AI Portable Chest X-ray Triage Device Market
The AI Portable Chest X-ray Triage Device is segmented by Product type (Handheld systems, Cart systems, Backpack systems), AI function (TB screening, Critical triage, Full analysis), Deployment mode (Cloud-connected, Edge offline, Hybrid mode), End user (Public programs, Hospitals, Mobile clinics, NGOs), Sales model (Bundled systems, Device lease, Software license, Pay-per-scan), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
AI Portable Chest X-ray Triage Device Market Size, Market Forecast and Outlook By FMI
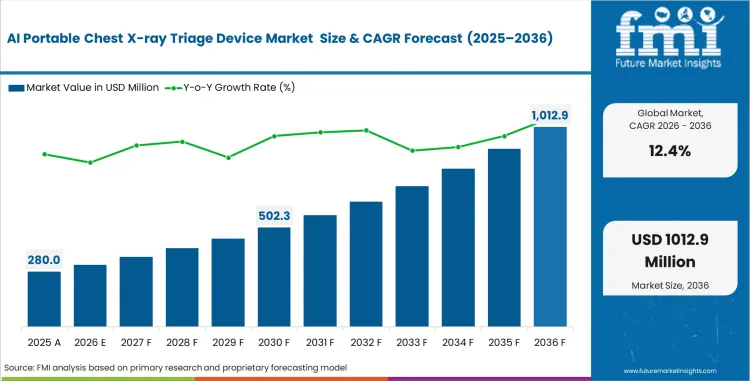
The AI Portable Chest X-ray Triage Device market was valued at USD 250.0 million in 2025 and is projected to reach USD 900 million by 2036. Industry valuation is anticipated to rise to USD 280 million in 2026, with the sector advancing at a 12.4% CAGR over the 2026 to 2036 period.
Summary of AI Portable Chest X-ray Triage Device Market
- The market is forecast to reach USD 900 million by 2036.
- The market is expected to grow at a CAGR of 12.4% from 2026 to 2036.
- The market was estimated at USD 250.0 million in 2025.
- The forecast period represents an incremental opportunity of USD 620 million.
- Handheld systems lead the product segment with a 44% share, driven by strong demand for mobility in outreach and remote screening.
- TB screening dominates the AI function segment with a 38% share, supported by large-scale adoption in national and donor-funded programs.
- Cloud-connected systems account for 46% share, enabling centralized monitoring, reporting, and multi-site device management.
- Public programs represent 34% share of the end-user segment, reflecting the role of government-led screening initiatives.
- Bundled systems hold a 42% share, as buyers prefer integrated packages including hardware, AI, training, and service support.
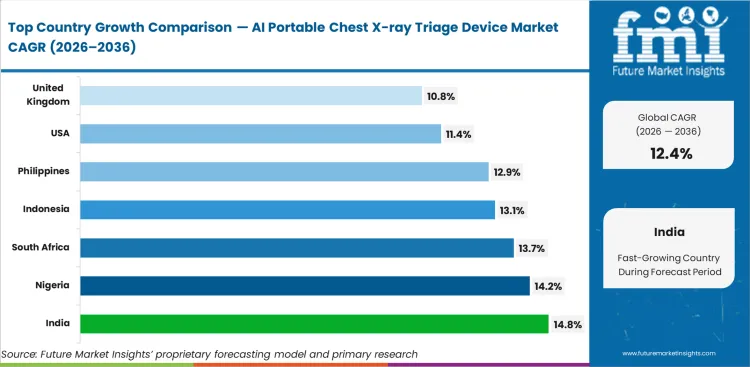
- India, Nigeria, South Africa, Indonesia, and the Philippines are the fastest-growing markets, while the United States and the United Kingdom remain key high-value regions.
- Market growth is driven by expanding TB screening programs, increasing demand for rapid triage in emergency care, and the need for AI-supported diagnostics in low-radiologist settings.
AI Portable Chest X-ray Triage Device Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 280 million |
| Industry Value (2036) | USD 900 million |
| CAGR (2026 to 2036) | 12.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Growth remains tied to point-of-care chest imaging in infectious disease screening, particularly where earlier triage can reduce pressure on centralized review pathways.
Radiologist shortages remain a practical constraint for regional health systems, particularly where rural clinics cannot wait for centralized image review before acting. Hand held X-rays are becoming more relevant because delays in first-line screening extend patient waiting time and raise operational pressure in facilities handling suspected airborne infections. Automated triage changes the workflow at an early stage by allowing clinical teams to act on algorithm-based prioritization before the first specialist read. Integration with broader X-ray systems also makes deployment more workable in settings that need speed without adding a full diagnostic layer at the front end.
