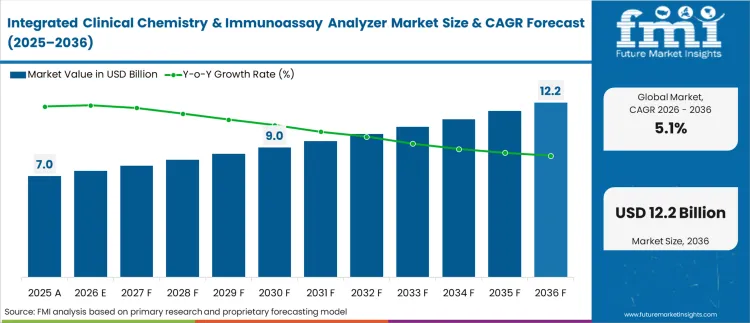
In 2026, the integrated clinical chemistry & immunoassay analyzer market was valued at USD 7.4 billion. Based on Future Market Insights analysis, demand for integrated clinical chemistry & immunoassay analyzers is estimated to grow to USD 12.2 billion by 2036. FMI projects a CAGR of 5.1% during the forecast period.
Absolute dollar growth of USD 4.8 billion over the decade signals steady scale-up rather than a broad replacement wave. As per FMI, growth is expected to be supported by high-volume labs shifting assays onto integrated platforms to reduce manual handoffs, while budget approvals and method verification workload are expected to keep upgrade cadence controlled in mature networks. “The development of the DxC 500i Analyzer is yet another example of how Beckman Coulter is investing in the needs of a broad range of laboratories across healthcare systems, The DxC 500i Clinical Analyzer powers both clinical chemistry and immunoassay testing in one space-saving package. With steadfast performance, practical simplicity, and trusted clinical quality, this analyzer meets the specific demands of the low-volume laboratory customer, standalone laboratories and community hospitals.” said Kathleen Orland, Senior Vice President, Business Unit, General Manager, Chemistry and Immunoassay for Beckman Coulter Diagnostics. [1]
Purchasing behaviour is being shaped by analyser standardisation and reagent continuity planning, with buyers prioritising uptime, calibration stability, and service response time that reduces disruption risk. Multi-year reagent and service agreements are being used to stabilise operating cost, while instrument refresh is being aligned to throughput growth and automation line integration needs.
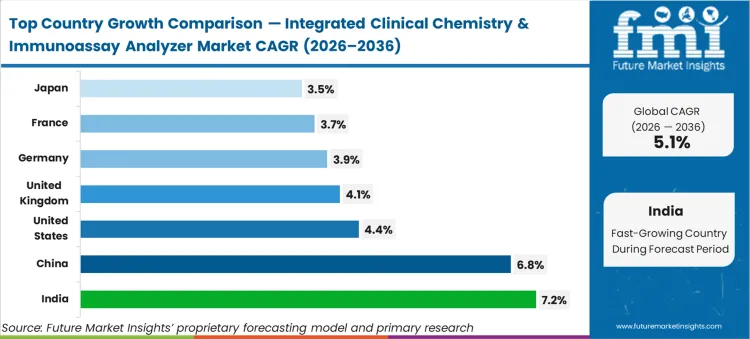
India (7.2% CAGR, supported by expanding private lab networks and rising test volumes) and China (6.8% CAGR, driven by hospital capacity growth and higher immunoassay utilisation) are expected to lead growth. The USA (4.4% CAGR) and the UK (4.1% CAGR) are expected to expand through platform consolidation and replacement purchasing. Mature markets including Germany (3.9%), France (3.7%), and Japan (3.5%) are expected to contribute more through replacement demand, constrained by procurement controls and verification timelines.
The market covers integrated laboratory platforms that run clinical chemistry and immunoassay testing within a single system or integrated configuration. These platforms are purchased by hospital laboratories, reference laboratories, independent or private labs, and academic or research settings to deliver routine and specialised testing with controlled turnaround time and QC discipline. Offerings include instruments, reagents and consumables, and software supporting workflow and connectivity. Analyzer configurations range from compact integrated systems to automation line or workcell integrations, with throughput classes spanning low to ultra-high. Demand is shaped by rising test volumes, staffing efficiency needs, and menu consolidation.
The report includes global and regional market sizes and a 10-year forecast for 2026 to 2036. Segment-level sizing is provided by product type, analyzer configuration, immunoassay detection, throughput class, and end user, supported by country-level CAGR comparisons. Coverage includes assessment of procurement patterns tied to analyser standardisation, automation line adoption, reagent contracting, and service coverage expectations, alongside competitive benchmarking of leading system providers, as per FMI analysis.
