Non-NGS Predictive Cancer Diagnostics Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Non-NGS Predictive Cancer Diagnostics Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Non-NGS Predictive Cancer Diagnostics Market Forecast and Outlook 2025 to 2035
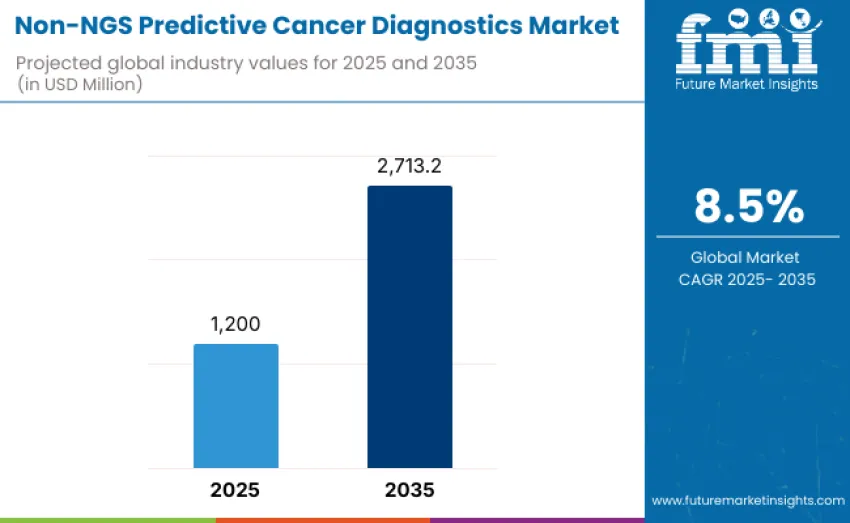
The global Non-NGS predictive cancer diagnostics market is projected to reach USD 2,713.2 million by 2035, recording an absolute increase of USD 1,513.2 million over the forecast period. This market is valued at USD 1,200 million in 2025 and is set to rise at a CAGR of 8.5% during the assessment period. The overall size is expected to grow by nearly 2.3 times, supported by increasing adoption of precision oncology solutions, rising demand for early cancer detection and risk prediction tools, and expanding healthcare infrastructure in emerging and developed regions.
Technological advancements in assay platforms, biomarker detection methods, and automated analysis systems are reshaping the non-NGS predictive diagnostics landscape. Modern tests incorporate multiplex immunoassays, PCR-based panels, and protein expression profiling that enable high-throughput, reliable, and reproducible results. Integration with hospital information systems, electronic health records, and clinical decision support platforms allows seamless workflow compatibility across oncology care teams. Advanced analytics and reporting capabilities provide clinicians with risk scores, predictive outcomes, and therapy guidance.
Quick Stats for Non-NGS Predictive Cancer Diagnostics Market
- Non-NGS Predictive Cancer Diagnostics Market Value (2025): USD 1,200 million
- Non-NGS Predictive Cancer Diagnostics Market Forecast Value (2035): USD 2,713.2 million
- Non-NGS Predictive Cancer Diagnostics Market Forecast CAGR: 8.5%
- Leading Product Type in Non-NGS Predictive Cancer Diagnostics Market: PCR-based companion diagnostics (30% share)
- Key Growth Regions in Non-NGS Predictive Cancer Diagnostics Market: Asia Pacific, Europe, and North America
- Top Players in Non-NGS Predictive Cancer Diagnostics Market: Roche Diagnostics, Abbott Laboratories, Thermo Fisher, Siemens Healthineers, Bio-Rad, Others
Government healthcare initiatives, cancer screening programs, and precision medicine policies accelerate growth. Educational and training programs for clinicians and laboratory personnel expand adoption by improving awareness of predictive diagnostics. Hospitals, diagnostic networks, and research institutions invest in non-NGS platforms to enhance diagnostic capacity and integrate risk prediction tools into standard clinical practice. Industry adoption of value-based care models drives sustained investment in predictive diagnostics that improve patient outcomes, optimize treatment costs, and enable data-driven cancer management.
Non-NGS Predictive Cancer Diagnostics Market Year-over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the Non-NGS predictive cancer diagnostics market is projected to expand from USD 1,200 million to USD 1,804.4 million, resulting in a value increase of USD 604.4 million, which represents 39.9% of total forecast growth for the decade. This phase will be shaped by rising demand for early cancer detection and risk prediction tools, increasing adoption of precision oncology solutions, product innovation in multiplex assay platforms, and expanding integration with hospital information systems and clinical decision support workflows.
From 2030 to 2035, growth is forecast from USD 1,804.4 million to USD 2,713.2 million, adding USD 908.8 million, which constitutes 60.1% of overall ten-year expansion. This period is expected to be characterized by introduction of advanced non-NGS predictive diagnostics with expanded biomarker panels, automated high-throughput testing systems, and integration with personalized treatment planning and precision medicine initiatives. Strategic collaborations between diagnostic companies and healthcare providers, along with increased government funding for cancer screening programs, will drive adoption.
Non-NGS Predictive Cancer Diagnostics Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 1,200 million |
| Market Forecast Value (2035) | USD 2,713.2 million |
| Forecast CAGR (2025 to 2035) | 8.5% |
Why Is the Non-NGS Predictive Cancer Diagnostics Market Growing Worldwide?
The Non-NGS predictive cancer diagnostics market grows by enabling healthcare providers to deliver personalized cancer care through rapid, reliable, and cost-effective predictive testing solutions. Oncologists and clinical laboratories face mounting pressure to accurately stratify patients, optimize therapy selection, and improve clinical outcomes within constrained treatment timelines, with non-NGS predictive diagnostics typically reducing decision-making time compared with conventional approaches. The rising prevalence of cancer and increasing emphasis on early detection drive demand for predictive diagnostics that provide actionable insights.
Government healthcare initiatives, cancer screening programs, and precision medicine policies accelerate adoption across hospitals, diagnostic laboratories, and specialized cancer centers. Educational programs and clinician training initiatives enhance awareness of predictive testing benefits, facilitating integration into standard clinical workflows. The global shift toward value-based care and patient-centric treatment models accelerates demand, as predictive diagnostics help reduce treatment delays, optimize therapy outcomes, and improve cost-effectiveness across diverse healthcare settings.
How Is the Non-NGS Predictive Cancer Diagnostics Market Segmented by Product Type, Application, and Region?
