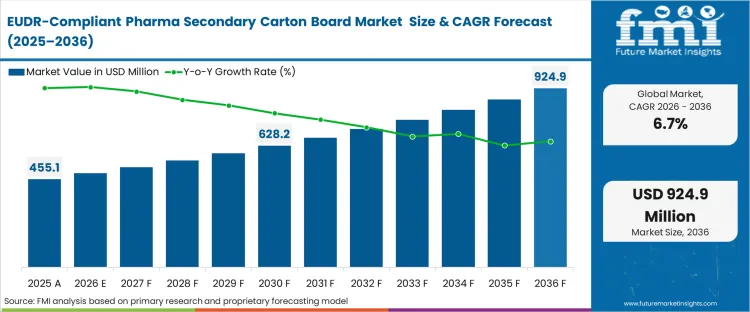
The EUDR-compliant pharma secondary carton board market crossed a valuation of USD 450.5 million in 2025. The sector is expected to reach USD 485.4 million in 2026 at a CAGR of 6.66% during the forecast period. Sustained investment propels the valuation to USD 925.3 million through 2036 as the non-optional nature of deforestation compliance for pharmaceutical products entering European borders creates a long-term lock-in for verified board suppliers.
Procurement directors in the life sciences sector are moving away from broad certification logos toward granular geolocation data verification. This shift forces a decision between maintaining diverse, opaque fiber sources or consolidating spend with a handful of vertically integrated mills capable of providing plot-level coordinates. The stakes for delay are severe, as non-compliant secondary packaging can result in entire drug batches being impounded at EU points of entry. While the industry views this as an environmental mandate, it is functionally a digital audit challenge where the carton board solutions are only as valuable as the metadata accompanying them.
Market acceleration depends on the structural stabilization of satellite-based remote sensing as a primary proof of compliance. With EU regulatory bodies having operationalized the TRACES-NT Information System for geolocation data, the manual burden of verifying thousands of smallholder fiber plots will diminish. This transition is being triggered by a coalition of tier-1 packaging converters and software providers who are building the "digital twin" of forest origins, making the next unit of compliant board significantly easier to qualify than the first.
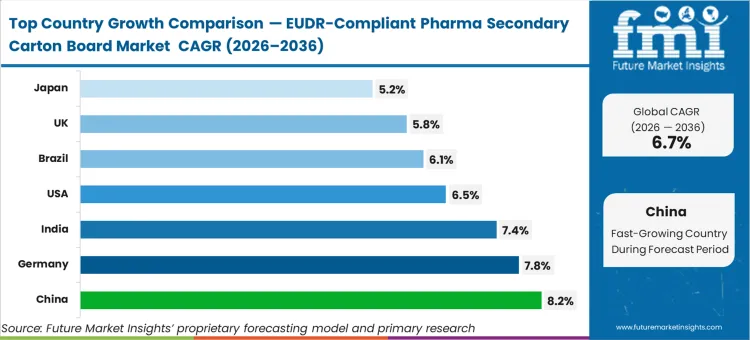
China is anticipated to record a CAGR of 8.2% as its massive contract manufacturing base aligns with European export requirements. Germany is expected to post 7.8% growth driven by its role as the primary logistics gateway for the European Union's pharmaceutical supply chain. India follows with a projected 7.4% increase, reflecting the modernization of its pharmaceutical export hubs. The USA sector is poised to expand at 6.5%, while Brazil is set to track a 6.1% growth rate. The United Kingdom is likely to follow at 5.8%, and Japan is estimated to garner a 5.2% CAGR through 2036. The divergence across these regions stems from the varying maturity of national forest tracking infrastructures rather than pharmaceutical demand alone.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 485.4 million |
| Industry Value (2036) | USD 925.3 million |
| CAGR (2026-2036) | 6.66% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The EUDR-Compliant Pharma Secondary Carton Board Market comprises specialized paperboard materials used for drug outer packaging that meet the strict requirements of the EU Deforestation Regulation. This involves a functional boundary where every square inch of board must be traceable to a specific, non-deforested plot of land through a verified chain of custody. Unlike standard carton board, this market is defined by the integration of geolocation data and legal due diligence into the material's value proposition.
