Wearable Healthcare Devices Market
The wearable healthcare devices market is segmented by Product (Trackers, Smartwatches, Patches, Smart Clothing), Device (Diagnostic & Monitoring, Therapeutic), Grade (Consumer-Grade, Clinical-Grade), Application (General Health & Fitness, Remote Patient Monitoring, Home Healthcare), Distribution Channel (Pharmacies, Online Channel, Hypermarkets), and Region. Forecast period: 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Wearable Healthcare Devices Market Size, Market Forecast and Outlook By FMI
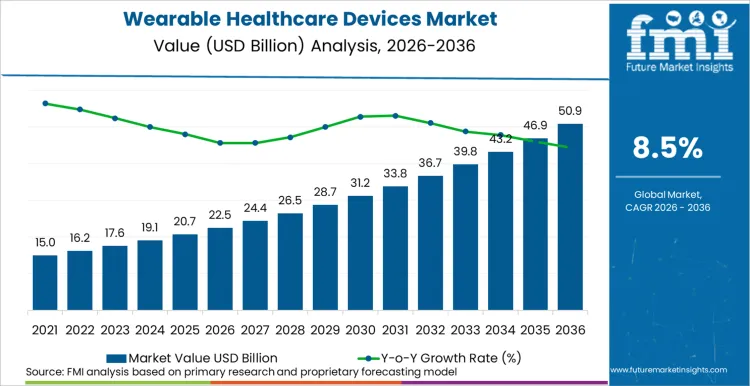
The wearable healthcare devices market is anticipated to expand from USD 22.57 billion in 2026 to USD 50.99 billion by 2036. Consumer-grade wearables are expected to dominate early adoption, while clinical-grade devices gain traction in chronic disease management. Online channels are projected to capture the largest sales volume, reflecting growing direct-to-consumer engagement.
Summary of the Wearable Healthcare Devices Market
- Demand and Growth Drivers
- Increasing prevalence of chronic diseases and aging populations is fueling demand for continuous monitoring devices.
- Integration of AI and predictive analytics is enhancing home-based patient monitoring.
- Government incentives and local manufacturing programs in Asia Pacific are supporting cost-effective adoption.
- Product and Segment View
- Trackers and smartwatches are likely to lead the product segment due to wide consumer adoption.
- Clinical-grade devices are gaining traction in diabetes, cardiovascular, and remote patient monitoring applications.
- Therapeutic wearables, including insulin pumps and rehabilitation devices, are emerging as high-value solutions.
- Geography and Competitive Outlook
- Asia Pacific is expected to be the fastest-growing region, led by India and China.
- North America maintains the highest market value, driven by regulatory-compliant integration into healthcare IT systems.
- Europe’s growth is supported by telemedicine adoption and reimbursement programs in digital health.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI, states, “Wearable healthcare devices are shifting toward interoperable medical ecosystems. Success will depend on regulatory compliance, AI-enabled analytics, and integration with clinical workflows.”
- Wearable Healthcare Devices Market Value Analysis
- The market is transitioning from consumer fitness tracking to integrated clinical monitoring ecosystems.
- Adoption is driven by chronic disease management, AI-enabled diagnostics, and preventive health programs.
- Spending is expected to increase as advanced devices integrate therapy, monitoring, and telehealth features.
- Online sales channels and digital health platforms are expanding access and recurring revenue potential.
Wearable Healthcare Devices Market Definition
Wearable healthcare devices are non-invasive or minimally invasive technologies worn on the body to monitor physiological data, track fitness metrics, or deliver therapeutic interventions. These devices encompass a range of form factors, including wristbands, smartwatches, bio-patches, and smart clothing, equipped with sensors to measure vitals such as heart rate, blood pressure, oxygen saturation, and glucose levels. The definition extends to software platforms that interpret this data for clinical or consumer use.
