Life Science Trade Lane Design and Qualification Services Market
The life science trade lane design and qualification services is segmented by Service Type (Lane design, Lane qualification, Requalification, Packaging qualification, Deviation review), Transport Mode (Air, Road, Ocean, Multimodal), Temperature Range (2–8°C, Controlled ambient, Frozen, Cryogenic), End Use (Commercial biologics, Clinical trials, Vaccines, Cell therapies, Diagnostics), Customer Type (Pharma manufacturers, Biotech firms, CDMOs, CROs, 3PLs), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Life Science Trade Lane Design and Qualification Services Market Size, Market Forecast, and Outlook By FMI
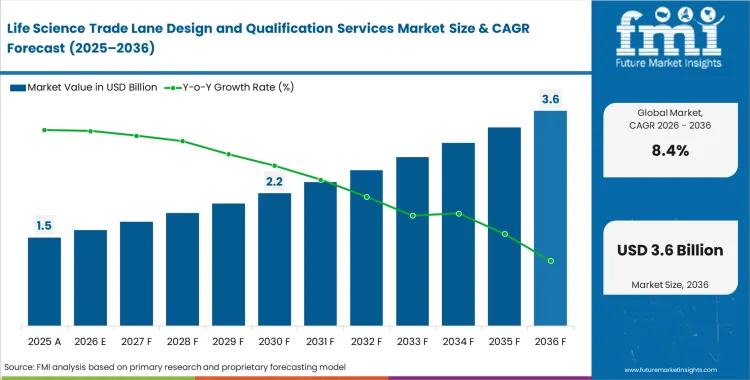
The life science trade lane design and qualification services market reached USD 1.4 billion in 2025 and is expected to exceed USD 1.5 billion in 2026, expanding at a CAGR of 8.40% as companies intensify distribution logistics assessment cycles. Continued investment is set to drive the market to approximately USD 3.3 billion by 2036, supported by a regulatory shift that prioritizes real-time transit route verification over traditional, facility-based temperature mapping.
Summary of Life Science Trade Lane Design and Qualification Services Market
- Market Snapshot
- The Life Science Trade Lane Design and Qualification Services Market is valued at USD 1.36 billion in 2025 and is projected to reach USD 3.32 billion by 2036.
- The industry is expected to grow at an 8.4% CAGR from 2026 to 2036, creating an incremental opportunity of USD 1.84 billion over the period.
- The market is a compliance-led specialist logistics services category covering route design, lane qualification, requalification, mapping, packaging validation, and deviation review for GDP- and CEIV-sensitive life-science shipments.
- Structural demand remains tied to certification, risk management, and repeat revalidation, because CEIV Pharma covers route planning, supplier management, audits, mapping, monitoring, and operational controls rather than simple freight execution.
- Demand and Growth Drivers
- Demand is rising because pharmaceutical air and cold-chain networks must prove route suitability across origin, transit, and destination conditions, with UPS explicitly framing lane qualification around temperature performance, airport infrastructure, reliability, and handling conditions.
- Adoption is also supported by the expansion of the certification base, with IATA reporting more than 500 pharma-certified stations inside a CEIV network of more than 700 stations, increasing the installed base for qualification and requalification services.
- Growth is being reinforced by network expansion and healthcare-capability investments, including DHL’s EUR 2 billion sector investment plan and FedEx’s 2025 CEIV Pharma milestone across Asia-Pacific hubs and ramps.
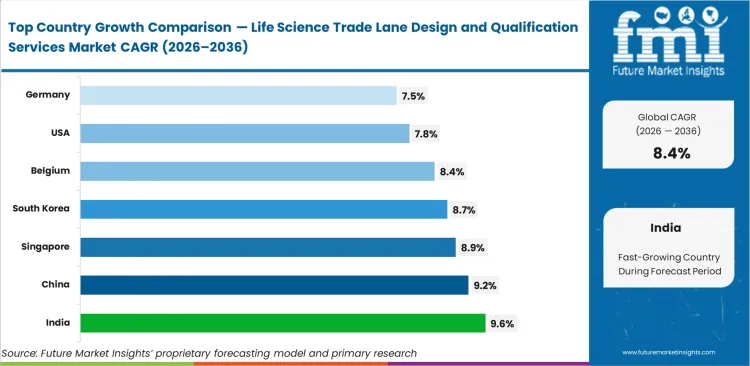
- India leads at 9.6% CAGR, followed by China at 9.2%, Singapore at 8.9%, South Korea at 8.7%, Belgium at 8.4%, the United States at 7.8%, and Germany at 7.5%. These are modelled from export intensity, certified infrastructure, and biopharma complexity rather than copied from syndicated reports.
- Growth is moderated by the fact that qualification is project-based and often bundled with wider logistics programs, which keeps it below the expansion rates seen in more disruptive therapy-specific cold-chain markets.
- Product and Segment View
- The market includes trade-lane engineering and validation services of air, road, ocean and multimodal pharmaceutical movements of GDP, CEIV Pharma and temperature-control operating models.
- These services are used across commercial biologics, clinical trials, vaccines, cell therapies, and diagnostics, where route reliability and documented control directly affect release and compliance decisions.