The Non-NGS predictive cancer diagnostics market is segmented by product type, application, and region. By product type, division includes PCR-based companion diagnostics, IHC & protein assays, proteomic assays, liquid biopsy non-NGS (qPCR), and others. Based on application, categorization covers therapy selection/companion Dx, prognostics, recurrence monitoring, and research use. Regionally, segmentation spans Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
By Product Type, PCR-Based Companion Diagnostics Account for Largest Share
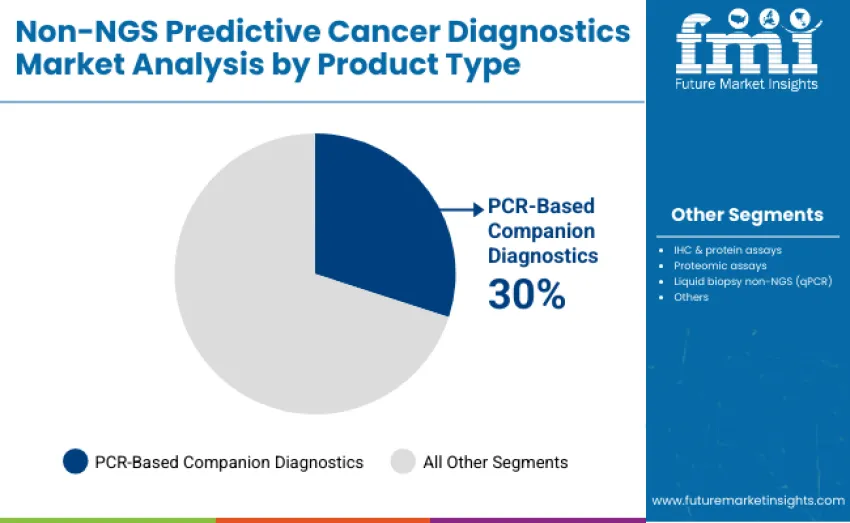
The PCR-based companion diagnostics segment represents the dominant force, capturing 30.0% of total share in 2025. This category includes high-sensitivity PCR assays used for therapy selection, mutation detection, and companion diagnostic purposes, forming a critical component of precision oncology workflows. Leadership stems from its essential role in enabling clinicians to identify actionable biomarkers, guide targeted therapies, and improve patient outcomes in a timely and cost-effective manner.
The IHC & protein assays segment holds a substantial 25.0% share, serving hospitals and diagnostic laboratories requiring protein expression profiling and immunohistochemistry-based analysis for prognostic and therapeutic decision-making. Liquid biopsy non-NGS (qPCR) accounts for 20.0%, offering minimally invasive testing options for therapy monitoring and early detection. Proteomic assays contribute 15.0%, while others represent 10.0% of revenue. Key advantages driving the PCR-based companion diagnostics segment include high analytical sensitivity and specificity for detecting clinically relevant mutations and biomarkers.
By Application, Therapy Selection/Companion Dx Leads
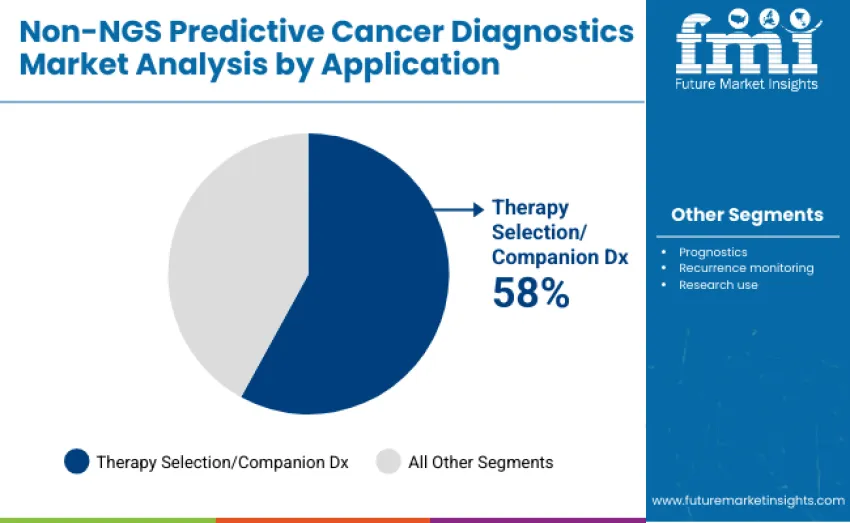
Therapy selection/companion Dx applications dominate with 58.0% share in 2025, reflecting the critical role of predictive diagnostics in guiding personalized cancer treatment. Leadership is reinforced by widespread adoption in hospitals, oncology clinics, and diagnostic laboratories where clinicians rely on companion diagnostics to identify actionable biomarkers, select targeted therapies, and optimize patient outcomes.
The prognostics segment represents 15.0% share, supporting risk assessment and treatment planning by providing predictive insights on disease progression and patient survival. Recurrence monitoring accounts for 12.0%, enabling early detection of cancer relapse and timely intervention. Research use contributes 15.0% of revenue, facilitating biomarker discovery, novel assay validation, and integration into clinical trial workflows. Key dynamics include therapy selection adoption driven by rising demand for precision oncology and personalized treatment strategies.
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Non-NGS Predictive Cancer Diagnostics Market?
The Non-NGS predictive cancer diagnostics market is driven by three concrete demand factors tied to precision oncology evolution and healthcare transformation. Rising cancer prevalence and aging populations create increasing requirements for early detection and risk prediction tools, with global cancer incidence expanding by 6-8% annually, requiring proven diagnostic technologies that support treatment decision-making in oncology, biomarker assessment, and therapeutic monitoring applications. Precision medicine adoption drives healthcare providers toward companion diagnostic investments, with 70-80% of oncology centers now integrating predictive testing that necessitates professional-grade non-NGS platforms.
What Are the Major Restraints Limiting the Adoption of Non-NGS Predictive Cancer Diagnostics?
Restraints include premium platform pricing creating adoption barriers, as professional laboratory systems with automation represent significant capital investments compared to basic testing equipment priced at one-fifth to one-tenth the cost of comprehensive diagnostic platforms. Technical complexity and workflow integration requirements pose challenges for laboratories transitioning from conventional testing, as non-NGS predictive diagnostic mastery requires extensive staff training to achieve proficiency levels comparable to traditional laboratory skills.
What Emerging Trends Are Shaping the Non-NGS Predictive Cancer Diagnostics Market?
Key trends indicate rapid adoption of diagnostic platforms in Asia-Pacific, particularly in China and India. Government programs supporting healthcare modernization and the expansion of precision medicine are driving this shift, with funding subsidies and training grants incentivizing healthcare providers to adopt advanced diagnostic technologies. These initiatives aim to enhance healthcare infrastructure, improve diagnostic accuracy, and support professional development. As a result, both countries are emerging as key growth hubs for diagnostic platform adoption in the region, fostering widespread industry advancement.
How Are Leading Countries Driving Growth in the Non-NGS Predictive Cancer Diagnostics Market?
