Digital Insomnia (DTx) Market
Digital Insomnia (DTx) Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Digital Insomnia (DTx) Market Forecast and Outlook 2026 to 2036
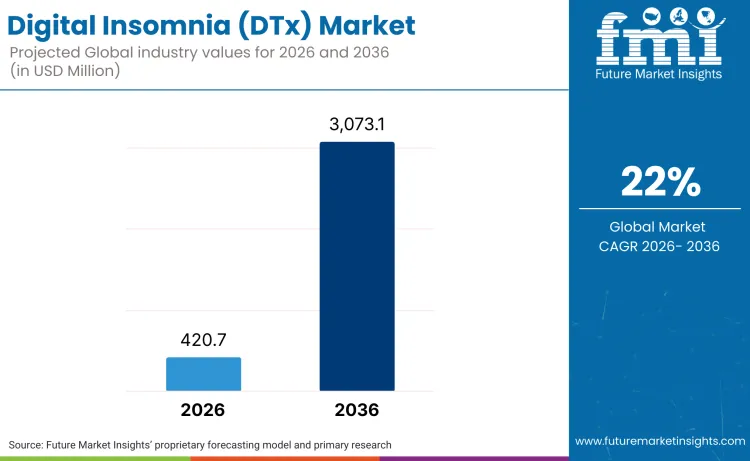
The digital insomnia therapeutics market is positioned to expand from USD 420.7 million in 2026 toward USD 3,073.1 million by 2036, driven by 22% CAGR. As per FMI, growth stems from payer recognition that cognitive behavioral therapy for insomnia delivered through regulated digital therapeutics reduces pharmaceutical spending while addressing root causes of chronic sleep disorders. As per FMI's projection, this expansion is supported by FDA software-as-a-medical-device guidance that establishes clear regulatory pathways for prescription DTx, converting experimental apps into reimbursable treatments.
As more individuals turn to digital health solutions, there has been a growing demand for accessible, non-pharmacological treatments for improving sleep quality. DTx platforms provide personalized, evidence-based interventions that combine cognitive behavioral therapy (CBT), mindfulness techniques, and sleep education, offering an effective alternative to traditional drug-based therapies. With the increasing prevalence of sleep disturbances, especially in urban populations where stress, technology use, and poor sleep hygiene contribute to the condition, DTx solutions are gaining traction as a sustainable and scalable option for managing insomnia.
The market is shifting from a focus on symptom management to a more integrated approach that emphasizes long-term behavioral change. This shift is driven by advancements in artificial intelligence, which allows for more personalized, real-time interventions based on user data. Moreover, healthcare systems are increasingly incorporating digital therapeutics into broader mental health and wellness programs. This trend is leading to a more proactive approach in treating insomnia, with digital health solutions becoming a mainstream part of the treatment landscape, expanding their reach to global markets and addressing diverse patient needs across various demographics.Companies are focusing on securing FDA approvals for their products to boost their customer base. For instance, in August 2024, Big Health secured the USA FDA approval for its flagship digital therapeutic, SleepioRx.
"The FDA's clearance of SleepioRx is a landmark milestone for Big Health and provides accessible, guideline-recommended, first-line treatment for insomnia. This clearance, coupled with the new proposed reimbursement codes, will for the first time motivate USA healthcare providers to prescribe safe and effective treatment alternatives to traditional medication."- Big Health CEO Yael Berman
Summary of the Digital Insomnia (DTx) Market
What is Growth Outlook on Digital Insomnia (DTx) Market as per Future Market Insights Projection?
Future Market Insights projects the digital insomnia therapeutics market to grow at a CAGR of 22% from 2026 to 2036, expanding from USD 420.7 million to USD 3,073.1 million.
FMI research approach: Bottom-up revenue modeling by DTx product type and payer channel, primary interviews with digital health platforms and behavioral health directors, and reimbursement policy analysis across Medicare Advantage and commercial plans.
How do FMI Analysts Perceive Digital Insomnia (DTx) Market to Evolve?
FMI analysts perceive the market transitioning from consumer wellness subscriptions toward prescription-grade DTx integrated into provider workflows and employer benefits, where outcomes data validates reimbursement and pharmaceutical displacement.
FMI research approach: Comparative analysis of FDA-cleared versus non-prescription products, adoption patterns across health systems and payers, and regulatory guidance tracking for software as medical device classification.
