Bioinductive Implant Market
Bioinductive Implant Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Bioinductive Implant Market Forecast and Outlook 2026 to 2036
Surging clinical preference for biological scaffolds is propelling the global bioinductive implant market toward a valuation of USD 3.4 billion by 2036, expanding from a 2026 baseline of USD 1.1 billion at a robust CAGR of 12.1%. Momentum is underpinned by a decisive clinical shift from mechanical fixation to soft tissue healing, as surgeons prioritize reducing re-tear rates in degenerative cases. The industry currently reflects a bifurcation between high-volume adoption in mature economies and rapid infrastructure-led growth in emerging regions like India.
'China volume based procurement (VBP) was a significant headwind, that masked sports medicine's strong performance across the rest of the world.' - Deepak Nath, CEO, Smith+Nephew. According to FMI's estimates, the manufacturer focus is shifting toward securing premium reimbursement by demonstrating superior long-term economic value. Financial burdens from revision surgeries, which cost healthcare systems billions annually, have created an urgent need for implants that ensure first-time healing success.
Data released by the FDA in March 2024 highlights that safety concerns regarding synthetic meshes are accelerating the transition to bio-inductive alternatives in shoulder and achilles repairs. Companies are responding to market pressure by funding rigorous randomized controlled trials to prove efficacy to payers. Establishing these clinical advantages is now the primary lever for sustaining margins in a competitive, value-based healthcare environment.
Quick Stats for Bioinductive Implant Market
- Bioinductive Implant Market Value (2026): USD 1.1 billion
- Bioinductive Implant Market Value (2036): USD 3.4 billion
- Bioinductive Implant Industry Forecast CAGR: 12.1%
- Bioinductive Implant Industry Leading Segment: Rotator Cuff Repair (42%)
- Bioinductive Implant Industry Leading Channel: Hospitals (55%)
- Bioinductive Implant Industry Key Growth Regions: India (15.6%), Brazil (14.8%)
- Bioinductive Implant Industry Key Players: Smith+Nephew, Zimmer Biomet, Stryker, Integra LifeSciences, Arthrex
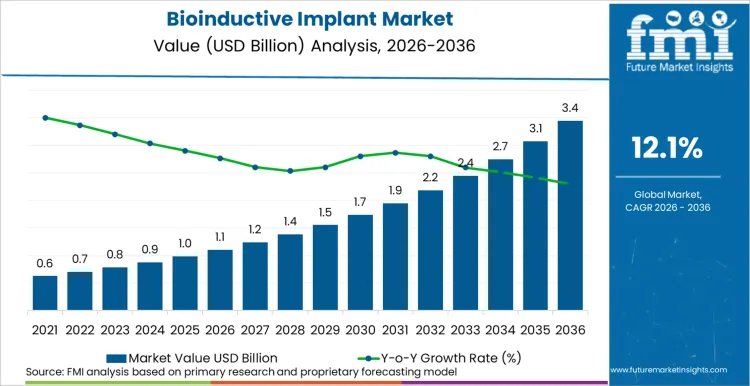
Bioinductive Implant Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.1 billion |
| Industry Value (2036) | USD 3.4 billion |
| CAGR (2026-2036) | 12.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Drivers Are Fueling the Shift from Mechanical to Biological Repair?
Sports-related injuries act as a primary catalyst for industry expansion, necessitating advanced soft tissue repair solutions. Increasing participation in high-impact activities by aging populations has led to a surge in complex tendon tears that resist traditional suturing. FMI analysts opine that standard mechanical fixation often fails in degenerative tissue, driving surgeons toward bio-inductive scaffolds that promote cellular ingrowth. Data from the American Academy of Orthopaedic Surgeons presented in March 2025 indicates that biological augmentation can reduce rotator cuff re-tear rates by over 30%. Such compelling clinical evidence is forcing hospital procurement departments to prioritize regenerative implants despite their higher upfront costs compared to sports medicine sutures.
Why is regulatory harmonization accelerating market access?