Adoption usually becomes easier when national screening protocols define how algorithm-assisted triage should be used. Where computer-aided detection scores are used for preliminary triage, field programs and decentralized care networks can move suspected cases faster without treating AI output as a stand-alone diagnosis. That matters most in settings where referral queues are long and specialist review remains limited.
India is projected to witness 14.8% CAGR through 2036, supported by government-funded TB screening and broad rural outreach deployment. Nigeria follows at 14.2% where battery-powered systems fit areas with unreliable grid access, while South Africa is projected to expand at 13.7% on the back of established infectious-disease screening pathways. Indonesia at 13.1% and the Philippines at 12.9% benefit from decentralized diagnostics and municipal health upgrades. Growth is more moderate in the United States at 11.4% and the United Kingdom at 10.8%, where the buying case is shaped more by workflow efficiency than by first-time access.
Segmental Analysis
AI Portable Chest X-ray Triage Device Market Analysis by Product Type
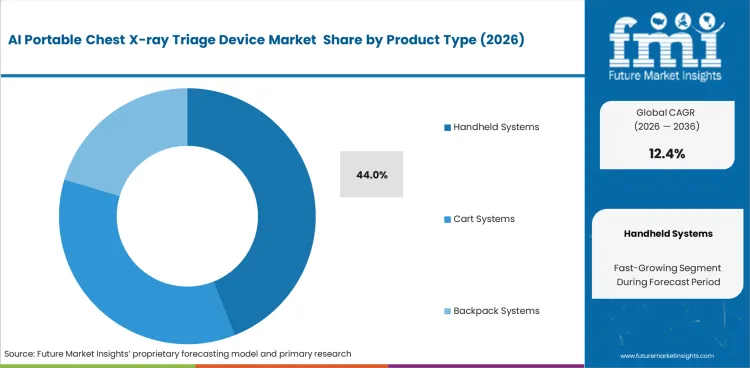
The Handheld systems segment is expected to account for 44.0% share in 2026 because extreme miniaturization allows single-user deployment across challenging terrain. Remote screening campaigns demand equipment capable of reaching populations located far from paved roads. Public health coordinators prioritize this form factor since it eliminates logistical hurdles associated with transporting larger X-ray device alternatives. The field technicians value battery autonomy over peak image resolution when conducting high-volume community screening. Standard specifications highlight weight reduction. Field realities dictate that ergonomic balance prevents user fatigue during continuous scanning sessions. Sourcing leads who select heavier cart formats for remote outreach immediately encounter transportation barriers that reduce screening throughput. Advanced neural networks embedded directly into these ultra-mobile units represent a critical application of artificial intelligence in healthcare, allowing instant triage without internet connectivity. Such capabilities prove essential for decentralized medical teams operating far from urban hospitals.
- Deployment flexibility: Battery-operated generators allow screening inside community centers lacking dedicated electrical supply. Field technicians avoid facility modification costs while establishing pop-up triage centers.
- Workflow acceleration: Instant imaging combined with automated scoring eliminates callback requirements for patients. Rural residents receive immediate care decisions, measurably reducing loss-to-follow-up rates.
- Component durability: Frequent transport exposes sensitive imaging plates to vibration and impact. Hardware failures halt entire mobile screening operations until replacement parts arrive from centralized depots.
AI Portable Chest X-ray Triage Device Market Analysis by AI Function
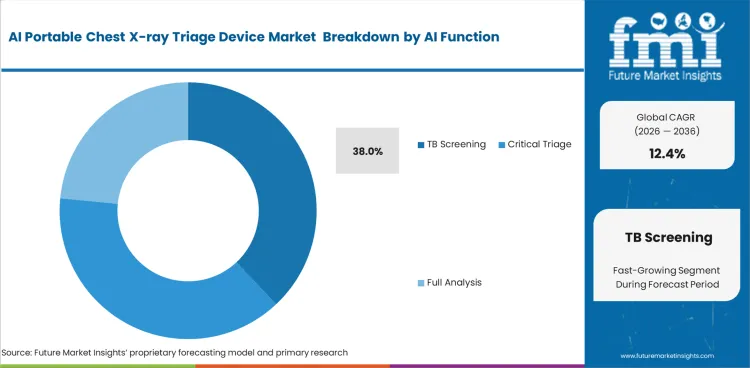
National eradication strategies dictate technological priorities across developing regions. TB screening is projected to secure 38.0% share in 2026 as international funding heavily subsidizes computer-aided detection for respiratory infectious diseases. Program directors at non-governmental organizations specify this functionality to satisfy donor metrics. It has been indicated that widespread integration of an ai radiology tool for tuberculosis specifically addresses radiologist shortages across high-burden countries. Surface metrics emphasize screening volume. Actual clinical value emerges from measurably reducing false-negative rates during asymptomatic patient evaluations. Diagnostic heads delaying implementation of automated respiratory screening face escalating case backlogs that overwhelm limited central reading facilities. Utilizing dedicated healthcare ai computer vision algorithms ensures consistent interpretation accuracy regardless of technician fatigue levels. Consistent diagnostic performance remains crucial for scaling intervention efforts securely.
