Orthopedic Trauma Device Market
The Orthopedic Trauma Device Market is segmented by Product (Internal Fixators and External Fixators); End User (Hospitals, Orthopedic and Trauma Centers and Ambulatory Surgical Centers); and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Orthopedic Trauma Device Market Size, Market Forecast and Outlook By FMI
The orthopedic trauma device market surpassed a valuation of USD 11.5 billion in 2025. The market is projected to reach USD 12.2 billion by 2026-end and expand at a CAGR of 6.4% between 2026 and 2036 to reach USD 22.6 billion by 2036. Internal fixators are expected to dominate with an 80.6% product share, and hospitals are expected to lead the end user segment with a 56.4% share.
Summary of the Orthopedic Trauma Device Market
- Demand and Growth Drivers
- Road crash injury care keeps demand steady as fracture patients need plates, screws and fixation sets for surgical repair.
- Geriatric fall care expands the trauma procedure base as hip and wrist fractures need dependable fixation options.
- Hospital trauma systems favor established implant sets as emergency cases need fast tray access and surgeon familiarity.
- Product and Segment View
- Internal fixators are expected to lead the product segment with 80.6% share in 2026, reflecting broad fracture repair use.
- Hospitals are expected to lead end user demand with 56.4% share in 2026, reflecting operating room access and trauma care depth.
- External fixators are expected to hold specialist demand as staged trauma care needs temporary stabilization before final fixation.
- Geography and Competitive Outlook
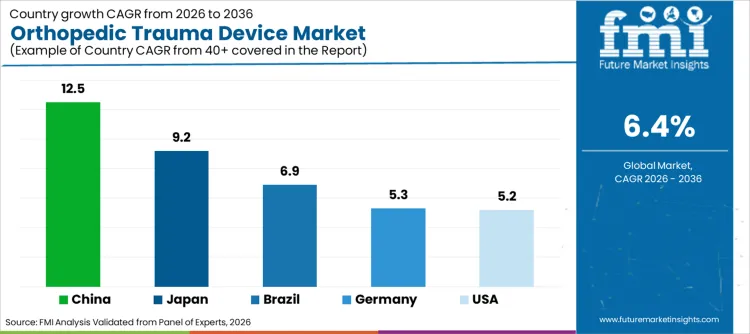
- China is expected to record a CAGR of 12.5% through 2036, driven by hospital expansion and trauma care investment.
- Japan is projected to expand at 9.2% CAGR through 2036, as older adult fracture care needs high-quality fixation.
- Companies combining broad implant portfolios with surgeon training are strengthening market position during the forecast period.
- Analyst Opinion
- Anurag Sharma, Principal Consultant at FMI, says, “Orthopedic trauma devices are becoming evidence-based surgical systems for fracture repair. Hospital teams need familiar instruments with dependable case support. Internal fixation stays central through plates and nails. Public tenders pressure pricing while broad nailing coverage reduces urgent tray gaps.”
- Orthopedic Trauma Device Market Value Analysis
- The orthopedic trauma device market is moving from basic fracture hardware into procedure-specific implant systems.
- Adoption is being driven by fracture repair needs across road trauma and older adult fall injuries.
- The market is benefiting from deeper use of procedure-specific fixation products across hospital trauma programs.
- Demand is backed by the need for implant systems meeting safety and surgical workflow expectations.

Orthopedic trauma device makers face two practical pressures: stronger approval files and easier hospital use during urgent fracture surgery. The FDA issued guidance in November 2024 that defines expectations for 510(k) submissions covering non spinal plates and screws. Manufacturers must develop stronger testing records before finalizing product launch plans and regulatory submissions. Stryker’s February 2026 T2 Alpha Humerus Nailing System includes active intraoperative compression of up to 6 mm and uses its wider T2 Alpha platform, which can help hospitals reduce training gaps and tray variation. Johnson & Johnson MedTech launched VOLT wrist and proximal humerus plating systems
in June 2025 for two common fracture sites, pointing to a stronger focus on upper-extremity fixation. Zimmer Biomet’s October 2025 launch for pilon and hindfoot trauma adds depth in complex foot and ankle cases, with pilon fractures accounting for about 7% to 10% of tibial fractures.
