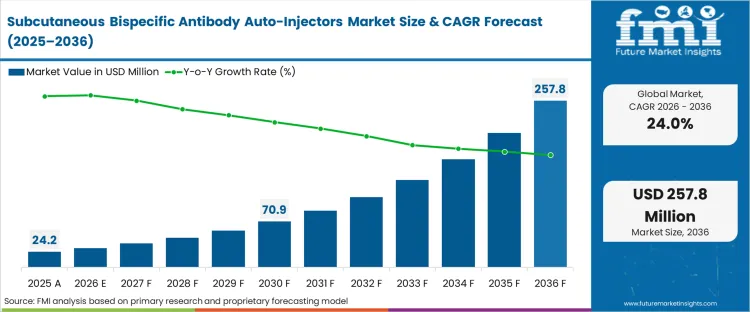
The subcutaneous bispecific antibody auto-injectors market was valued at USD 24.2 million in 2025. The industry is poised to cross USD 30.0 million in 2026 at a CAGR of 24.0% during the forecast period. Demand outlook is expected to be USD 257.8 million through 2036 as pharmaceutical developers remove complex biologics delivery away from intravenous infusion suites to localised subcutaneous administration formats.
Biopharmaceutical manufacturers are being forced to redesign their entire clinical delivery strategies, moving away from post-formulation device selection toward parallel drug-device co-development. The stakes for delaying this integration involve severe late-stage clinical hold-ups and forfeiture of outpatient administration approvals within the market. While standard engineering focuses on expanding payload capacities, the actual friction point centers on the molecular rheology of the payload itself. Early integrators of wearable injectors gain a critical advantage by matching the device power curve to the specific viscosity profile of bispecific agents, validating the need for a high-viscosity bispecific auto-injector.
The qualification of a large-volume bispecific injection device that can reliably extrude viscous co-formulations without tissue damage must be achieved before growth becomes self-reinforcing. Primary packaging suppliers trigger this gate by commercialising advanced container architectures capable of withstanding extreme mechanical stress. Once this threshold is crossed, the transition from clinical monitoring to at-home maintenance dosing accelerates rapidly, elevating the subcutaneous bispecific antibody injector forecast.
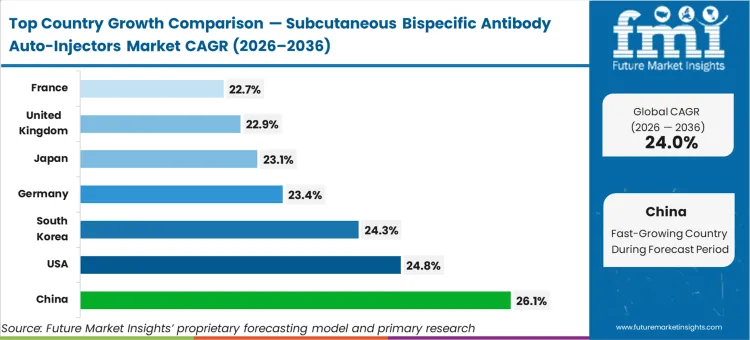
China advances at 26.1%, driven by a hyper-accelerated pipeline of dual-targeting agents and rapidly scaling domestic manufacturing capacity. The United States follows closely at 24.8%, where aggressive transitions toward decentralised oncology care compel rapid adoption. South Korea tracks at 24.3%, leveraging its concentrated biomanufacturing infrastructure. Germany, Japan, the United Kingdom, and France are expected to register 23.4%, 23.1%, 22.9%, and 22.7%, respectively. The differences across regions highlight how prepared each national healthcare system is to transition the management of severe conditions management from monitored hospital settings into outpatient frameworks, defining overall growth.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 30.0 million |
| Industry Value (2036) | USD 257.8 million |
| CAGR (2026-2036) | 24.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
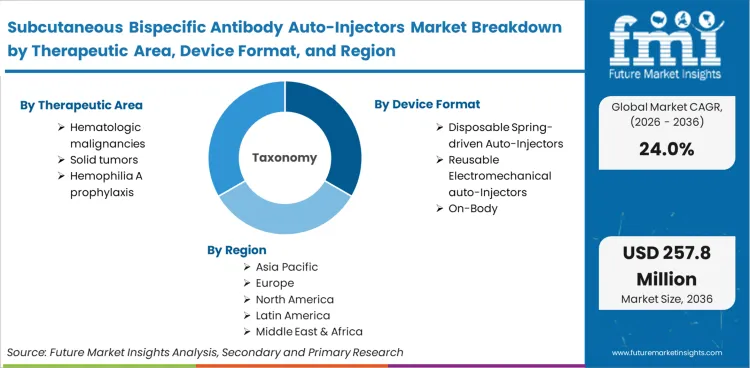
The subcutaneous bispecific antibody auto-injectors market comprises specialised mechanical and electromechanical delivery devices engineered specifically for the self-administration or outpatient delivery of dual-targeting monoclonal antibodies. It is functionally distinct from standard biologic pens due to the specific power, volume, and viscosity requirements dictated by complex bispecific formulations and their unique pharmacokinetic delivery profiles.
