Rupture of Membranes Tests Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Rupture of Membranes Tests Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Rupture of Membranes Tests Market Forecast and Outlook 2026 to 2036
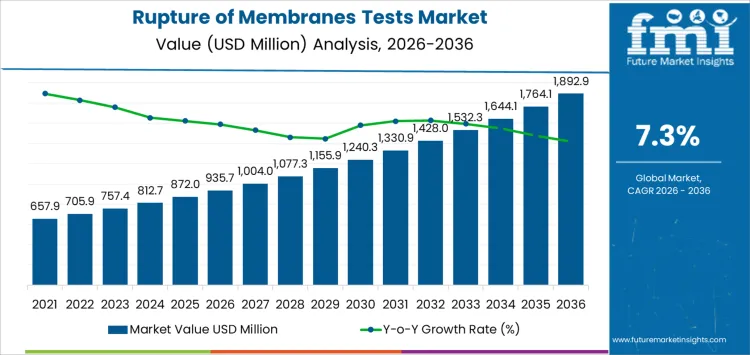
The rupture of membranes tests market in clinical maternity units is projected to reach USD 935.7 million in spending by 2026, with an anticipated growth to USD 1,892.9 million by 2036. This growth reflects a compound annual growth rate (CAGR) of 7.3%. The adoption of these tests is heavily influenced by their accuracy, turnaround time, and compatibility with laboratory or point-of-care workflows, rather than brand recognition. Once a hospital or birthing center validates a test for routine use, any substitutions must undergo parallel testing and quality verification.
Staff members evaluate factors such as ease of sample collection, shelf life, and storage conditions. At the same time, procurement teams monitor lot consistency, regulatory compliance, and technical support. Integration with electronic health records and reporting systems further enhances workflow efficiency.
Market growth depends more on procedural volume and the adoption of protocols than on promotional campaigns. Manufacturers focus on ensuring reagent stability, device calibration, and packaging protection, with the release of new tests requiring verification of sensitivity, specificity, and reproducibility. Distributors position kits near maternity units and regional laboratories to meet time-sensitive demands effectively.
Quick Stats for Rupture of Membranes Tests Market
- Rupture of Membranes Tests Market Value (2026): USD 935.7 million
- Rupture of Membranes Tests Market Forecast Value (2036): USD 1,892.9 million
- Rupture of Membranes Tests Market Forecast CAGR (2026-2036): 7.3%
- Leading Type in Rupture of Membranes Tests Market: Immunoassay-based tests
- Key Growth Regions in Rupture of Membranes Tests Market: Asia Pacific, North America, Europe
- Top Players in Rupture of Membranes Tests Market: Qiagen, Hologic, Abbott, Roche, Artron
What Is the Growth Forecast for the Rupture of Membranes Tests Market through 2036?
Hospital protocol adoption and maternal care guidelines shape the rupture of membranes tests market more than overall birth rates. In 2026, at about USD 935.7 million, demand is concentrated in maternity wards, obstetrics centers, and perinatal networks where early detection of membrane rupture influences intervention timing and reduces infection risk. Test selection is integrated into care pathways and remains consistent across multiple delivery cycles, which ties purchasing to the number of equipped labor and delivery units rather than short term fluctuations in patient volume. Uptake rises as hospitals standardize testing protocols and expand capacity for high risk pregnancies. The growth reflects progressive integration into maternal health programs rather than replacement of existing diagnostic methods.
Supply chain readiness and procedural efficiency guide the later phase of the rupture of membranes tests market. As value approaches roughly USD 1,892.9 million by 2036, buyers focus on test reliability, shelf life, and ease of use by clinical staff. Manufacturers invest in batch consistency, rapid turnaround formats, and distribution systems to support multi-site deployment. The move from under one billion into the low two billion reflects cumulative adoption across hospitals and maternal health networks rather than a single regulatory or guideline change. Suppliers that combine predictable quality, stable delivery, and responsive support tend to secure long term positions in hospital diagnostic portfolios.
Rupture of Membranes Tests Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 935.7 million |
| Forecast Value (2036) | USD 1,892.9 million |
| Forecast CAGR (2026-2036) | 7.3% |
How Is the Rupture of Membranes Tests Market Improving Clinical Accuracy and Operational Workflow?