Orthopedic Trauma Device Market Definition
The orthopedic trauma device market includes implants used to stabilize fractured bones after injury. Scope includes internal fixators, external fixators, plates, screws, rods and trauma fixation systems. Trauma products serve emergency fracture repair and planned reconstruction after bone injury caused by accidents. Joint replacement implants, spinal devices and sports medicine anchors are outside the scope.
Orthopedic Trauma Device Market Inclusions
Market scope covers all commercially traded orthopedic trauma devices categorized by product type, including internal fixators and external fixators; materials, including titanium, stainless steel, and bioabsorbable materials; fixation sites, including lower extremity, upper extremity, pelvic fixation, and cranial fixation; and end users, including hospitals, orthopedic and trauma centers, and ambulatory surgical centers. Revenue scope covers 2026 to 2036. Key stakeholders include orthopedic trauma device manufacturers, implant material suppliers, component providers, hospitals, trauma care centers, orthopedic surgeons, distributors, group purchasing organizations, regulatory bodies, and healthcare reimbursement agencies.
Orthopedic Trauma Device Market Exclusions
Scope does not include joint reconstruction implants, spinal implants, sports medicine repair systems, orthopedic braces, dental implants or bone growth stimulators.
Orthopedic Trauma Device Market Research Methodology
- Primary Research: FMI analysts spoke to orthopedic surgeons, hospital buying teams, trauma device makers and distributor partners in important markets.
- Desk Research: Combined data from regulatory guidance, public injury datasets, company product portfolios and orthopedic device classifications.
- Market sizing and forecasting: Added trauma fixation use across product groups and end user channels from the bottom up.
- Data validation: Cross-checked every three months against fracture care patterns, company product coverage and country-level trauma indicators.
Why is the Orthopedic Trauma Device Market Growing?
- Road injury cases create recurring demand for internal fixation devices across emergency orthopedic surgery.
- Older adult fall injuries increase the need for hip nails and wrist plate systems.
- Hospitals are standardizing trauma sets to reduce case delay and improve operating room readiness.
Road trauma raises a large recurring surgical base for orthopedic trauma devices across emergency systems. Road trauma enhances steady need for fracture repair across emergency care systems. Patients arriving after serious crashes often need implants for bone stabilization and surgical recovery. Internal fixation sets gain preference as surgeons need plates, screws, and nails suited to different bone sites. Hospitals value consistent instruments and tray layouts because trauma surgery often allows limited planning time.
Older adult falls add a separate fracture pathway in established health systems across trauma departments. CDC reported in February 2026 over 14 million adults aged 65 years and older fall each year in the United States. CDC notes 37% of people who fall report injury needing medical treatment or activity limits. This boosts demand for hip fixation nails and distal radius plates across hospitals. Suppliers with geriatric fracture portfolios can protect share through simpler instrumentation and dependable logistics.
Market Segmentation Analysis
- Internal fixators are forecast to hold 80.6% of product demand in 2026 as plates and nails cover most operative fracture repair needs.
- Hospitals are projected to account for 56.4% of end user use in 2026, reflecting the need for imaging access and operating room readiness.
- External fixators serve a specialist role across open fractures and staged trauma cases requiring temporary bone stabilization before final repair.
- Orthopedic and trauma centers are expected to capture focused procedure volume as complex fracture cases need dedicated surgical teams and deeper fixation planning.
- Ambulatory surgical centers are projected to handle selective trauma procedures as lower-acuity fracture cases move into planned outpatient care.
The market for orthopedic trauma devices is divided into groups based on product type, material, fixation site, end user, sales channel, and region. Under product type, the market includes internal fixators and external fixators used for fracture stabilization. By material, the market covers titanium, stainless steel, and bioabsorbable materials used across different implant requirements. Based on fixation site, it includes lower extremity, upper extremity, pelvic fixation, and cranial fixation applications. By end user, the market includes hospitals, orthopedic and trauma centers, and ambulatory surgical centers. By sales channel, the market is segmented into direct sales, distributor sales, and group purchasing.