- Targeted identification: Algorithms trained specifically on localized tuberculosis presentations outperform generalized software. Clinicians achieve higher confidence in positive triaging outcomes, accelerating isolation protocols.
- Threshold tuning: Software allows administrators to adjust sensitivity parameters based on local prevalence rates. Public health officials control follow-up testing burdens by calibrating algorithmic strictness dynamically.
- Protocol standardization: Automated scoring forces uniformity across widely distributed screening sites. National health ministries gain reliable epidemiological data sets, enabling targeted resource allocation.
AI Portable Chest X-ray Triage Device Market Analysis by Deployment Mode
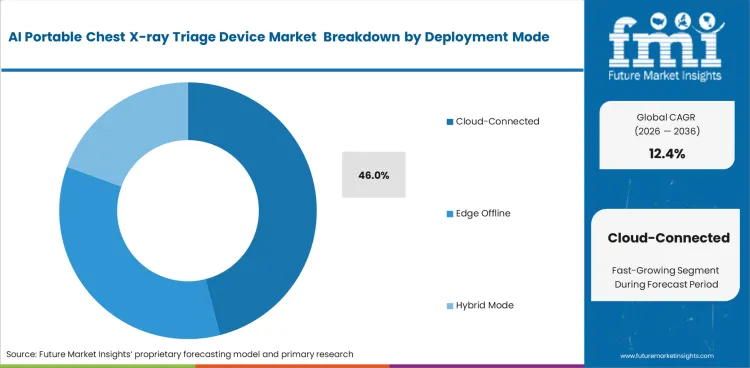
Institutional IT policies heavily influence how medical devices interact with broader hospital networks. The Cloud-connected category is likely to represent 46.0% of the market in 2026 because centralized data aggregation enables continuous algorithm performance monitoring and fleet management. Chief information officers favor this architecture to ensure software updates deploy simultaneously across all active units. Reliance on constant connectivity introduces vulnerabilities during network outages. Marketing materials emphasize imaging interoperability middleware integration. IT administrators recognize that local bandwidth limitations frequently throttle image transmission speeds. Facilities choosing exclusively offline modes sacrifice real-time central oversight and long-term predictive analytics capabilities. Connecting field devices directly to a central radiology information system ris accelerates downstream billing and patient tracking workflows. Streamlined data transfer speeds up the entire revenue cycle for connected facilities.
- Centralized oversight: Remote dashboards allow administrators to track scanning volumes and device health across multiple clinic locations. Maintenance teams predict hardware failures before they interrupt clinical scheduling.
- Algorithm refinement: Continuous data uploading provides developers with diverse training images. Future software iterations achieve higher accuracy across varied patient demographics, directly improving diagnostic yields.
- Security compliance: Centralized data storage minimizes patient information residing on easily stolen mobile devices. Risk officers maintain privacy regulations while expanding remote diagnostic capabilities.
AI Portable Chest X-ray Triage Device Market Analysis by End User
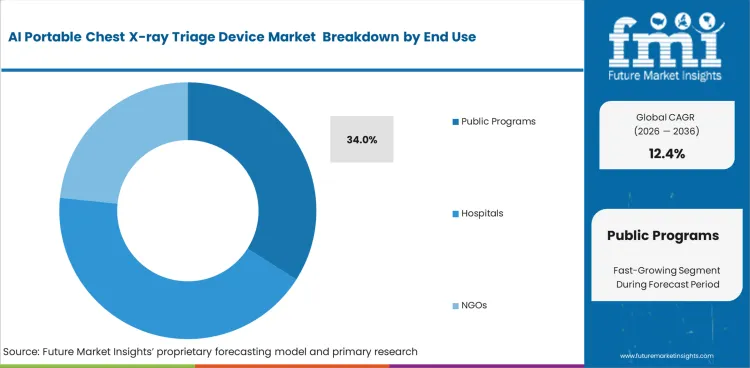
National health directors utilize these portable units to execute outreach campaigns across underserved districts. Population screening mandates require coordinated institutional responses. In 2026, Public programs are expected to contribute 34.0% of total market share due to governmental investments targeting communicable disease eradication at a national scale. According to estimations, institutional buyers prioritize equipment lifespan and vendor support over cutting-edge interface features. Total cost calculations rarely account for intensive technician training required to maintain high diagnostic yields during community deployment. Delaying purchases forces regional health authorities to rely on costly centralized diagnostic imaging services, limiting overall population coverage. Implementing diagnostic X-ray system technology at community levels directly reduces late-stage disease presentations at tertiary hospitals. Early intervention capabilities reshape municipal health economics.