Wearable Healthcare Devices Market Inclusions
The report includes a comprehensive analysis of devices used for both general wellness and specific medical conditions like diabetes, cardiovascular diseases, and sleep apnea. It covers the full ecosystem of connected health, including the hardware sensors, the companion mobile applications, and the cloud-based analytics platforms used by providers. Market sizing encompasses revenue from device sales across consumer retail, pharmacy, and hospital procurement channels.
Wearable Healthcare Devices Market Exclusions
Excluded from the scope are implantable medical devices such as pacemakers or internal defibrillators that are not worn externally. The report also omits non-connected traditional medical equipment, such as standard analog thermometers or non-digital blood pressure cuffs, and general-purpose smartphones that lack dedicated medical-grade sensors. Revenue from standalone fitness apps without paired proprietary hardware is also excluded.
Wearable Healthcare Devices Market Research Methodology
- Primary Research: Validated through interviews with R&D heads at major device manufacturers, regulatory consultants, and procurement managers in hospital networks.
- Desk Research: Analyzed 510(k) clearances, corporate annual reports, World Health Organization digital health surveys, and national health ministry budget allocations.
- Market-Sizing and Forecasting: Utilized a bottom-up approach aggregating shipment volumes of smart watch and medical trackers, cross-referenced with sensor unit pricing and regional adoption rates.
- Data Validation and Update Cycle: Forecasts are calibrated against quarterly earnings from top-tier players and adjusted for new regulatory approvals or reimbursement code changes.
Segmental Analysis
Wearable Healthcare Devices Market Analysis by Grade
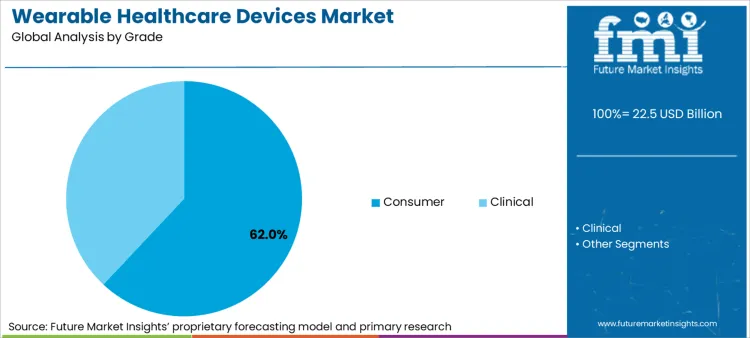
Consumer-grade devices command 62.0 percent of the market in 2026, driven by the mass adoption of smartwatches and fitness bands that now incorporate advanced FDA-cleared sensors. FMI analysts opine that this dominance is being challenged by the rapid miniaturization of clinical-grade technologies that offer higher accuracy for chronic disease management.
- Regulatory conversion: Demand is reinforced by consumer tech companies securing medical clearances (like De Novo classifications for sleep apnea), which validates smart wearables for insurance-backed health programs. [3]
- Adoption mechanism: Buyers are transitioning from simple step-counting to outcome-based monitoring because devices now offer irregular heart rhythm notifications and blood oxygen tracking suitable for clinical triage. [3]
- Data utility: Growth is anchored in the ability of these devices to export longitudinal health data to physicians, enabling proactive care adjustments before acute events occur. [3]
Wearable Healthcare Devices Market Analysis by Application
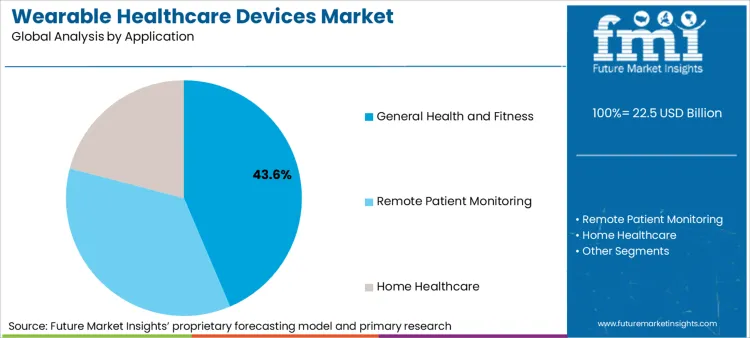
General health and fitness applications account for 43.6 percent of the market, serving as the entry point for most users before they graduate to specific disease monitoring tools. The segment is evolving from passive tracking to active lifestyle management powered by algorithmic coaching.