Rupture of membranes (ROM) tests are employed to identify amniotic fluid leakage during labor or suspected preterm membrane rupture. Traditional detection relied on visual inspection or nitrazine testing, which often produced false positives and increased infection risk. Modern tests use biochemical markers, immunoassays, or fluorescence-based detection to provide rapid, objective results at the point of care. Hospitals and birthing centers evaluate devices by detection accuracy, speed, shelf stability, and ease of integration with labor room protocols. Adoption is concentrated in high-volume maternity wards and tertiary obstetric units, where reliable early detection reduces unnecessary interventions, supports appropriate antibiotic administration, and improves neonatal outcomes. Device performance also influences staff workflow efficiency and bed utilization in labor and delivery units.
Market growth and competitive positioning focus on reagent consistency, lot traceability, and regulatory compliance rather than price alone. Early deployments were limited to pilot sites, which restricted data on performance across diverse patient populations. Current demand favors standardized test formats that simplify inventory, training, and reporting for multiple wards or hospitals. Manufacturers compete on assay sensitivity, reproducibility, and operational robustness under variable clinical conditions. Hospitals and health systems evaluate total operational value through reduced repeat testing, improved patient throughput, and fewer adverse events, rather than per-test cost. Distributors add value by providing technical training, supply chain continuity, and quality documentation to support accreditation and audits.
What Factors Are Shaping the Demand for Rupture of Membranes Tests Market in 2026 by Product Type and Application?
Demand in the Rupture of Membranes (ROM) Tests Market is primarily influenced by early detection requirements, clinical workflow integration, and hospital compliance standards rather than short-term procurement trends. Hospitals and maternity units require tests that provide rapid, accurate results to guide timely interventions and reduce complications. Segment demand is shaped by sensitivity, specificity, and ease of use in labor and delivery settings. Suppliers providing robust quality assurance, reliable lot-to-lot consistency, and training support can secure long-term adoption. Market growth is concentrated in preterm and term PROM diagnosis, creating predictable volumes for manufacturers capable of scaling production while maintaining regulatory compliance.
How Does Product Type Influence Supplier Responsibilities and Clinical Integration in the Rupture of Membranes Tests Market?
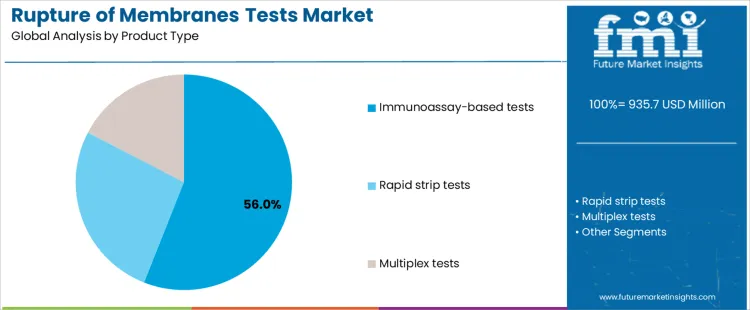
Immunoassay-based tests represent approximately 56% of demand due to their high sensitivity and suitability for point-of-care diagnostics in maternity wards. Suppliers must ensure batch consistency, regulatory compliance documentation, and training programs for clinical staff to maintain accuracy and confidence. Rapid strip tests are deployed for fast bedside decision-making, emphasizing portability, ease of use, and minimal operator training, yet they require supplier support for quality control and consumable supply. Multiplex tests address specialized clinical requirements where multiple biomarkers are assessed simultaneously, increasing complexity in validation, handling, and training. Product type selection defines the level of supplier engagement, technical support, and consumable management needed for successful adoption.
Operational and clinical workflow considerations further differentiate product types. Immunoassay systems require integration into lab workflows, calibration verification, and ongoing quality checks to sustain performance in high-volume maternity settings. Rapid strip tests minimize procedure time but require consistent supply and user guidance to ensure reliability. Multiplex tests, though lower in volume, necessitate additional technical oversight, data interpretation training, and validation to satisfy hospital standards. Once a hospital standardizes on a test type, changing systems is limited due to revalidation requirements, training, and quality assurance, making supplier reliability and service critical for retention.
Why Does Application Mix Define Volume Concentration and Supplier Strategy in the Rupture of Membranes Tests Market?

Preterm PROM diagnosis accounts for approximately 66% of market demand because early detection is critical to prevent neonatal complications and guide timely interventions. Devices used in this application must deliver accurate, rapid results to inform clinical decisions. Term PROM procedures, while performed at lower frequency, require reliable testing to prevent infection and reduce unnecessary interventions, creating niche opportunities for specialized tests. Application-specific requirements influence procurement patterns, inventory planning, and supplier support. Companies that align product performance with high-impact preterm PROM use and provide consumables and training for term PROM procedures can maximize adoption and account penetration.