- Scale execution: Bulk supply contracts allow health ministries to standardize screening protocols across entire provinces. Implementation coordinators simplify maintenance channels by deploying uniform hardware fleets.
- Funding alignment: Donor organizations specifically finance equipment designed for population-level interventions. Program directors secure capital for ongoing operations by proving high screening throughput.
- Impact measurement: Standardized algorithmic reporting provides governments with verifiable disease prevalence data. Public health officials justify continued funding requests using concrete automated screening statistics.
AI Portable Chest X-ray Triage Device Market Analysis by Sales Model
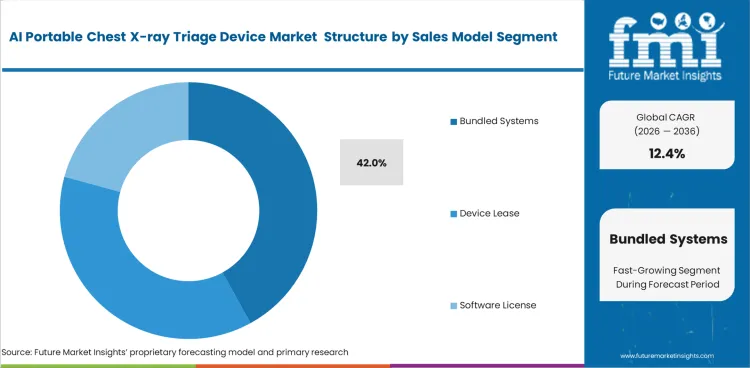
Financial unpredictability within healthcare budgets forces alternative acquisition strategies. The market is expected to see Bundled systems account for 42.0% share in 2026 because packaging hardware, software, and maintenance into a single contract simplifies convoluted purchasing processes. Sourcing managers prefer fixed-cost agreements to avoid unexpected software licensing fees disrupting annual operating budgets. The combined contracts often mask inflated long-term maintenance costs beneath attractive initial hardware pricing. Vendor documentation highlights integrated support. Hospital financial officers recognize that hardware-software coupling creates barriers when attempting to upgrade individual components later. Administrators opting for separate unbundled acquisitions face complex integration challenges that delay actual clinical deployment. Utilizing bundled packages ensures complete compatibility with existing digital X-ray equipment standards. Reliable integration prevents costly workflow interruptions in high-volume triage environments.
- Budget predictability: Single-line-item pricing allows clinics to forecast total ownership costs accurately. Financial directors avoid requesting supplemental funding mid-year to cover unexpected licensing renewals.
- Accountability consolidation: One vendor handles all hardware failures and software glitches. IT support teams avoid troubleshooting disputes between multiple providers, restoring clinical operations rapidly.
- Upgrade restrictions: Locked contracts prevent organizations from utilizing superior algorithms developed by competing software firms. Clinical directors remain tied to incumbent capabilities until complete system replacement occurs.
AI Portable Chest X-ray Triage Device Market Drivers, Restraints, and Opportunities
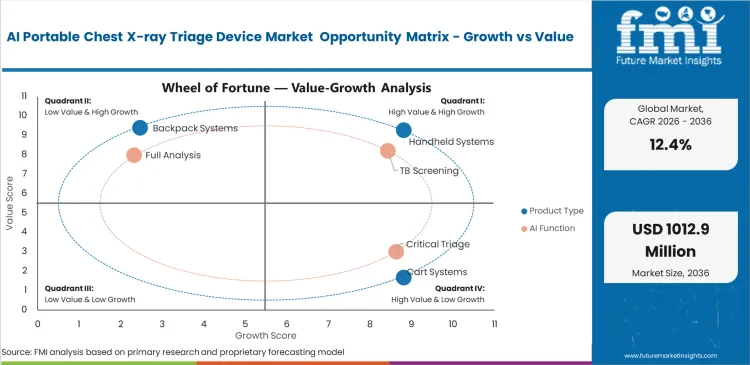
Diagnostic bottlenecks push hospital administrators to rethink traditional imaging pathways. Emergency department overcrowding pushes clinical directors toward immediate point-of-care triaging solutions to accelerate patient disposition. Waiting for centralized radiological review delays critical interventions. Deploying mobile units equipped with automated scoring allows attending physicians to make faster preliminary isolation or discharge decisions. This operational shift directly relieves pressure on fixed imaging suites. Implementing AI-enabled medical devices at patient bedsides can reduce transport-related complications and frees valuable staff time.
Inconsistent electrical supply can restrict mobile scanning expansion across developing regions. Battery degradation under temperature variations limits operational scanning hours. Mobile clinic teams frequently pause operations to recharge units using unreliable local power grids. While improved lithium-ion cells extend daily capacity, fundamental limitations in high-voltage power generation for x-ray emission persist. Connecting these units to broad teleradiology networks becomes difficult where rural telecommunications remain unreliable.