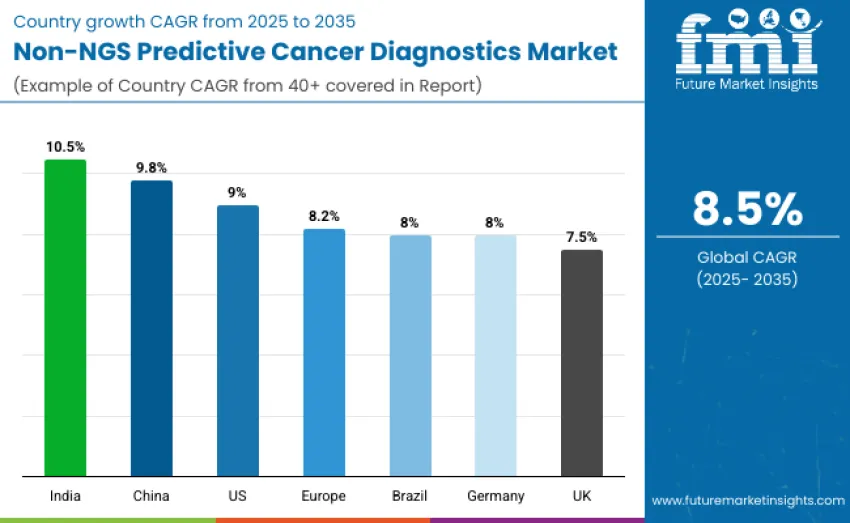
| Country | CAGR (2025 to 2035)% |
|---|---|
| United States | 9.0 |
| Brazil | 8.0 |
| China | 9.8 |
| India | 10.5 |
| Europe | 8.2 |
| Germany | 8.0 |
| United Kingdom | 7.5 |
The Non-NGS predictive cancer diagnostics market is expanding globally, with India leading at a 10.5% CAGR, driven by growing precision oncology adoption, government screening programs, and increased investment in diagnostic infrastructure. China follows at 9.8%, benefiting from rising healthcare expenditure and rapid hospital network expansion. Germany records 8.0% growth, supported by clinical excellence and targeted oncology initiatives. Brazil grows at 8.0%, driven by expanding cancer care and early detection programs. The USA sees 9.0% growth, while Europe grows at 8.2%, with the UK at 7.5%, supported by national cancer strategies and predictive diagnostics integration.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Is Healthcare Infrastructure Accelerating Growth in China?
China demonstrates the strongest growth potential with a CAGR of 9.8% through 2035. Leadership stems from comprehensive precision oncology initiatives, expanding hospital and diagnostic laboratory networks, and government cancer screening programs driving adoption of predictive diagnostics. Growth is concentrated in major healthcare hubs, including Beijing, Shanghai, Shenzhen, and Guangzhou, where leading hospitals, cancer centers, and diagnostic laboratories are implementing PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS platforms to support therapy selection.
Distribution channels include hospital networks, commercial diagnostic laboratories, and direct manufacturer partnerships, enabling wide deployment across clinical and research applications. Government healthcare policies, including funding for laboratory infrastructure, training programs, and precision medicine initiatives, provide support for adoption of non-NGS predictive diagnostics. Expansion of oncology care infrastructure includes increasing numbers of tertiary hospitals and specialized cancer centers deploying non-NGS predictive diagnostics across major provinces. Government support through healthcare modernization programs provides funding assistance for diagnostic infrastructure.
Why Are Healthcare Infrastructure and Precision Medicine Programs Enhancing Adoption in India?
The Non-NGS predictive cancer diagnostics market demonstrates strong growth momentum with a CAGR of 10.5% through 2035, linked to rising cancer prevalence, expanding hospital networks, and increasing investment in early detection and risk prediction capabilities. Indian healthcare providers are implementing PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS workflows to support therapy selection, prognostics, and recurrence monitoring, improving patient outcomes while meeting global oncology standards. Leading healthcare clusters, including Bangalore, Hyderabad, Mumbai, and Delhi, drive diagnostic platform adoption.
Government incentive programs enable funding support for laboratory infrastructure, assay deployment, and personnel training through national and state healthcare schemes. Technology collaboration agreements accelerate adoption with international and domestic diagnostic manufacturers establishing distribution and technical support networks. Policy support through Digital India health initiatives and precision medicine programs provides access to advanced diagnostics, clinical training, and laboratory modernization resources. The country's precision oncology and cancer screening programs create sustained demand for non-NGS predictive diagnostics.
How Does Healthcare Infrastructure and Oncology Expertise Sustain Leadership in Germany?
Germany's advanced healthcare system demonstrates sophisticated implementation of non-NGS predictive cancer diagnostics, with clinical studies showing improvement in diagnostic workflow efficiency through adoption of PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS platforms across hospitals, cancer centers, and specialized laboratories. Healthcare infrastructure in major medical hubs, including Berlin, Munich, Hamburg, and Frankfurt, showcases integration of predictive diagnostics into clinical workflows, leveraging expertise in precision medicine, oncology research, and standardized laboratory practices.
German healthcare providers emphasize diagnostic accuracy, reliability, and regulatory compliance, creating demand for premium, high-performance non-NGS assays that support patient management and meet rigorous clinical standards. Growth maintains strong momentum through focus on hospital and specialty oncology segments, with a CAGR of 8.0% through 2035. Specialized oncology centers implement predictive diagnostic workflows with comprehensive staff training programs. Strategic partnerships between European distributors and diagnostic manufacturers ensure reliable product availability and technical support services.
Why Is Expanding Oncology Infrastructure Fueling Demand in Brazil?
The Brazilian Non-NGS predictive cancer diagnostics market leads Latin American adoption, driven by expanding hospital networks, oncology centers, and increasing cancer screening initiatives supporting early detection and personalized therapy programs. Growth shows solid potential with a CAGR of 8.0% through 2035, fueled by healthcare modernization efforts and rising demand for precision oncology solutions across São Paulo, Rio de Janeiro, Brasília, and Porto Alegre metropolitan areas. Brazilian healthcare providers are adopting PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS platforms for therapy selection.
Distribution channels through regional hospital networks, commercial laboratories, and direct manufacturer partnerships expand coverage across public and private healthcare facilities. Hospitals and oncology centers implement predictive diagnostic workflows for patient stratification, therapy selection, and clinical monitoring. Technology partnerships with international and domestic diagnostic manufacturers enable Brazilian access through localized pricing strategies and financing options for laboratory equipment. Strategic collaborations between healthcare associations and diagnostic suppliers enable group procurement programs and professional training initiatives.
How Do Established Healthcare Systems Strengthen Performance in the United States?
The USA Non-NGS predictive cancer diagnostics market demonstrates mature implementation focused on large hospital networks, specialty oncology centers, and independent clinical laboratories, with documented integration of PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS platforms across major healthcare hubs including California, New York, Texas, and Massachusetts. Growth maintains steady momentum with a CAGR of 9.0% through 2035, driven by high cancer prevalence, advanced precision medicine initiatives, and a well-established clinical diagnostics ecosystem.
Major diagnostic manufacturers maintain headquarters and development operations in the USA, creating strong integration with clinical laboratory workflows and electronic health record systems to optimize diagnostic efficiency and reporting accuracy. Large hospital and oncology center adoption includes standardized testing protocols ensuring consistent workflow capabilities, regulatory compliance, and clinical accuracy. Established distribution infrastructure through regional diagnostic networks, commercial laboratories, and direct manufacturer partnerships provides reliable product supply, training, and technical support.