The scope excludes standalone clinical chemistry analysers and standalone immunoassay analysers sold outside integrated platform configurations. Point-of-care immunoassay devices, lateral flow tests, and manual ELISA workflows are excluded. Laboratory information systems and middleware sold as independent products are excluded unless packaged as part of the integrated analyser offering. General lab consumables not tied to analyser operation and testing services revenue are excluded. The focus remains on integrated chemistry and immunoassay analyser platforms and their associated reagent, instrument, and software components.

Based on FMI’s report, reagents and consumables are estimated to hold 54% share in 2026, followed by instruments at 32% share. Reagents lead because routine diagnostics depend on continuous replenishment tied to daily test volumes, which creates stable recurring purchasing once an analyzer is embedded. Instruments remain the second position since placements and replacements happen in cycles, with purchasing paced by capex approvals and lab consolidation plans.
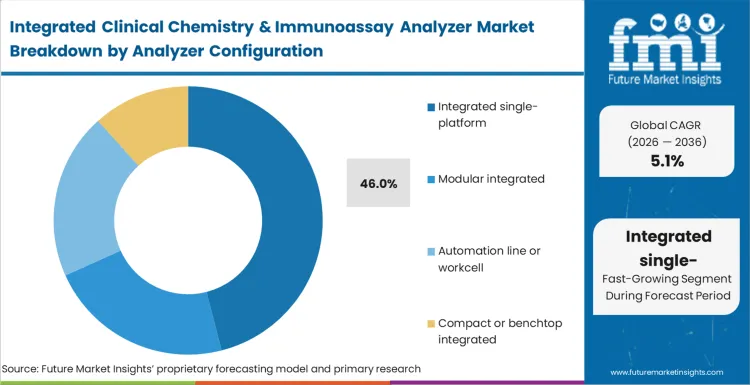
Integrated single-platform systems account for 46% share in 2026, followed by modular integrated at 24%, based on FMI’s report. Single-platform leadership is supported by workflow simplification where one integrated system reduces sample routing, staffing touchpoints, and cross-instrument reconciliation. Modular systems follow because large labs value scalability, adding modules to expand lanes without rebuilding the full setup, though integration effort and footprint planning shape adoption.
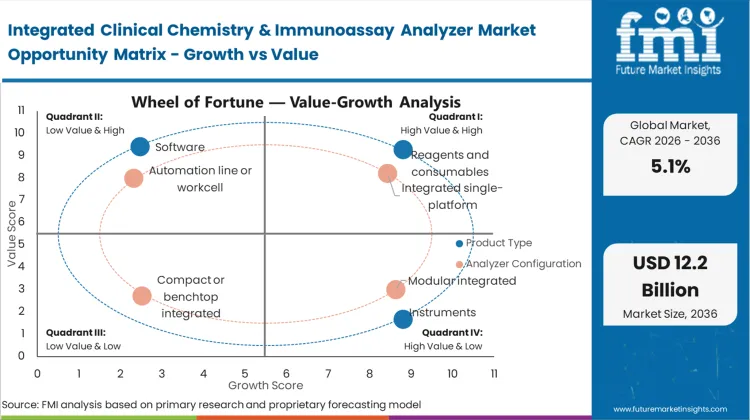
Future Market Insights analysis that historical patterns point at a workflow-consolidation diagnostics category where demand is anchored in high routine testing volumes and the need to reduce sample handling across chemistry and immunoassay lanes. Current demand is being shaped by staffing constraints, turnaround expectations, and the operational benefit of running consolidated menus through fewer platforms, as per FMI.
While growth is being restrained by long replacement cycles, capital budgeting discipline, and switching friction linked to assay validation and LIS integration, value expansion is being supported by recurring reagent pull-through and wider consolidation in hospital and reference laboratories. Based on FMI’s report, purchasing decisions are increasingly being filtered by uptime performance, service reach, and reagent continuity rather than by instrument price alone.
Based on the regional analysis, integrated clinical chemistry & immunoassay analyzer market is segmented into North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East & Africa across 40+ countries. Regional performance is interpreted through replacement cycles for integrated and modular analyzers, expansion of CLIA and ECLIA testing, throughput-driven automation investments, and reagent-consumables pull across hospital and reference labs, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 4.4% |
| China | 6.8% |
| Germany | 3.9% |
| Japan | 3.5% |
| India | 7.2% |
| United Kingdom | 4.1% |
| France | 3.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America is driven by network-level lab consolidation, where reagent and consumables contracts determine platform continuity and where throughput class selection is linked to hub-and-spoke testing models. Roche Diagnostics remains strongly placed in integrated single-platform systems with recurring reagent pull. Abbott maintains broad share through immunoassay coverage and hospital lab placements. Siemens Healthineers is positioned where automation lines and workcells are adopted in higher-volume reference labs. FMI analysts note that hospital laboratories remain the core end users, with reference labs driving high and ultra-high throughput demand.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA, Canada and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia is defined by rapid lab modernisation and expanding routine testing volumes, where modular integrated systems gain share in large hospitals and automation workcells rise in high-throughput environments. Roche Diagnostics and Abbott participate through installed base and reagent economics, while Mindray expands placements where procurement favours cost efficiency and local service coverage. Sysmex and Fujirebio add regional strength in diagnostics ecosystems that support immunoassay adoption. FMI is of the opinion that growth remains led by reagents and consumables as installed base expands.