Streamlined approval pathways are significantly reducing time-to-market for novel devices, reshaping the global launch landscape. Regulatory bodies are increasingly recognizing the distinct safety profile of bio-inductive materials compared to permanent synthetics, facilitating faster clearances. Recent guidance updates from ANVISA in Brazil, specifically RDC 848/2024, have harmonized local standards with global norms, lowering entry barriers for international manufacturers. This regulatory clarity encourages investment in next-generation orthobiologics pipelines. Consequently, manufacturers can now execute simultaneous product launches across multiple geographies, maximizing return on R&D investment and accelerating global adoption rates.
What financial pressures are driving value-based adoption?
Healthcare systems are intensifying their scrutiny of long-term costs, favoring interventions that prevent expensive revision surgeries. The economic argument for bioinductive implants rests on their ability to lower total cost of care by ensuring successful healing in the primary procedure. FMI analysis indicates that payers are increasingly willing to reimburse premium implants if they demonstrate a reduction in readmission rates. A 2024 industry report by BVMed highlighted that hospitals in Germany are shifting procurement budgets toward technologies that offer proven health-economic benefits. This trend is compelling manufacturers to bundle products with health economic data, moving the sales conversation from price to patient outcomes.
What characterizes the current segmentation landscape?
The market is fundamentally segmented by application, involving rotator cuff repair, Achilles tendon repair, and knee ligament augmentation, alongside material types such as biological grafts and synthetic scaffolds. Rotator cuff repair currently dominates the landscape due to high procedural volumes, while material innovation is pivoting toward hybrid implants that combine durability with biology. End-use segmentation highlights a split between traditional hospitals and fast-growing ambulatory surgery centers. Understanding these specific categories is crucial for stakeholders, as growth rates vary significantly between the mature shoulder segment and emerging foot and ankle applications.
Why does rotator cuff repair hold a dominant share?
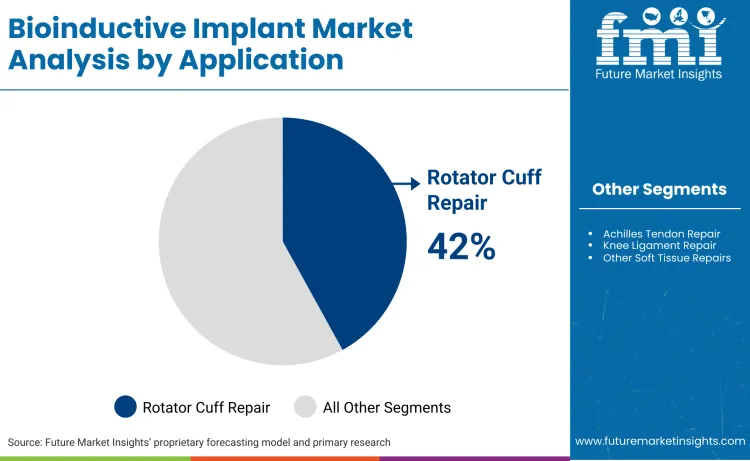
Rotator cuff repair commands a dominant 42% market share, fueled by high failure rates in traditional surgical repairs. Degenerative changes in tendon tissue often preclude successful healing with sutures alone, creating a clinical necessity for augmentation using augmented glenoid systems and patches. Bioinductive implants provide a scaffold for new tissue growth, significantly improving structural integrity. Clinical studies cited by Smith+Nephew in their January 2026 update demonstrate that these implants effectively reverse disease progression in partial-thickness tears. This application acts as the standard-of-care entry point for most manufacturers. As a result, R&D budgets are heavily skewed toward optimizing scaffolds specifically for shoulder anatomy.
Why do hospitals lead in end-user adoption?