Insights into Orthopedic Trauma Device Market by Product
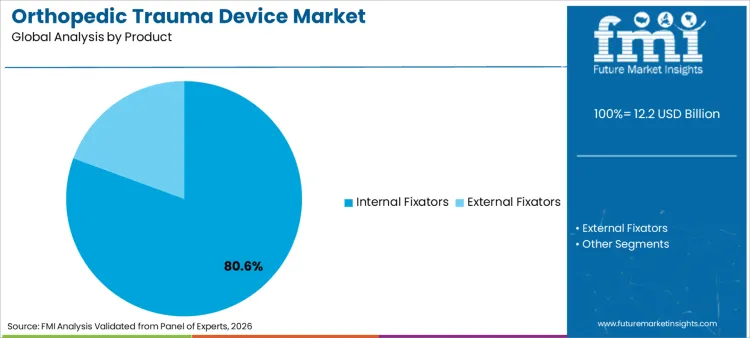
- In 2026, internal fixators are expected to contribute 80.6% of the product segment. Plates, screws and intramedullary nails support stable fracture repair across high-volume trauma procedures.
- External fixation products address selected open fractures and staged trauma cases. Their use improves in injuries with swelling, soft tissue damage or infection risk before final fixation.
Insights into Orthopedic Trauma Device Market by End User
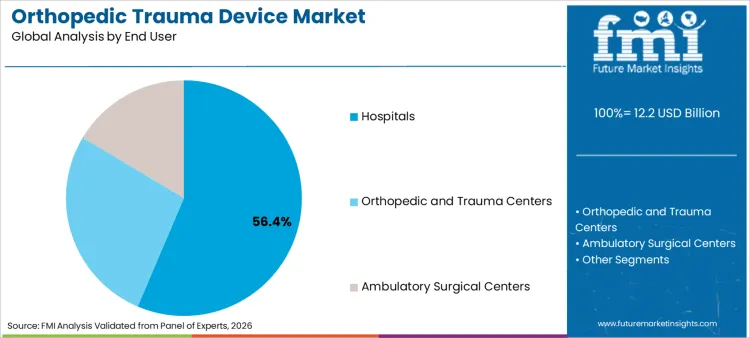
- Hospitals are projected to account for 56.4% of end user demand in 2026. Emergency care access and operating room readiness make hospitals the main trauma fixation setting.
- Orthopedic and trauma centers address complex referral cases needing focused fracture care. Their value improves in procedures requiring dedicated surgical teams and intraoperative imaging access.
Insights into Orthopedic Trauma Device Market by Material
- Titanium is expected to hold 48.7% of material demand in 2026. Its use is supported by strength and compatibility across plates, screws and nailing systems.
- Stainless steel remains important in cost-sensitive trauma programs. Bioabsorbable materials serve selected cases where temporary fixation and reduced implant removal needs influence use.
Insights into Orthopedic Trauma Device Market by Fixation Site
- Lower extremity fixation is projected to account for 52.1% of fixation site demand in 2026. Femur, tibia and ankle fractures create steady procedure volume across trauma hospitals.
- Upper extremity fixation gains demand from wrist and humerus fracture repair. Pelvic and cranial fixation remain specialist areas requiring deeper surgical planning and focused implant sets.
Insights into Orthopedic Trauma Device Market by Sales Channel
- Direct sales are expected to account for 46.3% of sales channel value in 2026. Hospital systems prefer direct supplier support for implant trays and urgent surgical case coverage.
- Distributor sales remain relevant in regional hospitals where local inventory access matters. Group purchasing channels influence pricing across large hospital networks and trauma care systems.
Orthopedic Trauma Device Market Drivers, Restraints, and Opportunities
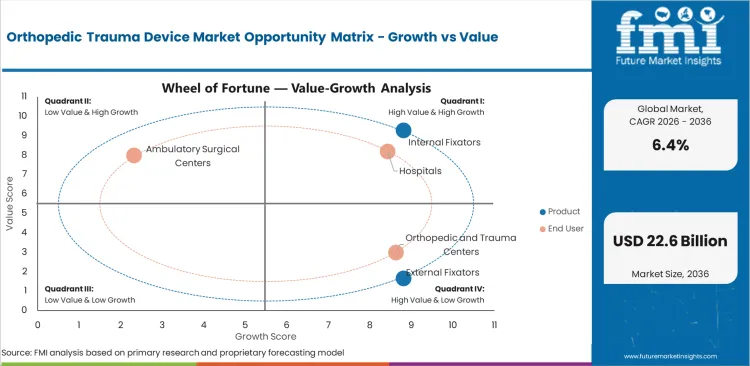
- Fracture repair volume raises demand for internal fixation products across hospital trauma departments.
- FDA evidence expectations raise submission cost for smaller makers of plates and screws.