Which Country Holds Largest Share in the Global Digital Insomnia (DTx) Market?
The United States holds the largest share of the global digital insomnia DTx market by value, accounting for 52% of 2026 revenue, driven by advanced digital health infrastructure and payer willingness to reimburse evidence-based behavioral interventions.
FMI research approach: Country-level revenue modeling by end-user channel and regulatory status, analysis of prescription DTx coverage policies, and FDA clearance pathway mapping for insomnia indications.
How Large Will the Digital Insomnia (DTx) Market be by 2036?
The global digital insomnia DTx market is estimated to reach USD 3,073.1 million by 2036, with the USA market reaching USD 1,734.2 million.
FMI research approach: Long-term revenue forecast based on chronic insomnia prevalence trends, pharmaceutical substitution rates, and payer coverage expansion timelines for behavioral health DTx.
What is Definition of Digital Insomnia (DTx) Market?
The digital insomnia therapeutics market comprises software-based interventions, predominantly cognitive behavioral therapy for insomnia, delivered through mobile applications and web platforms to treat chronic sleep disorders through structured therapeutic programs.
FMI research approach: Market taxonomy validation across prescription versus consumer products, alignment with FDA software as medical device guidance, and exclusion mapping for sleep tracking devices without therapeutic intervention protocols.
What are Globally Unique Trends Shaping Digital Insomnia (DTx) Market?
Globally unique trends include regulatory pathways for prescription DTx creating medical necessity frameworks, integration with wearable health monitoring devices for continuous sleep data, and employer benefits prioritizing non-pharmaceutical interventions to reduce sedative dependency and next-day impairment.
FMI research approach: Analysis of FDA and EMA digital health guidance evolution, partnership tracking between DTx platforms and device manufacturers, and employer benefit design trends for behavioral health coverage.
What is Driving Demand for Digital Insomnia (DTx)?
Rising awareness of sedative risks is fundamentally reshaping sleep medicine, creating demand for evidence-based alternatives that address insomnia's cognitive and behavioral roots. Regulatory acceptance of prescription-grade software as medical devices has converted experimental apps into FDA-cleared therapeutics eligible for insurance coverage. Clinical validation through randomized controlled trials published in peer-reviewed journals is converting skeptical providers into prescribers, creating sustained demand for DTx platforms with proven efficacy data. The sleep aids market is experiencing disruption as payers recognize DTx's potential to reduce long-term pharmaceutical spending.
Payer economics are accelerating adoption, as chronic insomnia drives medical costs through comorbidity exacerbation and productivity loss. Health plans covering CBT-I DTx report medication cost offsets and reduced emergency utilization, creating financial incentives to expand coverage beyond traditional sleep center referrals. Employers are incorporating DTx into wellness benefits to address workforce productivity, targeting shift workers and high-stress occupations where insomnia prevalence is elevated.
Telehealth expansion during the COVID-19 pandemic normalized remote behavioral health interventions, reducing barriers to digital-only sleep treatment. Patients now expect on-demand access to therapeutic programs without clinic visits, a preference DTx platforms satisfy through asynchronous delivery models. This infrastructure acceptance is positioning digital insomnia therapeutics for mainstream adoption as primary care providers gain comfort prescribing software-based interventions.
How is the Digital Insomnia (DTx) Market Segmented?
The digital insomnia therapeutics market has been segmented based on product type, use case, end-user channel, and region. In terms of product type, the market is divided into CBT-I digital therapeutics (prescription/regulated), CBT-I consumer apps (non-prescription), sleep coaching and tele-psychology bundles, comorbidity modules for anxiety/pain/menopause, and data integrations with outcomes reporting. By use case, the market encompasses chronic insomnia disorder treatment, insomnia with anxiety/depression overlap, insomnia in cardiometabolic patients, shift-work/occupational insomnia, and post-acute/post-COVID sleep support. By end-user channel, the market includes payers/employers through benefit programs, provider-prescribed DTx channel, direct-to-consumer subscriptions, telehealth platforms, and others. Regionally, the market spans North America, Europe, East Asia, South Asia, Latin America, and Middle East and Africa.
Why Do CBT-I Digital Therapeutics Dominate the Market?