Supplier strategy is also shaped by clinical use patterns. High-volume preterm PROM testing favors bulk procurement, ongoing staff training, and validated quality assurance processes. Term PROM programs often require modular testing solutions, portable formats, and rapid replenishment of consumables. Multiplex or specialized PROM tests are deployed in tertiary centers, emphasizing documentation, calibration, and technical support. Segment demand therefore moves with clinical guidelines, hospital delivery volumes, and maternity care protocols rather than short-term purchasing trends. Suppliers that provide comprehensive device support, consumable reliability, and training can secure long-term contracts across multiple applications.
How is the Rupture of Membranes Tests Market Being Defined by Clinical Accuracy and Workflow Integration?
The Rupture of Membranes Tests Market is shaped by hospital and maternity unit requirements for rapid, reliable confirmation of membrane status during labor. Devices and test kits are evaluated for sensitivity, specificity, ease of use, and compatibility with routine prenatal procedures. Selection decisions involve obstetricians, nurses, and laboratory staff. Suppliers that offer validated products with minimal handling steps and reliable readouts are preferred. This positions rupture of membranes tests as essential diagnostic tools rather than optional screening methods, emphasizing clinical accuracy, confidence, and consistent supply for high-volume maternity care.
How Are Standardized Labor and Delivery Protocols Influencing Supplier Expectations in the Rupture of Membranes Tests Market?
Hospitals increasingly follow standardized labor and delivery protocols to reduce variability and ensure patient safety. This drives demand in the Rupture of Membranes Tests Market toward kits and devices that provide consistent results, require minimal procedural steps, and integrate smoothly into existing workflows. Buyers prefer tests with clear instructions, predictable handling requirements, and minimal contamination risk. Suppliers are evaluated on batch-to-batch consistency, regulatory compliance, and technical support rather than novel or niche features. Standardized devices improve adoption across multiple units and hospital networks.
Where is Recurring Demand Being Built in the Rupture of Membranes Tests Market?
Recurring demand is generated in hospitals, maternity centers, and obstetrics clinics that manage high patient throughput and routine labor monitoring. Each delivery or prenatal assessment requires accurate and timely testing. The Rupture of Membranes Tests Market also benefits when multi-site hospital systems standardize on a single supplier or test model. Suppliers that provide training, quality assurance, and reliable inventory management secure repeat orders. Embedding tests into standardized labor protocols ensures predictable demand tied to patient volume and delivery frequency.
Why do Cost and Clinical Risk Considerations Limit Broader Adoption in the Rupture of Membranes Tests Market?
Some facilities continue using traditional methods due to cost sensitivity or familiarity with existing procedures. The Rupture of Membranes Tests Market also faces adoption constraints from staff training requirements, validation processes, and procurement approval cycles. Hospitals prioritize devices that demonstrate reliability and minimize clinical risk, which can slow the introduction of new test models. Suppliers entering the market must provide robust technical support, training, and documentation to overcome adoption barriers. As a result, usage is concentrated in high-volume maternity units and well-resourced hospitals.
What is the Demand for Rupture of Membranes Tests by Country?
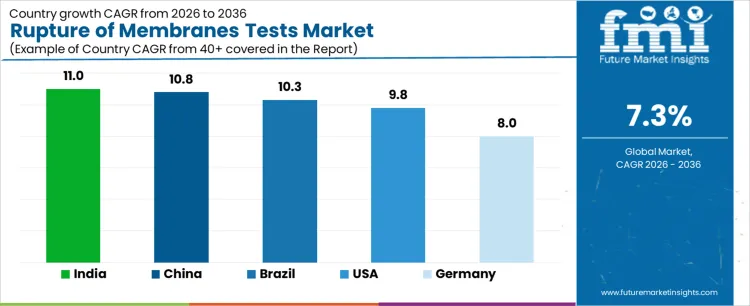
| Country | CAGR |
|---|---|
| USA | 9.8% |
| Brazil | 10.3% |
| China | 10.8% |
| India | 11.0% |
| Germany | 8.0% |
Demand for rupture of membranes tests is increasing as hospitals, maternity clinics, and diagnostic laboratories adopt rapid and accurate methods for assessing membrane integrity during pregnancy and labor. India leads with an 11.0% CAGR, supported by expanding maternal healthcare infrastructure, higher hospital deliveries, and increased awareness of prenatal diagnostics. China follows at 10.8%, driven by large maternity care networks, advanced prenatal screening programs, and hospital modernization. Brazil records 10.3%, reflecting adoption in both public and private healthcare systems. The USA grows at 9.8%, shaped by routine use in obstetrics departments and integration with labor monitoring protocols. Germany, at 8.0%, reflects a mature market with steady, protocol driven adoption in maternity care.