The scope of the bispecific antibody auto-injector market includes disposable mechanical pens, reusable electromechanical drivers, and high-volume wearable modules designed exclusively for bispecific agents. It incorporates primary container components heavily integrated into these devices, as well as systems utilizing large volume injectors tailored for co-formulated enzymatic dispersion technologies. Equipment validated specifically for these complex therapeutic payloads falls entirely within this boundary.
Standard insulin pens, traditional single-target monoclonal antibody injectors, and general-purpose intravenous infusion pumps are explicitly excluded. These generic self administered parenteral formats are excluded because they lack the specific power mechanisms, container durability, and volume capacities required to handle the extreme rheological demands of bispecific antibody therapies.
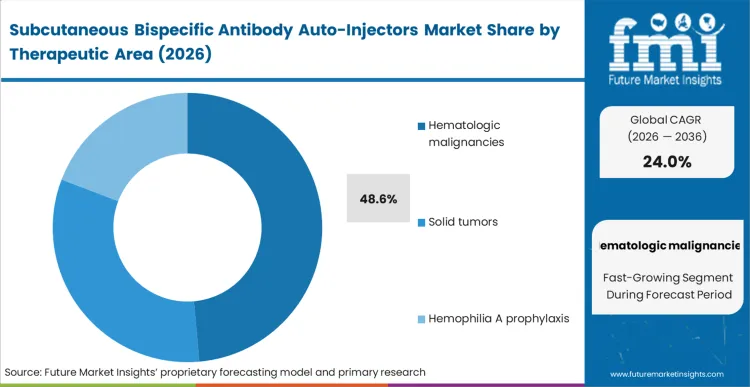
The reason hematologic malignancies hold 48.6% of this market comes down to a single operational reality: the first wave of approved subcutaneous bispecific therapies specifically targets blood cancers like multiple myeloma and lymphomas.
According to FMI's estimates, clinical developers prioritized these indications because frequent dosing regimens create an acute bottleneck in hospital infusion suites. Oncology pathway directors are altering their care models to route these specific patients away from intravenous chairs. The integration of connected drug delivery devices validates patient adherence without requiring physical hospital visits. Developers who delay the transition of their haematology pipelines to subcutaneous formats face immediate exclusion from decentralised procurement frameworks.
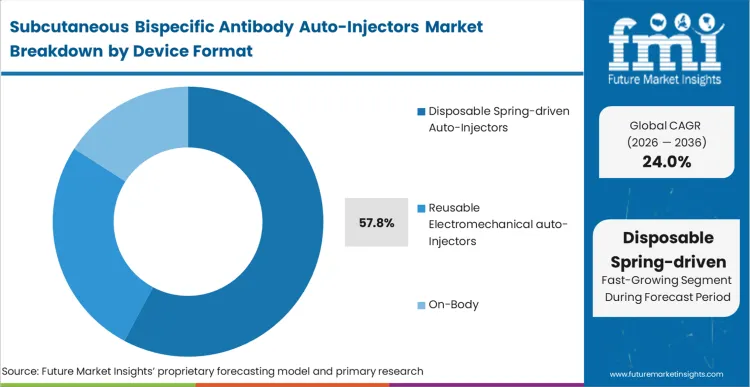
Disposable spring-driven auto-injectors command a 57.8% share because they represent the most mature, regulatorily validated pathway for primary container integration. Device engineers at leading biopharmaceutical firms rely on this format to avoid introducing novel mechanical risks during late-stage clinical reviews.