How Is Healthcare Infrastructure and Precision Medicine Expansion Supporting Growth in the United Kingdom?
In London, Manchester, Bristol, and Edinburgh healthcare clusters, hospitals, oncology centers, and diagnostic laboratories are implementing non-NGS predictive cancer diagnostics workflows, including PCR-based companion diagnostics, IHC & protein assays, and liquid biopsy non-NGS platforms, to support therapy selection, prognostics, and recurrence monitoring. Growth shows solid potential with a CAGR of 7.5% through 2035, linked to national precision medicine initiatives, increasing cancer screening programs, and growing emphasis on clinical laboratory workforce development.
Hospital and oncology center adoption across public and private healthcare systems implements standardized diagnostic workflows with staff training and assay validation programs. Educational partnerships between clinical laboratories, medical schools, and diagnostic manufacturers provide training and technical support for laboratory personnel. Strategic collaborations between healthcare associations and technology suppliers enable product validation, professional feedback integration, and innovation in assay development. Emphasis on accuracy, reliability, and workflow efficiency drives preference for high-performance non-NGS diagnostics.
How Are Healthcare Infrastructure Investments Advancing Expansion Across Europe?
The Non-NGS predictive cancer diagnostics market in Europe is projected to grow from USD 360 million in 2025 to USD 759.7 million by 2035, registering a CAGR of 8.9% over the forecast period. Germany maintains leadership with a 22.5% share in 2025, declining to 21.0% by 2035, supported by advanced healthcare infrastructure, precision oncology programs, and established hospital and laboratory networks. France follows with a 16.5% share in 2025, reaching 15.5% by 2035, driven by national cancer screening initiatives.
Italy commands an 11.5% share in 2025, decreasing to 11.0% by 2035, backed by regional diagnostic center development and public-private collaborations in oncology care. Spain accounts for 8.5% in 2025, declining to 8.0% by 2035 due to consolidation of diagnostic services. The BENELUX region grows from 6.5% in 2025 to 7.5% in 2035, reflecting focused investments in laboratory modernization. Nordic countries hold 8.5% in 2025, rising to 9.5% by 2035, supported by government cancer programs.
How Competitive Is the Non-NGS Predictive Cancer Diagnostics Market and What Defines Its Structure?
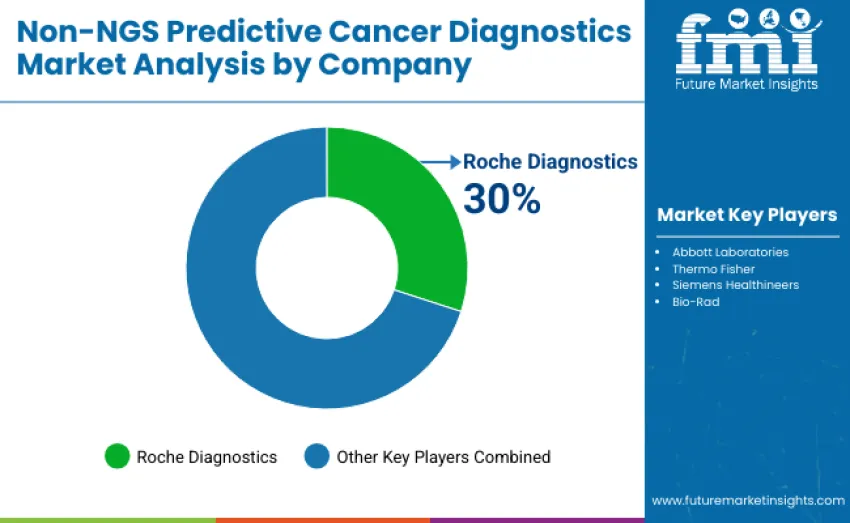
The Non-NGS predictive cancer diagnostics market is moderately concentrated, with 12-15 players. The top five companies collectively control 45% of the global market share. Roche Diagnostics leads with a 30% share, supported by its broad product offering and established leadership in clinical diagnostics. Competition primarily revolves around assay accuracy, platform integration, turnaround time, and regulatory compliance, with less emphasis on price competition.
Other major players, including Abbott Laboratories, Thermo Fisher, Siemens Healthineers, and Bio-Rad, maintain competitive advantages through proprietary assay technologies, extensive clinical validation, and strong relationships with hospitals, oncology centers, and research institutions worldwide. These companies continually invest in research and development to enhance assay sensitivity, multiplexing capabilities, automation systems, and laboratory workflow integration, defending their positions while expanding into emerging clinical and research applications.
Challengers and niche players target specific diagnostic segments or regions, offering differentiated capabilities such as liquid biopsy non-NGS platforms, proteomic assays, and companion diagnostics for therapy selection, prognostics, and recurrence monitoring. Emerging manufacturers from Asia, particularly in India, China, and Latin America, exert competitive pressure through aggressive pricing strategies and rapid product development, catering to cost-sensitive markets while demanding high diagnostic accuracy.
Key Players in the Non-NGS Predictive Cancer Diagnostics Market
- Roche Diagnostics
- Abbott Laboratories
- Thermo Fisher
- Siemens Healthineers
- Bio-Rad
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 1,200 million |
| Product Type | PCR-based companion diagnostics, IHC & protein assays, Proteomic assays, Liquid biopsy non-NGS (qPCR) and Others |
| Application | Therapy selection/companion Dx, Prognostics, Recurrence monitoring and Research use |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | Roche Diagnostics, Abbott Laboratories, Thermo Fisher, Siemens Healthineers , Bio-Rad, Others |
| Additional Attributes | Dollar sales by product type and application, regional trends across Asia Pacific, Europe, North America, Latin America, and Middle East & Africa, competitive landscape of diagnostic companies, technical specifications, integration with LIMS, innovations in non-NGS assay technologies, and rapid diagnostic platform development |
Non-NGS Predictive Cancer Diagnostics Market by Segments
By Product Type:
- PCR-based companion diagnostics
- IHC & protein assays
- Proteomic assays
- Liquid biopsy non-NGS (qPCR)
- Others
By Application:
- Therapy selection / companion Dx
- Prognostics
- Recurrence monitoring
- Research use
By Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the Non-NGS Predictive Cancer Diagnostics Market in 2025?
The global Non-NGS predictive cancer diagnostics market is valued at USD 1,200 million in 2025.
What will be the size of the Non-NGS Predictive Cancer Diagnostics Market in 2035?
The market is projected to reach USD 2,713.2 million by 2035.
How fast will the Non-NGS Predictive Cancer Diagnostics Market grow between 2025 and 2035?
The market will grow at a CAGR of 8.5% from 2025 to 2035.
Which product type leads the Non-NGS Predictive Cancer Diagnostics Market in 2025?
PCR-based companion diagnostics lead the market with a 30% share in 2025.
Who are the major players in the Non-NGS Predictive Cancer Diagnostics Market?