Opportunities in the AI Portable Chest X-ray Triage Device Market
- Occupational health screening: Mining and manufacturing companies require rapid respiratory evaluation for workers exposed to hazardous particulates. Occupational health directors deploy portable units to conduct on-site compliance monitoring efficiently.
- Disaster response deployment: Emergency relief organizations need immediate diagnostic capabilities in resource-depleted disaster zones. Logistics coordinators utilize digital radiography sensor technologies to establish temporary triage centers instantly.
- Elderly care integration: Nursing facilities demand bedside imaging to avoid transporting frail residents to hospitals. Geriatric administrators implement mobile screening to evaluate acute respiratory conditions safely without risking transit-induced trauma.
Regional Analysis
Based on regional analysis, AI portable chest x-ray triage device industry is segmented into North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 14.8% |
| Nigeria | 14.2% |
| South Africa | 13.7% |
| Indonesia | 13.1% |
| Philippines | 12.9% |
| United States | 11.4% |
| United Kingdom | 10.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
South Asia & Pacific AI Portable Chest X-ray Triage Device Market Analysis
South Asia & Pacific remains a key deployment base for portable chest imaging because large populations, uneven healthcare access, and high respiratory screening needs favor mobile diagnostic formats. Health ministries across the region are funding outreach programs that need lightweight systems, fast setup, and dependable field use. Rural facilities are skipping older film-based systems and moving directly toward digital and AI-supported workflows. Central health authorities also value structured reporting and connected screening data because both improve visibility into regional disease patterns.
- India: India is projected to expand at a CAGR of 14.8% through 2036 in this market. Government-backed tuberculosis screening programs continue to support large-volume mobile deployment across rural and semi-urban areas. Automated triage helps offset specialist shortages and improves screening throughput where reading capacity is limited. Local manufacturing also strengthens the country’s position by supporting rugged product designs suited to operating conditions on the ground.
- Indonesia: Indonesia’s market remains commercially relevant because its island geography keeps decentralized diagnostics essential across dispersed communities. Regional health systems rely on portable units that can be moved without depending on fixed hospital setting, and backpack systems are particularly useful where boat transport and remote access shape delivery planning. This market is forecast to register a 13.1% CAGR over the study period, reflecting continued reliance on field-ready screening rather than hospital-led expansion alone.
- Philippines: Municipal health modernization programs continue to support point-of-care imaging across the Philippines, especially where older local facilities need practical upgrades. Portable systems help clinical teams run broader respiratory screening activity at community level, and donor-backed procurement still plays a visible role in equipment expansion. Adoption in the Philippines is anticipated to move ahead at a CAGR of 12.9% during the assessment period. That keeps the market on a positive trend as local care networks push screening capacity closer to the patient base.
FMI’s report covers additional countries across the region. Interoperability remains an important regional issue because island-based health systems need portable imaging devices to connect smoothly with central referral hospitals.
Middle East & Africa AI Portable Chest X-ray Triage Device Market Analysis
Middle East & Africa presents a strong use case for portable chest X-ray triage devices because equipment gaps, unreliable infrastructure, and outreach-led screening models all support off-grid operation. NGOs, public health agencies, and donor-backed programs continue to shape procurement decisions across several African markets. The buyers place durability, battery performance, and field reliability ahead of advanced software layering when operating conditions are difficult. Teleradiology linkage is still useful, but network inconsistency can limit how smoothly these devices connect with centralized reporting systems.
- Nigeria: Nigeria’s industry outlook points to 14.2% CAGR through 2036. Battery-operated systems are particularly relevant where grid instability affects routine imaging access, especially in high-density urban and peri-urban screening environments. Portable AI-enabled units help programme teams run active tuberculosis case-finding with less dependence on fixed diagnostic centers. The country also serves as an important proof point for how these systems perform in demanding off-grid healthcare settings.
- South Africa: Public health programs in South Africa continue to integrate automated chest screening into wider infectious disease management pathways. Portable systems are also useful in facilities managing heavy respiratory caseloads, where faster triage supports workflow discipline and earlier escalation decisions. South Africa is likely to post 13.7% CAGR by 2036 in this market. Local validation work also matters here because software performance must align with regional patient profiles and clinical expectations.
FMI’s report covers additional countries across the region. Continental health systems continue to require localized algorithm training data so software accuracy aligns more closely with regional disease presentation patterns.