In FMI's view, the decision to utilise disposable mechanical systems is not driven by performance superiority, but by the necessity of moving rapidly through combination product regulatory gates. These systems integrate seamlessly into existing fill-finish architectures, minimising capital expenditure on novel assembly lines. Manufacturers clinging to complex, unvalidated delivery modules face severe commercial consequences as competitors reach the market years earlier with familiar subcutaneous drug delivery devices.
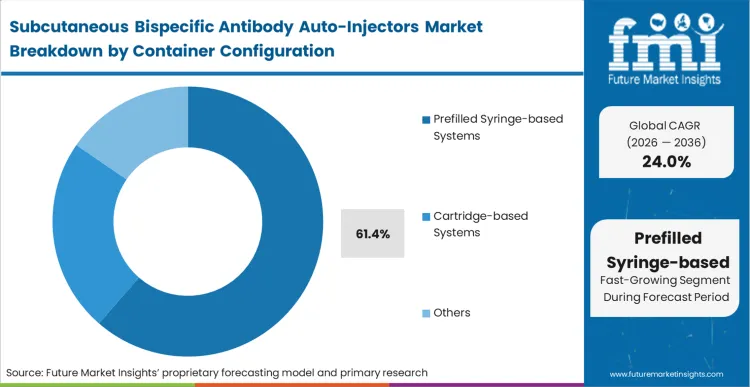
The specific choice facing primary packaging procurement heads right now is whether to push prefilled syringe architectures to their limits or pivot entirely to cartridge-based platforms. Prefilled syringe-based systems capture 61.4% share precisely because the global fill-finish ecosystem is overwhelmingly optimised for this format.
As per FMI's projection, abandoning this infrastructure introduces unacceptable risk and delay into the commercialisation timeline. The syringe acts as the building block of the combination product, requiring only minor adaptations to the external drug delivery technology housing. Biopharma companies that attempt to qualify novel container configurations late in the clinical process routinely experience multi-year regulatory delays.
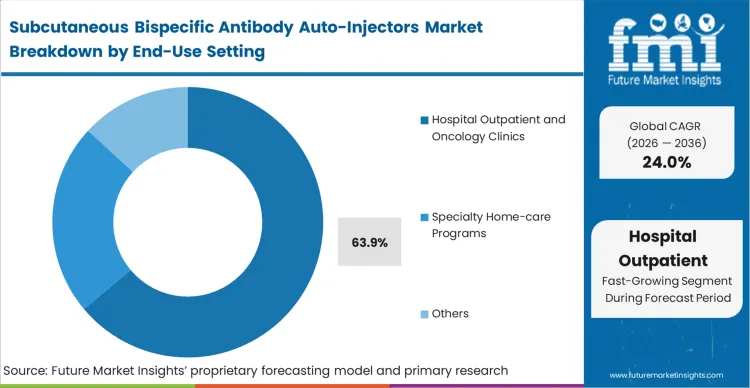
Hospital outpatient and oncology clinics dominate with a 63.9% share, reflecting the reality that bispecific antibodies carry severe immunologic monitoring requirements. Despite the physical capability of the device to enable home use, clinical pathway directors compel administration in monitored settings to manage cytokine release syndrome risks during step-up dosing.
Based on FMI's assessment, this setting provides the necessary clinical bridge, allowing healthcare systems to capture the efficiency of subcutaneous administration while maintaining acute monitoring capabilities. The integration of advanced drug delivery solutions within these clinics drastically reduces patient processing time. Institutions that fail to establish dedicated outpatient pathways for these therapies severely restrict their patient capacity.