Specialty extremity trauma systems improve opportunity as surgeons seek anatomical plates and dedicated nails.
Orthopedic trauma devices keep expanding as hospitals need stable fracture fixation systems and reliable implant trays for emergency surgery. Cost pressure and limits on premium titanium systems can slow broader use. Demand continues through internal fixators and geriatric fracture repair applications.
Bundled Inpatient Payments Are Tightening Implant Price Headroom
Orthopedic trauma device pricing is being squeezed by the gap between hospital input costs and fixed-case reimbursement. In January 2026, CMS stated that FY 2026 IPPS operating payment rates rise 2.6% for qualifying acute-care hospitals, based on a 3.3% hospital market basket update less a 0.7 percentage-point productivity adjustment; CMS noted 772 MS-DRGs for FY 2026. For trauma fixation cases, plates and screws as well as intramedullary nails and external fixation systems are included in episode based payment models. This structure creates stronger resistance to implant price increases, as suppliers must justify higher costs. They can show value through shorter procedure time or reduced revision risk, or they can demonstrate meaningful inventory savings. This keeps price movement selective instead of broad-based.
FDA Fees Are Raising Commercialization Cost Floors
Regulatory cost inflation is creating a clear minimum threshold for orthopedic trauma device pricing. This effect is especially evident for manufacturers introducing new fixation designs or biologic adjacent trauma products or modified implants. In September 2025, FDA listed FY 2026 MDUFA fees effective October 1, 2025, through September 30, 2026, including an annual establishment registration fee of US$11,423, a standard 510(k) fee of US$26,067, De Novo fee of US$173,782, and PMA/PDP/PMR/BLA fee of US$579,272. For commodity trauma hardware, these costs pressure margins; for differentiated systems, they encourage premium pricing and tighter launch discipline. Smaller trauma device firms may rely on reduced small-business fees, but scale players can spread approval costs across higher volumes.
Analysis of Orthopedic Trauma Device Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 12.5% |
| Japan | 9.2% |
| Brazil | 6.9% |
| Germany | 5.3% |
| USA | 5.2% |
Source: Future Market Insights, 2026.
Orthopedic Trauma Device Market CAGR Analysis By Country
- China is projected to lead country-level expansion at 12.5% CAGR through 2036, with hospital capacity improving access to trauma fixation procedures.
- Japan’s 9.2% CAGR reflects older adult fracture care and stronger preference for precision implant systems during the forecast period.
- Brazil is expected to grow at 6.9% CAGR as road injury treatment keeps trauma fixation use active across public hospitals.
- Germany at 5.3% CAGR and the USA at 5.2% CAGR reflect established hospital systems focused on controlled implant upgrades by 2036.
Demand for orthopedic trauma devices is forecast to rise at 6.4% CAGR from 2026 to 2036. Country-level analysis covers the major markets contributing to this forecast, with the key markets listed below.
Demand Outlook for Orthopedic Trauma Device Market in China
China’s orthopedic trauma device industry is projected to record a CAGR of 12.5% through 2036. The September 2025 Asian Transport Observatory profile reported China’s road crash fatality rate at 17.4 per 100,000 population in 2021. This burden keeps fracture repair capacity relevant across trauma hospitals. Local demand favors suppliers with broad plate sets and dependable nailing systems. Domestic makers gain room as volume-based buying puts pressure on imported premium implants.
- Tier 2 hospitals in inland provinces need trauma trays suited to higher emergency case throughput.
- National road safety targets through 2025 place more attention on injury prevention and emergency response.
- Provincial trauma centers need implant availability across femur, tibia and ankle fixation programs.
Sales Analysis of Orthopedic Trauma Device Market in Japan
The orthopedic trauma device sector in Japan is expected to expand at a CAGR of 9.2% during the assessment period. Japan’s National Police Agency reported in January 2026 road traffic fatalities of 2,547 in 2025. The number was the lowest under the current formula since 1948. Trauma device demand is shaped less by road expansion and more by older patient fracture repair. Suppliers benefit from precision implants suited to smaller anatomical profiles.
- Tokyo and Osaka hospitals need implant systems suited to complex fractures in older adults.
- National Police Agency safety records point to lower crash deaths but persistent injury treatment needs.
- University hospitals often prefer supramalleolar osteotomy implants for complex lower limb care.
