorly classified into seven categories including diagnostic imaging devices, drug delivery devices, molecular diagnostic, surgical devices, monitoring devices, bioimplants and neurostimulation, and others.
The medical devices market is showing continuous growth driven by various factors such as rise in aging population, innovations in medical devices, changing lifestyles and increasing awareness about the medical conditions and available treatments.
Diagnostic imaging devices segment is the major revenue generating market for medical devices market due to extensive research and development. Other major revenue segment for medical devices market is bioimplants and neurostimulation specifically in markets of North America and Europe due to increasing demand for cosmetic implants and economic stability in these regions.
North America leads in the medical devices market followed by the Europe majorly because of aging population. According to the data of Centers for Disease Control and Prevention (CDS), in 2010 40 million people aged 65 and above in United States accounting for 13% of the total population and CDC projected that around 20% of the population in the country would be under this category by 2036.
Market of Asia-Pacific is also growing at faster pace for medical devices majorly driven by the growing economies of Japan, China and India. The key players in the market include Stryker Corporation, Johnson & Johnson, Philips Healthcare, Siemens Healthcare, GE Healthcare, Toshiba Medical Systems, Boston Scientific Corporation, St. Jude Medical, Inc., Biomet, Inc. and Smith and Nephew. Among all of the above Stryker Corporation and Johnson & Johnson are currently dominating medical devices markets.
This research report presents a comprehensive assessment of the market and contains thoughtful insights, facts, historical data and statistically-supported and industry-validated market data and projections with a suitable set of assumptions and methodology. It provides analysis and information by categories such as market segments, regions, product types and distribution channels.
The report covers exhaustive analysis on
-
Market Segments
-
Market Dynamics
-
Historical Actual Market Size, 2026 - 2026
-
Market Size & Forecast 2026 to 2026
-
Supply & Demand Value Chain
-
Current Trends/Issues/Challenges
-
Competition & Companies involved
-
Technology
-
Value Chain
-
Market Drivers and Restraints
Regional analysis includes
- North America
- US & Canada
- Latin America
- Brazil, Argentina & Others
- Western Europe
- EU5
- Nordics
- Benelux
- Eastern Europe
- Asia Pacific
- Australia and New Zealand (ANZ)
- Greater China
- India
- ASEAN
- Rest of Asia Pacific
- Japan
- Middle East and Africa
- GCC Countries
- Other Middle East
- North Africa
- South Africa
- Other Africa
The report is a compilation of first-hand information, qualitative and quantitative assessment by industry analysts, inputs from industry experts and industry participants across the value chain.
The report provides in-depth analysis of parent market trends, macro-economic indicators and governing factors along with market attractiveness as per segments. The report also maps the qualitative impact of various market factors on market segments and geographies.
Analysis of Medical Devices Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 8.1% |
| India | 7.5% |
| Germany | 6.9% |
| France | 6.3% |
| UK | 5.7% |
| USA | 5.1% |
| Brazil | 4.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
Country-wise Analysis
The Medical Devices Market is expected to register a CAGR of 6.0% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 8.1%, followed by India at 7.5%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Brazil posts the lowest CAGR at 4.5%, yet still underscores a broadly positive trajectory for the global Medical Devices Market. In 2024, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 6.9%. The USA Medical Devices Market is estimated to be valued at USD 234.7 billion in 2026 and is anticipated to reach a valuation of USD 385.9 billion by 2036. Sales are projected to rise at a CAGR of 5.1% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 31.5 billion and USD 18.0 billion respectively in 2026.