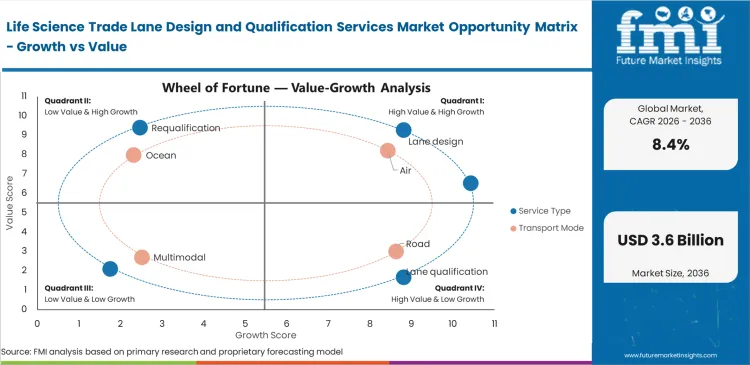
- Lane design leads the Service Type segment with 34.1% share, because initial route configuration and SOP design usually precede field qualification and later requalification work.
- Air leads the Transport Mode segment with 46.3% share, reflecting the concentration of qualification intensity in cross-border, time-sensitive, temperature-controlled pharmaceutical freight.
- 2–8°C leads the Temperature Range segment with 41.7% share, as this remains the broadest qualified range across vaccines, specialty drugs, and conventional cold-chain biologics.
- Commercial biologics leads the End Use segment with 38.4% share, because it combines recurring launch lanes, global distribution complexity, and strict excursion sensitivity.
- Pharma manufacturers lead the Customer Type segment with 44.6% share, as they own release risk, supplier qualification, and transport-validation accountability even when operations are outsourced.
- The scope includes route design, shipping-lane verification, qualification protocols, requalification, mapping, and associated compliance consulting, but excludes linehaul revenue, warehousing revenue, and the full value of packaging hardware or monitored shipments themselves.
- Geography and Competitive Outlook
- The models constitute India, China, and Singapore as the most rapidly developing of the national markets, and the United States as the largest of the stable demand bases due to its combination of high volumes of life-science shipments and developed GDP/quality systems.
- Integrated healthcare networks, the ability to combine qualification work with managed transport, and the ability to compete with each other shape competition, and the major players are DHL Health Logistics, UPS Healthcare, Marken, World Courier, Kuehne+Nagel, FedEx, and Envirotainer.
- The market is moderately fragmented, and the leading player’s share is estimated in the mid-teens rather than at highly concentrated levels because route qualification is still split across global integrators, specialty medical couriers, packaging-validation firms, and cold-chain platform providers.
Quality assurance leaders within pharmaceutical companies are operating with increasingly limited tolerance for error when selecting specialized logistics partners. It is no longer sufficient to confirm that packaging maintains temperature integrity; firms must now document ambient conditions, transit delays, and handling practices at every stage of the journey. Any delay in executing comprehensive route mapping raises the risk of significant cargo loss and potential regulatory actions, including clinical holds. Notably, procurement teams often underestimate the importance of independent trade lane qualification services, which tend to carry greater weight in regulatory audits than internal performance metrics from freight partners.
As customs authorities begin to require detailed, node-level temperature records for import approvals, trade lane mapping is shifting from a best practice to a baseline compliance requirement. This development is placing pressure on mid-sized cold chain logistics providers, many of whom are absorbing the cost of rigorous route risk assessments to retain pharmaceutical clients. Companies that adopt full route transparency early are gaining an advantage, securing preferential handling arrangements at increasingly congested air cargo hubs.
API export growth is pushing capacity expansion faster in India, where the market is projected to grow at a CAGR of 9.6% through 2036 as suppliers work to meet stricter international compliance requirements. China remains close behind at 9.2%, supported by domestic distribution networks that are increasingly aligned with global good distribution practice standards. Advanced therapy transshipment is sustaining Singapore’s momentum, with growth reaching 8.9%, while South Korea is expected to expand at 8.7% as biologics logistics require tighter specialized handling. Belgium continues to benefit from its role as a major European entry point and is forecast to post 8.4% CAGR over the study period. Existing network density supports a steadier path in the United States and Germany, where growth rates of 7.8% and 7.5%, respectively, are reinforced by recurring requalification requirements across established logistics systems.
Segmental Analysis
Life Science Trade Lane Design and Qualification Services Market Analysis by Service Type
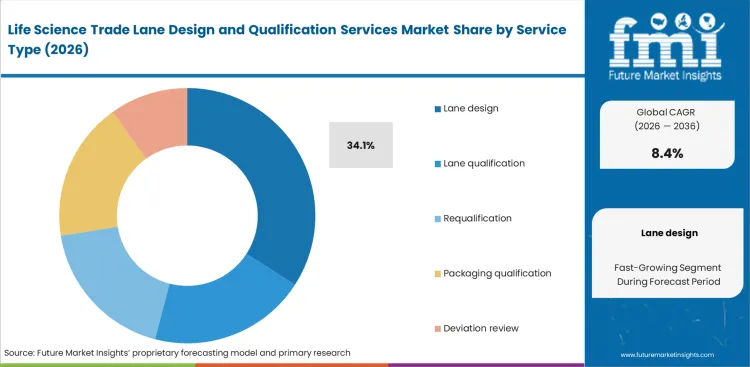
Initial route architectures tend to break down when shippers attempt to apply legacy clinical trial packaging to trade lanes that have not been properly mapped. Lane design is expected to account for 34.1% share of the market because it defines the compliance framework that shapes all downstream operations. Within biopharmaceutical firms, quality assurance leaders depend on specialized trade lane qualification providers to conduct detailed design assessments that identify thermal risks before any shipment is initiated. Early-stage hazard identification significantly reduces the likelihood of costly deviations during time-sensitive clinical trials. A common gap among sourcing teams is underestimating that effective lane design is built around thermal stability and predictable transit nodes, which ultimately underpin the broader trade lane design services market. Poorly structured routes often force manufacturers to rely on high-cost active containers to compensate for inherent transit weaknesses. Sponsors that bypass formal design phases typically encounter higher excursion rates and increased regulatory scrutiny.