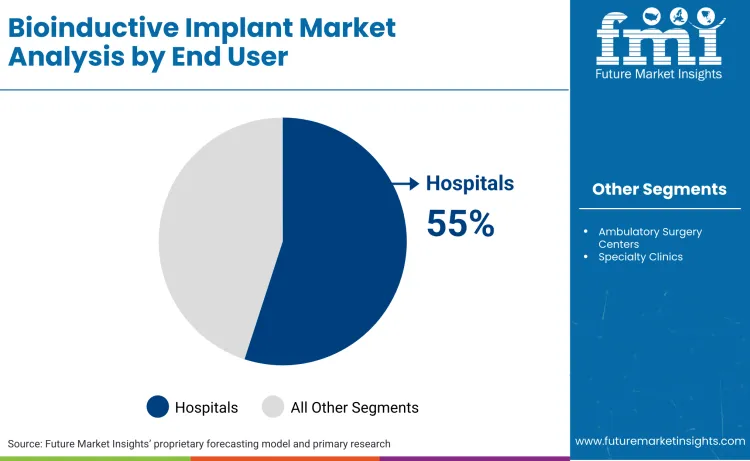
Hospitals maintain a leading position with a 55% share, serving as the primary centers for complex orthopedic procedures. These institutions possess the necessary infrastructure and specialist teams to handle high-acuity cases requiring biological augmentation and advanced arthroscopy devices. Large hospital networks benefit from centralized procurement, allowing them to negotiate favorable pricing for high-volume implant orders. Paul Tornetta III, President of AAOS, noted during his March 2025 address that 'we have the tools to contribute to the greater good,' highlighting the pivotal role of institutional support. Hospitals are also early adopters of robotic-assisted surgery. This creates a sticky ecosystem where bio-implants are integrated into broader surgical protocols.
How is the shift to ambulatory care impacting product design?
Procedures are migrating from inpatient settings to ASCs, driven by payer pressure for cost containment and patient preference for same-day discharge. Sports medicine companies are designing simplified delivery systems tailored for this outpatient environment, prioritizing speed and ease of use. Integra LifeSciences launched its Micro-Matrix system in early 2025 specifically to address efficiency needs in smaller surgical suites. This operational shift demands that manufacturers create "procedure-in-a-box" solutions that minimize inventory complexity for space-constrained centers.
Why are hybrid biologics gaining traction over pure synthetics?
Innovation is focusing on combining synthetic strength with biologic healing properties to address the limitations of singular materials. Manufacturers are developing hybrid scaffolds utilizing recombinant collagen that offer immediate mechanical support while facilitating long-term tissue regeneration. Zimmer Biomet's 2025 annual report emphasizes a strategic pivot toward these "active healing" technologies to differentiate from generic collagen patches. By merging durability with bio-integration, these hybrids offer a "best of both worlds" solution that appeals to surgeons managing active patients.
What role does personalized medicine play in implant evolution?
Advances in 3D bioprinting are enabling patient-specific implant designs that conform perfectly to individual anatomical defects. This trend allows for precise anatomical matching, reducing surgical time and improving graft integration in complex cartilage repair cases. Research from major universities published in 2024 has validated the efficacy of custom-printed scaffolds in reducing operative risks. FMI observes that the industry is gradually moving away from off-the-shelf sizing toward a model of mass customization, driven by digital imaging integration.
How Will Bioinductive Implant Market Expansion Unfold Across Key Global Regions?
Global bioinductive implant markets reflect a fundamental divergence between developed and emerging economies, with the former battling reimbursement constraints while the latter race to expand access for rapidly aging populations. Developed nations like the US and Germany prioritize value-based care, demanding rigorous clinical evidence to justify premium pricing. Conversely, emerging markets in Asia and Latin America are driven by volume expansion and infrastructure modernization. As per FMI's projection, this duality forces multinational corporations to adopt bifurcated strategies—delivering high-tech innovation to mature markets while optimizing supply chains for volume in developing regions. This geographic nuance is defining global asset allocation strategies.
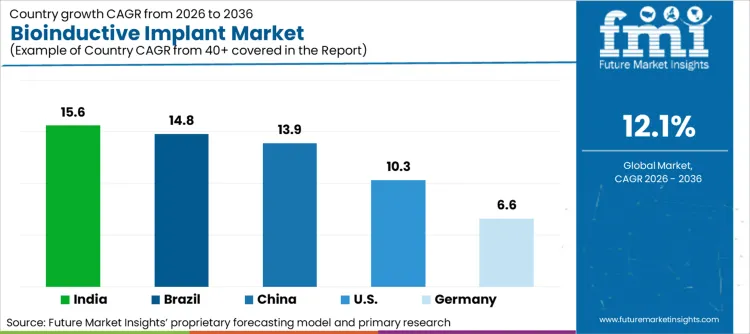
| Country | CAGR (2026–2036) |
|---|---|
| India | 15.6% |
| Brazil | 14.8% |
| China | 13.9% |
| United States | 10.3% |
| Germany | 6.6% |
Source: FMI historical analysis and forecast data.