Key Players in the Medical Devices Market
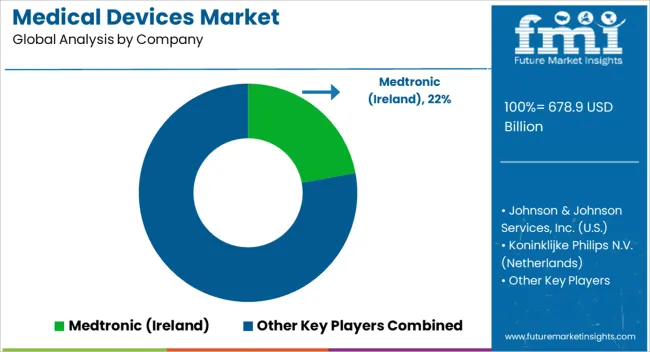
- Medtronic (Ireland)
- Johnson & Johnson Services, Inc. (USA)
- Koninklijke Philips N.V. (Netherlands)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Boston Scientific Corporation (USA)
- Fresenius Medical Care AG (Germany)
- GE Healthcare (USA)
- Siemens Healthineers AG (Germany)
- Stryker (USA)
- Abbott (USA)
- BD (USA)
- Cardinal Health (USA)
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD Billion 719.63 to USD Billion 1288.75, at a CAGR of 6% |
| Market Definition | Medical devices encompass instruments, apparatus, machines, implants, and in-vitro reagents intended for diagnosis, prevention, monitoring, treatment, or alleviation of disease. The market covers the full spectrum of device classes from low-risk consumables through high-risk implantable systems, spanning cardiovascular, orthopedic, diagnostic imaging, surgical, diabetes care, ophthalmic, dental, nephrology, and wound management categories. Device selection is governed by clinical indication, regulatory classification, facility accreditation requirements, and payer reimbursement eligibility. |
| Segmentation | Type (In-vitro Diagnostic, Orthopedic Devices, Cardiovascular Devices, Diagnostic Imaging Devices, Minimally Invasive Surgery Devices, Wound Management, Diabetes Care Devices, Ophthalmic Devices, Dental Devices, Nephrology Devices, General Surgery, Others), End-User (Hospitals & ASCs, Clinics, Others), and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | Medtronic, Johnson & Johnson Services, Inc., Koninklijke Philips N.V., F. Hoffmann-La Roche Ltd., Boston Scientific Corporation, Fresenius Medical Care AG, GE Healthcare, Siemens Healthineers AG, Stryker, Abbott, BD, Cardinal Health |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a hybrid bottom-up and top-down methodology starting with company-level revenue and regional health expenditure benchmarks to project adoption and growth trajectories. |
Medical Devices Market by Segments
By Type:
- In-vitro Diagnostic (IVD)
- Orthopedic Devices
- Cardiovascular Devices
- Diagnostic Imaging Devices
- Minimally Invasive Surgery Devices
- Wound Management
- Diabetes Care Devices
- Ophthalmic Devices
- Dental Devices
- Nephrology Devices
- General Surgery
- Others
By End-User:
- Hospitals & ASCs
- Clinics
- Others
By Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Health Organization. (2024). Global atlas of medical devices. WHO.
- 2. Organisation for Economic Co-operation and Development. (2024). Health at a glance 2024: OECD indicators. OECD Publishing.
- 3. USA Food and Drug Administration. (2025). Annual report on medical device approvals and clearances, fiscal year 2024. FDA.
- 4. European Commission. (2024). Implementation of Regulation (EU) 2017/745 on medical devices: Progress report. European Commission.
- 5. World Bank. (2024). Health nutrition and population statistics: Health expenditure. World Bank Group.
- 6. International Organization for Standardization. (2024). ISO 13485:2016 medical devices quality management systems. ISO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Medical Devices in the global market in 2026?
Demand for medical devices in the global market is estimated to be valued at USD Billion 719.63 in 2026.
What will be the market size of Medical Devices in the global market by 2036?
Market size for medical devices is projected to reach USD Billion 1288.75 by 2036.
What is the expected demand growth for Medical Devices in the global market between 2026 and 2036?
Demand for medical devices is expected to grow at a CAGR of 6% between 2026 and 2036.
Which Type is poised to lead global sales by 2026?
In-vitro Diagnostic accounts for 21.2% share in 2026.
How is the Medical Devices market segmented by End-User?
Hospitals & ASCs leads by End-User with 42.3% share in 2026.
Which country is projected to grow fastest in the Medical Devices market?
China is projected to grow at a CAGR of 8.1% during 2026 to 2036.
What is included in the scope of this Medical Devices market report?
Market scope includes global and regional revenue sizing for all regulated medical device classes, forecast projections from 2026 to 2036, segmentation by device type and end-user, country-level demand analysis, and competitive landscape mapping across diagnostic, therapeutic, surgical, and monitoring device categories.
What is excluded from the scope of this report?
The scope excludes pharmaceutical products, consumable laboratory reagents sold independently of device platforms, health information technology software-only solutions, veterinary devices, and consumer wellness wearables not classified under medical device regulations.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- In-vitro Diagnostic
- Orthopedic Devices
- Cardiovascular Devices
- Diagnostic Imaging Devices
- Minimally Invasive Surgery Devices
- Wound Management
- Diabetes Care Devices
- Ophthalmic Devices
- Dental Devices
- Nephrology Devices
- General Surgery
- In-vitro Diagnostic
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-User, 2026 to 2036
- Hospitals & ASCs
- Clinics
- Others
- Hospitals & ASCs
- Y to o to Y Growth Trend Analysis By End-User, 2021 to 2025
- Absolute $ Opportunity Analysis By End-User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End-User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End-User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By End-User
- Competition Analysis
- Competition Deep Dive
- Medtronic (Ireland)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson Services, Inc. (USA)
- Koninklijke Philips N.V. (Netherlands)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Boston Scientific Corporation (USA)
- Fresenius Medical Care AG (Germany)
- GE Healthcare (USA)
- Siemens Healthineers AG (Germany)
- Stryker (USA)
- Abbott (USA)
- BD (USA)
- Cardinal Health (USA)
- Medtronic (Ireland)
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End-User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End-User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End-User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End-User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End-User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End-User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End-User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End-User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End-User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End-User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

