Demand for non-alcoholic steatohepatitis clinical trials in Japan is expected to grow from USD 161.6 million in 2025 to USD 269.6 million by 2035, reflecting a compound annual growth rate (CAGR) of 5.3%. Growth is driven by the increasing prevalence of NASH, a liver disease that is closely associated with obesity, diabetes, and metabolic syndrome. As the global focus shifts towards understanding and treating chronic liver diseases, Japan’s strong healthcare infrastructure and research capabilities position it as a key player in developing therapies for NASH. Ongoing advancements in medical research, increasing investment in clinical trials, and growing awareness of the impact of NASH will contribute to this expansion.
Growth is supported by rise in regulatory approvals for new treatments aimed at managing NASH and the growing number of pharmaceutical companies conducting clinical trials for potential therapies. As more patients are diagnosed with NASH and the need for effective treatments becomes more urgent, the demand for NASH clinical trials is expected to increase steadily, with Japan continuing to play an important role in the global clinical trial landscape.
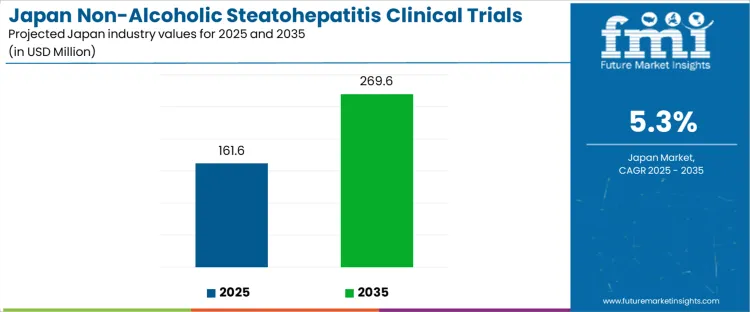
The rolling compound annual growth rate (CAGR) analysis for the NASH clinical trials market in Japan shows consistent and gradual growth over the forecast period. Starting at USD 161.6 million in 2025, the market grows to USD 170.1 million in 2026 and USD 179.0 million in 2027, reflecting early-stage increases driven by an expanding clinical focus on liver diseases and the rising number of NASH clinical trial studies.
From 2027 to 2030, the market continues to expand steadily, reaching USD 188.4 million in 2028, USD 198.3 million in 2029, and USD 208.7 million in 2030. During this phase, the market is driven by more pharmaceutical companies investing in NASH treatments and expanding their clinical trial pipelines. By 2035, the market will reach USD 269.6 million, reflecting significant growth as NASH-related clinical trials gain momentum, treatments are refined, and more global stakeholders invest in the Japanese market. The rolling CAGR analysis shows that growth is sustained at a steady pace throughout the decade, with incremental increases year-on-year, driven by clinical trial expansions and the pressing demand for effective NASH therapies.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 161.6 million |
| Industry Forecast Value (2035) | USD 269.6 million |
| Industry Forecast CAGR (2025-2035) | 5.3% |
Demand for clinical trials targeting NASH in Japan is driven by the rising prevalence of Nonalcoholic Fatty Liver Disease (NAFLD) and Nonalcoholic Steatohepatitis (NASH) in the population. Studies show that a significant portion of the Japanese adult population is affected by NAFLD, and a meaningful subset of these individuals progress to NASH, which can lead to liver fibrosis, cirrhosis, or hepatocellular carcinoma. The increasing burden of metabolic disorders such as diabetes, obesity, and dyslipidemia in Japan contributes to the growing number of individuals at risk for NASH. The clinical and public health challenges presented by NASH create a pressing need for effective therapies and improved diagnostic tools.
Future demand for NASH clinical trials in Japan is expected to rise as both the medical and research communities seek new treatment options for NASH and associated liver conditions. The lack of fully approved and effective therapies for NASH motivates drug developers to test new compounds and treatments. Additionally, the unique genetic, lifestyle, and comorbidity patterns in Japan make it an attractive site for trials. As the population continues to age and the prevalence of metabolic diseases remains high, the number of patients eligible for NASH trials will increase. Success in these trials could lead to the broader adoption of new therapies, which will further sustain research demand. The future pace of trials will depend on regulatory support, funding, and collaboration between public and private sectors.
The Non-Alcoholic Steatohepatitis (NASH) clinical trials market in Japan is segmented by study design and phase. Interventional studies dominate the study design segment, holding 45% of the market share. In terms of phase, Phase 3 trials lead, representing 40% of the market. The increasing focus on developing effective treatments for NASH and the rising prevalence of liver disease are driving the demand for clinical trials in Japan.