North America AI Portable Chest X-ray Triage Device Market Analysis
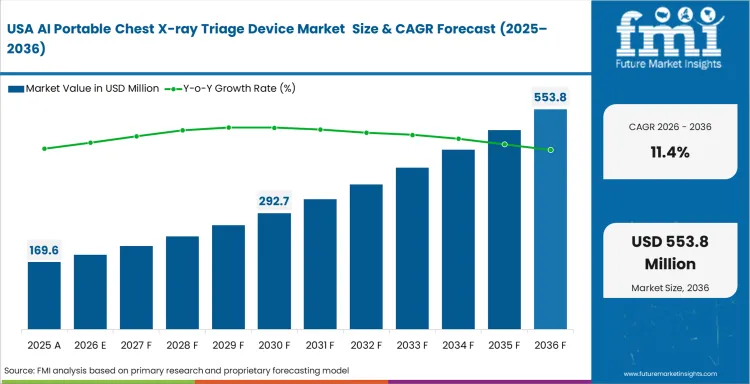
North America remains a workflow-driven market where adoption is tied closely to hospital throughput, emergency department pressure, and integration with established clinical systems. Healthcare providers use portable AI-enabled screening tools to speed up patient sorting and reduce delays in reading priority cases. The regulatory scrutiny makes rollout slower for newly introduced algorithms than in many developing markets. Purchasing teams also expect these systems to connect cleanly with existing hospital networks, which keeps interoperability and IT fit central to buying decisions.
- United States: Emergency departments across the United States continue to adopt algorithm-supported portable chest imaging to improve triage speed and support faster case prioritization. Digital mobile X-ray units paired with critical triage software are particularly relevant where patient volumes stay high and radiology teams need better queue management. The market in the United States is expected to record 11.4% CAGR during the forecast period. Mature hospital facilities gives the country a solid base for deployment, but buyers still expect strong workflow compatibility before scaling adoption.
FMI's report includes Canada. Private healthcare networks prioritize software modules capable of identifying multiple incidental findings alongside primary respiratory screening.
Europe AI Portable Chest X-ray Triage Device Market Analysis
Europe’s market is being shaped by public health backlog reduction, radiology capacity pressure, and the need to use specialist time more efficiently. Hospitals and community diagnostic networks are bringing AI into chest imaging workflows to separate routine scans from cases that need faster review. The data privacy expectations across Europe often support edge-based deployment models, especially where cloud use faces tighter governance review. Clinical utility evidence also remains central to adoption because reimbursement and hospital approval standards are relatively strict.
- United Kingdom: The United Kingdom is projected to post a 10.8% CAGR over the forecast period in this market. Public trusts are using portable systems in community diagnostic settings to help reduce outpatient pressure and move basic imaging activity beyond large hospitals. Automated preliminary screening supports faster handling of routine chest cases and helps specialist teams focus on more urgent or complex scans. This gives the market a practical role in service reorganization rather than simple equipment replacement.
FMI’s report includes Germany, France, Italy, and Spain. European health systems continue to look for stronger real-world clinical evidence before widening reimbursement support for algorithm-based triage tools.
Competitive Aligners for Market Players
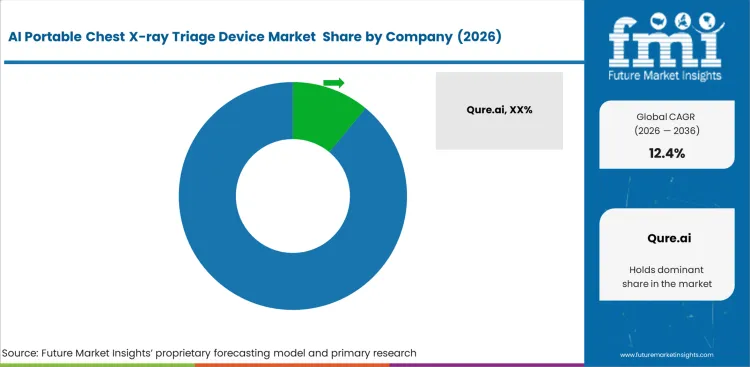
Regulatory clearance pathways shape competitive positioning more than algorithm performance alone. Vendors with broader clearance coverage, stronger deployment history, and integration support tend to fare better in institutional and public-health tenders. Global health buyers typically require recognized regulatory or policy alignment before award, which raises the evidence burden for smaller entrants. New suppliers often find that multicenter validation, documentation, and post-deployment support carry heavier costs than the model itself.
Incumbent software developers possess vast proprietary datasets compiled across diverse global populations. This data diversity ensures algorithm stability across different ethnicities and equipment types. Challengers utilizing narrow training datasets frequently experience accuracy drops when their software encounters novel hardware configurations. Established players secure exclusive integration partnerships with major hardware manufacturers, blocking new entrants from deploying software on widely distributed imaging platforms.