Why is the Indian market evolving?
Demand for bioinductive implants in India is anticipated to grow at 15.6% CAGR. Government investment in Tier-1 healthcare infrastructure has expanded capacity by 34% as of July 2024, while corporate chains like Apollo and Max Healthcare are aggressively integrating regenerative medicine technologies. The "Heal in India" initiative is effectively turning the country into a medical tourism hub for orthopedics, necessitating world-class implant inventory. Dr. Prathap C. Reddy, Chairman of Apollo Hospitals, stated in the September 2025 Integrated Annual Report: 'At the heart of this effort is a simple truth; the only way to future-proof an organisation like ours, is to stay rooted in timeless values while embracing transformative innovation.' Such a commitment to high-tech adoption signals a departure from purely cost-driven procurement. India is rapidly emerging as a center for quality-focused orthopedic excellence rather than just a volume market.
What fuels growth in Brazil?
Sales of bioinductive implants in Brazil are set to rise at a solid 14.8% CAGR. The implementation of RDC 848/2024 has lowered entry barriers for high-tech implants by aligning safety requirements with international standards. This regulatory clarity is triggering a surge in imports; the bio-implants and advanced device segments alone grew over 30% in 2024, signaling a robust appetite for foreign medical technology. José Fernando Dantas from ABIMO noted in a January 2025 trade statement: 'The positive performance shows that the Brazilian Health Devices project continues to be efficient in its goal of promoting the Brazil brand in the international medical device market.' This successful integration into the global supply chain has revitalized local distribution networks. Manufacturers can now leverage Brazil as a streamlined, regulatory-friendly gateway to the broader Latin American region.
How is China balancing volume and value?
The bioinductive implant industry in China is projected to expand at 13.9% CAGR. While the sheer patient volume drives demand, Volume-Based Procurement (VBP) policies have forced a pricing reset. Global players are navigating this by pivoting to premium segments not yet covered by VBP, while domestic firms capitalize on state support. The market dynamics are currently defined by this friction between price caps and the clinical need for advanced soft tissue repair. Deepak Nath, CEO of Smith+Nephew, observed in a January 2026 trading update: 'China volume based procurement (VBP) was a significant headwind, that masked sports medicine's strong performance across the rest of the world.' Navigating these headwinds requires a nuanced "local partnership" approach. Foreign entrants must adapt to lower margins by focusing on premium, non-VBP procedure categories to sustain profitability.
Why does the USA remain the innovation hub?
Bioinductive implant market in the USA is poised to register a 10.3% CAGR. The FDA's rigorous focus on safety, exemplified by the CDRH 2024 Safety Report, favors established players with deep clinical data. With over 60 million Americans participating in fitness activities, the "weekend warrior" demographic is driving procedure volumes in Ambulatory Surgery Centers, where efficient, proven bio-interventions are preferred over complex revisions. Paul Tornetta III, President of AAOS, stated in his March 2025 address: “As orthopaedic surgeons, we share a strong bond based in helping others. We have the tools to contribute to the greater good – to make a positive impact on the well-being of others that few people can” This clinical optimism reinforces the market's resilience against economic pressures. The region stands firm as the global benchmark for value-based reimbursement and technological adoption.
What drives technology adoption in Germany?
Bioinductive implant sector in Germany is forecast to grow at 6.6% CAGR. As the largest EU market, Germany is battling rising energy and manufacturing costs (BVMed 2024 Report), yet it remains aggressive in adopting value-based care. The healthcare system's focus is shifting toward technologies that offer long-term cost savings through reduced revision rates, perfectly aligning with the value proposition of bioinductive implants. Bernd Montag, CEO of Siemens Healthineers, remarked in an April 2025 interview: 'Real meaningful innovations make healthcare better and cheaper... I am always a bit careful when it comes to this notion of expensive technology. You can look at it as an investment, and not only as a cost.' This economic mindset prioritizes efficiency over raw purchase price. Consequently, suppliers must position their devices as long-term assets that alleviate the financial strain on statutory insurance funds.
What Strategic Moves Are Defining Competitive Landscape?