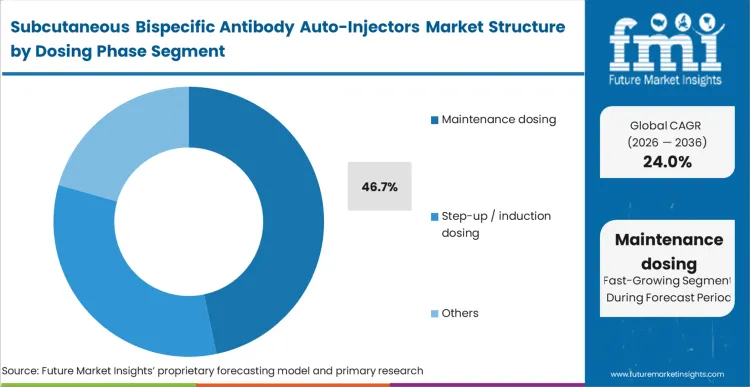
The commercial outcome of moving patients to maintenance dosing formats dictates the long-term profitability of the therapeutic agent. Maintenance dosing accounts for 46.7% share, representing the phase where the operational and economic benefits of auto-injectors are fully realized.
FMI analysts opine that once the acute monitoring of the induction phase is cleared, the requirement for localised administration becomes the primary determinant of patient adherence. Pharmaceutical developers design their delivery systems specifically to secure this long-tail revenue stream, utilizing injectable drugs packaged for maximum patient autonomy. Companies that fail to provide a viable auto-injector option for the maintenance phase routinely lose market share to competitors offering superior patient convenience.
The pressure of oncology care decentralization forces hospital administrators to transition maintenance therapies to outpatient administration formats. Biopharmaceutical developers are compelled to integrate delivery mechanisms directly into their clinical lifecycles to secure regulatory approval for these decentralized pathways. The commercial stakes are absolute: a dual-targeting agent without a validated subcutaneous delivery mechanism cannot compete in a market optimizing for patient throughput and reduced healthcare resource utilization. This change mandates the parallel development of highly specialized combination products tailored for complex biologics and creates massive demand for a reliable contract development for bispecific drug-device combination.
The extreme viscosity and massive molecular payload of bispecific agents create a critical physical friction point that cannot be solved by simply increasing spring tension. High-force mechanical drivers routinely shatter standard glass primary containers, while standard springs stall mid-injection, leading to incomplete dosing. This limitation requires extensive, costly re-engineering of the container-closure system and the integration of advanced polymers. While specialised biosimilars and follow on biologics developers explore enzymatic co-formulations to temporarily reduce viscosity, this adds profound complexity to the stability and regulatory qualification of the final product.
Opportunities in the Subcutaneous Bispecific Antibody Auto-Injectors Market
The regional breakdown shows that the Subcutaneous Bispecific Antibody Auto-Injectors market spans North America, Europe, Asia Pacific, and the rest of the world, covering more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 26.1% |
| United States | 24.8% |
| South Korea | 24.3% |
| Germany | 23.4% |
| Japan | 23.1% |
| United Kingdom | 22.9% |
| France | 22.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The hyper-accelerated expansion of domestic CDMO capacity for advanced combination products shapes the adoption curve across the Asia Pacific region. Rather than relying on imported delivery technology, regional biomanufacturing hubs are vertically integrating device engineering with biologic formulation. In FMI's view, this concentrated industrial strategy allows for the rapid iteration of delivery mechanisms tailored specifically to the unique rheological properties of dual-targeting agents. The regional deployment of next generation immunotherapies heavily depends on this localized production scale-up, insulating the market from global supply chain bottlenecks.
FMI's report includes secondary markets across Southeast Asia and Australasia. Infrastructure readiness and the establishment of reliable cold chain logistics dictate the pace at which these nations can import and distribute complex biologic combination products.