How Are Obstetrics Service Expansions Driving Demand in the Rupture of Membranes Tests Market in the United States?
Expanding maternity services and labor monitoring programs correspond to a 9.8% CAGR for rupture of membranes tests demand in United States. Hospital obstetrics departments, labor and delivery units, and procurement committees control most purchases. Adoption focuses on rapid diagnostics, accuracy, and safety for high volume birthing centers. Once validated, tests are standardized across multiple hospital networks. Device choice emphasizes reliability, ease of use, and integration into electronic health records. Import systems dominate advanced testing kits while domestic suppliers provide routine assays. Training programs influence usage and adoption. Capital allocations align with maternity unit expansions and procedure growth. Replacement cycles follow usage intensity. Commercial success depends on inclusion in hospital protocols and network agreements rather than isolated department purchases.
Why Are Private Hospital Networks Influencing Demand in the Rupture of Membranes Tests Market in Brazil?
Growth of private maternity facilities supports a 10.3% CAGR for rupture of membranes tests demand in Brazil. Procurement authority resides with hospital network purchasing offices and obstetrics service committees. Adoption focuses on rapid detection of membrane rupture to optimize labor and delivery workflows. Device selection emphasizes accuracy, shelf life, and ease of use in high volume obstetrics wards. Import kits dominate premium centers while domestic suppliers cover routine birthing units. Once approved, deployment extends across affiliated hospitals. Training programs and workflow protocols affect adoption. Capital allocation aligns with network expansion. Replacement cycles track usage and test frequency. Commercial success depends on inclusion in network approved protocols and reference site adoption rather than ad hoc individual hospital orders.
What Institutional Programs Are Driving Growth in the Rupture of Membranes Tests Market in China?
Expansion of tertiary hospitals and standardized maternal care programs supports a 10.8% CAGR for rupture of membranes tests demand in China. Large hospital networks and obstetrics units control procurement. Adoption focuses on rapid, accurate diagnostics to manage labor progression and reduce complications. Domestic suppliers provide standard kits while international brands retain positions in high precision assays. Tender cycles and multi-year hospital planning govern procurement timing. Once approved, tests are implemented across multiple facilities. Training and clinical guideline adherence influence utilization. Replacement cycles follow procedure volume rather than shelf life alone. Capital allocation aligns with hospital expansion and maternity unit growth. Commercial success depends on inclusion in provincial and national hospital catalogs rather than individual clinic purchases.
Why Are Maternity Unit Expansions Driving Demand in the Rupture of Membranes Tests Market in India?
Growth of organized maternity services and maternal health programs corresponds to an 11% CAGR for rupture of membranes tests demand in India. Large hospital chains and specialty maternity centers lead procurement. Device selection emphasizes rapid results, safety, and integration with obstetrics protocols. Import kits serve high acuity facilities while domestic suppliers cover standard birthing wards. Procurement authority rests with hospital obstetrics committees and capital boards. Once approved, deployment spreads across multiple affiliated hospitals. Training, workflow alignment, and laboratory support affect utilization. Replacement cycles track usage intensity. Budget allocation aligns with maternity unit expansion and new clinic openings. Commercial success depends on inclusion in network approved equipment lists and reference site adoption rather than isolated hospital purchases.
How Are Regulatory Guidelines and Clinical Protocols Shaping Demand in the Rupture of Membranes Tests Market in Germany?
Hospital compliance and obstetric guideline adherence support an 8% CAGR for rupture of membranes tests demand in Germany. University hospitals and regional maternity centers control most procurement. Adoption focuses on early detection of membrane rupture and workflow integration into labor management protocols. Device evaluation emphasizes accuracy, reliability, and patient safety. Procurement follows structured health technology assessment and capital approval processes. Import suppliers dominate premium assays while domestic suppliers provide routine kits. Once approved, deployment spreads across multiple departments. Training, protocol adherence, and laboratory compatibility affect utilization. Replacement cycles track usage. Budget allocation aligns with hospital unit expansions. Commercial success depends on framework inclusion and reference site adoption rather than isolated clinic purchases.
How Do Suppliers Compete for Clinical Adoption in the Rupture of Membranes Tests Market?