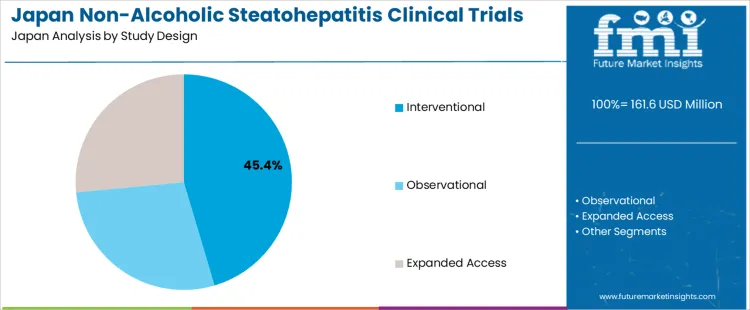
Interventional studies are the most prevalent study design in NASH clinical trials in Japan, accounting for 45% of the market share. Interventional trials involve actively administering a treatment or procedure to participants to assess its effects on the disease, making them critical in the development of new therapies for NASH. These trials are essential for testing the efficacy and safety of potential NASH treatments, including pharmacological agents and non-pharmacological interventions. The demand for interventional studies is driven by the need to identify effective treatments for NASH, as the condition is associated with increased risk of liver cirrhosis and liver cancer. While observational and expanded access studies are also used, interventional studies are the key focus for advancing treatment options in clinical settings.
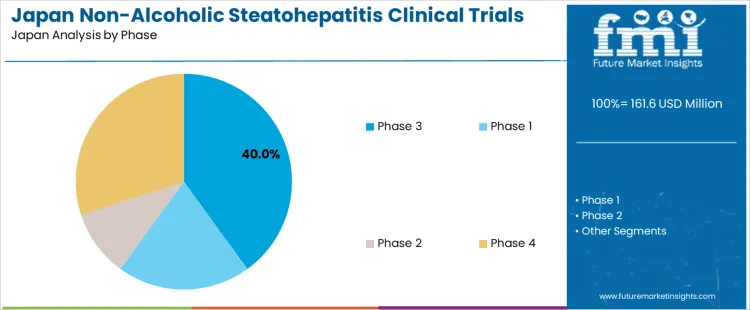
Phase 3 trials are the leading phase in the NASH clinical trials market in Japan, representing 40% of the market share. Phase 3 trials are crucial for testing the effectiveness of new drugs or treatments in large patient populations and comparing them against current standard treatments. They are essential for obtaining the data required for regulatory approval and market entry. The focus on Phase 3 trials reflects the need to validate potential therapies for NASH before they can be widely used in clinical practice. Phase 1 and Phase 2 trials also contribute to the market, but Phase 3 trials dominate because they are pivotal to bringing new treatments to market. Phase 4 trials, conducted after a drug has been approved, also contribute to the market by monitoring long-term safety and efficacy.
Demand for non-alcoholic steatohepatitis (NASH) clinical trials in Japan is rising due to the increasing prevalence of liver diseases, particularly NASH, a condition that can lead to liver cirrhosis and other complications. NASH is a major public health concern, driven by factors such as obesity, diabetes, and metabolic syndrome, which are becoming more common in Japan. As the healthcare system in Japan seeks to address the rising burden of liver diseases, there is a growing need for clinical trials focused on the development of effective treatments for NASH. Advances in clinical research, along with increasing healthcare awareness, are further driving the demand for NASH clinical trials.
What are the Drivers of Demand for NASH Clinical Trials in Japan?
Several factors are driving the demand for NASH clinical trials in Japan. Growing prevalence of obesity, diabetes, and metabolic syndrome is leading to an increase in NASH cases, making it a critical area of focus for healthcare professionals and researchers. Lack of approved pharmacological treatments for NASH and the need for effective therapeutic options are pushing the demand for clinical trials that can test new drugs and interventions. Japan’s aging population is contributing to the rise in liver diseases, as older adults are more susceptible to chronic liver conditions. Growing awareness of the importance of early diagnosis and intervention in managing NASH is also encouraging the development of clinical trials in this area. Japan’s well-established healthcare infrastructure and regulatory environment support the growth of clinical research, making it a key player in NASH clinical trials.
What are the Restraints on Demand for NASH Clinical Trials in Japan?