Included within this scope are all virgin and recycled fiber board grades utilized for folding cartons, display packs, and wallet cards in pharmaceutical applications. This encompasses solid bleached sulfate (SBS), folding boxboard (FBB), and coated recycled board (CRB) that specifically carry EUDR compliance documentation. The scope also covers pharma packaging formats like tamper-evident cartons and multi-pack outers that act as the secondary layer of protection for primary medical containers.
Explicitly excluded from this market are primary packaging materials such as glass vials, plastic bottles, and aluminum blister foils, as these do not utilize wood-fiber board substrates. Furthermore, general-purpose carton boards lacking the specific serialization-grade surface finish required for pharmaceutical Datamatrix coding are excluded. These are omitted because they do not face the same stringent qualification and regulatory validation cycles as medicinal secondary packaging, even if they claim basic deforestation compliance.
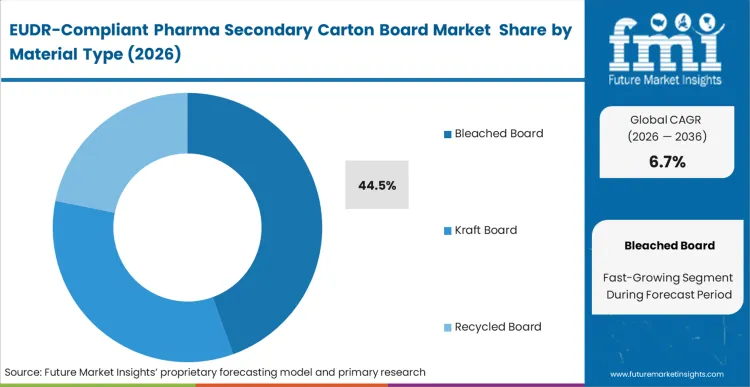
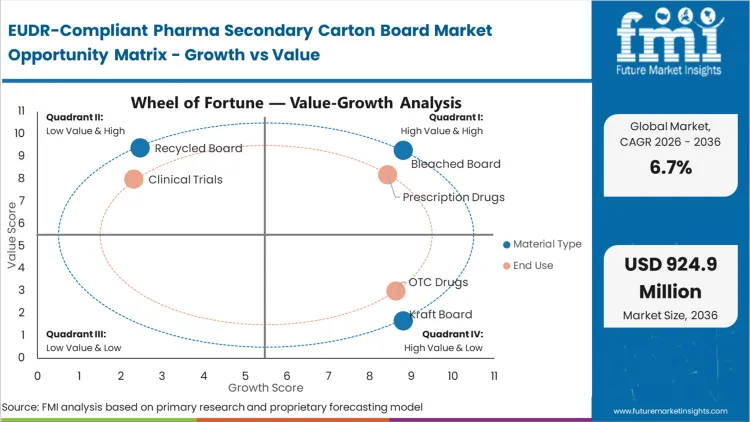
The reason bleached board holds a dominant 44.5% share comes down to the operational requirement for absolute serialization legibility. Pharmaceutical converters cannot risk the inconsistent surface brightness or high "noise" of lower-grade recycled boards when high-speed cameras must verify 2D Datamatrix codes at 400 cartons per minute. This material is not chosen merely for its strength; it is the structural substrate for the drug's digital identity. According to FMI's assessment, the transition to bleached board types is accelerating because virgin fiber offers a simpler path to EUDR compliance than the complex, mixed-origin footprint of recycled pulp. Buyers who delay this transition find themselves unable to meet the traceability standards required by tier-1 European distributors, who are increasingly refusing shipments that lack plot-level documentation.