- Initial hazard assessment: Procurement teams begin with baseline evaluations of transit nodes to screen out logistics providers that lack adequate tarmac handling capabilities. This step helps pharmaceutical manufacturers mitigate the risk of temperature excursions before committing to long-term freight contracts.
- Thermal mapping execution: Supply chain leaders assess key transit points against seasonal temperature variations to determine appropriate packaging configurations. Without this validation, products are exposed to conditions that can compromise their efficacy by the time they reach their destination.
- Standard operating procedure generation: Quality managers establish clear handling protocols for all stakeholders involved across approved routes. The absence of documented procedures creates immediate compliance gaps and often leads to failures during routine regulatory audits.
Life Science Trade Lane Design and Qualification Services Market Analysis by Transport Mode
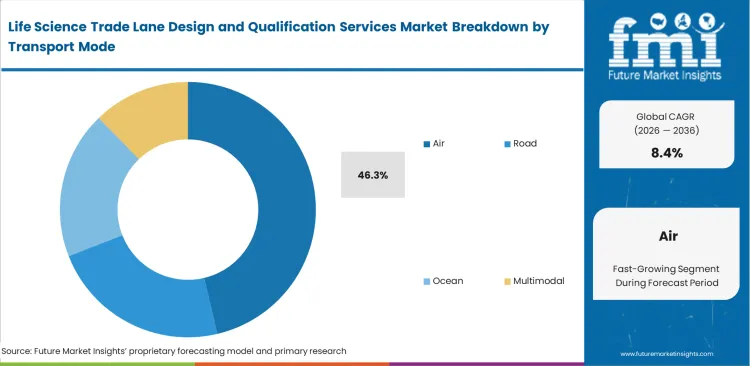
Intercontinental movement of high-value biologics requires speed, making lane qualification across multiple transfer points essential. Air freight remains the primary mode for global pharmaceutical and radiopharmaceutical logistics, holding an industry share of 46.3%, as it helps limit out-of-refrigeration exposure during transit. Logistics teams at global manufacturers depend on this mode to preserve product integrity within tight timelines. Lane qualification efforts are largely concentrated in air networks, where conditions vary widely, from freezing cargo holds to high heat during tarmac handling. Qualification is less about in-flight conditions and more about managing origin build-up, destination handling, and customs clearance delays. Teams working without specialized external support often underestimate variability in ground handling. Misjudging tarmac exposure windows can result in full shipment rejection and costly requalification.
- Tarmac delay mitigation: Logistics teams define transfer windows at congested hubs to reduce dependence on active containers. Extended delays increase leasing costs and put product stability at risk.
- Customs clearance prediction: Supply chain teams anticipate import hold times and adjust passive cooling levels accordingly. Overuse of coolant reduces payload efficiency and adds weight.
- Airline capability auditing: Quality teams verify whether carriers consistently meet required thermal handling standards before approving shipments. Selecting unverified carriers increases the risk of temperature excursions and insurance claims.
Life Science Trade Lane Design and Qualification Services Market Analysis by Temperature Range
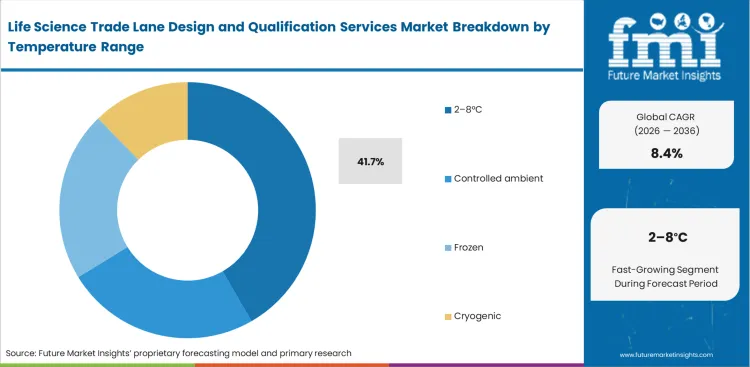
Strict control of refrigerated conditions, supported by temperature-controlled pharma packaging, continues to expand qualification requirements across established biologics portfolios, shaping demand within the GDP lane qualification services market. Most vaccines and monoclonal antibodies operate within a tightly defined 2–8°C range, making it the standard reference point for thermal compliance. Packaging engineers in biopharmaceutical companies devote significant effort to mapping trade lanes that can consistently maintain this narrow band. Even slight deviations trigger detailed quality investigations and, in many cases, lead to batch rejection. What is less apparent is that validating 2–8°C lanes is more complex than working with deep-frozen conditions, as shipments are exposed to both high and low ambient temperatures within the same journey. This requires packaging systems that can simultaneously protect against heat and cold. Without properly mapping these dual risks, products remain vulnerable to freezing during colder transit conditions. The segment accounts for a 41.7% share, reflecting its central role across routine biologics distribution. Its scale also means that even marginal improvements in lane stability can significantly reduce overall product loss and compliance risk.