Institutional purchasing committees strongly resist software licensing models that lock hospitals into single-vendor contracts. Financial officers demand interoperability standards allowing hospitals to swap algorithms without replacing underlying imaging hardware. Software firms prioritizing open architecture protocols capture significant attention from large hospital networks. Developers focus heavily on expanding detection capabilities beyond respiratory infections to include cardiovascular anomalies and opportunistic diseases.
Key Players in AI Portable Chest X-ray Triage Device Market
- Qure.ai
- Lunit
- VUNO
- Annalise.ai
- Aidoc
- Delft Imaging
- Oxipit
Scope of the Report
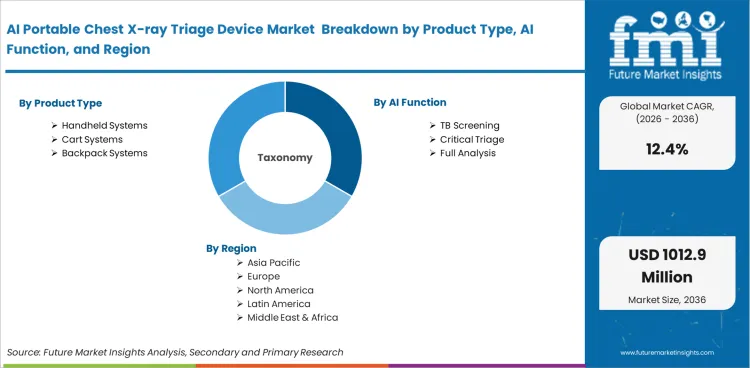
| Metric | Value |
|---|---|
| Quantitative Units | USD 280 million to USD 900 million, at a CAGR of 12.4% |
| Market Definition | AI Portable Chest X-ray Triage Device encompasses compact radiological imaging hardware integrated with algorithmic software designed to acquire and analyze thoracic scans at point of care, automatically identifying specific pathologies to prioritize patient care pathways without immediate human radiological review. |
| Segmentation | Product type, AI function, Deployment mode, End user, Sales model |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, Nigeria, South Africa, Indonesia, Philippines, United States, United Kingdom |
| Key Companies Profiled | Qure.ai, Lunit, VUNO, Annalise.ai, Aidoc, Delft Imaging, Oxipit |
| Forecast Period | 2026 to 2036 |
| Approach | Hardware deployment contracts awarded by non-governmental organizations and regional health trusts established starting valuation parameters. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
AI Portable Chest X-ray Triage Device Market Analysis by Segments
Product type:
- Handheld systems
- Cart systems
- Backpack systems
AI function:
- TB screening
- Critical triage
- Full analysis
Deployment mode:
- Cloud-connected
- Edge offline
- Hybrid mode
End user:
- Public programs
- Hospitals
- Mobile clinics
- NGOs
Sales model:
- Bundled systems
- Device lease
- Software license
- Pay-per-scan
Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- Alege, A., Ogundiran, A., Onazi, O., Alege, P., et al. (2025, June). Implementation costs and cost-effectiveness of ultraportable chest X-ray with artificial intelligence in active case finding for tuberculosis in Nigeria. PLOS Global Public Health, 5(6), e0004408.
- Dakum, P., Agbaje, A., Daniel, O., Anyaike, C., Chijoke-Akaniro, O., Okpokoro, E., Akingbesote, S., Anyomi, C., Adekunle, A., Alege, A., et al. (2024, August 14). Implementation of Portable Digital Chest X-ray Machine for Tuberculosis Contact Tracing in Oyo and Osun States, Nigeria: A Formative Assessment. Journal of Respiration, 4(3), 163-176.
- NHS England. (2025, October 23). Diagnostic Imaging Dataset Statistical Release.
- Stop TB Partnership. (2025, June 10). Use of computer-aided detection software for tuberculosis-related abnormalities on chest radiography in individuals aged 15 years and older: Policy statement. World Health Organization.
- The Global Fund. (2024, October 30). Request for Proposal (RFP) Template: TB CXR mapping.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- What underlying limitations force public health directors to deploy handheld screening units rather than relying on cart-based systems.
- How bundled sales contracts create long-term vendor lock-in for hospital purchasing committees evaluating algorithm upgrades.
- Why off-grid electrical reliability dictates equipment selection across Nigerian and South African diagnostic campaigns.
- What specific parameters algorithms use to execute critical triage workflows prior to radiologist verification.
- How centralized cloud architectures improve overarching algorithmic accuracy despite introducing local bandwidth vulnerabilities.
- Why United Kingdom public trusts utilize automated screening to actively reduce outpatient waiting lists.
- What functional mechanisms allow computer-aided detection scores to trigger immediate isolation orders confidently.
- How component durability requirements supersede peak image resolution specifications in rural screening deployments.
Frequently Asked Questions
What was AI Portable Chest X-ray Triage Device valued at in 2025?