Despite the increasing demand, several challenges may limit the growth of NASH clinical trials in Japan. Key restraints include high cost and complexity associated with conducting clinical trials, particularly those that involve long-term treatment and monitoring of patients. These trials require significant financial investment, which may be a barrier for smaller research institutions or pharmaceutical companies. Another challenge is the relatively slow recruitment of patients for NASH trials, as many individuals with early-stage NASH may not be aware of their condition or may not meet the strict inclusion criteria for clinical trials. The lack of standardized diagnostic criteria for NASH and the difficulty in identifying patients at the right stage for treatment can also hinder the development and recruitment for clinical trials. Additionally, regulatory hurdles and the need for extensive data to prove the safety and efficacy of new treatments may delay the progress of clinical trials in NASH.
What are the Key Trends Influencing Demand for NASH Clinical Trials in Japan?
Several key trends are shaping the demand for NASH clinical trials in Japan. One major trend is the increasing focus on precision medicine, which allows for tailored treatments based on individual patient profiles and genetic data. This trend is contributing to the development of more targeted therapies for NASH, driving the need for clinical trials that can evaluate their efficacy. Another trend is the rising interest in non-invasive diagnostic techniques for NASH, facilitating patient recruitment for clinical trials and speeding early diagnosis. Advances in biomarker discovery and imaging technologies are also enabling more accurate identification of patients who would benefit from clinical trials. There is growing collaboration between Japanese research institutions, pharmaceutical companies, and global organizations to address the unmet medical needs in NASH, further stimulating clinical trials. As the focus on liver disease research intensifies, government support for clinical trials and healthcare innovations is likely to increase, contributing to the growth of NASH research in Japan.
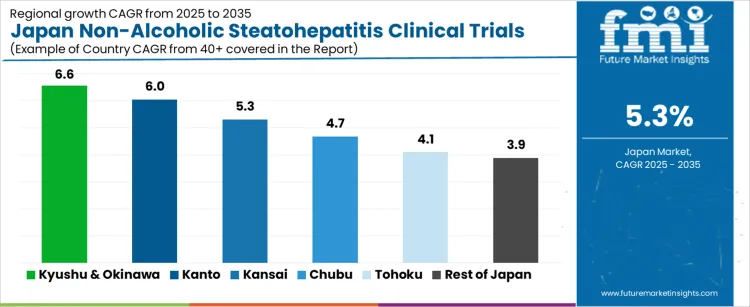
Demand for Non-Alcoholic Steatohepatitis (NASH) clinical trials in Japan is expected to experience steady growth, with Kyushu & Okinawa leading the way at a projected CAGR of 6.6%. Kanto follows with a growth rate of 6.0%, while Kansai is expected to grow at 5.3%. Chubu is projected to grow at a rate of 4.7%, while Tohoku and Rest of Japan show more moderate growth, with CAGRs of 4.1% and 3.9%, respectively. The rising prevalence of NASH, a chronic liver disease, and the growing focus on innovative treatments for liver conditions are key drivers of the demand for clinical trials in Japan.
| Region | CAGR (2025-2035) |
|---|---|
| Kyushu & Okinawa | 6.6% |
| Kanto | 6.0% |
| Kansai | 5.3% |
| Chubu | 4.7% |
| Tohoku | 4.1% |
| Rest of Japan | 3.9% |
Kyushu & Okinawa are projected to experience the highest growth in the NASH clinical trials market, with a projected CAGR of 6.6%. The region’s increasing burden of liver diseases, particularly NASH, combined with the growing demand for innovative treatment options, is a key driver of this growth. The aging population in Kyushu & Okinawa, which is more prone to chronic diseases such as NASH, contributes significantly to the demand for clinical trials focused on liver health. Additionally, the region’s focus on improving healthcare infrastructure and the rising interest in the development of new therapeutics for liver diseases is driving the growth of NASH clinical trials. As healthcare systems in Kyushu & Okinawa continue to prioritize the management and treatment of liver diseases, the market for NASH clinical trials is expected to expand at a strong pace.
In Kanto, the demand for NASH clinical trials is projected to grow at a CAGR of 6.0%. Kanto, which includes Tokyo, is Japan’s most populous and urbanized region and a hub for medical research and clinical trials. The region’s well-established healthcare infrastructure, combined with an increasing prevalence of NASH, is driving the growth of clinical trials focused on liver health. The high incidence of metabolic conditions like obesity and diabetes, which are closely linked to NASH, contributes to the rising demand for NASH trials in Kanto. Healthcare providers in the region are increasingly adopting advanced diagnostics and therapies to treat liver diseases, which in turn drives the need for more clinical trials to evaluate new treatments. As the region continues to invest in clinical research and development, the market for NASH clinical trials is expected to grow steadily.