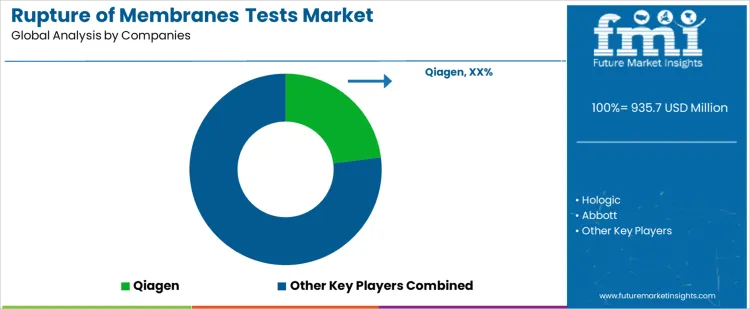
Rupture of membranes test selection follows obstetric workflow design, sample handling, and diagnostic protocol definition rather than routine catalog review. Clinicians specify sensitivity, specificity, turnaround time, and sample volume requirements before approving kits. Qiagen, Hologic, Abbott, Roche, Artron compete at the evaluation stage through different assay formats and integration strategies. Once a test enters hospital protocols, validation files, quality control charts, and staff training anchor the selection. Qiagen emphasizes molecular and nucleic acid detection for high accuracy. Hologic provides rapid immunoassays with simplified processing. Abbott focuses on analyzer compatibility and standardized reporting. Roche integrates tests with broader laboratory platforms. Artron targets cost sensitive clinics requiring ease of use and minimal workflow disruption.
Competitive outcomes reflect assay reliability, documentation completeness, and regulatory compliance rather than pricing. Laboratory teams evaluate sensitivity under clinical conditions, specificity against contaminants, stability, and lot to lot consistency during validation. Procurement favors suppliers with predictable supply, service support, and traceable batch records. Qiagen benefits from molecular assay expertise and technical support. Hologic secures adoption through rapid turnaround and consistent clinical performance. Abbott holds share where instrument standardization governs laboratory workflow. Roche competes through platform integration and multi analyte testing. Artron serves smaller hospitals and clinics with operational simplicity. Market positions vary by hospital type, patient volume, and regulatory oversight.
Key Players in the Rupture of Membranes Tests Market
- Qiagen
- Hologic
- Abbott
- Roche
- Artron
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Product Type | Immunoassay-based tests; Rapid strip tests; Multiplex tests |
| Application | Preterm PROM; Term PROM |
| End User | Hospitals; Maternity clinics; Laboratories |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Qiagen; Hologic; Abbott; Roche; Artron |
| Additional Attributes | Dollar by sales by product and application, immunoassay versus rapid test mix, shelf life and lot consistency, turnaround time and workflow integration, training and interpretation requirements, inventory rotation cycles, regulatory and quality compliance burden, multi-site protocol standardization patterns |
Rupture of Membranes Tests Market Segmentation
Product Type:
- Immunoassay-based tests
- Rapid strip tests
- Multiplex tests
Application:
- Preterm PROM
- Term PROM
End User:
- Hospitals
- Maternity clinics
- Labs
Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- American Gastroenterological Association. (2025). AGA clinical practice guideline on the management of gastroesophageal reflux disease. Gastroenterology.
- Katz, P. O., & Dunbar, K. B. (2024). Over-the-counter use of proton pump inhibitors: Appropriate use and misuse. Alimentary Pharmacology & Therapeutics.
- Mermelstein, J., & Chait, M. M. (2025). The evolving landscape of acid suppression therapy for gastroesophageal reflux disease. Journal of Clinical Gastroenterology.
- World Health Organization. (2024). WHO model list of essential medicines: Gastrointestinal medicines and rational use. World Health Organization.
- Organisation for Economic Co-operation and Development. (2024). Health system impacts of self-care and non-prescription medicines in advanced economies. OECD Publishing.
Frequently Asked Questions
How big is the rupture of membranes tests market in 2026?
The global rupture of membranes tests market is estimated to be valued at USD 935.7 million in 2026.
What will be the size of rupture of membranes tests market in 2036?
The market size for the rupture of membranes tests market is projected to reach USD 1,892.9 million by 2036.
How much will be the rupture of membranes tests market growth between 2026 and 2036?
The rupture of membranes tests market is expected to grow at a 7.3% CAGR between 2026 and 2036.
What are the key product types in the rupture of membranes tests market?
The key product types in rupture of membranes tests market are immunoassay-based tests, rapid strip tests and multiplex tests.
Which application segment to contribute significant share in the rupture of membranes tests market in 2026?
In terms of application, preterm prom segment to command 66.0% share in the rupture of membranes tests market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Immunoassay-based tests
- Rapid strip tests
- Multiplex tests
- Immunoassay-based tests
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Preterm PROM
- Term PROM
- Preterm PROM
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Maternity clinics
- Labs
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Qiagen
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Hologic
- Abbott
- Roche
- Artron
- Others
- Qiagen
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