FMI’s report includes a detailed analysis of the growth in the East Asia region, along with a country-wise assessment that includes China, Japan and South Korea. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the East Asia region.
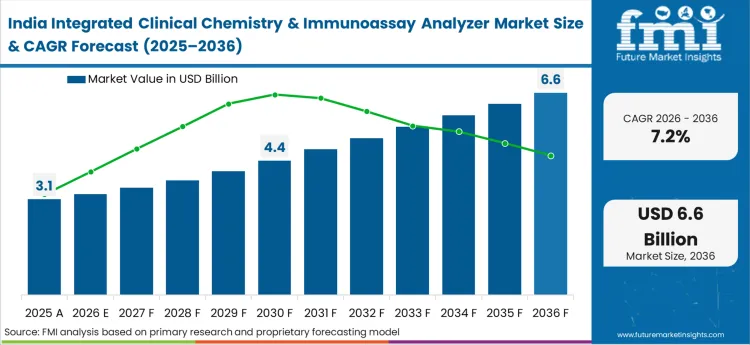
South Asia & Pacific is characterised by rapid expansion of diagnostic chains and rising test volumes beyond tier-1 cities, which supports mid throughput adoption and gradual movement toward automation workcells in hub labs. Roche Diagnostics and Abbott retain premium placements in hospitals, while Mindray and other value-focused suppliers gain share where service response times and distributor coverage influence uptime. QuidelOrtho and Randox participate through immunoassay and chemistry offerings aligned to routine testing. FMI analysts note that reagent and consumables remain the largest revenue stream, reinforcing long-term supply contracts.
FMI’s report includes a detailed analysis of the growth in the South Asia & Pacific region, along with a country-wise assessment that includes India, ASEAN Countries, Australia & New Zealand and Rest of South Asia. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the South Asia & Pacific region.
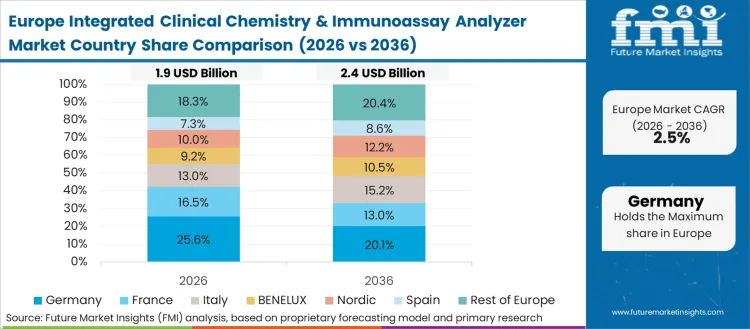
Western Europe is procurement-led and replacement-driven, where platform choice is shaped by tender frameworks, total cost of ownership, and long-term reagent economics. Roche Diagnostics retains strong continuity in integrated single-platform systems. Siemens Healthineers is positioned in automation line and workcell deployments for high-volume sites. Abbott remains relevant through broad immunoassay menus and hospital reach. Beckman Coulter (Danaher) supports adoption where automation integration and throughput upgrades are prioritised. FMI is of the opinion that value remains driven by reagents and consumables, with instruments tied to scheduled refresh cycles.