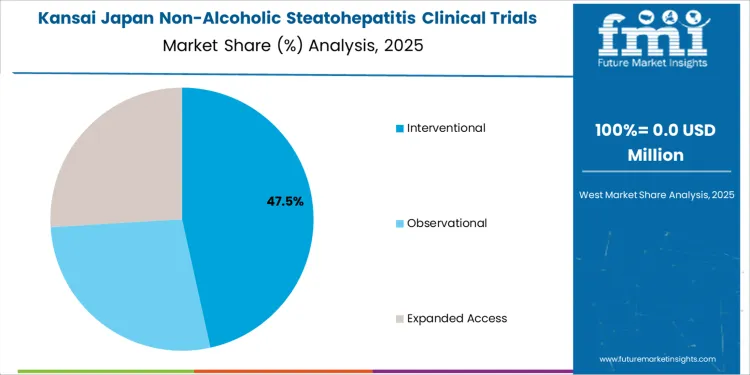
In Kansai, the NASH clinical trials market is projected to grow at a CAGR of 5.3%. The region, which includes major cities like Osaka and Kyoto, has a strong healthcare infrastructure that is increasingly focused on addressing chronic diseases such as NASH. With a rising number of NASH diagnoses, especially among the elderly and individuals with metabolic syndrome, Kansai’s healthcare systems are becoming more engaged in conducting clinical trials to assess new treatments. The region’s healthcare providers are partnering with pharmaceutical companies to expand clinical trial opportunities for NASH, as there is a significant demand for effective therapies. Kansai’s focus on improving treatment options for liver diseases is helping to drive market growth. As the region continues to develop its medical research capabilities, the demand for NASH clinical trials is expected to rise steadily.
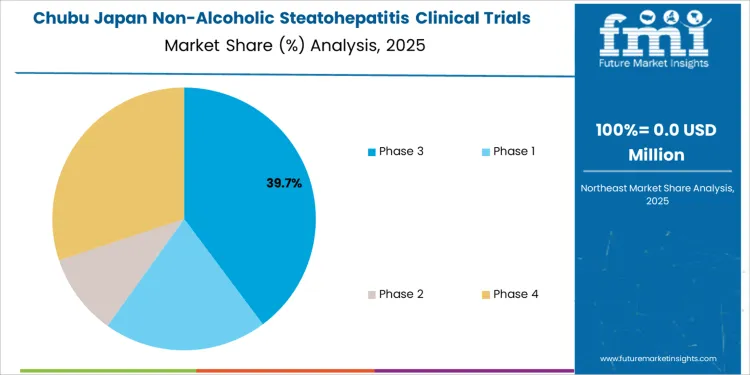
In Chubu, the NASH clinical trials market is expected to grow at a CAGR of 4.7%. The region’s increasing focus on healthcare innovation and the rising incidence of liver diseases are key drivers of this growth. Chubu, with cities like Nagoya, has a growing healthcare sector, and the adoption of advanced medical technologies is driving the expansion of clinical trials, including those focused on NASH. The region’s aging population, which is more susceptible to liver conditions, is contributing to the demand for clinical trials to test new therapies for NASH. Additionally, Chubu’s emphasis on improving patient care and increasing the availability of clinical trials is helping to meet the growing need for new treatment options. As healthcare providers in Chubu continue to embrace cutting-edge research and development, the market for NASH clinical trials is expected to expand gradually.
In Tohoku, the NASH clinical trials market is projected to grow at a CAGR of 4.1%. While the growth rate in Tohoku is more moderate compared to other regions, the demand for clinical trials focused on NASH is still increasing due to the region’s aging population and rising prevalence of liver diseases. As the healthcare systems in Tohoku continue to adopt new technologies for the diagnosis and treatment of liver diseases, there is a growing need for clinical trials to evaluate new NASH treatments. The region’s healthcare providers are becoming more involved in conducting trials to assess new therapies for liver conditions, contributing to the steady growth of the market. As access to clinical research opportunities improves and awareness of NASH grows in Tohoku, the market is expected to continue expanding at a moderate pace.
In Rest of Japan, the NASH clinical trials market is expected to grow at a more moderate rate of 3.9%. Although the demand for NASH clinical trials is smaller compared to major metropolitan regions, the increasing awareness of liver diseases and the need for effective treatments are driving market growth. Healthcare providers in Rest of Japan are gradually adopting advanced diagnostic tools and expanding access to clinical trials, which is contributing to the demand for new NASH therapies. As the region’s healthcare systems continue to develop and improve access to specialized care, the market for NASH clinical trials is expected to grow steadily. Additionally, the rise in chronic liver diseases and metabolic conditions in Rest of Japan is contributing to the continued expansion of the market.