Demand Outlook for Orthopedic Trauma Device Market in Brazil
The Brazil orthopedic trauma device industry is forecast to grow at a CAGR of 6.9% by 2036. The World Bank reported in August 2025 over 34,000 road traffic injury fatalities in Brazil during 2023. This road trauma burden creates continued demand for fracture fixation in public hospitals. Lower-cost internal fixation systems gain importance as SUS-linked facilities manage high caseloads. External fixation retains value in severe injury stabilization.
- São Paulo and Minas Gerais trauma centers carry high procedure volumes across road injury admissions.
- Federal road safety programs keep attention on motorcycle injury reduction and emergency care access.
- Public hospitals need durable implants and dependable distributor coverage outside the largest cities.
Opportunity Analysis of Orthopedic Trauma Device Market in Germany
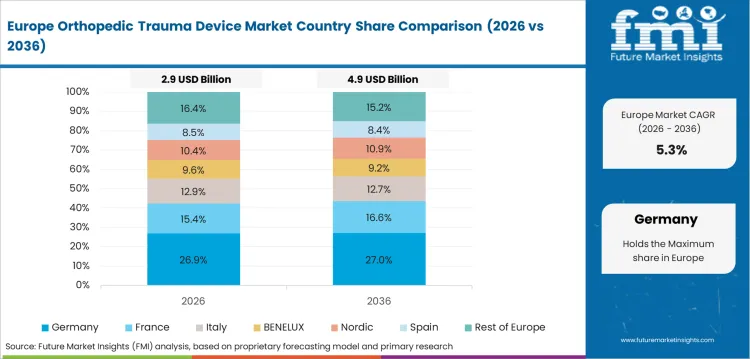
The orthopedic trauma device segment in Germany is forecast to expand at 5.3% CAGR over the forecast years. Destatis listed 2,770 road traffic deaths and 50,601 serious injuries in 2024. This injury base sustains trauma procedure demand in a established surgical system. German buying teams place strong weight on documentation, metallurgy and proven clinical performance. Premium knee arthrodesis implant use reflects preference for complex reconstructive options.
- Bavaria and North Rhine-Westphalia hold large hospital networks with specialized orthopedic trauma capacity.
- BG trauma clinics increase demand for workplace and road injury fracture repair.
- German hospitals favor validated implants with precise instruments and stable after-sales service.
Future Outlook for Orthopedic Trauma Device Market in the United States
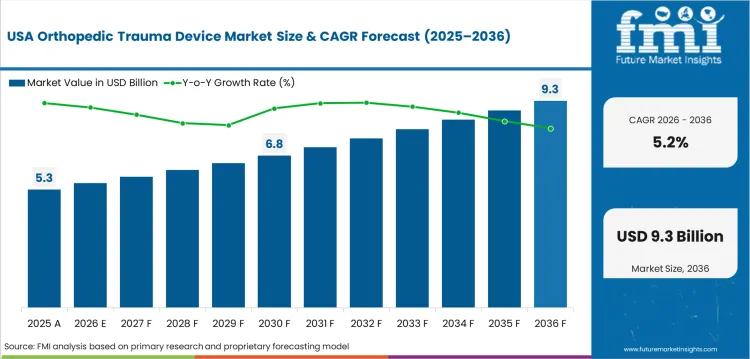
The USA orthopedic trauma device industry is expected to post 5.2% CAGR through 2036. NHTSA estimated in April 2026 traffic fatalities of 36,640 in 2025, with a fatality rate of 1.10 per 100 million vehicle miles traveled. This reflects a large trauma care base despite better road safety performance. Hospitals value suppliers with full trauma trays and field service for urgent cases. Value committees watch implant cost closely as reimbursement pressure affects case economics.
- Level I trauma centers in California and Texas need rapid implant access across high-acuity fracture cases.
- Medicare payment pressure favors implant families with proven operating room efficiency.
- Large group purchasing organizations influence trauma device contracts across multi-hospital systems.