Key players include Roche Diagnostics, Abbott Laboratories, Thermo Fisher, Siemens Healthineers, Bio-Rad, and others.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assessment
- Products Market Assessment
- Value Chain Analysis
- Participants and their Roles
- Raw Material Suppliers
- Manufacturers
- Mid-Level Participants
- Wholesalers and Distributors
- Profit Margin Analysis
- Operating Margins By Region
- Notes on Value Added at Each Node in the Value Chain
- Participants and their Roles
- Strategic Divestments/ Investments
- Market Trade Assessment
- Import-Export Analysis
- Supply Demand Gap Assessment
- White -Space Opportunity Assessment and Mapping
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Product Launches & Development
- Technological Advancement
- Market Dynamics
- Regional Level Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size USD Million Analysis, 2020 to 2024
- Current and Future Market Size USD Million Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Product Type, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Product Type, 2025 to 2035
- PCR-based companion diagnostics
- IHC & protein assays
- Proteomic assays
- Liquid biopsy non-NGS (qPCR)
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By Application, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By Application, 2025 to 2035
- Therapy selection / companion Dx
- Prognostics
- Recurrence monitoring
- Research use
- Y-o-Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million Analysis By End User, 2020 to 2024
- Current and Future Market Size USD Million Analysis and Forecast By End User, 2025 to 2035
- Hospitals & Oncology centers
- Diagnostic labs
- Academic R&D
- POC/Community
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Regional Level Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size USD Million Analysis By Region, 2020 to 2024
- Current Market Size USD Million Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size USD Million Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size USD Million Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Roche Diagnostics
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Abbott Laboratories
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Thermo Fisher
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Siemens Healthineers
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Bio-Rad
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Roche Diagnostics
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million), By Product Type, 2020-2035
- Table 2: Global Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 3: Global Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 4: Global Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 5: Global Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 6: Global Market Size (USD Million), by Others, 2020-2035
- Table 7: Global Market Size (USD Million), by Application, 2020-2035
- Table 8: Global Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 9: Global Market Size (USD Million), by Prognostics, 2020-2035
- Table 10: Global Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 11: Global Market Size (USD Million), by Research use, 2020-2035
- Table 12: Global Market Size (USD Million), by End User, 2020-2035
- Table 13: Global Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 14: Global Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 15: Global Market Size (USD Million), by Academic R&D, 2020-2035
- Table 16: Global Market Size (USD Million), by POC/Community, 2020-2035
- Table 17: North America Market Size (USD Million), By Product Type, 2020-2035
- Table 18: North America Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 19: North America Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 20: North America Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 21: North America Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 22: North America Market Size (USD Million), by Others, 2020-2035
- Table 23: North America Market Size (USD Million), by Application, 2020-2035
- Table 24: North America Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 25: North America Market Size (USD Million), by Prognostics, 2020-2035
- Table 26: North America Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 27: North America Market Size (USD Million), by Research use, 2020-2035
- Table 28: North America Market Size (USD Million), by End User, 2020-2035
- Table 29: North America Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 30: North America Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 31: North America Market Size (USD Million), by Academic R&D, 2020-2035
- Table 32: North America Market Size (USD Million), by POC/Community, 2020-2035
- Table 33: USA Market Size (USD Million), By Product Type, 2020-2035
- Table 34: USA Market Size (USD Million), by Application, 2020-2035
- Table 35: USA Market Size (USD Million), by End User, 2020-2035
- Table 36: Canada Market Size (USD Million), By Product Type, 2020-2035
- Table 37: Canada Market Size (USD Million), by Application, 2020-2035
- Table 38: Canada Market Size (USD Million), by End User, 2020-2035
- Table 39: Mexico Market Size (USD Million), By Product Type, 2020-2035
- Table 40: Mexico Market Size (USD Million), by Application, 2020-2035
- Table 41: Mexico Market Size (USD Million), by End User, 2020-2035
- Table 42: Latin America Market Size (USD Million), By Product Type, 2020-2035
- Table 43: Latin America Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 44: Latin America Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 45: Latin America Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 46: Latin America Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 47: Latin America Market Size (USD Million), by Others, 2020-2035
- Table 48: Latin America Market Size (USD Million), by Application, 2020-2035
- Table 49: Latin America Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 50: Latin America Market Size (USD Million), by Prognostics, 2020-2035
- Table 51: Latin America Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 52: Latin America Market Size (USD Million), by Research use, 2020-2035
- Table 53: Latin America Market Size (USD Million), by End User, 2020-2035
- Table 54: Latin America Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 55: Latin America Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 56: Latin America Market Size (USD Million), by Academic R&D, 2020-2035
- Table 57: Latin America Market Size (USD Million), by POC/Community, 2020-2035
- Table 58: Brazil Market Size (USD Million), By Product Type, 2020-2035
- Table 59: Brazil Market Size (USD Million), by Application, 2020-2035
- Table 60: Brazil Market Size (USD Million), by End User, 2020-2035
- Table 61: Argentina Market Size (USD Million), By Product Type, 2020-2035
- Table 62: Argentina Market Size (USD Million), by Application, 2020-2035
- Table 63: Argentina Market Size (USD Million), by End User, 2020-2035
- Table 64: Rest of Latin America Market Size (USD Million), By Product Type, 2020-2035
- Table 65: Rest of Latin America Market Size (USD Million), by Application, 2020-2035
- Table 66: Rest of Latin America Market Size (USD Million), by End User, 2020-2035
- Table 67: East Asia Market Size (USD Million), By Product Type, 2020-2035
- Table 68: East Asia Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 69: East Asia Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 70: East Asia Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 71: East Asia Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 72: East Asia Market Size (USD Million), by Others, 2020-2035
- Table 73: East Asia Market Size (USD Million), by Application, 2020-2035
- Table 74: East Asia Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 75: East Asia Market Size (USD Million), by Prognostics, 2020-2035
- Table 76: East Asia Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 77: East Asia Market Size (USD Million), by Research use, 2020-2035
- Table 78: East Asia Market Size (USD Million), by End User, 2020-2035
- Table 79: East Asia Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 80: East Asia Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 81: East Asia Market Size (USD Million), by Academic R&D, 2020-2035
- Table 82: East Asia Market Size (USD Million), by POC/Community, 2020-2035
- Table 83: China Market Size (USD Million), By Product Type, 2020-2035
- Table 84: China Market Size (USD Million), by Application, 2020-2035
- Table 85: China Market Size (USD Million), by End User, 2020-2035
- Table 86: Japan Market Size (USD Million), By Product Type, 2020-2035
- Table 87: Japan Market Size (USD Million), by Application, 2020-2035
- Table 88: Japan Market Size (USD Million), by End User, 2020-2035
- Table 89: South Korea Market Size (USD Million), By Product Type, 2020-2035
- Table 90: South Korea Market Size (USD Million), by Application, 2020-2035
- Table 91: South Korea Market Size (USD Million), by End User, 2020-2035
- Table 92: South Asia Market Size (USD Million), By Product Type, 2020-2035
- Table 93: South Asia Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 94: South Asia Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 95: South Asia Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 96: South Asia Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 97: South Asia Market Size (USD Million), by Others, 2020-2035
- Table 98: South Asia Market Size (USD Million), by Application, 2020-2035
- Table 99: South Asia Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 100: South Asia Market Size (USD Million), by Prognostics, 2020-2035
- Table 101: South Asia Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 102: South Asia Market Size (USD Million), by Research use, 2020-2035
- Table 103: South Asia Market Size (USD Million), by End User, 2020-2035
- Table 104: South Asia Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 105: South Asia Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 106: South Asia Market Size (USD Million), by Academic R&D, 2020-2035
- Table 107: South Asia Market Size (USD Million), by POC/Community, 2020-2035
- Table 108: India Market Size (USD Million), By Product Type, 2020-2035
- Table 109: India Market Size (USD Million), by Application, 2020-2035
- Table 110: India Market Size (USD Million), by End User, 2020-2035