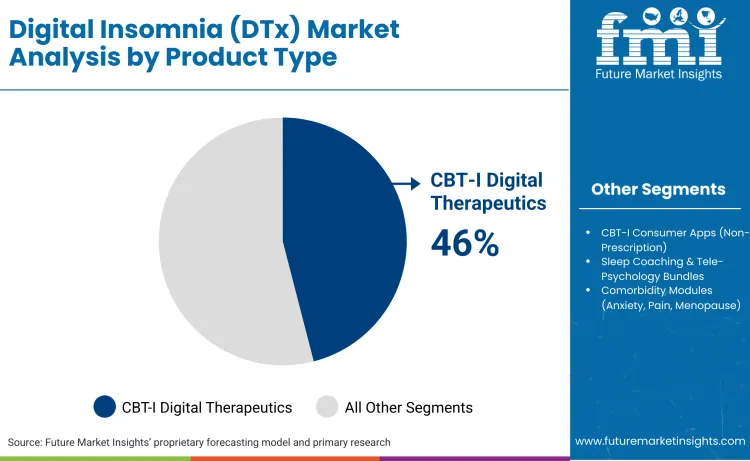
CBT-I digital therapeutics command 46% market share in 2026, driven by prescription status that qualifies products for medical billing and insurance coverage. FDA clearance as software medical devices establishes clinical credibility that consumer wellness apps cannot replicate, enabling provider adoption within evidence-based treatment protocols. Reimbursement through behavioral health benefits is concentrated on prescription DTx, creating revenue stability that supports clinical trial investments and outcomes data collection.
Efficacy data from randomized controlled trials demonstrates that digital CBT-I produces comparable outcomes to in-person therapy, a validation threshold that satisfies payer medical necessity criteria. This evidence base allows prescription DTx to position as pharmaceutical alternatives rather than complementary wellness tools, capturing patients who would otherwise receive benzodiazepines or Z-drugs. Health systems integrate prescription DTx into insomnia care pathways, embedding software into electronic health records as standard treatment options. Companies, such as Big Health offer digital CBT-I through its signature program SleepioRx, an evidence-based digital treatment that delivers structured cognitive behavioral therapy for insomnia through interactive sessions on mobile and web platforms.
How Do Use Cases Influence Digital Insomnia DTx Adoption?
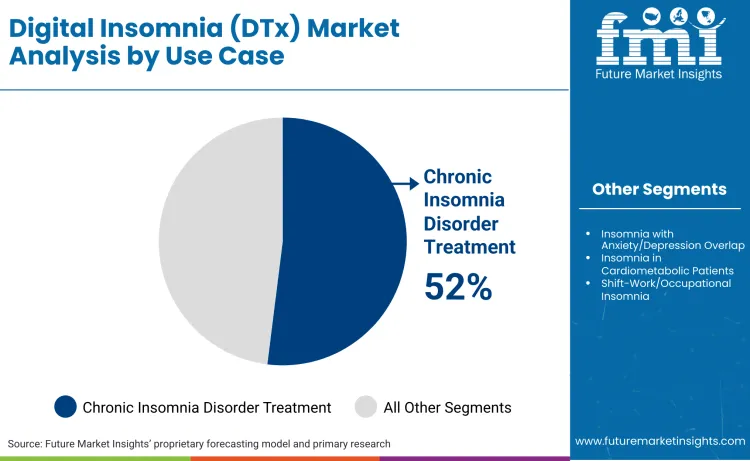
Chronic insomnia disorder treatment leads with 52% share, driven by FDA recognition as a distinct medical condition eligible for therapeutic intervention. As per the Sleep Foundation, 10-15% of people are affected by chronic insomnia. This indication provides the clearest reimbursement pathway and strongest clinical evidence base, concentrating market activity among patients meeting diagnostic criteria for persistent sleep difficulty. Providers prescribe DTx for chronic insomnia with confidence supported by guideline recommendations and outcomes studies.
Insomnia overlapping with anxiety or depression accounts for 18%, creating demand for integrated behavioral health platforms that address multiple conditions simultaneously. These comorbid presentations represent high-cost patients where successful insomnia treatment improves mental health outcomes, a value proposition that resonates with behavioral health carve-out plans. DTx vendors are developing modules that combine CBT-I with anxiety management and mood monitoring to serve this complex population.
How is Technological Evolution Reshaping Digital Insomnia (DTx)?