Competitive Landscape and Strategic Positioning
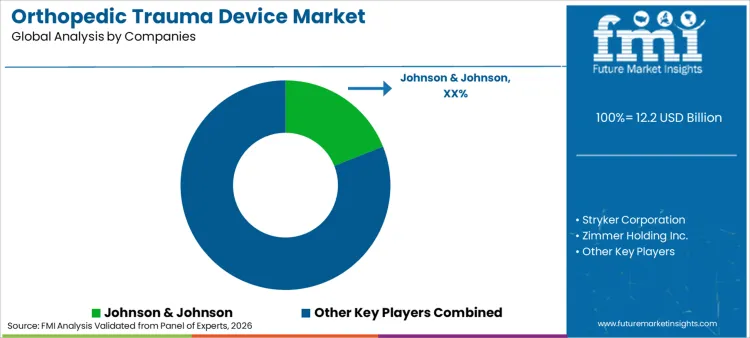
- Johnson & Johnson leads through DePuy Synthes trauma systems and broad surgeon training coverage across major hospital networks.
- Stryker Corporation and Zimmer Biomet compete on global trauma portfolios and specialty extremity systems for complex fracture procedures.
- Specialist providers focus on anatomical plates and targeted fixation products with procedure-specific depth across selected fracture sites.
Competition in the orthopedic trauma device market is shaped by implant breadth emphasize on complete plate and nail systems helping hospitals reduce tray gaps during emergency cases. Focused players focus on anatomical plates and extremity fixation products aimed at selected procedure needs.
Johnson & Johnson holds a strong position through integrated trauma platforms covering plates and nails across hospital accounts. Stryker offers deep fixation coverage and adjacent orthopedic power tools for surgical teams managing fracture repair. Zimmer Biomet has expanded foot and ankle capability through Paragon 28 and related specialty systems. Smith+Nephew competes on EVOS plating and TRIGEN nailing systems across major trauma workflows. Arthrex holds a specialist role across extremity repair and surgical tool networks.
High implant set cost and strict regulatory documentation create barriers across trauma device submissions. Competitive priorities center on locking plate depth and faster tray replenishment across hospital networks. Hospital buying teams favor suppliers offering internal fixation breadth and dependable case help during urgent fracture repair. Companies with broad trauma portfolios and stronger surgeon training are positioned to win long-cycle contracts.
Key Companies in the Orthopedic Trauma Device Market
Key global companies leading the orthopedic trauma device market include:
- Johnson & Johnson, Stryker Corporation and Zimmer Biomet have broad trauma fixation infrastructure and global surgeon education reach.
- Smith+Nephew and Arthrex serve hospitals through extremity systems and trauma repair products.
- Invibio Ltd. holds an adjacent position through surgical visualization and implant material capability.
Competitive Benchmarking: Orthopedic Trauma Device Market
| Company | Locking Plate Depth | Nailing System Coverage | Surgeon Training Reach | Geographic Footprint |
|---|---|---|---|---|
| Johnson & Johnson | High | High | Strong | Global presence through DePuy Synthes, with broad orthopaedics coverage across North America, Europe and Asia Pacific. |
| Stryker Corporation | High | High | Strong | Global medical technology footprint, with strong orthopedic reach across the United States, Europe and key Asia Pacific markets. |
| Zimmer Biomet | High | Medium | Strong | Operations in 25+ countries and sales in 100+ countries, with a strong base across the United States and Europe. |
| Smith+Nephew | High | High | Strong | Operates in around 100 countries, with orthopedic coverage across the United States, Europe and selected Asia Pacific markets. |
| Arthrex Inc. | Medium | Medium | Strong | Headquartered in Florida, with regional offices in Germany and Singapore serving North America, EMEA and Asia Pacific. |
| Invibio Ltd. | Low | Low | Moderate | UK-based global biomaterials supplier, with teams in Europe, the United States and Asia for implant material customers. |
Source: Future Market Insights, 2026.
Key Developments in Orthopedic Trauma Device Market
- In June 2025, Johnson & Johnson MedTech launched the VOLT™ Distal Radius Wrist and VOLT™ Proximal Humerus 3.5 Plating Systems in the USA The development is directly relevant to the Orthopedic Trauma Device Market because both systems target common fracture sites and expand the company’s trauma plating portfolio. The systems use Variable Angle Optimized Locking Technology and are designed to improve stability, expand surgical options, and support treatment of upper-extremity fractures.
- In February 2026, Smith+Nephew added the A’TOMIC™ Nitinol Fixation System to its Trauma, Foot & Ankle, and Hand & Wrist portfolio through an exclusive USA distribution agreement with RMR Ortho. The development is relevant to the Orthopedic Trauma Device Market because the system is designed for dynamic compression fixation across fusion, fracture, and osteotomy sites. It strengthens Smith+Nephew’s extremities and trauma fixation offering in high-frequency fracture procedures across USA surgical channels.