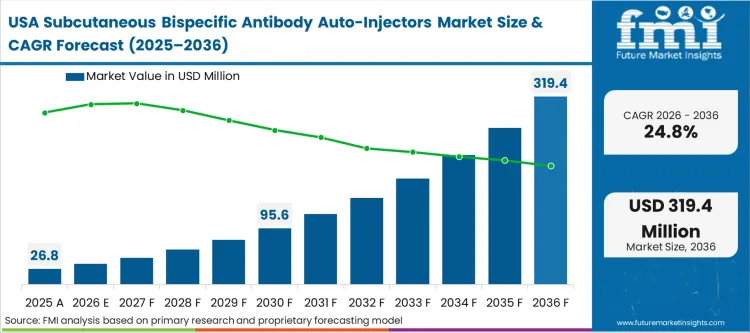
The intense push by commercial payers to decentralise oncology care and drastically reduce hospital resource utilization defines the North American trajectory. Reimbursement frameworks systematically penalise prolonged intravenous chair times, effectively forcing clinical pathway directors to mandate subcutaneous formats wherever clinically viable. FMI analysts opine that this dynamic creates a massive, immediate demand for validated delivery devices capable of handling complex payloads. The integration of specialised cancer biological therapy modules directly into outpatient procurement networks accelerates the transition at a scale unmatched globally.
FMI's report includes Canada and regional healthcare networks. The alignment of regulatory standards with the USA FDA allows for seamless cross-border commercialization of validated combination products.
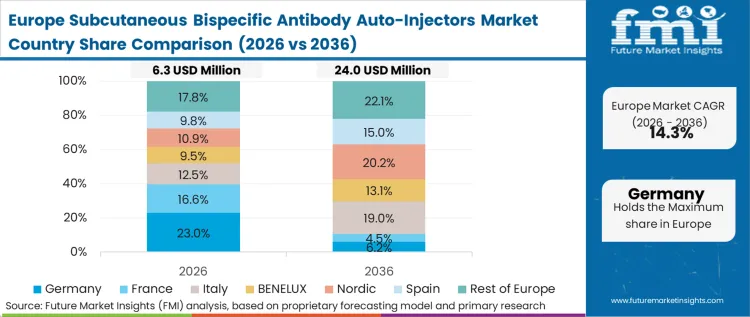
Extended producer responsibility and stringent medical device regulations (MDR) shape the qualification pathways for combination products across European jurisdictions. The regulatory environment forces device manufacturers to exhaustively document the human factors engineering and material safety profiles of high-power injection systems before commercialization is permitted. According to FMI's estimates, this rigorous scrutiny extends the development cycle but ultimately creates a highly resilient, deeply validated device ecosystem. The reliance on prefilled syringes drug molecules as the core container architecture mitigates some regulatory friction, providing a known baseline for safety assessments.
FMI's report includes Italy, Spain, and the broader EU bloc. Harmonized medical device regulations across these nations dictate a unified approach to combination product registration, simplifying the regulatory strategy for global device manufacturers.
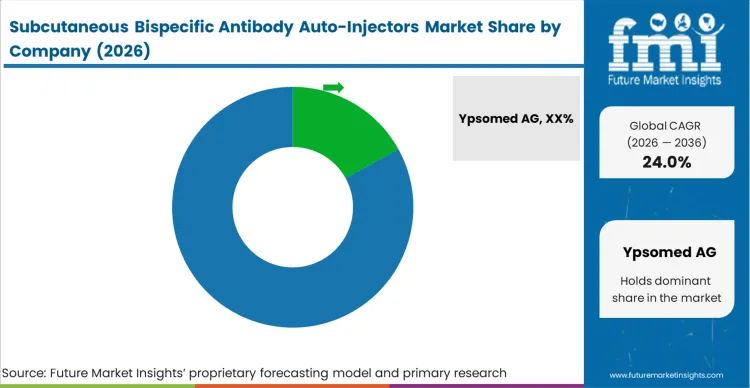
The extreme intellectual property barriers surrounding combination product engineering and the massive capital required for precision manufacturing dictate the highly concentrated scenario of this market. Buyers, specifically device engineering leads at major pharma companies evaluating an RFQ bispecific auto-injector platform, stinguish qualified from unqualified vendors based entirely on the vendor's documented track record of successfully guiding combination products through FDA and EMA approvals.
Top bispecific antibody auto-injector suppliers possess deeply entrenched, regulatorily validated platform architectures that biopharma companies can adopt with minimal customization risk. This advantage persists because changing the delivery device mid-clinical trial introduces catastrophic regulatory delays. A challenger must build a fully verified, massively scalable manufacturing infrastructure capable of producing zero-defect syringes and complex power mechanisms before a biopharma partner will even consider engaging in a pilot program. The capital requirement to build this capability from scratch virtually eliminates low-tier entrants.