Leading manufacturers are aggressively consolidating the market by acquiring complementary technologies to offer end-to-end soft tissue solutions. FMI predicts that the industry is moving away from standalone product offerings toward integrated surgical platforms that combine implants, fixation devices, and visualization tools. Stryker's acquisition of SERF SAS in March 2024 exemplifies this strategy, aiming to fortify its European joint replacement footprint which serves as a distribution channel for its broader sports medicine portfolio. By controlling the entire procedural stack, from suture anchor devices to biologic grafts, companies like Stryker and Zimmer Biomet can lock out niche competitors and bundle pricing for hospital networks.
Differentiation is increasingly driven by rigorous clinical data rather than just material novelty. As payers tighten reimbursement criteria, companies are forced to prove that their bioinductive implants statistically reduce revision rates compared to standard of care. Smith+Nephew has spearheaded this approach by publishing pivotal RCT data for its Regeneten portfolio, validating a 68% reduction in re-tear rates. Deepak Nath, CEO of Smith+Nephew, emphasized the importance of this performance in January 2026: 'Sports medicine's strong performance across the rest of the world continues to drive portfolio growth.' FMI observes that competitors lacking such Level 1 evidence are facing rapid commoditization. The market is evolving into a "prove it or lose it" landscape where clinical validity dictates market share.
Key Players in Bioinductive Implant Market
- Smith+Nephew
- Zimmer Biomet
- Stryker
- Integra LifeSciences
- Arthrex
- Conmed Corporation
- MiMedx
- Organogenesis
- Rotation Medical
- Cokogel
Market Definition
The bioinductive implant market represents revenue generated from implantable medical devices designed to stimulate biological tissue regeneration and healing in orthopedic and soft-tissue repair procedures. As operationally defined in the article, the market measures commercial sales of bioinductive scaffolds, patches, and hybrid implants that promote cellular ingrowth and tendon or ligament regeneration, primarily used to reduce re-tear rates and improve long-term surgical outcomes. Market sizing reflects manufacturer revenues from these implants, analysed by application, material type, end user, and region, and expressed in USD billion.
The market includes bioinductive implants used across rotator cuff repair, Achilles tendon repair, knee ligament augmentation, and other soft-tissue reinforcement applications explicitly referenced in the article. It covers products based on biological grafts, synthetic scaffolds, and hybrid biomaterials incorporating collagen or regenerative matrices. Revenue generated from use in hospitals and ambulatory surgical centers is included, with hospitals identified as the dominant end-user segment. The scope counts implants deployed in sports medicine, orthopedic surgery, and related minimally invasive procedures, including products bundled with arthroscopy systems, fixation devices, or delivery tools as part of integrated surgical platforms. Market coverage spans North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa.
The market excludes revenues from traditional orthopedic fixation devices such as sutures, anchors, screws, and plates sold without bioinductive or regenerative functionality. Surgical procedures, physician fees, hospital stays, rehabilitation services, and physical therapy are not included. Non-implant biologics such as injectable PRP, stem cell therapies, or pharmaceutical biologics are outside scope unless integrated into implantable scaffolds. General wound care products, meshes not positioned as bioinductive, and experimental research implants not commercially marketed are excluded. Government healthcare spending, reimbursement payments, regulatory fees, and public research funding referenced contextually are not counted as market revenue.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.1 Billion |
| Solution Type | Rotator Cuff Repair, Achilles Tendon Repair, Knee Ligament Repair, Other Soft Tissue Repairs |
| Material Type | Biological Grafts (Bovine, Porcine), Synthetic Scaffolds, Hybrid Implants |
| End User | Hospitals, Ambulatory Surgery Centers, Specialty Clinics |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, United Kingdom, Germany, India, China, Japan, Brazil, and 15+ others |
| Key Companies Profiled | Smith+Nephew, Zimmer Biomet, Stryker, Integra LifeSciences, Arthrex, Conmed, MiMedx, Organogenesis |
| Additional Attributes | Dollar sales by application and material, regulatory impact analysis (RDC 848/2024, FDA Safety Reports), reimbursement landscape assessment, supply chain evolution, and competitive market share analysis |
Source: FMI historical analysis and forecast data.