Key Players in the Orthopedic Trauma Device Market
Major Global Players:
- Johnson & Johnson
- Stryker Corporation
- Zimmer Biomet
- Smith+Nephew
- Arthrex Inc.
Specialist Players:
- Invibio Ltd.
- Acumed
- Orthofix Medical
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 12.2 billion to USD 22.6 billion, at a CAGR of 6.4% |
| Market Definition | The orthopedic trauma device market encompasses implants used to stabilize fractured bones after injury or traumatic damage. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, Germany, China, India, Japan, Brazil, and 30 plus countries |
| Key Companies Profiled | Johnson & Johnson, Stryker Corporation, Zimmer Biomet, Smith+Nephew, Arthrex Inc., Invibio Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with fixation product demand and verified trauma care patterns. |
Orthopedic Trauma Device Market by Segments
Orthopedic Trauma Device Market Segmented by Product Type:
- Internal Fixators
- External Fixators
Orthopedic Trauma Device Market Segmented by Material:
- Titanium
- Stainless Steel
- Bioabsorbable Materials
Orthopedic Trauma Device Market Segmented by Fixation Site:
- Lower Extremity
- Upper Extremity
- Pelvic Fixation
- Cranial Fixation
Orthopedic Trauma Device Market Segmented by End User:
- Hospitals
- Orthopedic and Trauma Centers
- Ambulatory Surgical Centers
Orthopedic Trauma Device Market Segmented by Sales Channel:
- Direct Sales
- Distributor Sales
- Group Purchasing
Orthopedic Trauma Device Market Segmented by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- Centers for Disease Control and Prevention. (2026, February 26). Older adult falls data. Centers for Disease Control and Prevention.CDC older adult falls data
- Food and Drug Administration. (2024, November 21). Orthopedic non-spinal bone plates, screws, and washers - premarket notification submissions. USA Food and Drug Administration.FDA orthopedic non-spinal bone plates, screws, and washers guidance
- Asian Transport Observatory. (2025, September). People’s Republic of China road safety profile 2025. Asian Transport Observatory.Asian Transport Observatory China road safety profile 2025
- National Police Agency. (2026, January). Traffic Bureau overview. National Police Agency.Japan National Police Agency Traffic Bureau
- World Bank Global Road Safety Facility. (2025, August 26). GRSF-funded study reveals alarming costs of road traffic injuries in Brazil. World Bank.World Bank GRSF road traffic injuries in Brazil
- Federal Statistical Office. (2026, February 25). Accidents registered by the police: Specification. Destatis.Destatis accidents registered by the police
- National Highway Traffic Safety Administration. (2026, April). Early estimate of motor vehicle traffic fatalities and fatality rate in 2025. NHTSA.NHTSA 2025 traffic death estimates
- Stryker. (2026, February 11). Stryker launches T2 Alpha Humerus Nailing System. Stryker.Stryker T2 Alpha Humerus Nailing System launch
- Zimmer Biomet. (2025, October 8). Zimmer Biomet launches two new foot and ankle solutions for complex trauma cases. Zimmer Biomet.Zimmer Biomet foot and ankle trauma solutions launch
- Zimmer Biomet. (2025, April 21). Zimmer Biomet completes acquisition of Paragon 28. Zimmer Biomet.Zimmer Biomet completes Paragon 28 acquisition
- Smith+Nephew. (2026, February 24). Smith+Nephew signs exclusive US distribution agreement with RMR Ortho. Smith+Nephew.Smith+Nephew RMR Ortho distribution agreement
- USA Food and Drug Administration. (2025, September 30). Medical Device User Fee Amendments (MDUFA): Fees. FDA. Medical Device User Fee Amendments (MDUFA): Fees | FDA
- Centers for Medicare & Medicaid Services. (2026, January 1). MLN6922507 - Medicare Payment Systems. CMS. MLN6922507 - Medicare Payment Systems
- Johnson & Johnson MedTech. (2025, June 19). Johnson & Johnson launches VOLT™ wrist and proximal humerus plating systems in the USA [Press release]. Johnson & Johnson Launches VOLT™ Wrist and Proximal Humerus Plating Systems in the USA | DePuy Synthes
- Food and Drug Administration. (2024, November 21). Orthopedic non-spinal bone plates, screws, and washers - premarket notification (510(k)) submissions. USA Food and Drug Administration.FDA orthopedic non-spinal bone plates, screws, and washers guidance
- Stryker. (2026, February 11). Stryker launches T2 Alpha Humerus Nailing System, supporting surgical workflow and care for complex fractures. Stryker.Stryker T2 Alpha Humerus Nailing System launch
This Report Answers
- What is the current and future size of the orthopedic trauma device market?