- Preventive trigger: Uptake is accelerated by rising global inactivity rates, where 81 percent of adolescents and 31 percent of adults fail to meet recommended physical activity levels, prompting individuals to purchase wearable fitness trackers for behavioral nudging. [5]
- Clinical bridge: Usage expands as fitness devices integrate with remote patient monitoring devices, allowing healthcare providers to utilize consumer-generated data for managing pre-chronic conditions. [7]
- Efficiency gain: Expansion is linked to the reduction of manual vital sign checks, as automated continuous monitoring proves more reliable than sporadic nurse assessments in detecting patient deterioration. [7]
Wearable Healthcare Devices Market Analysis by Distribution Channel
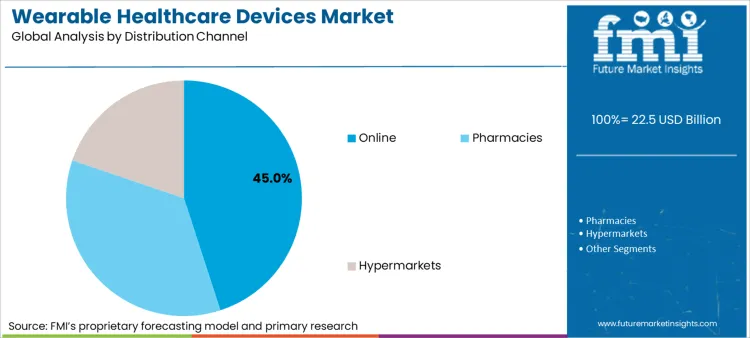
The online channel captures 45.0 percent of sales, leveraging direct-to-consumer models that offer customization and subscription-based coaching services. This channel is critical for reaching younger demographics and facilitating the recurring revenue models preferred by device makers.
- Access shift: Sales volume migrates to digital platforms because manufacturers can bundle hardware with premium health insights and replacement services directly to the end user. [6]
- Availability logistics: Growth tracks the expansion of e-commerce logistics, which ensures that specialized medical wearables are accessible even in regions with limited brick-and-mortar pharmacy infrastructure. [6]
- Pricing leverage: The online model allows companies to offer competitive pricing by bypassing traditional retail markups, making advanced health tracking affordable for a broader population. [6]
Wearable Healthcare Devices Market Drivers, Restraints, and Opportunities
Drivers and Restraints
The primary demand driver is the urgent need for scalable chronic disease management, particularly for diabetes and cardiovascular conditions. As healthcare systems face staffing shortages, the ability to automate monitoring through continuous glucose monitoring device ecosystems and connected sensors becomes essential. This mechanism allows patients to manage conditions at home, reducing hospital readmissions. For instance, Abbott reported a 19 percent organic increase in its diabetes care sales in Q2 2025, validating the massive shift toward sensor-based disease management. [2]
Growth is restrained by significant data reliability and privacy concerns that hinder full clinical trust. Physicians are often hesitant to base treatment decisions on wearable data due to inconsistencies in signal quality and the lack of standardized validation protocols. A study by GE HealthCare highlighted that fewer than half of vital signs were monitored according to physician orders in manual workflows, and while wearables aim to fix this, early implementations often suffer from signal noise and interoperability gaps. [7] This friction slows the transition from pilot programs to hospital-wide deployments, forcing vendors to invest heavily in clinical validation studies.