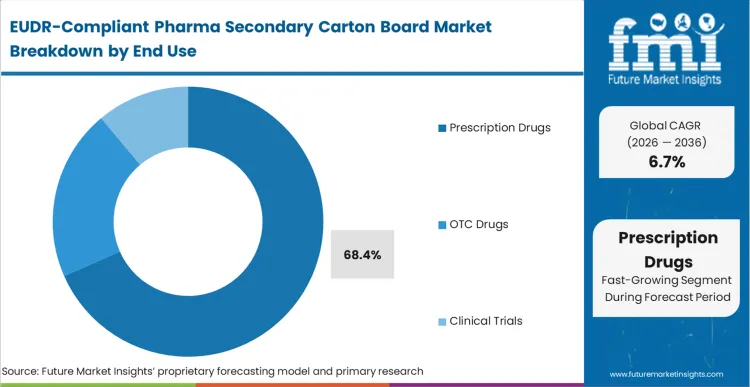
The incumbent approach of using generic, multi-origin carton board for prescription medication is failing as European regulators move toward zero-tolerance for "blind" fiber sourcing. Prescription drugs account for the majority of the market with 68.4% share because the financial stakes of a shipment rejection are highest in this segment. A single impounded batch of a biologic medication can represent millions in lost revenue, making the unit cost of compliant board a negligible insurance premium. In FMI's view, drug packaging needs are evolving from simple containment to regulatory gatekeeping. Pharmaceutical firms are now prioritizing suppliers who can offer "traceability as a service" alongside the physical material. Those who continue to source board through fragmented, unverified channels risk immediate exclusion from the major European hospital and pharmacy procurement networks that now mandate EUDR documentation.
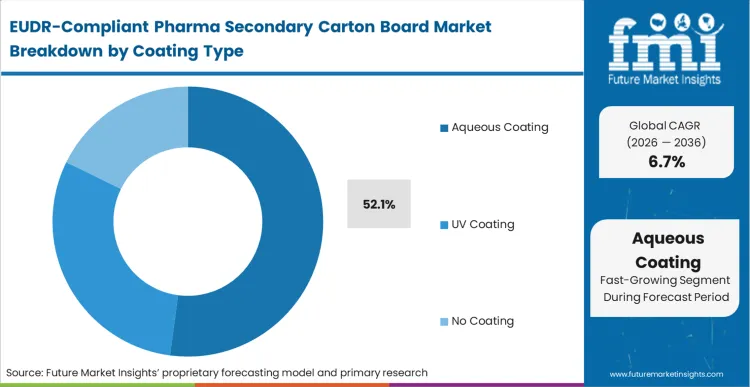
The commercial consequence of choosing the wrong coating in an EUDR-compliant environment is often the total loss of the board's recyclability profile. Aqueous coating holds 52.1% of the market because it protects the board's surface without interfering with the repulpability that European circular economy mandates demand. As per FMI's projection, coated board applications are shifting toward water-based systems because they avoid the chemical complexity of UV-cured alternatives, which can complicate the "virgin fiber" status of future recycled streams. Buyers who select non-compliant or hard-to-recycle coatings face escalating "eco-modulated" fees in several European jurisdictions, which can double the effective cost of the packaging over the product lifecycle. This forces a move toward aqueous solutions that satisfy both the physical protection of the drug carton and the environmental compliance profile required for the European market.
The structural forcing condition driving this market is the non-negotiable legal deadline for EUDR compliance, which requires every pharmaceutical company exporting to the Union to provide geolocation data for their packaging. This is not a voluntary ESG goal; it is a market-access requirement that forces procurement directors to choose between compliant, traceable fiber or total exclusion from European distribution. The stakes are particularly high for global brands whose centralized manufacturing hubs serve both European and non-European markets, as they must decide whether to dual-track their inventory or move the entire global supply to compliant board.
