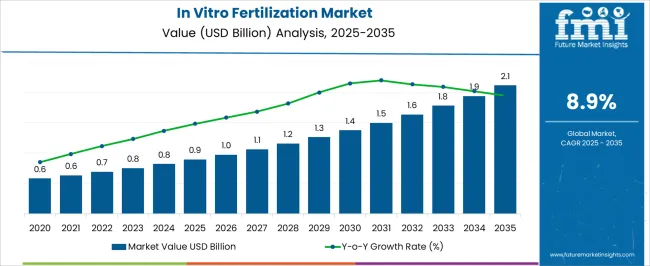
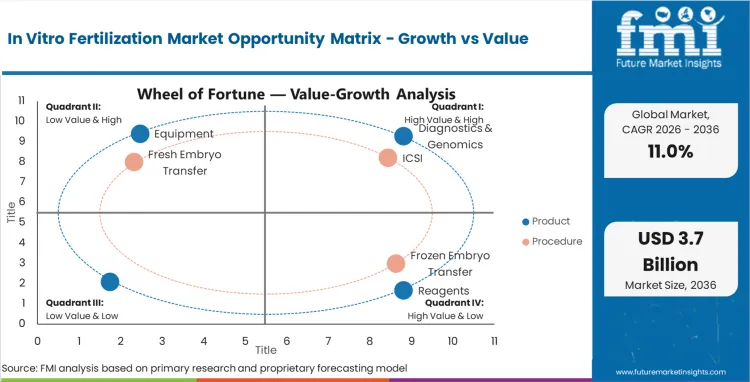
The in vitro fertilization (IVF) market was valued at USD 1.17 billion in 2025, and projections show it will increase to USD 1.30 billion in 2026 before reaching USD 3.80 billion by 2036. This growth, reflected in a sustained CAGR of 11.0%, is driven by a notable shift in reproductive healthcare dynamics. As per FMI's projection, the demand is stabilizing around high-fidelity procedures like ICSI which now command dominant volume shares due to higher success rates.
Compliance gate and retrofit capex dynamics are reshaping the industry's cost structure. As clinics invest in advanced incubators and AI-powered selection tools to meet stricter success-rate benchmarks. In June 2025, CooperSurgical invested USD 15 million to upgrade its genomic testing facilities, signaling a market-wide pivot toward premium diagnostic integration to justify higher cycle costs [2]. This capital intensity forces smaller clinics to consolidate or upgrade, effectively setting a new floor for operational viability.
"The demand for precision in assisted reproductive technology is forcing a transition from volume-based care to outcome-based pricing models," notes the Chief Medical Officer of Vitrolife in a 2025 industry address [3]. Suppliers should invest in on-site service hubs to support this transition, as clinics increasingly demand 24/7 distinct technical support for complex embryo culture systems.
Global demand is distributed across mature and emerging hubs, with India and China leading growth velocity. The market in India is projected to expand at 11.3% CAGR, while China follows closely at 10.5% CAGR, and North American markets like the USA (11.0% CAGR) and Canada (10.7% CAGR) sustain momentum through reimbursement reforms. FMI analysts opine that the harmonization of cross-border reproductive travel will further balance regional utilization rates.
The In Vitro Fertilization (IVF) market comprises the global ecosystem of assisted reproductive technologies, including the instruments, consumables, and reagents used to facilitate fertilization outside the body. It encompasses the complete procedural workflow from oocyte retrieval to embryo transfer, serving patients facing infertility due to genetic, age-related, or physiological factors.
The report includes a comprehensive analysis of the core technologies enabling IVF, specifically focusing on micromanipulation equipment, culture media, and cryopreservation systems. It covers revenue streams generated by fertility clinics, surgical centers, and clinical research institutes that procure these specialized medical devices and consumables.
The scope excludes general gynecological devices not specific to the IVF workflow, such as standard ultrasound machines used for routine obstetric monitoring unless integrated with follicle tracking software. It also omits the pharmaceutical sales of hormonal stimulation drugs, focusing strictly on the procedural hardware, reagents, and laboratory infrastructure.