- Winter freezing prevention: Packaging teams qualify phase change materials that prevent unintended freezing during low-temperature tarmac exposure. Once frozen, biologics undergo irreversible protein damage and cannot be recovered.
- Summer excursion modeling: Quality teams assess extreme heat exposure at key transit points to define safe shipment durations. Inadequate modeling of summer conditions leads to predictable thermal failures during peak periods.
- Multi-season validation: Supply chain teams conduct separate validation exercises for summer and winter to ensure consistent lane performance throughout the year. Skipping seasonal validation often forces shipment restrictions during extreme weather conditions.
Life Science Trade Lane Design and Qualification Services Market Analysis by End Use
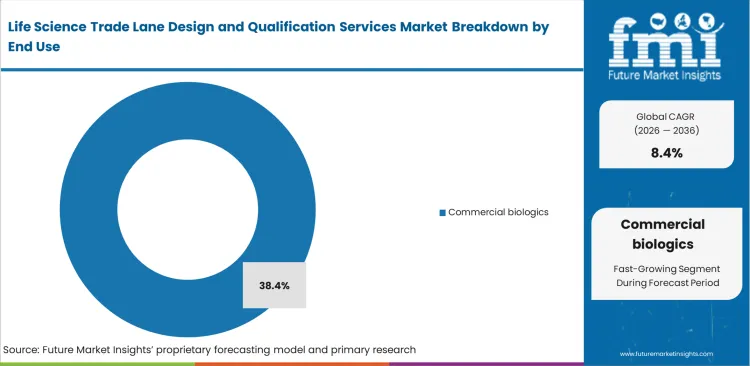
Global distribution volumes are pushing supply chains toward standardized routing structures, yet product-specific stability requirements continue to demand tailored handling protocols. Commercial biologics remain central to the global pharmaceutical movement because routine pharmacy replenishment depends on stable, fully validated international lanes, unlike clinical trial shipments or cell therapy programs that operate on temporary, route-specific designs. Supply chain leaders at major pharmaceutical companies depend on detailed lane mapping to preserve product shelf life through to delivery. The high financial value of these shipments supports ongoing requalification and strict node-level audits. Commercial biologics is anticipated to account for a 38.4% share, reflecting their dominance within continuous, high-volume distribution flows. A persistent operational challenge remains: commercial lanes frequently rely on shared freight networks, where forwarders consolidate multiple cold chain pallets, reducing direct control over transit conditions. This makes it a specific temperature-controlled packaging solution to validate how their packaging performs alongside other cargo. Overreliance on standard forwarder data increases the risk of gradual product degradation.
- High-volume throughput validation: Logistics teams assess major trade routes to confirm that ground handling facilities can manage multi-pallet volumes. Where origin infrastructure falls short, shipments must be split, increasing cost and operational complexity.
- Node capacity verification: Supply chain teams evaluate destination airport cold storage to ensure sufficient plug-in points for active containers. Lack of confirmed capacity at arrival can quickly lead to battery depletion and temperature excursions.
- Last-mile distribution mapping: Quality teams extend qualification beyond primary hubs to include regional trucking routes that deliver to wholesalers. Overlooking this final leg creates exposure, as a large share of temperature deviations occurs during last-mile transit.
Life Science Trade Lane Design and Qualification Services Market Analysis by Customer Type
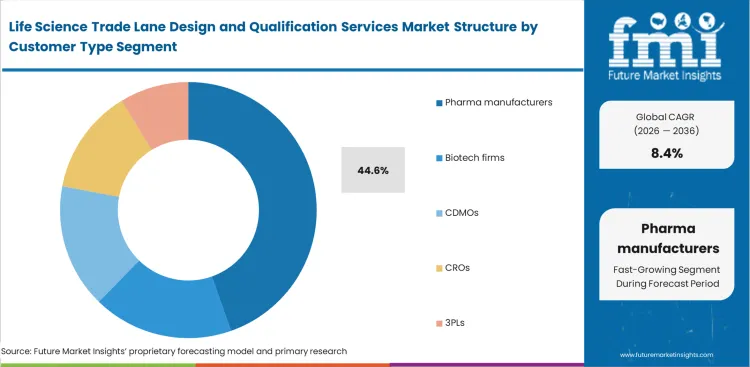
Primary drug sponsors constantly balance the cost of detailed route analysis against the regulatory risks of moving inadequately documented shipments. Pharmaceutical manufacturers, which estimates to account for a 44.6% share, remain at the center of this process as they carry ultimate legal responsibility for product quality under Good Distribution Practice requirements. Quality assurance leaders within these organizations require thorough lane qualification to meet the expectations of regulatory inspectors. Many sponsors choose to outsource these assessments to specialized GDP route validation providers, allowing them to access technical expertise while keeping internal focus on drug production. A less visible challenge is the lack of data sharing between pharmaceutical packagers and their contract partners, which leads to repeated qualification of the same trade lanes. This fragmented approach forces logistics providers to revalidate identical routes using slightly varied packaging configurations. Companies that rely solely on internal qualification teams often struggle to keep pace with ongoing changes in global aviation infrastructure, increasing the risk of gaps in transport readiness.
- Regulatory audit defense: Quality teams rely on independently generated route qualification reports to demonstrate structured risk management during regulatory inspections. Weak or incomplete documentation can lead to warning letters and, in severe cases, product recalls.
- Vendor agnostic assessment: Procurement teams engage independent consultants to evaluate logistics providers without commercial bias. Depending solely on forwarder-reported data can conceal recurring handling issues and delay corrective measures.