The primary restraint is the structural lack of digital integration between forest managers and pharmaceutical converters. Most global board mills still operate on paper-based or siloed chain-of-custody systems that cannot easily output the CSV or blockchain-ready coordinates required for an EUDR "Due Diligence Statement." This friction is organizational rather than technical; the data often exists but is trapped in a multi-tier supply chain where transparency is traditionally resisted. While satellite monitoring is emerging as a partial solution, the cost of verifying smallholder plots in complex sourcing regions like Brazil or Southeast Asia remains a significant barrier to total market coverage.
Opportunities in the EUDR-Compliant Pharma Secondary Carton Board Market
Based on the regional analysis, the EUDR-Compliant Pharma Secondary Carton Board Market is segmented into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.2% |
| Germany | 7.8% |
| India | 7.4% |
| USA | 6.5% |
| Brazil | 6.1% |
| United Kingdom | 5.8% |
| Japan | 5.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
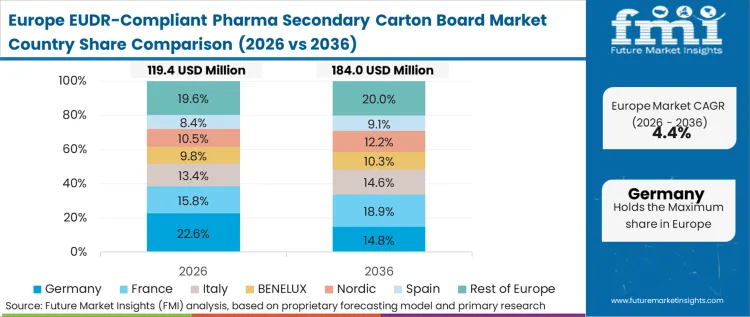
Europe's adoption pattern is driven by its role as the regulatory epicenter where compliance is the price of admission. The policy-led environment here makes the transition a legal imperative rather than a market choice, with the European Medicines Agency (EMA) standards increasingly referencing packaging sustainability. Based on FMI's assessment, pharmaceutical firms in this region are prioritizing the stabilization of their supply chains against the 2026 deadline, leading to a surge in long-term contracts for verified recycled board demand where traceability can be proven. This region serves as the global testbed for geolocation verification technologies.
The structural lens for Asia Pacific is infrastructure-led, specifically the modernization of forest-tracking systems in major export hubs. According to FMI's estimates, the region is responding to the procurement specification updates that Western brand customers push down through each annual sourcing review. This region is the primary manufacturing engine for global generics, and its board mills are under intense pressure to digitize their sourcing records to maintain access to the European market. The growth here reflects a rate of compliance-led capital refresh that tracks the brand sourcing review calendar rather than a domestic regulatory deadline.
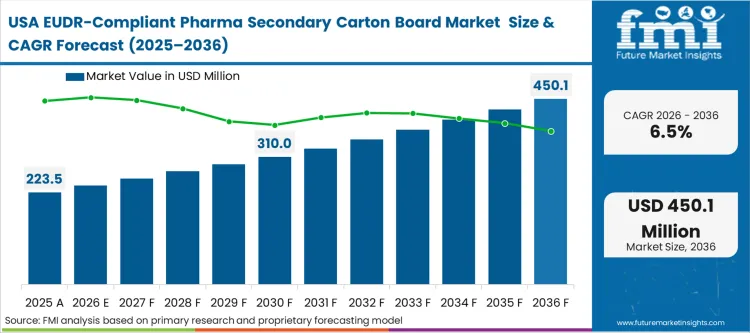
North America's market dynamic is buyer behavior-led, with global pharmaceutical giants headquartered in the US applying European standards to their global operations to avoid dual-stocking costs. FMI analysts opine that US-based procurement directors are the primary drivers of this market, as they force their domestic board suppliers to adopt EUDR-like traceability even for products sold outside of Europe. This ensures a unified global packaging specification that simplifies logistics and quality control across their international manufacturing sites.
FMI's report includes detailed analysis of additional markets in Southeast Asia, the Middle East, and Scandinavia. These regions are forming "traceability clusters" where shared infrastructure for forest monitoring is lowering the entry barrier for smaller board converters.