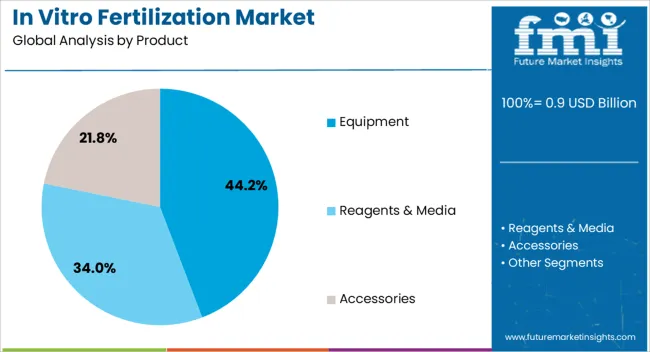
Consumables and reagents represent the recurring revenue backbone of the industry, driven by the high frequency of single-use plastics and culture media required per cycle. Reagents and media specifically command a premium, as maintaining optimal pH and osmolarity is non-negotiable for embryo survival, insulating this sub-segment from broader economic downturns.
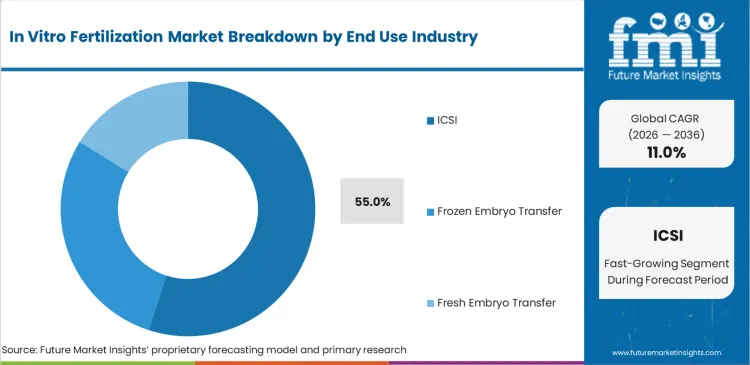
Based on FMI’s estimates, Intracytoplasmic Sperm Injection (ICSI) holds 55.0% of the procedural market share in 2026. This dominance stems from its superior fertilization rates in cases of male factor infertility, which is rising globally, making ICSI the default standard of care in many advanced clinics.
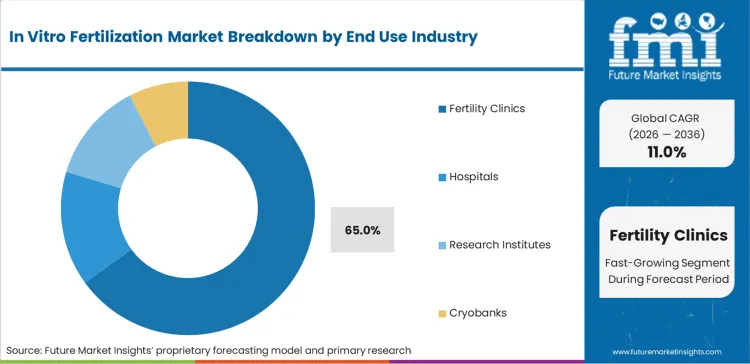
Fertility clinics command 65.0% of the market volume in 2026, serving as the primary point of care for patients seeking specialized reproductive solutions. While hospitals maintain a presence, the specialized infrastructure and focused expertise of standalone fertility centers allow for higher throughput and more personalized patient management.
The primary driver propelling the In Vitro Fertilization market is the global shift toward delayed childbearing, which significantly increases the incidence of age-related infertility. As more couples choose to conceive later in life, the natural decline in gamete quality necessitates medical intervention, positioning IVF not as a luxury but as a medical necessity. According to FMI's estimates, this demographic shift translates into a sustained increase in cycle volumes, anchored by data from national health registries showing a 5% year-on-year rise in patients over 35 initiating treatment in 2025 [8].
Conversely, the high cost of treatment remains a formidable restraint, limiting access to a fraction of the eligible population in non-reimbursed markets. A single IVF cycle can exceed USD 15,000 in the USA, creating a financial barrier that restricts market penetration to high-income demographics and forces suppliers to compete aggressively on price-performance ratios. Clinics mitigate this by offering tiered financing packages and shared-risk programs, yet the financial burden remains the primary cause of patient drop-out before successful conception [1].