- Table 111: ASEAN Countries Market Size (USD Million), By Product Type, 2020-2035
- Table 112: ASEAN Countries Market Size (USD Million), by Application, 2020-2035
- Table 113: ASEAN Countries Market Size (USD Million), by End User, 2020-2035
- Table 114: Australia & New Zealand Market Size (USD Million), By Product Type, 2020-2035
- Table 115: Australia & New Zealand Market Size (USD Million), by Application, 2020-2035
- Table 116: Australia & New Zealand Market Size (USD Million), by End User, 2020-2035
- Table 117: Rest of South Asia Market Size (USD Million), By Product Type, 2020-2035
- Table 118: Rest of South Asia Market Size (USD Million), by Application, 2020-2035
- Table 119: Rest of South Asia Market Size (USD Million), by End User, 2020-2035
- Table 120: Western Europe Market Size (USD Million), By Product Type, 2020-2035
- Table 121: Western Europe Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 122: Western Europe Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 123: Western Europe Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 124: Western Europe Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 125: Western Europe Market Size (USD Million), by Others, 2020-2035
- Table 126: Western Europe Market Size (USD Million), by Application, 2020-2035
- Table 127: Western Europe Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 128: Western Europe Market Size (USD Million), by Prognostics, 2020-2035
- Table 129: Western Europe Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 130: Western Europe Market Size (USD Million), by Research use, 2020-2035
- Table 131: Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 132: Western Europe Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 133: Western Europe Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 134: Western Europe Market Size (USD Million), by Academic R&D, 2020-2035
- Table 135: Western Europe Market Size (USD Million), by POC/Community, 2020-2035
- Table 136: Germany Market Size (USD Million), By Product Type, 2020-2035
- Table 137: Germany Market Size (USD Million), by Application, 2020-2035
- Table 138: Germany Market Size (USD Million), by End User, 2020-2035
- Table 139: UK Market Size (USD Million), By Product Type, 2020-2035
- Table 140: UK Market Size (USD Million), by Application, 2020-2035
- Table 141: UK Market Size (USD Million), by End User, 2020-2035
- Table 142: France Market Size (USD Million), By Product Type, 2020-2035
- Table 143: France Market Size (USD Million), by Application, 2020-2035
- Table 144: France Market Size (USD Million), by End User, 2020-2035
- Table 145: Italy Market Size (USD Million), By Product Type, 2020-2035
- Table 146: Italy Market Size (USD Million), by Application, 2020-2035
- Table 147: Italy Market Size (USD Million), by End User, 2020-2035
- Table 148: Spain Market Size (USD Million), By Product Type, 2020-2035
- Table 149: Spain Market Size (USD Million), by Application, 2020-2035
- Table 150: Spain Market Size (USD Million), by End User, 2020-2035
- Table 151: BENELUX Market Size (USD Million), By Product Type, 2020-2035
- Table 152: BENELUX Market Size (USD Million), by Application, 2020-2035
- Table 153: BENELUX Market Size (USD Million), by End User, 2020-2035
- Table 154: Nordic Countries Market Size (USD Million), By Product Type, 2020-2035
- Table 155: Nordic Countries Market Size (USD Million), by Application, 2020-2035
- Table 156: Nordic Countries Market Size (USD Million), by End User, 2020-2035
- Table 157: Rest of Western Europe Market Size (USD Million), By Product Type, 2020-2035
- Table 158: Rest of Western Europe Market Size (USD Million), by Application, 2020-2035
- Table 159: Rest of Western Europe Market Size (USD Million), by End User, 2020-2035
- Table 160: Eastern Europe Market Size (USD Million), By Product Type, 2020-2035
- Table 161: Eastern Europe Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 162: Eastern Europe Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 163: Eastern Europe Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 164: Eastern Europe Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 165: Eastern Europe Market Size (USD Million), by Others, 2020-2035
- Table 166: Eastern Europe Market Size (USD Million), by Application, 2020-2035
- Table 167: Eastern Europe Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 168: Eastern Europe Market Size (USD Million), by Prognostics, 2020-2035
- Table 169: Eastern Europe Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 170: Eastern Europe Market Size (USD Million), by Research use, 2020-2035
- Table 171: Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 172: Eastern Europe Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 173: Eastern Europe Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 174: Eastern Europe Market Size (USD Million), by Academic R&D, 2020-2035
- Table 175: Eastern Europe Market Size (USD Million), by POC/Community, 2020-2035
- Table 176: Russia Market Size (USD Million), By Product Type, 2020-2035
- Table 177: Russia Market Size (USD Million), by Application, 2020-2035
- Table 178: Russia Market Size (USD Million), by End User, 2020-2035
- Table 179: Hungary Market Size (USD Million), By Product Type, 2020-2035
- Table 180: Hungary Market Size (USD Million), by Application, 2020-2035
- Table 181: Hungary Market Size (USD Million), by End User, 2020-2035
- Table 182: Poland Market Size (USD Million), By Product Type, 2020-2035
- Table 183: Poland Market Size (USD Million), by Application, 2020-2035
- Table 184: Poland Market Size (USD Million), by End User, 2020-2035
- Table 185: Rest of Eastern Europe Market Size (USD Million), By Product Type, 2020-2035
- Table 186: Rest of Eastern Europe Market Size (USD Million), by Application, 2020-2035
- Table 187: Rest of Eastern Europe Market Size (USD Million), by End User, 2020-2035
- Table 188: Middle East & Africa Market Size (USD Million), By Product Type, 2020-2035
- Table 189: Middle East & Africa Market Size (USD Million), by PCR-based companion diagnostics, 2020-2035
- Table 190: Middle East & Africa Market Size (USD Million), by IHC & protein assays, 2020-2035
- Table 191: Middle East & Africa Market Size (USD Million), by Proteomic assays, 2020-2035
- Table 192: Middle East & Africa Market Size (USD Million), by Liquid biopsy non-NGS (qPCR), 2020-2035
- Table 193: Middle East & Africa Market Size (USD Million), by Others, 2020-2035
- Table 194: Middle East & Africa Market Size (USD Million), by Application, 2020-2035
- Table 195: Middle East & Africa Market Size (USD Million), by Therapy selection / companion Dx, 2020-2035
- Table 196: Middle East & Africa Market Size (USD Million), by Prognostics, 2020-2035
- Table 197: Middle East & Africa Market Size (USD Million), by Recurrence monitoring, 2020-2035
- Table 198: Middle East & Africa Market Size (USD Million), by Research use, 2020-2035
- Table 199: Middle East & Africa Market Size (USD Million), by End User, 2020-2035
- Table 200: Middle East & Africa Market Size (USD Million), by Hospitals & Oncology centers, 2020-2035
- Table 201: Middle East & Africa Market Size (USD Million), by Diagnostic labs, 2020-2035
- Table 202: Middle East & Africa Market Size (USD Million), by Academic R&D, 2020-2035
- Table 203: Middle East & Africa Market Size (USD Million), by POC/Community, 2020-2035
- Table 204: Saudi Arabia Market Size (USD Million), By Product Type, 2020-2035
- Table 205: Saudi Arabia Market Size (USD Million), by Application, 2020-2035
- Table 206: Saudi Arabia Market Size (USD Million), by End User, 2020-2035
- Table 207: Other GCC Countries Market Size (USD Million), By Product Type, 2020-2035
- Table 208: Other GCC Countries Market Size (USD Million), by Application, 2020-2035
- Table 209: Other GCC Countries Market Size (USD Million), by End User, 2020-2035
- Table 210: Türkiye Market Size (USD Million), By Product Type, 2020-2035
- Table 211: Türkiye Market Size (USD Million), by Application, 2020-2035
- Table 212: Türkiye Market Size (USD Million), by End User, 2020-2035
- Table 213: South Africa Market Size (USD Million), By Product Type, 2020-2035
- Table 214: South Africa Market Size (USD Million), by Application, 2020-2035
- Table 215: South Africa Market Size (USD Million), by End User, 2020-2035
- Table 216: Rest of MEA Market Size (USD Million), By Product Type, 2020-2035
- Table 217: Rest of MEA Market Size (USD Million), by Application, 2020-2035
- Table 218: Rest of MEA Market Size (USD Million), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 2: Global Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 3: Global Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 4: Global Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 5: Global Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 6: Global Market (USD Million) Forecast, by Others, 2020-2035
- Figure 7: Global Market (USD Million) Forecast, by Application, 2020-2035
- Figure 8: Global Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 9: Global Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 10: Global Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 11: Global Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 12: Global Market (USD Million) Forecast, by End User, 2020-2035
- Figure 13: Global Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 14: Global Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 15: Global Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 16: Global Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 17: North America Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 18: North America Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 19: North America Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 20: North America Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 21: North America Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 22: North America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 23: North America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 24: North America Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 25: North America Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 26: North America Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 27: North America Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 28: North America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 29: North America Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 30: North America Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 31: North America Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 32: North America Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 33: USA Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 34: USA Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 35: USA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 36: USA Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 37: USA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 38: USA Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 39: Canada Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 40: Canada Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 41: Canada Market (USD Million) Forecast, by Application, 2020-2035