The concentrated-moderate structure of this market is a direct result of the high capital requirement for board mills and the sophisticated IT systems needed to manage geolocation metadata. Buyers distinguish between qualified and unqualified vendors not by the physical properties of the board, which are largely standardized, but by the robustness of their data verification pipelines. Companies like Stora Enso Oyj and Metsä Board Corporation have an inherent advantage because they control large portions of their fiber supply, allowing them to provide plot-level coordinates with higher reliability than a converter who buys pulp from fragmented sources.
Incumbents in the paperboard packaging leaders space are building structural advantages by integrating satellite monitoring directly into their customer portals. A challenger must build more than just a compliant board; they must develop a digital verification capability that can survive a rigorous pharmaceutical audit. Leading players like Mondi plc and WestRock are leveraging their global footprints to offer a "one-stop-shop" for compliant board, ensuring that a pharmaceutical client can use the same verified grade whether they are packaging in Ireland or the United States.
Buyer power is exerted through long-term sourcing agreements that penalize vendors for data gaps as heavily as for material defects. There is a structural tension between the buyers' need for total transparency and the board mills' desire to protect their proprietary sourcing networks. As we move toward 2036, the market is becoming more concentrated as smaller mills that cannot afford the "traceability tax" are either acquired or relegated to the less regulated industrial packaging segments. The competitive trajectory is defined by a race to automate compliance, where the winners are those who make the " Due Diligence Statement" an invisible part of the invoice.

| Metric | Value |
|---|---|
| Quantitative Units | USD 485.4 million to USD 925.3 million, at a CAGR of 6.64% |
| Market Definition | Secondary packaging board for pharmaceutical use that meets the mandatory geolocation and no-deforestation requirements of Regulation (EU) 2023/1115. |
| Material Type Segmentation | Bleached Board, Kraft Board, Recycled Board |
| End Use Segmentation | Prescription Drugs, OTC Drugs, Clinical Trials |
| Coating Type Segmentation | Aqueous Coating, UV Coating, No Coating |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | China, Germany, India, USA, Brazil, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Stora Enso Oyj, Metsä Board Corporation, Mondi plc, WestRock Company, Graphic Packaging Holding Company, Sappi Limited, Mayr-Melnhof Karton AG |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with pharmaceutical procurement leads and board mill sustainability officers, anchored to EUDR trade flow data and validated through satellite monitoring adoption rates. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the EUDR-Compliant Pharma Secondary Carton Board Market in 2025?
The market was valued at USD 450.5 million in 2025. This figure signals that the pharmaceutical industry has already begun the transition to compliant board ahead of the hard regulatory deadlines, effectively pricing in the cost of traceability.
What will it be valued at by 2036?
The market is projected to reach USD 925.3 million by 2036. This growth reflects the total absorption of EUDR compliance into the global pharmaceutical supply chain, where non-compliant board becomes structurally obsolete for high-value medicinal exports.
What CAGR is projected for the forecast period?
A CAGR of 6.64% is expected between 2026 and 2036. This rate is constrained by the multi-year qualification cycles of pharmaceutical packaging, which prevents overnight shifts but ensures that once a mill is qualified, it remains locked into the supply chain for the product lifecycle.
Which Material Type segment leads the market?
Bleached Board leads with a 44.5% share in 2026. This dominance is driven by the operational necessity of high contrast for serialization codes and the simpler audit trail afforded by virgin fiber compared to the fragmented origin of recycled materials.
Which End Use segment leads the market?
Prescription Drugs is the dominant end-use segment. This is because the regulatory and financial consequences of batch impoundment at EU borders are most severe for biologics and specialty medicines, making compliant board a non-negotiable procurement requirement.
Which Coating Type leads the market?
Aqueous Coating leads with a 52.1% share. The segment leads because water-based systems provide the necessary protection for pharma cartons while maintaining the easy repulpability required by European circular economy mandates.
What drives the rapid growth in this market?