FMI’s analysis of integrated clinical chemistry & immunoassay analyzer market in Western Europe consists of country-wise assessment that includes Germany, UK, France, Italy, Spain, BENELUX, Nordic Countries and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
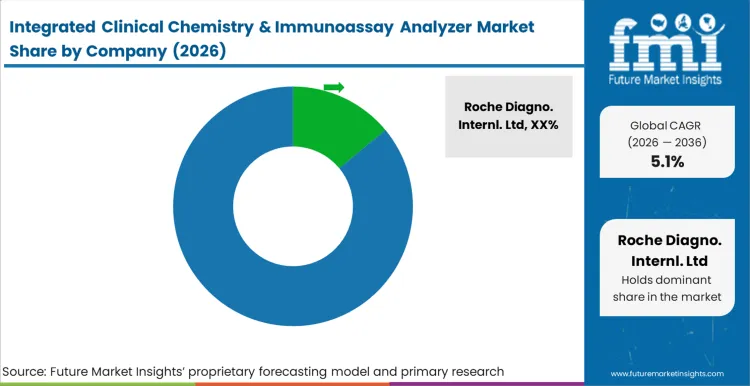
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can place analyzers at scale, protect uptime through dense field service coverage, and sustain recurring reagent pull-through under multi-year contracting. Competition is shaped by cost per reported result and operational continuity rather than short-cycle instrument pricing, since purchases are tied to platform standardisation, staff training, and workflow redesign in laboratories. The updated segmentation reinforces this dynamic because reagents and consumables remain the largest product value pool, so suppliers that control assays, calibrators, and QC ecosystems carry stronger competitive control, as per FMI.
Companies with structural advantages typically combine large installed bases with proprietary reagent menus and middleware layers that raise switching friction after validation. Integrated single-platform configurations are favored where mid throughput workloads dominate and footprint discipline is required, while modular integrated and automation line workcells are preferred in networks seeking higher throughput and consolidation. Suppliers able to support both ends of this configuration spectrum, with consistent assay performance and stable lot management, tend to retain accounts during contract renewal cycles. Firms with thinner assay breadth or weaker service reach face slower conversions because performance evidence, uptime history, and response time discipline are scrutinised before a network expands placements, based on FMI’s report.
Customer concentration reinforces buyer leverage. Hospital laboratories and reference labs typically run competitive tenders, qualify more than one vendor, and use performance-based contracts that include uptime commitments, response time terms, and reagent supply continuity clauses to reduce dependency. Independent and private labs often negotiate volume tiers tied to throughput class and keep alternates active for critical assays, which limits supplier ability to push through broad price increases. Pricing power is constrained for routine chemistry and immunoassay panels, while measured premiums are retained mainly where a supplier reduces hands-on time, improves turnaround consistency, or supports consolidation into fewer workcells without service disruption, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 7.4 Billion (2026) to USD 12.2 Billion (2036), at a CAGR of 5.1% |
| Market Definition | The integrated clinical chemistry and immunoassay analyzer market comprises global production and trade of analyzer systems that consolidate chemistry and immunoassay testing within integrated platforms, enabling laboratories to run routine and specialty panels under defined quality and regulatory requirements, where demand is shaped by test volume growth, automation priorities, and reagent continuity expectations. |
| Product Type Segmentation | Reagents and consumables, Instruments, Software |
| Analyzer Configuration Segmentation | Integrated single-platform, Modular integrated, Automation line or workcell, Compact or benchtop integrated |
| Immunoassay Detection Segmentation | CLIA, ECLIA, ELFA, Fluorescence immunoassay, Other |
| Throughput Class Segmentation | Low (<200 tests/hr), Mid (200-800), High (801-2000), Ultra-high (>2000) |
| End User Coverage | Hospital laboratories, Reference laboratories, Independent or private labs, Academic or research |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key Companies Profiled | Roche Diagnostics International Ltd, Abbott Laboratories, Siemens Healthineers AG, Beckman Coulter, Inc., Sysmex Corporation, QuidelOrtho Corporation, Mindray Bio-Medical Electronics Co., Ltd., Thermo Fisher Scientific Inc., Randox Laboratories Ltd, Fujirebio Holdings, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with laboratory directors, procurement stakeholders, and manufacturers, supported by installed base triangulation and reagent placement benchmarking, as per FMI. |
How large is the demand for Integrated Clinical Chemistry and Immunoassay Analyzers in the global market in 2026?
Demand for Integrated Clinical Chemistry and Immunoassay Analyzers in the global market is estimated to be valued at USD 7.4 Billion in 2026, as per FMI.
What will be the market size of Integrated Clinical Chemistry and Immunoassay Analyzers in the global market by 2036?
Market size for Integrated Clinical Chemistry and Immunoassay Analyzers is projected to reach USD 12.2 Billion by 2036.
What is the expected demand growth for Integrated Clinical Chemistry and Immunoassay Analyzers in the global market between 2026 and 2036?
Demand for Integrated Clinical Chemistry and Immunoassay Analyzers in the global market is expected to grow at a CAGR of 5.1% between 2026 and 2036.
Which immunoassay detection format is poised to lead global demand by 2026?
CLIA is expected to be the dominant immunoassay detection format, capturing 52% share in 2026.
Which analyzer configuration is expected to account for the largest share in 2026?
Integrated single-platform is expected to hold the highest share at 46% in 2026.
Which product type accounts for the largest share in 2026?
Reagents and consumables are projected to account for 54% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.