The digital insomnia therapeutics sector is transitioning from standalone apps toward integrated platforms that combine therapeutic content with wearable sleep data and telehealth coaching. Interoperability with consumer devices allows DTx to ingest objective sleep metrics, personalizing intervention intensity based on actual sleep patterns rather than self-reported assessments. Real-time data integration enables adaptive programs that adjust content difficulty and pacing to individual response patterns.
Machine learning algorithms are optimizing treatment pathways by predicting which patients require additional support beyond automated content delivery. Risk stratification models identify users at risk of dropout, triggering human coaching interventions that improve completion rates and outcomes. These predictive capabilities are converting DTx from static content libraries into responsive therapeutic systems that match intervention intensity to clinical need.
Outcomes reporting infrastructure is maturing, with DTx platforms generating structured data on sleep improvement, medication discontinuation, and quality-of-life changes. Standardized outcome measures allow payers to compare DTx efficacy across vendors and validate return-on-investment claims through medical cost trend analysis. This data infrastructure is essential for converting DTx from experimental benefits into standard formulary options with defined performance expectations.
How is the Digital Insomnia (DTx) Market Evolving Globally?
Global demand for digital insomnia therapeutics is accelerating as regulatory frameworks mature and payer reimbursement models validate digital interventions as medical treatments. Growth demonstrates increasing acceptance of software-based care for behavioral health conditions, with adoption patterns varying by healthcare system structure and pharmaceutical prescribing norms. Service selection emphasizes clinical validation, regulatory status, and integration capabilities across payer and provider workflows.
China records 24.5% CAGR, India records 23% CAGR, USA records 21.5% CAGR, Germany records 20% CAGR, France records 17% CAGR, and UK records 15.5% CAGR. Adoption patterns show regional differences in digital health infrastructure, reimbursement policy maturity, and cultural acceptance of behavioral therapy for sleep disorders.
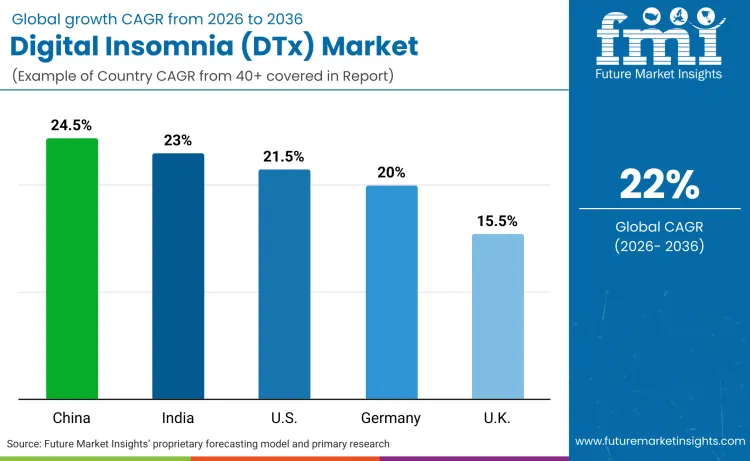
| Country | CAGR (2026-2036) |
|---|---|
| China | 24.5% |
| India | 23.0% |
| USA | 21.5% |
| Germany | 20.0% |
| UK | 15.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
China: How is Digital Health Infrastructure Enabling DTx Adoption?
China's digital insomnia therapeutics market is experiencing rapid expansion driven by widespread smartphone penetration and government initiatives supporting digital health innovation. Domestic platforms are developing CBT-I programs adapted for Chinese language and cultural contexts, addressing sleep disorders in populations facing high work stress and urbanization-related lifestyle factors. The Chinese digital insomnia DTx market is set to grow at 24.5% CAGR during the study period.
Regulatory pathways for digital therapeutics are evolving, with authorities establishing frameworks for software medical device classification and clinical validation requirements. Major tech companies are entering behavioral health markets, leveraging existing user bases to distribute sleep intervention programs at scale. Integration with national health data initiatives is creating opportunities for population-level sleep health monitoring and targeted intervention delivery.
India: Is Mobile-First Healthcare Accelerating DTx Access?
India's market growth shows expanding smartphone adoption and increasing recognition of sleep disorders in urban populations facing extended work hours and lifestyle stress. Telehealth platforms are incorporating sleep therapy modules into behavioral health offerings, providing access to structured interventions without requiring specialist sleep clinic referrals. The Indian digital insomnia DTx market is set to grow at 23% CAGR during the study period.