Demand for clinical trials targeting Non Alcoholic Steatohepatitis (NASH) in Japan has grown substantially as the prevalence of Non Alcoholic Fatty Liver Disease (NAFLD) and related metabolic disorders rises. Recent studies estimate that NAFLD affects roughly one quarter to a third of the general population in Japan. Among these, a significant proportion may progress to NASH, which is linked to obesity, diabetes and lifestyle related risk factors. The growing disease burden, combined with rising awareness among clinicians and patients and unmet medical need — due to lack of approved NASH specific therapies — fuels demand for clinical trials evaluating novel therapeutics. Pharmaceutical pipelines and research efforts are shifting toward anti fibrotic, metabolic and disease modifying candidates, increasing demand for trial enrolment and regulatory grade evidence in Japan.
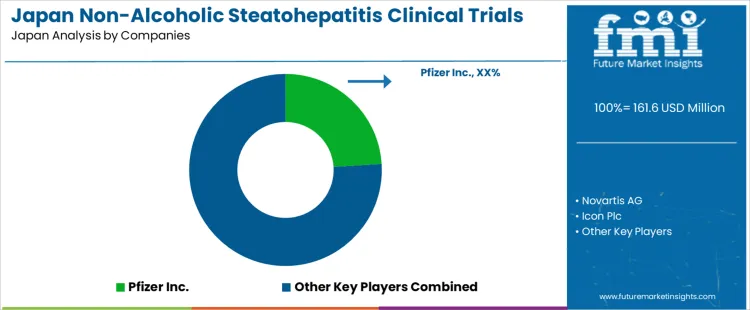
On the supply side, several multinational pharmaceutical and CRO affiliated companies are competing to lead NASH clinical trials in Japan. As per your list, Pfizer, Inc. holds a leading share among these players, followed by Novartis AG, Icon plc, AbbVie Inc., and F. Hoffmann La Roche AG. These companies are driving trials that explore new mechanisms — metabolic regulators, anti inflammatory or antifibrotic agents, and combination therapies. Competition centers on the strength of their drug pipelines, ability to recruit patients (given under diagnosis and need for liver biopsy or validated non invasive diagnostics), regulatory compliance, and ability to generate robust clinical data across liver disease severities. Firms that can efficiently run late stage trials, ensure safety and efficacy across Japanese populations, and navigate regulatory and reimbursement pathways are best positioned to lead as demand for NASH therapies and corresponding clinical evidence grows rapidly in Japan.
| Items | Details |
|---|---|
| Quantitative Units | USD Million |
| Regions Covered | Japan |
| Study Design | Interventional; Observational; Expanded Access |
| Phase | Phase 1; Phase 2; Phase 3; Phase 4 |
| Key Companies Profiled | Pfizer Inc.; Novartis AG; Icon Plc; AbbVie Inc.; F. Hoffmann La Roche Ltd |
| Additional Attributes | Dollar value driven by increasing NASH patient population, rising number of NASH trials, and growing interest in novel therapeutics. The shift toward interventional Phase 2 and Phase 3 trials — especially with emerging drug candidates — is boosting trial spend. Observational studies remain important for disease natural history, biomarkers, and long-term safety monitoring. |
How big is the demand for non-alcoholic steatohepatitis clinical trials in Japan in 2025?
The demand for non-alcoholic steatohepatitis clinical trials in Japan is estimated to be valued at USD 161.6 million in 2025.
What will be the size of non-alcoholic steatohepatitis clinical trials in Japan in 2035?
The market size for the non-alcoholic steatohepatitis clinical trials in Japan is projected to reach USD 269.6 million by 2035.
How much will be the demand for non-alcoholic steatohepatitis clinical trials in Japan growth between 2025 and 2035?
The demand for non-alcoholic steatohepatitis clinical trials in Japan is expected to grow at a 5.3% CAGR between 2025 and 2035.
What are the key product types in the non-alcoholic steatohepatitis clinical trials in Japan?
The key product types in non-alcoholic steatohepatitis clinical trials in Japan are interventional, observational and expanded access.
Which phase segment is expected to contribute significant share in the non-alcoholic steatohepatitis clinical trials in Japan in 2025?
In terms of phase, phase 3 segment is expected to command 40.0% share in the non-alcoholic steatohepatitis clinical trials in Japan in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.