Demand for aortic endografts in the UK is projected to be valued at USD 417.2 million in 2026 and is expected to reach USD 689.9 million by 2036, expanding at a 5.2% CAGR. This demand is shaped by how vascular surgery units, hospital networks, and device partners scale minimally invasive aneurysm repair capacity for abdominal and thoracic disease pathways.
Aortic endografts are positioned as implantable stent graft systems that enable catheter-based repair for aneurysms and related aortic conditions. Clinical teams evaluate performance through deployment control, conformability to anatomy, sealing reliability, long-term surveillance outcomes, and predictable imaging visibility during follow-up. These devices sit inside an operating model where patient selection, imaging planning, device sizing, and post-procedure surveillance determine both clinical outcomes and cost stability.
For healthcare leaders, the central operational question is procedure throughput and complication avoidance: how quickly eligible patients move from imaging workup to repair, how reliably teams can execute EVAR in routine workflows, and how efficiently post-repair monitoring can be managed. For technology service providers, value is tied to consistent delivery system performance, stable inventory availability, and training programs that reduce variation across operators and sites.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 417.2 million |
| Industry Forecast Value (2036) | USD 689.9 million |
| Forecast CAGR (2026 to 2036) | 5.2% |
Demand in the UK rises as health systems expand aneurysm identification, prioritise early intervention for high-risk patients, and strengthen vascular service line resilience. Aortic care pathways link screening, imaging surveillance, elective repair planning, emergency response readiness, and long-term follow-up. When those elements run smoothly, EVAR volumes stay stable and device utilisation becomes easier to forecast for procurement teams.
A key structural driver is the continued need to detect abdominal aortic aneurysm risk and manage surveillance. The NHS abdominal aortic aneurysm screening standards reporting highlights the scale of screening offers, uptake, surveillance caseload, and aneurysm identification outcomes in England. These system-level inputs influence downstream procedural flow because screening and surveillance are the upstream feeders of elective repair demand.
Clinical decision-making also strengthens device utilisation when teams align around consistent thresholds and treatment selection rules. Guidance frameworks shape when open repair is chosen versus endovascular repair and how imaging should be used for planning. NICE recommendations for abdominal aortic aneurysm diagnosis and management provide structured direction on monitoring and repair decision pathways.
Device selection is being shaped by a stronger focus on durability, instruction-for-use alignment, and evidence-backed outcomes. The 2024 ESVS clinical practice guidelines emphasise device durability considerations and long-term follow-up discipline after EVAR. This shifts stakeholder attention toward consistent implant performance and post-procedure monitoring infrastructure, not just short-term procedural success.
Stakeholder relevance extends beyond clinicians. CEOs and commercial leaders focus on predictable service capacity, stable margins, and reduced variability in outcomes. Technology partners focus on imaging integration, sizing workflow support, inventory readiness, and field training that shortens the learning curve for complex anatomies. End users often assess adjacent catheter-based intervention categories often use endovascular therapy devices as a benchmark to align investment priorities across minimally invasive care pathways.
Segmentation reflects differences in anatomy treated, workflow requirements, implant design priorities, and site-of-care economics. These dimensions influence how procurement teams structure contracts, how clinical teams set standard operating protocols, and how device suppliers position training and service support.
Abdominal aortic graft holds a 56.0% share, making it the leading product category. This position reflects the procedural flow tied to abdominal aortic aneurysm diagnosis and elective repair planning. Abdominal repair programs are built around repeatable imaging workups, sizing decisions, and standardised access approaches. Vascular teams value systems that support confident sealing, controlled deployment, and a predictable post-implant surveillance profile.
Suppliers competing in this segment also face high expectations for inventory management across diameters and lengths. A hospital network cannot keep elective lists stable without reliable access to a breadth of device configurations. Product managers and commercial heads therefore focus on demand planning, consignment strategies, and responsive logistics as much as clinical features.
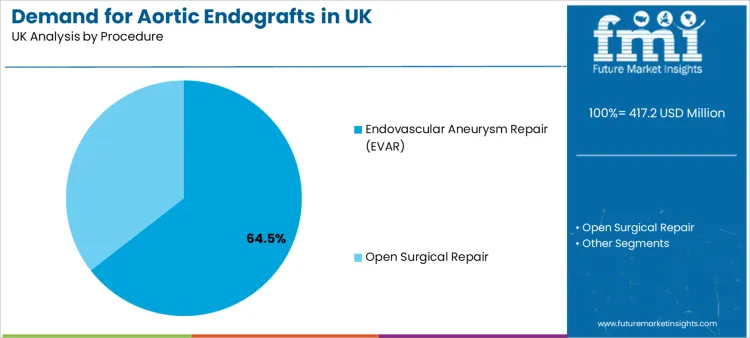
EVAR accounts for a 64.5% share, positioning it as the leading procedure. EVAR is preferred because it supports minimally invasive access, shorter recovery patterns, and scalable workflow design for many elective repair candidates. Hospitals can build repeatable pathways around imaging-led planning, device sizing, and standardised hybrid operating theatre routines.