Affordability considerations are driving development of lower-cost subscription models and freemium offerings that provide basic sleep education with paid tiers for therapeutic programs. English-language content accessibility combined with regional language adaptations is expanding addressable markets across diverse linguistic regions. Insurance coverage for digital therapeutics remains limited, concentrating growth in self-pay and employer-sponsored wellness programs.
USA: How is Regulatory Clarity Establishing Market Leadership?
The USA market maintains leadership through FDA clearance pathways that establish prescription DTx as medical devices eligible for insurance coverage. Health plans are adding CBT-I digital therapeutics to behavioral health formularies, driven by evidence of pharmaceutical cost offsets and improved outcomes compared to sedative monotherapy. The USA digital insomnia DTx market is set to grow at 21.5% CAGR during the study period.
Employer adoption is accelerating as benefits consultants position DTx as solutions for productivity loss and healthcare cost containment. Large self-insured employers are conducting internal outcomes analyses demonstrating return on investment through reduced absenteeism and disability claims. Provider integration into electronic health records enables prescription workflows comparable to pharmaceutical ordering, reducing friction in clinical adoption.
Germany: How is DiGA Framework Enabling Reimbursement?
Germany's market growth is supported by the Digital Health Applications (DiGA) framework that provides statutory health insurance reimbursement for approved digital therapeutics. This regulatory pathway establishes clear requirements for clinical evidence and data security, creating a defined route to market for insomnia DTx developers. The German digital insomnia DTx market is set to grow at 20% CAGR during the study period.
Physicians can prescribe DiGA-listed sleep therapy apps with reimbursement guaranteed for 12-month trial periods, eliminating cost barriers for patients. Evidence generation requirements are driving clinical trial activity, with vendors conducting German-population studies to satisfy regulatory standards. Insurance coverage is converting DTx from experimental to mainstream options, positioned alongside pharmaceutical and psychological therapies in treatment guidelines.
UK: How is NHS Adoption Validating DTx Efficacy?
The UK market growth is influenced by National Health Service evaluation and selective adoption of evidence-based digital therapeutics for insomnia management. NHS Digital has assessed multiple CBT-I platforms, with positive recommendations driving uptake across clinical commissioning groups. The UK digital insomnia DTx market is set to grow at 15.5% CAGR during the study period.
Big Health's Sleepio has achieved significant NHS deployment following clinical trials in UK populations, establishing a proof-of-concept for large-scale public health system adoption. General practitioners are increasingly comfortable referring patients to digital-first insomnia treatment, particularly for cases where sedative prescribing is clinically inappropriate. Formulary inclusion by NHS trusts provides volume scale that validates business models beyond direct-to-consumer channels.
Who are the Major Players Active in the Digital Insomnia (DTx) Market?
The competitive landscape is experiencing consolidation pressure as prescription DTx platforms with FDA clearance differentiate from consumer wellness apps, while health system acquisitions absorb smaller vendors into integrated care platforms. Major players are investing in clinical evidence generation, regulatory submissions, and payer contracting to establish formulary positioning. These companies are developing comprehensive data infrastructures that demonstrate outcomes to justify premium pricing and insurance coverage.
ResMed Inc. and Livongo Health, Inc. represent incumbents in the sleep and chronic condition management space, with ResMed’s expertise in sleep apnea and connected devices complementing its growing digital therapy offerings and Livongo’s data driven chronic care models supporting sleep related care. Omada Health, Inc. focuses on preventive health and behavior change, integrating sleep improvement into its portfolio of digital health programs that address lifestyle factors. Click Therapeutics, Inc. develops software based therapeutic programs that target sleep and other conditions with clinical validation frameworks. Akili Interactive Labs, Inc. brings a unique orientation with its focus on digital therapeutics that merge therapeutic intent with engaging interactive experiences, exploring how cognitive training can support overall wellness including sleep.
Key Industry Developments:
- In April 2025, Teladoc Health acquired UpLift, an innovative and tech-enabled provider of virtual mental health therapy, psychiatry and medication management services to broaden its portfolio.
- In February 2025, Teladoc Health acquired Catapult Health, a leading provider of virtual preventive care services to boost its customer base.
Key Players in the Digital Insomnia (DTx) Market
- Big Health, Inc.
- Teladoc Health, Inc.