The primary driver is the mandatory legal requirement of the EU Deforestation Regulation. This forces a structural shift where market access is contingent on providing geolocation data, moving compliance from a "nice to have" ESG goal to a core operational necessity for pharmaceutical exporters.
What is the primary restraint on market growth?
The primary restraint is the digital gap between forest data and pharmaceutical audit requirements. The structural friction of verifying thousands of fiber plots in complex, multi-tier supply chains creates an organizational bottleneck that slows the transition for smaller converters.
Which country is growing the fastest?
China is the fastest-growing market at an 8.2% CAGR. Structurally, Chinese board mills are modernizing faster than their domestic peers to protect their massive contract manufacturing exports to the European pharmaceutical market.
How does EUDR differ from previous certifications like FSC?
While FSC provides a broad "chain of custody" based on mass balance, EUDR requires specific geolocation coordinates for the exact plot of land where the fiber was harvested. This represents a shift from trust-based certification to data-verified evidence of no-deforestation.
Can recycled board ever be truly EUDR compliant?
Recycled board can be compliant if it consists of "pre-2020" material or if the post-consumer stream can be verified as deforestation-free. Structurally, this is much harder to prove than for virgin fiber, which is why the market is currently pivoting toward bleached virgin boards.
What happens if a drug carton is found to be non-compliant?
Under EUDR, non-compliant products can be seized at the border, and the importing company can face fines up to 4% of their annual EU turnover. This creates a zero-tolerance environment for pharmaceutical firms that forces them to use only verified, high-tier board suppliers.
How is China's forest tracking infrastructure evolving?
China is investing heavily in state-backed satellite monitoring and centralized timber tracking databases. This infrastructure-led approach allows Chinese mills to provide the "digital twin" of their board origins more efficiently than regions with fragmented private forest ownership.
Why is Germany growing faster than the UK in this market?
Germany's growth is driven by its role as the primary physical gateway and warehousing hub for the EU pharmaceutical sector. The operational outcome of having compliant board at the point of manufacture in Germany is a total reduction in customs delays across the entire Union.
What is the structural advantage of vertically integrated mills?
Mills that own their forests have a shorter, more transparent data chain. They can provide coordinates with absolute certainty, whereas a non-integrated mill must aggregate data from hundreds of external suppliers, increasing the risk of an audit failure.
How does aqueous coating improve procurement efficiency?
Aqueous coatings allow for faster line speeds on folding carton machines and do not require specialized UV waste management. This clears a bottleneck in the secondary packaging process while ensuring the final product meets EU recyclability guidelines.
What is the "traceability tax" in the board market?
The "traceability tax" refers to the overhead cost of maintaining the IT systems and audit trails required for EUDR. This cost favors large-scale producers, as they can amortize the digital investment over millions of tons, whereas smaller mills struggle to remain competitive.
Will the US adopt EUDR-like standards?
While not legally mandated in the US, major American pharmaceutical firms are adopting these standards for their global operations to maintain a single packaging specification. The structural trajectory is toward a global unified grade for medicinal cartons.
How do satellite verification platforms work?
These platforms use automated remote sensing to detect land-use changes in near real-time. By cross-referencing harvest timestamps with satellite imagery, they provide the proof of "no-deforestation" required by the European Commission.
What is the practitioner reality for sourcing board in Brazil?
The reality is that Brazil is considered a high-risk region, requiring the most intensive "Due Diligence Statements." This forces pharmaceutical firms to establish exclusive, closed-loop supply corridors with a few verified large-scale plantations to avoid any contamination from unverified land.
How does this market affect drug pricing?
The unit cost of the board is a minor component of a drug's price, but the cost of compliance is significant. Most firms are absorbing this cost as a "market access insurance" rather than passing it directly to the consumer, especially in price-regulated European markets.
What will be the dominant competitive variable in 2036?
By 2036, the competitive variable will be the level of "traceability automation." The winners will be mills that have fully integrated their forest data with their clients' ERP systems, making compliance a real-time, zero-touch process.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.