- Figure 42: Canada Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 43: Canada Market (USD Million) Forecast, by End User, 2020-2035
- Figure 44: Canada Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 45: Mexico Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 46: Mexico Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 47: Mexico Market (USD Million) Forecast, by Application, 2020-2035
- Figure 48: Mexico Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 49: Mexico Market (USD Million) Forecast, by End User, 2020-2035
- Figure 50: Mexico Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 51: Latin America Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 52: Latin America Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 53: Latin America Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 54: Latin America Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 55: Latin America Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 56: Latin America Market (USD Million) Forecast, by Others, 2020-2035
- Figure 57: Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 58: Latin America Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 59: Latin America Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 60: Latin America Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 61: Latin America Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 62: Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 63: Latin America Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 64: Latin America Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 65: Latin America Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 66: Latin America Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 67: Brazil Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 68: Brazil Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 69: Brazil Market (USD Million) Forecast, by Application, 2020-2035
- Figure 70: Brazil Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 71: Brazil Market (USD Million) Forecast, by End User, 2020-2035
- Figure 72: Brazil Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 73: Argentina Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 74: Argentina Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 75: Argentina Market (USD Million) Forecast, by Application, 2020-2035
- Figure 76: Argentina Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 77: Argentina Market (USD Million) Forecast, by End User, 2020-2035
- Figure 78: Argentina Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 79: Rest of Latin America Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 80: Rest of Latin America Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 81: Rest of Latin America Market (USD Million) Forecast, by Application, 2020-2035
- Figure 82: Rest of Latin America Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 83: Rest of Latin America Market (USD Million) Forecast, by End User, 2020-2035
- Figure 84: Rest of Latin America Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 85: East Asia Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 86: East Asia Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 87: East Asia Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 88: East Asia Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 89: East Asia Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 90: East Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 91: East Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 92: East Asia Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 93: East Asia Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 94: East Asia Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 95: East Asia Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 96: East Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 97: East Asia Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 98: East Asia Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 99: East Asia Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 100: East Asia Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 101: China Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 102: China Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 103: China Market (USD Million) Forecast, by Application, 2020-2035
- Figure 104: China Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 105: China Market (USD Million) Forecast, by End User, 2020-2035
- Figure 106: China Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 107: Japan Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 108: Japan Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 109: Japan Market (USD Million) Forecast, by Application, 2020-2035
- Figure 110: Japan Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 111: Japan Market (USD Million) Forecast, by End User, 2020-2035
- Figure 112: Japan Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 113: South Korea Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 114: South Korea Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 115: South Korea Market (USD Million) Forecast, by Application, 2020-2035
- Figure 116: South Korea Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 117: South Korea Market (USD Million) Forecast, by End User, 2020-2035
- Figure 118: South Korea Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 119: South Asia Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 120: South Asia Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 121: South Asia Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 122: South Asia Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 123: South Asia Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 124: South Asia Market (USD Million) Forecast, by Others, 2020-2035
- Figure 125: South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 126: South Asia Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 127: South Asia Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 128: South Asia Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 129: South Asia Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 130: South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 131: South Asia Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 132: South Asia Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 133: South Asia Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 134: South Asia Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 135: India Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 136: India Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 137: India Market (USD Million) Forecast, by Application, 2020-2035
- Figure 138: India Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 139: India Market (USD Million) Forecast, by End User, 2020-2035
- Figure 140: India Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 141: ASEAN Countries Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 142: ASEAN Countries Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 143: ASEAN Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 144: ASEAN Countries Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 145: ASEAN Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 146: ASEAN Countries Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 147: Australia & New Zealand Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 148: Australia & New Zealand Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 149: Australia & New Zealand Market (USD Million) Forecast, by Application, 2020-2035
- Figure 150: Australia & New Zealand Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 151: Australia & New Zealand Market (USD Million) Forecast, by End User, 2020-2035
- Figure 152: Australia & New Zealand Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 153: Rest of South Asia Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 154: Rest of South Asia Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 155: Rest of South Asia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 156: Rest of South Asia Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 157: Rest of South Asia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 158: Rest of South Asia Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 159: Western Europe Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 160: Western Europe Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 161: Western Europe Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 162: Western Europe Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 163: Western Europe Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 164: Western Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 165: Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 166: Western Europe Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 167: Western Europe Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 168: Western Europe Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 169: Western Europe Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 170: Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 171: Western Europe Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 172: Western Europe Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 173: Western Europe Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 174: Western Europe Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 175: Germany Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 176: Germany Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 177: Germany Market (USD Million) Forecast, by Application, 2020-2035