.webp)
Based on the regional analysis, the In Vitro Fertilization market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.3% |
| United States | 11.0% |
| Canada | 10.7% |
| China | 10.5% |
| France | 9.7% |
| Germany | 9.2% |
| Japan | 9.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific is emerging as the volume engine of the global IVF sector, driven by immense population baselines and a cultural shift normalizing assisted reproduction. The region is characterized by a rapid proliferation of private fertility clinics, particularly in urban centers, which are adopting Western standards of care and technology to attract a growing middle class.
FMI’s report includes a detailed analysis of the growth in the Asia Pacific region, along with a country-wise assessment. The report identifies South Korea and Australia as key opportunistic markets; South Korea’s ultra-low birth rate is driving aggressive government intervention and subsidies for infertility drugs, while Australia’s mature market focuses on single-embryo transfer mandates to improve safety [3].
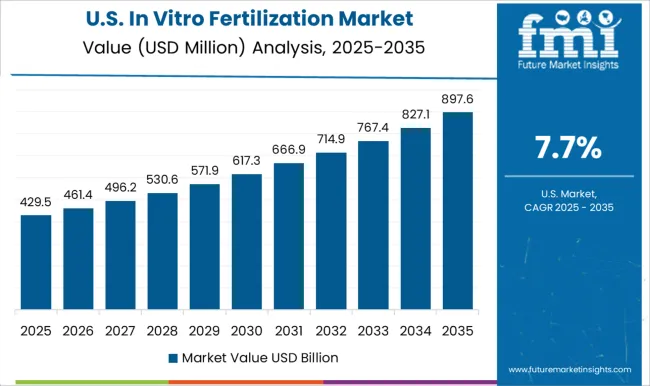
North America remains the value leader, characterized by high procedure costs and the rapid adoption of cutting-edge technologies like AI selection and non-invasive PGT. The region acts as a testing ground for premium innovations, with clinics competing on success rates and comprehensive care models rather than price.
FMI’s report includes a comprehensive evaluation of the North American landscape. Beyond the major economies, Mexico offers a burgeoning market for medical tourism, attracting US patients seeking cost-effective infertility treatment without compromising on quality standards; the report highlights cross-border partnerships as a key theme for suppliers [5].
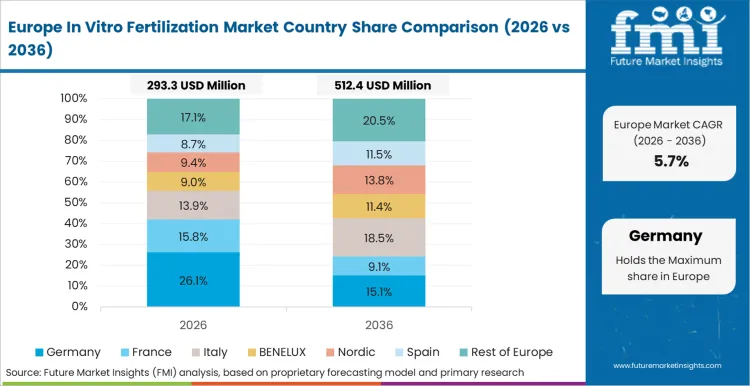
Europe represents a highly regulated and mature market where growth is driven by reimbursement policies and a strong focus on safety and ethics. The region leads in the adoption of single embryo transfer (SET) to reduce multiple birth rates, influencing demand for high-quality culture systems that ensure single embryo viability.
FMI’s report includes a detailed breakdown of the European fertility sector. Spain and the United Kingdom serve as crucial markets; Spain is a global hub for egg donation and semen warmer technologies due to favorable donor anonymity laws, while the UK’s HFEA regulation drives rigorous quality control standards that benefit premium equipment suppliers [4].
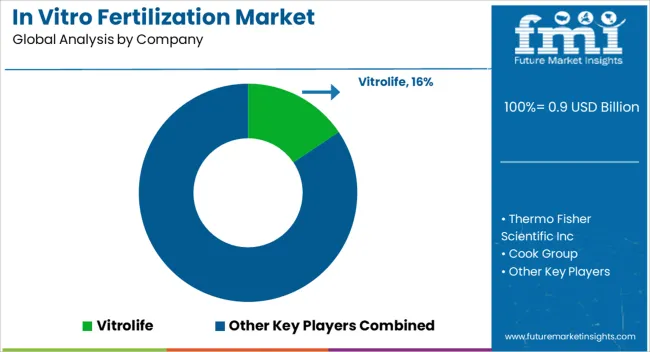
The competitive landscape is consolidated at the top, with players like CooperSurgical, Vitrolife, and Thermo Fisher Scientific dominating the high-value consumables and media segments. These market leaders leverage their scale to offer end-to-end lab solutions, creating high barriers to entry for smaller competitors who lack the breadth to supply entire clinic workflows [1].