Procedure leadership also reflects the maturity of team training and established perioperative protocols in many centres. Open surgical repair remains critical for select anatomies and clinical scenarios, yet EVAR retains the procedural lead where minimally invasive pathways align with patient selection needs and throughput targets. Stakeholders tracking EVAR evolution often cross-reference adjacent innovation themes through in the broader endovascular aneurysm repair landscape.
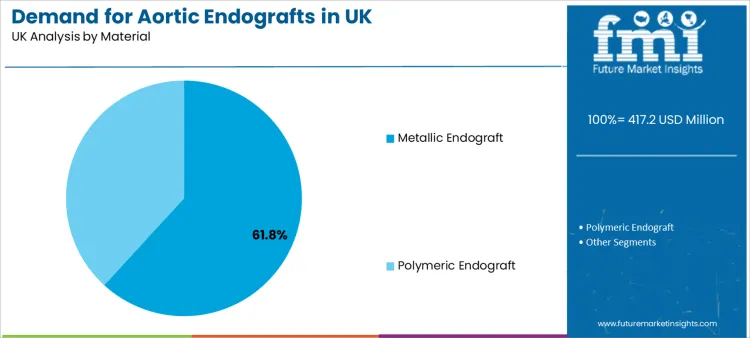
Metallic endograft holds a 61.8% share, making it the leading material category. This aligns with the engineering requirements of radial strength, long-term structural integrity, and consistent imaging visibility. Metallic frameworks support conformability while maintaining shape memory attributes that help positioning and fixation.
Engineering stakeholders evaluate metallic designs through deliverability, trackability, and sealing behaviour across varied anatomy. Procurement teams also review long-term performance signals because re-intervention risk affects operating costs, bed utilisation, and follow-up imaging burden.
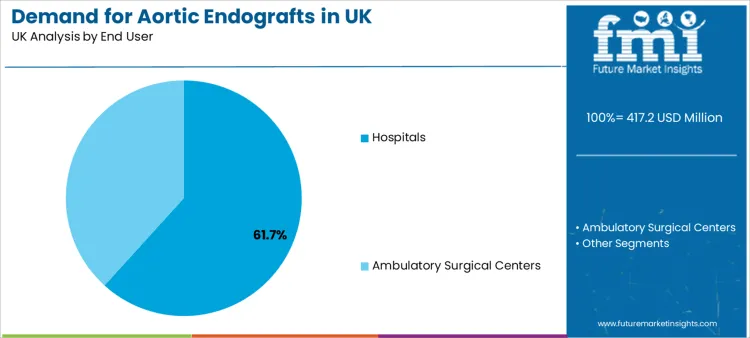
Hospitals represent a 61.7% share, making them the leading end user segment. Hospitals hold this position because they manage imaging workup, multidisciplinary case review, elective repair scheduling, and urgent response workflows under one coordinated system. Hybrid operating rooms, vascular teams, anaesthesia support, and post-implant surveillance infrastructure tend to be concentrated in hospital environments.
Ambulatory surgical centers play a selective role in elective procedures where pathway standardisation and risk profiles fit outpatient-oriented care delivery. Hospitals remain the anchor setting for complex anatomies, higher-acuity cases, and scenarios where intensive monitoring capacity is required.
Minimally invasive procedural preference supports steady utilisation. Screening and surveillance programs feed elective case flow, enabling capacity planning and procurement forecasting. Clinical guideline updates reinforce device durability expectations and long-term follow-up discipline, which keeps endograft selection tied to outcomes evidence.
A second driver is service line modernisation. Hospital groups standardise imaging protocols, introduce structured MDT decision models, and invest in hybrid operating capability. These shifts strengthen volume stability for EVAR pathways and reinforce demand for predictable device availability.
Capital allocation pressure remains a recurring restraint. Hospitals must prioritise across imaging equipment, theatre upgrades, staffing, and device budgets. Workforce capacity limits also constrain throughput. Aortic repair requires trained teams, imaging access, and post-procedure monitoring resources.
Regulatory and post-market surveillance requirements influence supplier obligations and buyer expectations. The MHRA has issued guidance to support implementation of new post-market surveillance requirements coming into force in June 2025, raising the bar for systematic monitoring and reporting. These requirements increase the importance of compliant documentation, traceability, and incident trend response.
Opportunity sits in workflow simplification and planning support. Software-enabled sizing support, imaging integration, and procedural standardisation reduce variation across operators. Training programs that accelerate competency expansion improve throughput and reduce schedule volatility.
Another opportunity sits in advanced anatomy solutions. Branched and fenestrated pathways push device requirements toward higher customisation and specialised support. Product portfolios aligned with complex anatomy strategies are frequently compared with broader developments in branched stent graft solutions.