- Figure 178: Germany Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 179: Germany Market (USD Million) Forecast, by End User, 2020-2035
- Figure 180: Germany Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 181: UK Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 182: UK Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 183: UK Market (USD Million) Forecast, by Application, 2020-2035
- Figure 184: UK Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 185: UK Market (USD Million) Forecast, by End User, 2020-2035
- Figure 186: UK Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 187: France Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 188: France Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 189: France Market (USD Million) Forecast, by Application, 2020-2035
- Figure 190: France Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 191: France Market (USD Million) Forecast, by End User, 2020-2035
- Figure 192: France Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 193: Italy Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 194: Italy Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 195: Italy Market (USD Million) Forecast, by Application, 2020-2035
- Figure 196: Italy Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 197: Italy Market (USD Million) Forecast, by End User, 2020-2035
- Figure 198: Italy Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 199: Spain Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 200: Spain Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 201: Spain Market (USD Million) Forecast, by Application, 2020-2035
- Figure 202: Spain Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 203: Spain Market (USD Million) Forecast, by End User, 2020-2035
- Figure 204: Spain Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 205: BENELUX Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 206: BENELUX Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 207: BENELUX Market (USD Million) Forecast, by Application, 2020-2035
- Figure 208: BENELUX Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 209: BENELUX Market (USD Million) Forecast, by End User, 2020-2035
- Figure 210: BENELUX Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 211: Nordic Countries Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 212: Nordic Countries Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 213: Nordic Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 214: Nordic Countries Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 215: Nordic Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 216: Nordic Countries Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 217: Rest of Western Europe Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 218: Rest of Western Europe Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 219: Rest of Western Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 220: Rest of Western Europe Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 221: Rest of Western Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 222: Rest of Western Europe Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 223: Eastern Europe Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 224: Eastern Europe Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 225: Eastern Europe Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 226: Eastern Europe Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 227: Eastern Europe Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 228: Eastern Europe Market (USD Million) Forecast, by Others, 2020-2035
- Figure 229: Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 230: Eastern Europe Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 231: Eastern Europe Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 232: Eastern Europe Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 233: Eastern Europe Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 234: Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 235: Eastern Europe Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 236: Eastern Europe Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 237: Eastern Europe Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 238: Eastern Europe Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 239: Russia Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 240: Russia Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 241: Russia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 242: Russia Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 243: Russia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 244: Russia Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 245: Hungary Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 246: Hungary Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 247: Hungary Market (USD Million) Forecast, by Application, 2020-2035
- Figure 248: Hungary Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 249: Hungary Market (USD Million) Forecast, by End User, 2020-2035
- Figure 250: Hungary Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 251: Poland Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 252: Poland Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 253: Poland Market (USD Million) Forecast, by Application, 2020-2035
- Figure 254: Poland Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 255: Poland Market (USD Million) Forecast, by End User, 2020-2035
- Figure 256: Poland Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 257: Rest of Eastern Europe Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 258: Rest of Eastern Europe Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 259: Rest of Eastern Europe Market (USD Million) Forecast, by Application, 2020-2035
- Figure 260: Rest of Eastern Europe Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 261: Rest of Eastern Europe Market (USD Million) Forecast, by End User, 2020-2035
- Figure 262: Rest of Eastern Europe Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 263: Middle East & Africa Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 264: Middle East & Africa Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 265: Middle East & Africa Market (USD Million) Forecast, by IHC & protein assays, 2020-2035
- Figure 266: Middle East & Africa Market (USD Million) Forecast, by Proteomic assays, 2020-2035
- Figure 267: Middle East & Africa Market (USD Million) Forecast, by Liquid biopsy non-NGS (qPCR), 2020-2035
- Figure 268: Middle East & Africa Market (USD Million) Forecast, by Others, 2020-2035
- Figure 269: Middle East & Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 270: Middle East & Africa Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 271: Middle East & Africa Market (USD Million) Forecast, by Prognostics, 2020-2035
- Figure 272: Middle East & Africa Market (USD Million) Forecast, by Recurrence monitoring, 2020-2035
- Figure 273: Middle East & Africa Market (USD Million) Forecast, by Research use, 2020-2035
- Figure 274: Middle East & Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 275: Middle East & Africa Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 276: Middle East & Africa Market (USD Million) Forecast, by Diagnostic labs, 2020-2035
- Figure 277: Middle East & Africa Market (USD Million) Forecast, by Academic R&D, 2020-2035
- Figure 278: Middle East & Africa Market (USD Million) Forecast, by POC/Community, 2020-2035
- Figure 279: Saudi Arabia Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 280: Saudi Arabia Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 281: Saudi Arabia Market (USD Million) Forecast, by Application, 2020-2035
- Figure 282: Saudi Arabia Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 283: Saudi Arabia Market (USD Million) Forecast, by End User, 2020-2035
- Figure 284: Saudi Arabia Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 285: Other GCC Countries Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 286: Other GCC Countries Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 287: Other GCC Countries Market (USD Million) Forecast, by Application, 2020-2035
- Figure 288: Other GCC Countries Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 289: Other GCC Countries Market (USD Million) Forecast, by End User, 2020-2035
- Figure 290: Other GCC Countries Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 291: Türkiye Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 292: Türkiye Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 293: Türkiye Market (USD Million) Forecast, by Application, 2020-2035
- Figure 294: Türkiye Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 295: Türkiye Market (USD Million) Forecast, by End User, 2020-2035
- Figure 296: Türkiye Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 297: South Africa Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 298: South Africa Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 299: South Africa Market (USD Million) Forecast, by Application, 2020-2035
- Figure 300: South Africa Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 301: South Africa Market (USD Million) Forecast, by End User, 2020-2035
- Figure 302: South Africa Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 303: Rest of MEA Market (USD Million) Forecast, By Product Type, 2020-2035
- Figure 304: Rest of MEA Market (USD Million) Forecast, by PCR-based companion diagnostics, 2020-2035
- Figure 305: Rest of MEA Market (USD Million) Forecast, by Application, 2020-2035
- Figure 306: Rest of MEA Market (USD Million) Forecast, by Therapy selection / companion Dx, 2020-2035
- Figure 307: Rest of MEA Market (USD Million) Forecast, by End User, 2020-2035
- Figure 308: Rest of MEA Market (USD Million) Forecast, by Hospitals & Oncology centers, 2020-2035
- Figure 309: Roche Diagnostics - Company Snapshot
- Figure 310: Roche Diagnostics - SWOT Analysis
- Figure 311: Abbott Laboratories - Company Snapshot
- Figure 312: Abbott Laboratories - SWOT Analysis
- Figure 313: Thermo Fisher - Company Snapshot
- Figure 314: Thermo Fisher - SWOT Analysis
- Figure 315: Siemens Healthineers - Company Snapshot
- Figure 316: Siemens Healthineers - SWOT Analysis
- Figure 317: Bio-Rad - Company Snapshot
- Figure 318: Bio-Rad - SWOT Analysis
- Figure 319: Others - Company Snapshot
- Figure 320: Others - SWOT Analysis