Sales reached USD 250.0 million in 2025. Specialist shortages across high-burden disease zones force organizations to adopt algorithmic screening tools to manage expanding caseloads.
What is the projected value for 2036?
Revenue forecasts indicate a USD 900 million valuation by 2036. National health ministries mandate automated digital evaluations to execute communicable disease eradication programs effectively.
What is the calculated compound annual growth rate?
Valuation expands at a 12.4% rate between 2026 and 2036. Rapid clearance pathways for computer-aided detection software accelerate widespread implementation across developing regions.
Which product type currently leads adoption?
Handheld systems capture 44.0% share. Public health coordinators prioritize this form factor because extreme miniaturization allows single-operator deployment across unpaved terrain.
Why does TB screening dominate AI functions?
This functionality holds 38.0% share because international funding directly subsidizes respiratory infectious disease tracking. Program directors utilize automated scoring to handle community screening volumes.
How do cloud-connected modes influence operational workflows?
Cloud configurations represent 46.0% of deployments. Centralized data aggregation provides developers with diverse training images while allowing administrators to track scanning volumes across scattered clinic locations.
What end user category generates maximum volume?
Public programs account for 34.0% share. National health directors execute outreach campaigns utilizing bulk-procured portable units to establish standardized triage protocols across underserved districts.
Why do bundled systems maintain commercial dominance?
Single-contract packages capture 42.0% share. Purchasing managers prefer fixed-cost agreements combining hardware and software to avoid unexpected licensing fees disrupting annual operating budgets.
What operational difference splits United States and United Kingdom adoption?
American facilities deploy algorithmic triage to handle emergency room volumes and accelerate disposition. British public trusts target preliminary screening specifically to reduce outpatient waiting lists.
Why does India lead regional growth rates?
The market expands at 14.8% due to government-funded tuberculosis eradication campaigns. State health directors mandate automated triage to compensate for specialist shortages across rural territories.
How do inconsistent power grids shape Nigerian procurement?
Non-governmental organizations deploy battery-operated units explicitly to circumvent widespread power grid unreliability. Implementation coordinators prioritize equipment capable of extended off-grid scanning inside urban slums.
What barrier restricts off-grid edge deployment?
Facilities choosing exclusively offline modes sacrifice real-time central oversight. Risk officers struggle to maintain privacy regulations when sensitive patient data resides locally on easily stolen mobile devices.
How do algorithmic sensitivity thresholds alter clinical burdens?
Software allows administrators to adjust operational strictness based on local prevalence rates. Calibrating these parameters controls follow-up testing volumes, preventing unnecessary secondary diagnostic bottlenecks.
Why do institutional buyers reject unbundled software acquisitions?
Separate unbundled acquisitions create complex integration challenges. IT support teams face troubleshooting disputes between multiple providers when algorithmic software fails to communicate with third-party digital generators.
What limits continuous algorithmic improvement in decentralized networks?
Disconnected units cannot upload diverse training images to central developers. Future software iterations fail to incorporate novel regional disease presentations, stagnating overall accuracy improvements.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Handheld Systems
- Cart Systems
- Backpack Systems
- Handheld Systems
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By AI Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By AI Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By AI Function, 2026 to 2036
- TB Screening
- Critical Triage
- Full Analysis
- TB Screening
- Y to o to Y Growth Trend Analysis By AI Function, 2021 to 2025
- Absolute $ Opportunity Analysis By AI Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Mode, 2026 to 2036
- Cloud-Connected
- Edge Offline
- Hybrid Mode
- Cloud-Connected
- Y to o to Y Growth Trend Analysis By Deployment Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Public Programs
- Hospitals
- NGOs
- Public Programs
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Model, 2026 to 2036
- Bundled Systems
- Device Lease
- Software License
- Bundled Systems
- Y to o to Y Growth Trend Analysis By Sales Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By AI Function
- By Deployment Mode
- By End Use
- By Sales Model
- Competition Analysis
- Competition Deep Dive
- Qure.ai
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Lunit
- VUNO
- Annalise.ai
- Aidoc
- Delft Imaging
- Qure.ai
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by AI Function, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Sales Model, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by AI Function
- Figure 9: Global Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Deployment Mode
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Sales Model
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by AI Function
- Figure 35: North America Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Deployment Mode
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Sales Model
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by AI Function
- Figure 51: Latin America Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Deployment Mode
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Sales Model
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by AI Function
- Figure 67: Western Europe Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Deployment Mode
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Sales Model
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by AI Function
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Deployment Mode
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Sales Model
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by AI Function
- Figure 99: East Asia Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Deployment Mode
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Sales Model
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by AI Function
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Deployment Mode
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Sales Model
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by AI Function, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by AI Function, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by AI Function
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Deployment Mode
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Sales Model, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Model, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Sales Model
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