Bioinductive Implant Market by Segments
By Application:
- Rotator Cuff Repair
- Achilles Tendon Repair
- Knee Ligament Repair
- Other Soft Tissue Repairs
By Material Type:
- Biological Grafts (Bovine, Porcine)
- Synthetic Scaffolds
- Hybrid Implants
By End User:
- Hospitals
- Ambulatory Surgery Centers
- Specialty Clinics
By Region
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- USA Food and Drug Administration, Center for Devices and Radiological Health. (2024). CDRH 2024 safety report. USA Department of Health and Human Services.
- American Academy of Orthopaedic Surgeons. (2025, March). Annual meeting address. AAOS.
- Brazilian Association of Medical and Dental Equipment, Articles and Supplies. (2025, January). Brazilian health devices industry statement (Press release). ABIMO.
- Bundesverband Medizintechnologie. (2024). BVMed annual report 2024. BVMed.
- Apollo Hospitals Enterprise Limited. (2025, September). Integrated annual report 2024–25. Apollo Hospitals.
- Smith & Nephew plc. (2026, January). Trading update. Smith & Nephew.
- Zimmer Biomet Holdings, Inc. (2025, February). Annual report 2025. Zimmer Biomet.
- Stryker Corporation. (2024, March). Press release: Acquisition of SERF SAS. Stryker
Frequently Asked Questions
How big is the bioinductive implant market in 2026?
The global bioinductive implant market is estimated to be valued at USD 1.1 billion in 2026.
What will be the size of bioinductive implant market in 2036?
The market size for the bioinductive implant market is projected to reach USD 3.4 billion by 2036.
How much will be the bioinductive implant market growth between 2026 and 2036?
The bioinductive implant market is expected to grow at a 12.1% CAGR between 2026 and 2036.
What are the key product types in the bioinductive implant market?
The key product types in bioinductive implant market are collagen bioinductive patches, ecm scaffolds (xenograft/allograft), synthetic bioinductive scaffolds, bioactive-coated repair adjuncts and fixation & delivery accessories.
Which applications / procedures segment to contribute significant share in the bioinductive implant market in 2026?
In terms of applications / procedures, rotator cuff repair augmentation segment to command 42.0% share in the bioinductive implant market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Bioinductive Implant Types
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Bioinductive Implant Types , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Bioinductive Implant Types , 2026 to 2036
- Collagen bioinductive patches
- ECM scaffolds (xenograft/allograft)
- Synthetic bioinductive scaffolds
- Bioactive-coated repair adjuncts
- Fixation & delivery accessories
- Collagen bioinductive patches
- Y to o to Y Growth Trend Analysis By Bioinductive Implant Types , 2021 to 2025
- Absolute $ Opportunity Analysis By Bioinductive Implant Types , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications / Procedures
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications / Procedures, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications / Procedures, 2026 to 2036
- Rotator cuff repair augmentation
- Tendon repair (Achilles/hand)
- Ligament repair & reconstruction adjuncts
- Meniscus/cartilage repair support
- Other sports medicine procedures
- Rotator cuff repair augmentation
- Y to o to Y Growth Trend Analysis By Applications / Procedures, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications / Procedures, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users / Sales Channels
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users / Sales Channels, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users / Sales Channels, 2026 to 2036
- Hospitals
- Ambulatory surgery centers (ASCs)
- Orthopedic specialty clinics
- Distributor channels
- GPO / tender procurement
- Hospitals
- Y to o to Y Growth Trend Analysis By End Users / Sales Channels, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users / Sales Channels, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Bioinductive Implant Types
- By Applications / Procedures
- By End Users / Sales Channels
- Competition Analysis
- Competition Deep Dive
- Smith+Nephew
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Arthrex
- Zimmer Biomet
- Stryker
- Integra LifeSciences
- Others
- Smith+Nephew
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Bioinductive Implant Types , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications / Procedures, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 6: Global Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Applications / Procedures
- Figure 9: Global Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Users / Sales Channels
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 26: North America Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Applications / Procedures
- Figure 29: North America Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Users / Sales Channels
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications / Procedures
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users / Sales Channels
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications / Procedures
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users / Sales Channels
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications / Procedures
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users / Sales Channels
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications / Procedures
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users / Sales Channels
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications / Procedures
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users / Sales Channels
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Bioinductive Implant Types , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Bioinductive Implant Types , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Bioinductive Implant Types
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications / Procedures, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications / Procedures, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications / Procedures
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users / Sales Channels
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