Differentiation strategies focus on technological integration and service support. Companies like Cook Medical and Fujifilm Irvine Scientific are investing heavily in R&D to develop "smart" lab ecosystems where incubators, media, and handling devices communicate data seamlessly. This connectivity addresses the clinic's need for traceability and quality assurance, which are becoming mandatory for accreditation in key markets [2].
Strategic partnerships and acquisitions remain the primary avenue for expansion. Manufacturers are actively acquiring niche technology providers—such as those specializing in AI selection or genetic testing-to protect their core media businesses from commoditization. By controlling the entire embryo culture environment, these integrators can guarantee success rates, which is the ultimate currency for fertility clinics competing for patients [3].
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.30 billion (2026) to USD 3.80 billion (2036), at a CAGR of 11.0% |
| Market Definition | The In Vitro Fertilization market comprises the global ecosystem of assisted reproductive technologies, including instruments, consumables, and reagents used to facilitate fertilization outside the body. |
| Product Segmentation | Equipment, Reagents, Consumables, Others |
| Procedure Segmentation | ICSI (Intracytoplasmic Sperm Injection), Frozen Embryo Transfer, Fresh Embryo Transfer |
| Application Coverage | Fertility Clinics, Hospitals, Surgical Centers, Research Institutes |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key Companies Profiled | CooperSurgical, Vitrolife, Thermo Fisher Scientific, Cook Medical, Fujifilm Irvine Scientific, Genea Limited, Esco Micro Pte. Ltd., IVFtech ApS, The Baker Company, Kitazato Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with resin producers and panel manufacturers, supported by trade data benchmarking and plant level capacity verification |
Demand for In Vitro Fertilization in the global market is estimated to be valued at USD 1.30 billion in 2026.
Market size for In Vitro Fertilization is projected to reach USD 3.80 billion by 2036.
Demand for In Vitro Fertilization in the global market is expected to grow at a CAGR of 11.0% between 2026 and 2036.
ICSI (Intracytoplasmic Sperm Injection) is expected to be the dominant procedure, capturing approximately 55.0% of global market share in 2026 due to its high efficacy in male factor infertility.
Fertility clinics represent a critical segment, projected to hold a substantial 65.0% share of the total market in 2026 as specialized care centers aggregate volume.
Demand is driven by the "Make in India" initiative and new government subsidies for fertility treatments which are increasing patient access.
The National Health Mission guidelines and subsidies for fertility support are referenced as key benchmarks.
India is projected to grow at a CAGR of 11.3% during 2026 to 2036.
Europe is a mature, highly regulated market focused on safety and ethics, leading the adoption of single embryo transfers and advanced quality control.
Replacement driven demand for high-quality, compliant culture media and incubators dominates regional consumption.
France is projected to expand at a CAGR of 9.7% during 2026 to 2036.
Yes, Germany is included within Europe under the regional scope of analysis.
Reports from the Federal Statistical Office (Destatis) on health expenditure and ART service utilization are cited as primary reference sources.
Germany's demand is associated with the expansion of statutory health insurance coverage for ART procedures.
Yes, Japan is included within Asia Pacific under the regional coverage framework.
Insurance coverage expansion for ART procedures is emphasized as a key driver lifting utilization rates.
Consumables and reagents are prioritized due to the high volume of cycles and the critical need for lot-to-lot consistency.
In Vitro Fertilization is an assisted reproductive technology where an egg is fertilized by sperm outside the body, used primarily to treat infertility.
The market refers to the global trade and consumption of instruments, consumables, and reagents used in IVF procedures.
Scope covers IVF products by type, procedures including ICSI and FET, and end users such as fertility clinics and hospitals.
General gynecological devices and pharmaceutical drugs for hormonal stimulation are excluded unless integrated with IVF hardware.
Market forecast represents a model based projection built on defined assumptions for strategic planning purposes.
Forecast is developed using hybrid top down and bottom up modeling validated through clinic volume data, manufacturer reports, and industry review.
Primary interviews with embryologists and verifiable public datasets are used instead of unverified syndicated market estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.