- How fast is the orthopedic trauma device market expected to expand between 2026 and 2036?
- Which product segment is expected to lead the market by 2026?
- Which end user segment is expected to account for the leading demand by 2026?
- What factors are driving demand for orthopedic trauma devices globally?
- How are road injury patterns influencing use of fixation implants?
- Why are hospitals the main demand base for trauma fixation systems?
- How are complex fractures creating demand for specialty trauma implants?
- Which countries are projected to record faster expansion through 2036?
- What is driving market expansion in China and Japan?
- Who are the key companies active in the orthopedic trauma device market?
- How does FMI estimate and validate the market forecast?
Frequently Asked Questions
What is the global market demand for Orthopedic Trauma Device in 2026?
In 2026, the global market for orthopedic trauma device is expected to be worth USD 12.2 billion.
How big will the market for Orthopedic Trauma Device be in 2036?
By 2036, the market for orthopedic trauma device is expected to be worth USD 22.6 billion.
How much is demand for Orthopedic Trauma Device expected to expand between 2026 and 2036?
Between 2026 and 2036, demand for orthopedic trauma device is expected to expand at a CAGR of 6.4%.
Which product segment is likely to be the leading seller globally by 2026?
Internal fixators are expected to account for 80.6% of the product segment in 2026.
What is causing demand to rise in China?
The China orthopedic trauma device market is projected to record 12.5% CAGR through 2036, backed by hospital expansion and trauma care capacity.
What is causing demand to rise in Japan?
The Japan orthopedic trauma device market is projected to expand at 9.2% CAGR through 2036, backed by older adult fracture care and precision implant use.
What does this report mean by Orthopedic Trauma Device Market definition?
The orthopedic trauma device market includes implants used to stabilize fractured bones after traumatic injury.
How does FMI make the Orthopedic Trauma Device forecast and check it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with fixation product demand and checking it against trauma care patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Control Arm Bushings
- Stabilizer Bushings
- Strut Mounts
- Control Arm Bushings
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material, 2026 to 2036
- Rubber
- Polyurethane
- Hydraulic
- Rubber
- Y to o to Y Growth Trend Analysis By Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Material, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Vehicle Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Vehicle Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Vehicle Type, 2026 to 2036
- Passenger Cars
- Light Commercial Vehicles
- Heavy Commercial Vehicles
- Passenger Cars
- Y to o to Y Growth Trend Analysis By Vehicle Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Vehicle Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Repair Workshops
- Retail Chains
- Online Platforms
- Repair Workshops
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Replacement Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Replacement Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Replacement Type, 2026 to 2036
- Direct Replacement
- Performance Upgrade
- Kit Assembly
- Direct Replacement
- Y to o to Y Growth Trend Analysis By Replacement Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Replacement Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Material
- By Vehicle Type
- By Sales Channel
- By Replacement Type
- Competition Analysis
- Competition Deep Dive
- ZF Friedrichshafen AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Tenneco Inc.
- Continental AG
- Vibracoustic SE
- Sumitomo Riko Company Limited
- ZF Friedrichshafen AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Vehicle Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Replacement Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Material
- Figure 9: Global Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Vehicle Type
- Figure 12: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Sales Channel
- Figure 15: Global Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Replacement Type
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Material
- Figure 35: North America Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Vehicle Type
- Figure 38: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Sales Channel
- Figure 41: North America Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Replacement Type
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Material
- Figure 51: Latin America Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Vehicle Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 57: Latin America Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Replacement Type
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Material
- Figure 67: Western Europe Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Vehicle Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 73: Western Europe Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Replacement Type
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Material
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Vehicle Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Replacement Type
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Material
- Figure 99: East Asia Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Vehicle Type
- Figure 102: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 105: East Asia Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Replacement Type
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Vehicle Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Replacement Type
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Vehicle Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Vehicle Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Vehicle Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Replacement Type, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Replacement Type, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Replacement Type
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