Supply disruption remains a direct threat because elective lists can be destabilised by inventory gaps. Adverse event trends also create reputational risk for hospitals and manufacturers. Long-term follow-up requirements increase the operational burden on health systems, which can slow expansion in regions with constrained imaging capacity.
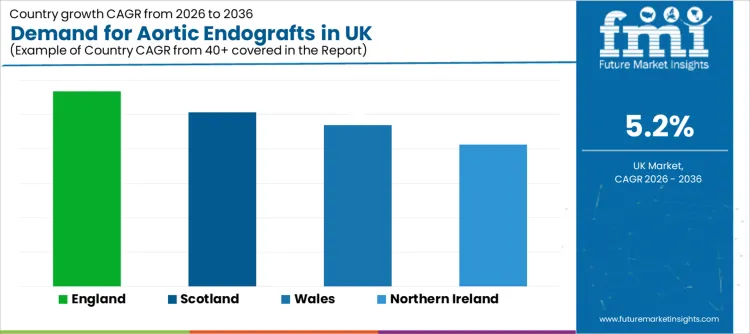
| Region | CAGR (2026-2036) |
|---|---|
| England | 5.7% |
| Scotland | 5.1% |
| Wales | 4.7% |
| Northern Ireland | 4.1% |
England grows at 5.7%, shaped by larger hospital networks and higher volumes moving through screening-to-repair pathways. Vascular service consolidation and hybrid operating capability support throughput and standardised protocols.
Device partners gain traction when they support reliable supply models, consistent training support, and responsive field services across multiple sites. England’s stakeholders also track adjacent aortic device developments in the wider aortic stent grafts space when aligning longer-term portfolio strategy.
Scotland increases at 5.1%, reflecting steady adoption tied to protocol-driven care delivery. Teams prioritise predictable outcomes, imaging-led planning, and controlled surveillance models. Purchasing decisions are influenced by service reliability, availability of sizing support, and product configurations that fit routine anatomy requirements.
Wales grows at 4.7%, shaped by capacity-building priorities that improve access to elective repair scheduling and imaging support. Hospitals that reduce referral delays and strengthen MDT alignment create more stable EVAR throughput. Suppliers that provide training pathways and dependable logistics fit well with these operational targets.
Northern Ireland rises at 4.1%, reflecting a measured expansion profile tied to staged investment cycles. Growth strengthens where hospitals maintain consistent theatre access, preserve trained team availability, and optimise surveillance scheduling. Device partners that reduce downtime risk and support predictable inventory availability help sustain utilisation growth.
Competition is shaped by deployment reliability, sealing confidence, long-term performance expectations, clinical evidence depth, and service responsiveness. Buyers evaluate how consistently systems deliver predictable outcomes across a range of anatomies, how well delivery platforms support stable procedural routines, and how rapidly suppliers can respond to field needs.
Medtronic plc competes through broad endovascular portfolio depth and a mature service footprint that aligns with hospital procurement requirements. Cook Medical holds strong positioning in graft platforms and catheter-based delivery systems, supporting vascular teams seeking standardised workflows. W. L. Gore & Associates competes through graft material capabilities and clinical familiarity in endovascular repair environments. Terumo Corporation contributes through device engineering and access system alignment for catheter-based intervention routines. Endologix LLC adds competitive differentiation through portfolio positioning that addresses specific procedural needs and device selection strategies.
Procurement leaders favour companies that support lifecycle stability: inventory reliability, training readiness, and post-implant surveillance support. Competitive evaluation often includes adjacent report comparisons such as the aortic endograft industry outlook when stakeholders benchmark broader category direction.
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product | Abdominal Aortic Graft; Thoracic Aortic Graft |
| Procedure | Endovascular Aneurysm Repair (EVAR); Open Surgical Repair |
| Material | Metallic Endograft; Polymeric Endograft |
| End User | Hospitals; Ambulatory Surgical Centers |
| Regions Covered | England; Scotland; Wales; Northern Ireland |
| Key Companies Profiled | Medtronic plc; Cook Medical; W. L. Gore & Associates; Terumo Corporation; Endologix LLC |
What is the outlook for Aortic Endografts demand in the UK for 2026?
Aortic endografts demand in the UK is expected to total USD 417.2 million in 2026.
What value is expected for Aortic Endografts demand in UK for 2036?
In 2036, demand for aortic endografts in UK will likely be valued at USD 689.9 million.
At what rate will Aortic Endografts demand in UK progress from 2026 to 2036?
Aortic endografts demand in UK is anticipated to advance at a 5.2% CAGR from 2026 to 2036.
Which product will constitute the bulk of aortic endograft utilisation in the UK?
Abdominal aortic graft is likely to be the dominant product category, capturing 56.0% of the total demand share.
Which procedure is expected to be most preferred in the UK?
Endovascular aneurysm repair (EVAR) will be most preferred, accounting for 64.5% of all procedures suported by endograft use.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.