- New route establishment: Clinical supply teams initiate rapid lane assessments to support product launches in new geographic markets. Entering these markets without proper qualification increases the likelihood of customs delays or shipment rejection, disrupting launch timelines.
Life Science Trade Lane Design and Qualification Services Market Drivers, Restraints, and Opportunities
Strict enforcement of Good Distribution Practice guidelines compels quality assurance directors to document specific thermal risks across every international trade route. Regulatory agencies demand active transit data proving payload integrity from origin to destination, evaluating the best pharma lane qualification providers based on exact node adherence. This compliance threshold forces massive investment in shipping lane verification pharma to prevent catastrophic commercial hold orders. Relying on basic pharmaceutical container specifications without understanding transit node capabilities guarantees audit failures.
Inherent misalignment between logistics provider quality systems and pharmaceutical risk profiles severely limits rapid route qualification. Forwarders build generic transit lanes designed for broad cargo types, while drug manufacturers require product-specific risk assessments. Bridging this gap requires extensive custom auditing, forcing companies to navigate complex CEIV Pharma vs GDP qualification frameworks that slow down new market access. Standardized qualification templates fail constantly because no two biologic stability profiles behave identically under identical transit stress.
Opportunities in the Life Science Trade Lane Design and Qualification Services Market
- Digital twin simulation: Supply chain directors deploy virtual lane modeling to predict thermal excursions before physical shipments occur. Procurement teams can request a precise pharma shipping lane assessment quote to evaluate physical qualification costs and accelerate new route approvals.
- Shared audit frameworks: Quality managers participate in standardized cold chain logistics registries to eliminate redundant node mapping. Pooling qualification data drastically lowers compliance overhead for mid-tier biotech firms.
- Real-time excursion routing: Logistics coordinators integrate active tracking data with active lane profiles to reroute threatened payloads mid-transit. Active intervention prevents total batch loss during unexpected ground delays.
Regional Analysis
.webp)
Based on regional analysis, Life Science Trade Lane Design and Qualification Services is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East and Africa across 40 plus countries, each displaying a distinct pharma lane qualification CAGR based on regulatory maturity.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.6% |
| China | 9.2% |
| Singapore | 8.9% |
| South Korea | 8.7% |
| Belgium | 8.4% |
| United States | 7.8% |
| Germany | 7.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Life Science Trade Lane Design and Qualification Services Market Analysis
Rapid growth in pharmaceutical export manufacturing is driving structural upgrades across transit points that were previously less regulated. Quality assurance teams across different regions are working to align local distribution networks with the stricter import standards enforced in Europe and North America. Securing consistent and reliable lane data remains challenging due to fragmented ground handling capabilities and significant climate differences between origin and transshipment locations. There is increasing emphasis on qualifying alternative routes to mitigate recurring customs delays and ensure continuity of supply.
- India: India’s pharmaceutical export ecosystem is increasingly shaped by the compliance thresholds imposed by Western destination markets, particularly for high-volume active pharmaceutical ingredient flows. This pressure is translating into an estimated 9.6% CAGR through 2036 for the India pharma trade lane qualification sector, as manufacturers are forced to document every step from factory exit to final overseas handoff. Quality leaders are responding by intensifying audits of regional airport handlers and third-party ground agents. As transit nodes are upgraded and revalidated, Indian exporters are protecting access to premium, long-term commercial contracts with regulated buyers.
- China: Domestic pharmaceutical distribution in China is undergoing a fundamental reset as internal transport networks are brought into line with international good distribution practice expectations. Regional regulators are newly empowered, compelling supply chain teams to requalify legacy trucking corridors and introduce uninterrupted thermal tracking across every movement. This recalibration is rendering older transport models obsolete, accelerating replacement demand. China’s pharma lane qualification services market is expected to advance at 9.2% CAGR by 2036, driven by the urgency to keep domestic routes export-eligible.
- Singapore: Singapore’s role as a transshipment hub for advanced therapies places disproportionate weight on very short but high-risk airport ground transfers. Route qualification services in Singapore are expanding at an estimated 8.9% CAGR during the forecast, reflecting the level of scrutiny required even for minutes-long handoffs. Logistics teams meticulously record tarmac dwell times and validate movement paths between aircraft holds and controlled cold rooms. This precision-driven approach has cemented Singapore’s position as a preferred gateway for cellular and personalized medicine distribution across the Asia-Pacific.
- South Korea: Rising biologics complexity in South Korea is forcing a redesign of packaging strategies, with phase‑change materials tailored to withstand multi‑leg international routes. Sourcing teams increasingly favor partners that bring pre‑qualified lanes and validated infrastructure to the table, particularly for time‑sensitive clinical programs. Within this environment, South Korea is set to advance at an 8.7% CAGR during the assessment period, as exporters face mounting biosimilar competition and tighter delivery expectations. Speed to route qualification has consequently emerged as a decisive commercial advantage over neighboring manufacturing hubs.
FMI's report includes extensive analysis of emerging Southeast Asian transit corridors. Sustained investment in specialized airport facilities continually reshapes qualification requirements across diverse geographic corridors. Vietnam is also examined, as rapid expansion of pharmaceutical manufacturing and upgraded cargo infrastructure around Ho Chi Minh City and Hanoi increase the need for validated outbound routes.