- Pear Therapeutics, Inc.
- Happify Health, Inc.
- ResMed Inc.
- Livongo Health, Inc.
- Omada Health, Inc.
- Click Therapeutics, Inc.
- Akili Interactive Labs, Inc.
- Propeller Health, Inc.
Market Definition
The digital insomnia therapeutics market represents revenue generated from software-based interventions that deliver structured therapeutic programs to treat chronic sleep disorders, predominantly through cognitive behavioral therapy for insomnia protocols. As operationally defined in the analysis, the market measures commercially deployed digital therapeutics sold to consumers, employers, health plans, and healthcare providers, analyzed by product type, use case, end-user channel, and region, and expressed in USD million.
Market Inclusion
Included in the digital insomnia therapeutics market are products explicitly segmented by regulatory status and delivery model: CBT-I digital therapeutics with prescription or FDA clearance, CBT-I consumer apps available without prescription, sleep coaching programs bundled with tele-psychology support, comorbidity modules addressing anxiety/pain/menopause-related insomnia, and data integration platforms for outcomes reporting. Use case coverage spans chronic insomnia disorder treatment, insomnia with anxiety/depression overlap, insomnia in cardiometabolic patients, shift-work and occupational insomnia, and post-acute sleep support including post-COVID populations.
Market Exclusion
Excluded from the market scope are sleep tracking devices and wearables that monitor sleep patterns without providing therapeutic intervention programs. Consumer sleep aids including supplements, aromatherapy products, and bedroom environment devices are not included. Pharmaceutical products for insomnia treatment including benzodiazepines, Z-drugs, and over-the-counter medications are outside market scope. In-person cognitive behavioral therapy delivered through traditional psychology or psychiatry practices is excluded, as are sleep center diagnostic services and continuous positive airway pressure devices for sleep apnea.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product Type | CBT-I Digital Therapeutics (Prescription/Regulated); CBT-I Consumer Apps (Non-Prescription); Sleep Coaching & Tele-Psychology Bundles; Comorbidity Modules (Anxiety, Pain, Menopause); Data Integrations & Outcomes Reporting |
| Use Case | Chronic Insomnia Disorder Treatment; Insomnia with Anxiety/Depression Overlap; Insomnia in Cardiometabolic patients; Shift-Work/Occupational Insomnia; Post-Acute/Post-COVID Sleep Support |
| End User Channel | Payers/Employers (Benefit Programs); Provider-Prescribed DTx Channel; Direct-to-Consumer Subscriptions; Telehealth Platforms; Others |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, United Kingdom, China, India, and 40+ countries |
| Key Companies Profiled | Big Health Inc.; Teladoc Health Inc.; Pear Therapeutics Inc.; ResMed Inc.; Omada Health Inc.; Others |
| Additional Attributes | Revenue analysis by product type, use case, and end-user channel; performance across prescription DTx, consumer apps, and integrated platforms; therapeutic efficacy validation, clinical outcomes measurement, and regulatory compliance across healthcare institutions, payer organizations, and direct-to-consumer channels; impact on pharmaceutical cost reduction, sleep quality improvement, and patient adherence during treatment protocols; compatibility with electronic health records and provider workflows; adoption dynamics driven by reimbursement policies, clinical evidence requirements, and digital health integration initiatives. |
Digital Insomnia (DTx) Market by Segments
Product Type:
- CBT-I Digital Therapeutics (Prescription/Regulated)
- CBT-I Consumer Apps (Non-Prescription)
- Sleep Coaching & Tele-Psychology Bundles
- Comorbidity Modules (Anxiety, Pain, Menopause)
- Data Integrations & Outcomes Reporting
Use Case:
- Chronic Insomnia Disorder Treatment
- Insomnia with Anxiety/Depression Overlap
- Insomnia in Cardiometabolic Patients
- Shift-Work/Occupational Insomnia
- Post-Acute/Post-COVID Sleep Support
End User Channel:
- Payers/Employers (Benefit Programs)
- Provider-Prescribed DTx Channel
- Direct-to-Consumer Subscriptions
- Telehealth Platforms
- Others
Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- National Institutes of Health. (2024). Cognitive behavioral therapy for insomnia (CBT-I): Clinical effectiveness and implementation in sleep disorders care. National Institute of Neurological Disorders and Stroke.