Large biopharmaceutical buyers actively resist lock-in by mandating dual-sourcing capabilities for all primary packaging and device components, forcing standardization across supplier ecosystems to support syringes and injectable drugs packaging. However, the unique mechanical requirements of bispecific agents often necessitate bespoke device configurations, clashing with the buyers' desire for genericized supply chains. Through 2036, the market will inherently consolidate around the few specialized engineering firms capable of reliably producing the high-force, large-volume modules required for the next generation of dual-targeting therapies.
| Metric | Value |
|---|---|
| Quantitative Units | USD 30.0 million to USD 257.8 million, at a CAGR of 24.0% |
| Market Definition | Specialized delivery devices engineered specifically for the self-administration or outpatient delivery of dual-targeting monoclonal antibodies, handling specific volume and viscosity requirements. |
| Therapeutic area Segmentation | Hematologic malignancies, Solid tumors, Hemophilia A prophylaxis, Autoimmune / immunology pipeline |
| Device format Segmentation | Disposable spring-driven auto-injectors, Reusable electromechanical auto-injectors, On-body / wearable auto-injectors |
| Container configuration Segmentation | Prefilled syringe-based systems, Cartridge-based systems, Hyaluronidase-enabled co-formulated systems |
| End-use setting Segmentation | Hospital outpatient / oncology clinics, Specialty home-care programs, Ambulatory care centers |
| Regions Covered | North America, Europe, Asia Pacific, Rest of the World |
| Countries Covered | China, United States, South Korea, Germany, Japan, United Kingdom, France, and 40 plus countries |
| Key Companies Profiled | Ypsomed AG, SHL Medical AG, Becton, Dickinson and Company, Gerresheimer AG, West Pharmaceutical Services, Inc., Johnson & Johnson Innovative Medicine, F. Hoffmann-La Roche Ltd |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews conducted with clinical trial directors and device engineering leads to establish adoption intent. Forecast baseline anchored to the commercialization volume of approved subcutaneous bispecific agents. Data validated against primary container manufacturing capacity expansions globally. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How big is the bispecific auto-injector market?
The market is valued at USD 30.0 million in 2026. This figure establishes the baseline revenue generated strictly from the first wave of approved haematology-focused bispecific combination products transitioning into outpatient settings.
What will it be valued at by 2036?
It is projected to reach USD 257.8 million by 2036. This massive expansion reflects the integration of wearable and high-volume delivery formats across both solid tumour and immunology pipelines globally.
What are the growth drivers for SC bispecific auto-injectors through 2036?
A 24.0% CAGR maps directly to the regulatory approval calendar of subcutaneous formulation variants currently in late-stage clinical trials. The decentralisation of oncology care forces hospital administrators to mandate outpatient administration formats, changing drug-delivery timelines.
Which companies are leading subcutaneous bispecific injector development?
Ypsomed AG, SHL Medical AG, and Becton, Dickinson and Company secure dominant positions not merely through design innovation, but through their ability to navigate complex regulatory qualification processes alongside their biopharmaceutical partners.
Which Therapeutic Area segment leads?
Hematologic malignancies account for 48.6% of the market. Oncology pathway directors prioritize these indications for device integration because frequent blood cancer dosing regimens create severe bottlenecks inprioritiseinfusion suites.
Which Device Format segment leads?
Disposable spring-driven auto-injectors capture 57.8% share. Biopharmaceutical developers rely on this format to avoid introducing novel electromechanical risks during the already complex combination product regulatory review process.
Which Container Configuration segment leads?
Prefilled syringe-based systems hold 61.4% share. The global fill-finish ecosystem is overwhelmingly optimized foroptimisedlling device engineers to build delivery modules around this established architecture to prevent launch delays.
Why are bispecific antibodies moving to subcutaneous delivery?
The decentralization of oncology care forces hospital administrators to mandate outpatient administration formats. Biopharmaceutical firms that fail to provide a validated subcutaneous delivery device face immediate exclusion from these streamlined clinical pathways that reduce intravenous chair time.
What limits auto-injector use for oncology bispecifics?