Europe Life Science Trade Lane Design and Qualification Services Market Analysis
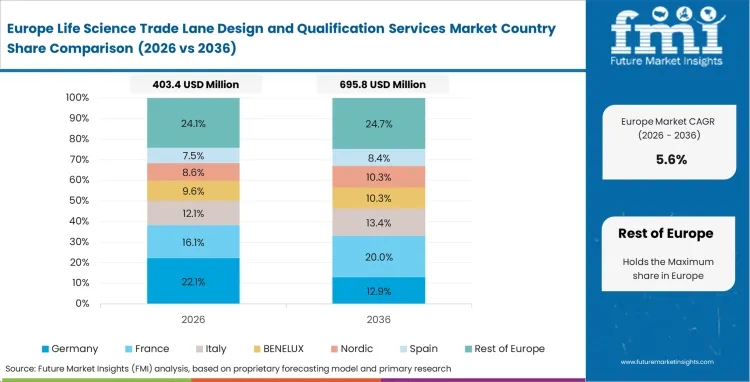
Legacy gateway infrastructure shifts the focus toward continuous requalification rather than initial route setup. Logistics teams operating across borders must comply with mature regulatory frameworks that require regular reassessment of existing trade lanes. Cross-border transit is now embedded within standard operating procedures, placing greater importance on advanced multimodal risk evaluation. Maintaining compliance in this environment depends on strong deviation tracking and review processes that can withstand detailed regulatory scrutiny.
- Belgium: Belgium’s long-standing position as a pharmaceutical airfreight gateway demands continual fine-tuning of ground operations as biologic shipment volumes climb. Supply chain leaders conduct detailed inspections of handling equipment to preserve uninterrupted movement between specialized pharma zones at major airports. Belgium pharma lane qualification sector is predicted to rise at an estimated 8.4% CAGR over the forecast, supported by operators that capitalize on decades of gateway reliability. Detailed node-level validation has effectively anchored Belgian forwarders deep within global life sciences supply chains.
- Germany: In Germany, the emphasis within pharma logistics has shifted toward periodic route requalification, particularly to account for subtle but recurring environmental changes. Quality teams focus heavily on winter trucking segments, running granular risk assessments to prevent temperature drift and excursion events. This disciplined approach translated to a moderate 7.5% CAGR expected during the forecast period in Germany’s pharma route validation sector which also reflects the stability of an already mature network. Overcoming local transit friction increasingly depends on highly specialized thermal packaging solutions rather than broad infrastructure expansion.
FMI's report includes broad coverage of Western and Eastern European distribution networks. Increasing reliance on rail and road integrations introduces fresh qualification challenges beyond traditional airfreight hubs, embedding reusable biologics cold chain shippers into routine operations. Poland also features in the analysis, where rising cross-border generics and biologics flows are accelerating the need for validated multimodal corridors connecting Central Europe to EU markets.
North America Life Science Trade Lane Design and Qualification Services Market Analysis
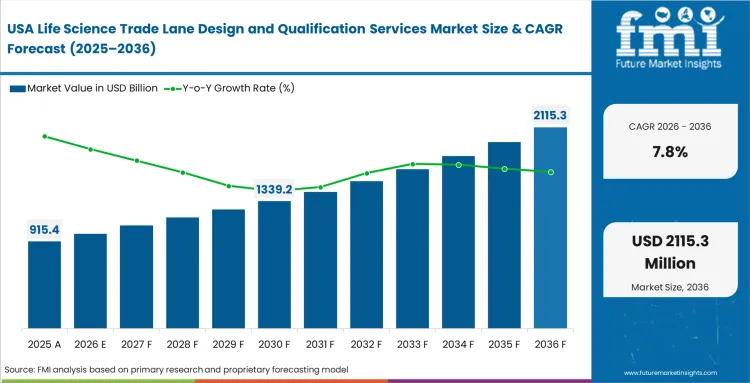
Large domestic markets with wide geographic spread require equally rigorous thermal mapping across diverse climate zones. Quality assurance teams oversee complex internal distribution networks that often mirror the challenges of international logistics. Strict oversight from federal regulators reinforces the need for ongoing route profiling and validation. Ensuring reliable movement across varied terrain depends on close coordination between transport providers and specialized quality partners.
- United States: The USA pharmaceutical logistics ecosystem is anchored in long‑established distribution corridors, where spending is increasingly directed toward replacement and requalification rather than greenfield expansion. Packaging engineers now routinely model extended domestic truck routes to anticipate severe summer heat exposure, particularly across southern transit lanes. Within this mature operating industry, expansion is predicted at 7.8% CAGR through 2036, supported by the sheer scale and frequency of internal biologics movements. Continuous refinement of baseline lane data has become critical, pushing providers to meet higher standards for specialized validation across nationwide networks.
FMI's report includes comprehensive assessments of Canadian and Mexican transit integrations. Consistent cross-border compliance relies entirely on harmonizing distinct national regulatory expectations into unified route profiles. Brazil is also examined in the report, where expanding biologics manufacturing and uneven internal infrastructure are driving demand for rigorously validated domestic and export lanes.