- World Health Organization. (2023). WHO guideline on digital health interventions for health system strengthening. World Health Organization.
- USA Food and Drug Administration. (2023). Digital health policy navigation tools. USA Department of Health and Human Services.
Frequently Asked Questions
What is the current global market size for digital insomnia therapeutics?
The global industry is valued at USD 420.7 million in 2026, driven by regulatory acceptance of prescription-grade software as medical devices.
What is the projected Compound Annual Growth Rate (CAGR) for the digital insomnia DTx market over the next 10 years?
Revenue is projected to grow at 22% CAGR from 2026 to 2036, reaching USD 3,073.1 million.
Which product types are the primary drivers of demand in the digital insomnia DTx market?
CBT-I digital therapeutics with prescription status lead with 46% share in 2026, followed by consumer apps at 22%.
What are the primary market risks or regulatory hurdles n the digital insomnia DTx market?
Reimbursement policy variation across payers creates revenue uncertainty, while clinical evidence requirements for FDA clearance impose substantial development costs.
Who are the leading companies in the digital insomnia DTx market?
ResMed Inc., Big Health Inc. are some of the leading companies.
Table of Content
- Digital Insomnia (DTx) Market Size and Share Forecast Outlook (2026 to 2036)
- Digital Insomnia (DTx) Market Forecast and Outlook (2026-2036)
- Digital Insomnia (DTx) Market Key Takeaways
- Key Shifts in the Digital Insomnia (DTx) Market from 2026 to 2036
- Digital Insomnia (DTx) Market Segmentation Analysis
- By Product Type
- CBT-I Digital Therapeutics (Prescription/Regulated)
- CBT-I Consumer Apps (Non-Prescription)
- Sleep Coaching & Tele-Psychology Bundles
- Comorbidity Modules (Anxiety, Pain, Menopause)
- Data Integrations & Outcomes Reporting
- By Use Case
- Chronic Insomnia Disorder Treatment
- Insomnia with Anxiety/Depression Overlap
- Insomnia in Cardiometabolic Patients
- Shift-Work/Occupational Insomnia
- Post-Acute/Post-COVID Sleep Support
- By End User Channel Channel
- Payers/Employers (Benefit Programs)
- Provider-Prescribed DTx Channel
- Direct-to-Consumer Subscriptions
- Telehealth Platforms
- Others
- By Region
- By Product Type
- Market Dynamics and Growth Analysis
- Growth Drivers
- Market Restraints
- Strategic Trends
- Digital Insomnia (DTx) Market Global Analysis
- East Asia
- South Asia
- North America
- Europe
- Latin America
- Middle East & Africa
- Competitive Landscape Analysis
- Key Players in the Digital Insomnia (DTx) Market
- Market Definition, Inclusion, and Exclusion Criteria
- Scope of the Digital Insomnia (DTx) Market Report
- Digital Insomnia (DTx) Market by Segment
- Bibliography
- Frequently Asked Questions (FAQs)
List of Tables
- Global Digital Insomnia (DTx) Market Size (USD Million), 2021-2036
- Digital Insomnia (DTx) Market Y-o-Y Growth (%), 2021-2036
- Global Market Share by Product Type, 2026
- Global Market Share by Use Case, 2026
- Global Market Share by End User Channel, 2026
- Digital Insomnia (DTx) Market Share by Region, 2026
- Market Share by Product Type (USD Million), 2026-2036
- Market Share by End User Channel (USD Million), 2026-2036
- Key Player Market Share in the Digital Insomnia (DTx) Market
- Digital Insomnia (DTx) Market Absolute $ Opportunity (USD Million), 2026-2036
- Digital Insomnia (DTx) Market CAGR (%) by Region, 2026-2036
List of Figures
- Global Digital Insomnia (DTx) Market Size (USD Million), 2021-2036
- Digital Insomnia (DTx) Market Y-o-Y Growth (%) from 2021-2036
- Digital Insomnia (DTx) Market Share by Product Type (%) in 2026
- Digital Insomnia (DTx) Market Share by Use Case (%) in 2026
- Global Digital Insomnia (DTx) Market by Region (%) 2026-2036
- Global Digital Insomnia (DTx) Market by End User Channel
- Competitive Landscape of Key Digital Insomnia (DTx) Market Players
- Market Trends in Digital Insomnia (DTx)
- Digital Insomnia (DTx) Adoption by Region