The extreme viscosity of bispecific combinations routinely shatters standard glass primary containers under high spring tension. This failure mode requires extensive, costly re-engineering of the device's internal power mechanisms before clinical trials can proceed.
Which country grows fastest?
China expands at 26.1%, outpacing the United States' 24.8%. China's hyper-accelerated pipeline of dual-targeting agents pairs with massive domestic CDMO expansion, enabling rapid, localized iteration of high-volume delivery modules.
Can bispecific antibodies be self-administered?
Yes, but specialty home-care programs arrive last in the adoption sequence. Large academic medical centers adopt these workflows first, converting to at-home use only after extensive post-market safety data completely de-risks the specific bispecific agent for unmonitored administration.
Which bispecific antibodies are given subcutaneously?
The first wave of approved subcutaneous bispecific therapies specifically targets blood cancers, such as epcoritamab and teclistamab, with solid tumor agents like amivantamab rapidly following as specialized delivery devices gain regulatory clearance.
Compare manual injection versus auto-injector use for bispecific antibodies
Manual injection struggles with the massive molecular payload of bispecific agents, often causing patient pain or incomplete dosing due to high viscosity. Auto-injectors standardize the delivery force, though they require extensive re-engineering of container-closure systems to withstand that same pressure without shattering.
How fast could bispecific antibodies shift from clinic injection to self-injection?
The timeline depends heavily on the qualification of high-payload delivery systems that can reliably extrude viscous co-formulations. Once primary packaging suppliers commercialize advanced container architectures capable of withstanding extreme mechanical stress, the transition accelerates rapidly.
Auto-injector vs manual injection for bispecific antibodies - how do buyers choose?
Buyers distinguish qualified from unqualified vendors based entirely on the vendor's documented track record of successfully guiding complex combination products through FDA and EMA regulatory gates, rather than merely evaluating mechanical design.
Why do hospital outpatient clinics hold the largest end-use share?
Despite the physical capability of the device to enable home use, clinical pathway directors compel administration in monitored settings to manage severe cytokine release syndrome risks during the initial step-up dosing phases.
What role do cartridge-based systems play?
Cartridge configurations are utilized when the volume of the bispecific formulation strictly exceeds the 2.25mL limits of standard bfs syringes, requiring specialized dual-chamber or wide-barrel designs to accommodate the payload.
How does South Korea leverage its biomanufacturing infrastructure?
South Korea's concentrated CDMO landscape enables rapid transition from clinical formulation to commercial-scale combination product assembly, allowing procurement directors to secure advanced delivery modules locally.
What changes operationally when a clinic adopts these devices?
Institutions eliminate lengthy intravenous chair times associated with systemic bispecific delivery, clearing massive backlogs in specialized oncology clinics and freeing critical resources for acute interventions.
Why is maintenance dosing the dominant application phase?
Maintenance dosing represents the phase where the economic benefits of auto-injectors are fully realized. Once acute induction monitoring is cleared, localized administration becomes the primary determinant of long-term patient adherence.
How do large-volume wearables alter the competitive landscape?
Device developers secure premium biopharma contracts by validating on-body modules capable of delivering 5mL+ payloads over extended durations, a capability standard hand-held mechanical pens physically cannot match.
What is the consequence of selecting a delivery device post-formulation?
Biopharmaceutical manufacturers face severe late-stage clinical hold-ups because the high-viscosity drug profile inevitably clashes with standard device limits, forcing a multi-year delay to redesign the primary container.
How does the United States' reimbursement framework drive device integration?
Commercial payers systematically penalize prolonged intravenous chair times, effectively forcing clinical pathway directors to mandate subcutaneous auto-injector formats wherever clinically viable to maintain facility profitability.
Why are smart or connected technologies gaining traction in this space?
Connected nfc integrated autoinjector secondary packaging modules validate patient adherence remotely, providing clinical pathway directors the necessary data to authorize specialty home-care programs without requiring physical hospital visits for compliance checks.
What defines the growth trajectory of the Japanese market?
Japan's stringent quality standards regarding patient comfort and injection pain force developers to extensively validate the user experience, resulting in a more measured but highly premium-priced move toward home-based administration.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.