Competitive Aligners for Market Players
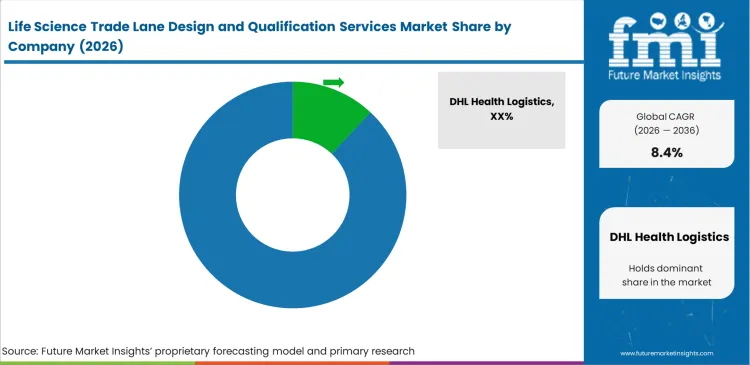
Specialized quality consultancies are increasingly positioning themselves against large logistics integrators by offering vendor-neutral route validation services. Companies such as DHL Health Logistics, UPS Healthcare, and Marken maintain strong market influence by combining transport capabilities with in-house compliance frameworks. Quality assurance leaders tend to assess these models based on their ability to withstand regulatory scrutiny rather than cost alone. Independent validation offers a clear separation of responsibilities, while integrated models can shorten timelines by aligning pharmaceutical cold-chain packaging qualification directly with transport execution.
Large global forwarders benefit from extensive proprietary datasets built on years of operational history across major trade routes. Firms such as World Courier, Kuehne+Nagel, and FedEx use detailed records of past temperature deviations to pre-qualify routes with a level of precision that independent consultants often cannot match without equivalent shipment volumes. For supply chain teams, partners that control physical infrastructure offer a practical advantage, as direct oversight of transit nodes simplifies subsequent risk assessments and supports GDP-aligned cold chain consulting.
Pharmaceutical manufacturers remain cautious about becoming too dependent on any single logistics provider. Many insist on qualification data that can be transferred across different forwarders without loss of validity. Companies like Envirotainer and other packaging specialists are influencing this shift by validating container performance alongside route conditions. This separation of packaging and transport responsibilities is forcing logistics providers to demonstrate that their handling processes deliver measurable improvements in product protection. The resulting tension between asset-heavy forwarders and independent quality assessors continues to shape procurement strategies across the sector.
Key Players in Life Science Trade Lane Design and Qualification Services Market
- DHL Health Logistics
- UPS Healthcare
- Marken
- World Courier
- Kuehne+Nagel
- FedEx
- Envirotainer
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.48 Billion in 2026 to USD 3.32 Billion by 2036, at a CAGR of 8.40% |
| Market Definition | This sector provides structural mapping, risk assessment, and validation of physical transport routes used for temperature-sensitive healthcare products. Service providers execute ambient profile analysis to establish standard operating procedures for specific pathways. |
| Segmentation | Service Type, Transport Mode, Temperature Range, End Use, Customer Type, Region |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Brazil, Mexico, Germany, United Kingdom, France, Spain, Italy, China, Japan, South Korea, India, Singapore, Australia, GCC Countries, South Africa |
| Key Companies Profiled | DHL Health Logistics, UPS Healthcare, Marken, World Courier, Kuehne+Nagel, FedEx, Envirotainer |
| Forecast Period | 2026 to 2036 |
| Approach | Annual lane assessment contracts signed by top-tier global forwarders. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Life Science Trade Lane Design and Qualification Services Market Analysis by Segments
Service Type
- Lane design
- Lane qualification
- Requalification
- Packaging qualification
- Deviation review
Transport Mode
- Air
- Road
- Ocean
- Multimodal
Temperature Range
- 2–8°C
- Controlled ambient
- Frozen
- Cryogenic
End Use
- Commercial biologics
- Clinical trials
- Vaccines
- Cell therapies
- Diagnostics
Customer Type
- Pharma manufacturers
- Biotech firms
- CDMOs
- CROs
- 3PLs
Region
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa
Bibliography
- Ciceri, C., Colicchia, C., Creazza, A., Dallari, F., & Melacini, M. (2025). Impact pathways: navigating risks in the pharmaceutical supply chain – a multi-tier, multi-regional perspective. International Journal of Operations & Production Management.
- European Medicines Agency. (2024, March). Good distribution practice for medicinal products for human use. EMA Europa.
- Federal Express Corporation. (2025, June 3). FedEx strengthens healthcare capabilities in Asia Pacific with CEIV Pharma certification.
- International Air Transport Association. (2025). IATA annual review 2025.
- Iyer, P., Jain, V., & Antony, J. (2025). Cold chain optimisation models: A systematic literature review from operational, sustainability and uncertainty lenses. International Journal of Production Economics, 286, 109689.
- Rong, Y., Yu, L., Liu, Y., Simic, V., & Pamucar, D. (2024). A pharmaceutical cold-chain logistics service quality model using a q-rung orthopair fuzzy framework with distance measure. Engineering Applications of Artificial Intelligence, 136, 109019.
- United States Pharmacopeia. (2024). India - Draft guidelines on good distribution practices for pharmaceutical products.
- WorldACD. (2025, December 19). Air cargo market data - November 2025.
- World Courier. (2024, August 12). Why quality matters when choosing a specialty logistics partner.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Outsourced qualification budgets specific to major pharmaceutical manufacturing centers.
- Node capacity verification protocols for highly congested global air cargo hubs.
- Standard operating procedure documentation requirements for Good Distribution Practice compliance.
- Tarmac delay mitigation strategies utilized by primary biologic sponsors.
- Baseline compliance requirements for emerging Asia Pacific transit corridors.
- Multi-season thermal mapping methodologies deployed across European and North American routes.
- Independent lane assessment capabilities compared against integrated freight forwarder services.
- Shared audit framework adoption rates among mid-tier biotechnology organizations.
Frequently Asked Questions
What limits rapid route qualification for clinical trials?
Inherent misalignment between logistics provider quality systems and pharmaceutical risk profiles severely delays validation. Forwarders build generic transit lanes designed for broad cargo types, while drug manufacturers require product-specific risk assessments before initiating sensitive trials.
Why does lane design hold significant share?
Initial route architecture establishes foundational compliance blueprints dictating all subsequent operational choices. Proactive hazard identification prevents costly shipment deviations and determines exact packaging requirements before physical transport begins.
How do tarmac delays impact air transit qualification?
Excessive delays drive up equipment rental fees and threaten payload viability. Logistics coordinators identify precise transfer windows at congested hubs to minimize active container leasing durations and maintain regulatory compliance.
What specific thermal hazard drives winter transit mapping?
Packaging engineers qualify phase change materials to prevent accidental payload freezing during cold-weather tarmac transfers. Frozen biologics suffer irreversible protein degradation and require immediate disposal.
Why do commercial biologics require continuous requalification?
Sheer financial value of commercial shipments justifies continuous route requalification. Commercial lanes often utilize shared freight networks where forwarders consolidate pallets, diluting direct shipper control over transit environments.
How do pharmaceutical manufacturers handle regulatory audits?
Quality directors utilize independent route mapping reports to demonstrate strict risk management during formal agency inspections. Insufficient documentation results in severe warning letters and potential product recalls.
What structural condition drives India's growth rate?
Massive active pharmaceutical ingredient export volumes hit stringent destination compliance gates, demanding rigorous origin-to-destination mapping. Upgrading these transit nodes secures highly lucrative commercial contracts with major Western buyers.
How do independent consultancies challenge global forwarders?
Specialized quality consultancies offer strictly vendor-agnostic route validation. Retaining independent auditors provides distinct separation of duties, ensuring qualification data remains untainted by freight execution incentives.
What specific capability do global forwarders possess?
Asset-heavy logistics integrators leverage granular operational history from thousands of historic temperature excursions to pre-qualify specific routes, a capability independent consultants struggle to replicate without actual physical shipment volumes.
How do shared audit frameworks benefit mid-tier biotech firms?
Quality managers participate in standardized registries to eliminate redundant node mapping. Pooling qualification data drastically lowers compliance overhead and removes massive barriers to international expansion.
Why are customs clearance delays modeled during qualification?
Supply chain directors map expected import holds to specify exact passive coolant quantities required for specific borders. Over-engineering passive systems severely limits volumetric payload capacity and increases overall shipping weight.
How does last-mile distribution impact commercial lanes?
Quality managers extend qualification protocols beyond major airports to cover regional trucking routes delivering direct to wholesalers. Ignoring final-mile logistics creates critical blind spots where majority of excursions actually occur.
What forces domestic trucking requalification in China?
Internal distribution networks rapidly align with global good distribution practice mandates. Supply chain managers mandate continuous thermal monitoring to satisfy newly empowered regional health authorities.
Why are customized phase-change profiles needed in South Korea?
Specialized biologic demands push packaging engineers to design complex multi-leg journeys supporting intense biosimilar export volumes. Securing qualified lanes provides distinct competitive advantages against neighboring manufacturing centers.
How does the United States manage sprawling geographic distances?
Packaging engineers map extensive domestic truck routes to anticipate extreme summer thermal hazards across southern corridors. Routine execution of these protocols directly shields manufacturers from catastrophic federal warning letters.
What drives demand for real-time excursion routing?
Logistics coordinators integrate active tracking data with active lane profiles to reroute threatened payloads mid-transit. Active intervention prevents total batch loss during unexpected ground delays.
How do airline capability audits protect payloads?
Quality managers verify carrier adherence to stated thermal protocols before authorizing high-value commercial shipments. Selecting unverified carriers leads to massive insurance claims following catastrophic temperature deviations.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Lane design
- Lane qualification
- Requalification
- Packaging qualification
- Deviation review
- Lane design
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Transport Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Transport Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Transport Mode, 2026 to 2036
- Air
- Road
- Ocean
- Multimodal
- Air
- Y to o to Y Growth Trend Analysis By Transport Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Transport Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Range
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Range, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Range, 2026 to 2036
- 2–8°C
- Controlled ambient
- Frozen
- Cryogenic
- 2–8°C
- Y to o to Y Growth Trend Analysis By Temperature Range, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Range, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Commercial biologics
- Commercial biologics
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- Pharma manufacturers
- Biotech firms
- CDMOs
- CROs
- 3PLs
- Pharma manufacturers
- Y to o to Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Transport Mode
- By Temperature Range
- By End Use
- By Customer Type
- Competition Analysis
- Competition Deep Dive
- DHL Health Logistics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- UPS Healthcare
- Marken
- World Courier
- Kuehne+Nagel
- FedEx
- Envirotainer
- DHL Health Logistics
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Transport Mode, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Temperature Range, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Transport Mode
- Figure 9: Global Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Temperature Range
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Customer Type
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Transport Mode
- Figure 35: North America Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Temperature Range
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Customer Type
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Transport Mode
- Figure 51: Latin America Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Temperature Range
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Customer Type
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Transport Mode
- Figure 67: Western Europe Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Temperature Range
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Transport Mode
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Temperature Range
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Transport Mode
- Figure 99: East Asia Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Temperature Range
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Customer Type
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Transport Mode
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Temperature Range
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Transport Mode, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Transport Mode, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Transport Mode
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Temperature Range, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Range, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Temperature Range
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