Market Opportunities
-
AI-Enhanced Diagnostics: Innovation lies in integrating on-device AI to interpret complex physiological signals locally, which Medtronic demonstrated by achieving a 0.7 percent HbA1c reduction in Type 2 diabetes trials using automated algorithms. [1]
- Emerging Market Localization: Opportunity exists in establishing local manufacturing hubs in high-growth regions, exemplified by India's approval for mass production of wearable components like HDI PCBs to lower device costs for domestic adoption. [8]
- Telehealth Integration: Expansion is favorable for platforms that combine hardware with virtual care services, as evidenced by WHO reporting that 77 percent of European nations now provide telemedicine services that rely on patient-generated data. [5]
Regional Analysis
Based on the regional analysis, the wearable healthcare devices market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
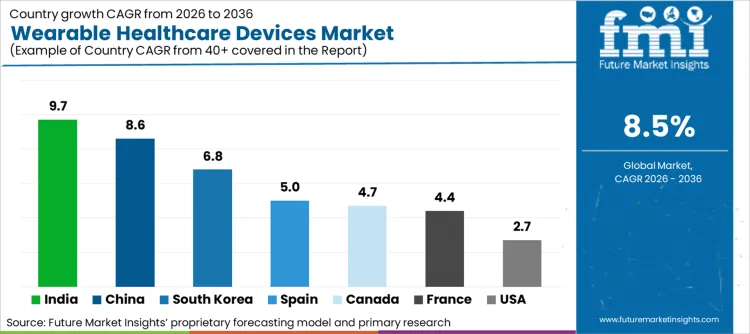
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.7% |
| China | 8.6% |
| South Korea | 6.8% |
| Spain | 5.0% |
| Canada | 4.7% |
| France | 4.4% |
| USA | 2.7% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Wearable Healthcare Devices Analysis
Asia Pacific is rapidly emerging as a global powerhouse for both consumption and production of medical wearables, driven by government initiatives to digitize public health infrastructure. The region benefits from a dual engine of aging populations in East Asia requiring monitoring and a young, tech-savvy demographic in South Asia adopting fitness tech.
- India: Demand for wearable blood pressure monitor tech in India is set to grow at 9.7% CAGR through 2036, supported by the National TB Elimination Programme's use of AI tools which reduced adverse outcomes by 27% before procurement cycles translate into higher deployment volumes. [8]
- China: Wearable sales in China are projected to rise at 8.6% CAGR from 2026 to 2036 as domestic tech giants integrate health metrics into "super-apps", creating a clearer pathway from policy or spending intent to purchase orders. [6]
- South Korea: The South Korean sector is poised to expand at 6.8% CAGR during 2026 to 2036 because 61% of adults fail to perform sufficient physical activity, which lifts utilisation rates and justifies technology refresh decisions. [9]
FMI’s report includes a detailed growth analysis for Asia Pacific, covering key markets like Japan and Australia. The region presents significant opportunities in Vietnam and Indonesia, where rising disposable incomes are fueling demand for affordable health tracking. Supply chain leaders should monitor Vietnam's growing role in electronics assembly, which offers a diversification option against regional geopolitical risks. [8]
Europe Wearable Healthcare Devices Analysis
Europe represents a sophisticated market where adoption is driven by national health services integrating digital therapeutics to combat aging-related costs. The focus here is on regulatory compliance and interoperability with established electronic health records.
- Spain: Spain is expected to record 5.0% CAGR in patient monitoring devices demand over 2026 to 2036 as the health ministry digitizes official statistical operations, tightening the link between operational constraints and adoption-led investment. [10]
- France: In France, the wearable market is forecast to advance at 4.4% CAGR through 2036, with growth anchored in the 77% availability of telemedicine services that converts into measurable budget allocation. [5]
FMI’s report includes analysis of the Europe market, highlighting Germany and the UK as central hubs for digital health reimbursement. Opportunities in Poland and Italy are emerging as these nations modernize their public health infrastructure. Suppliers should watch for EU-wide interoperability standards that will dictate market access for data-generating devices in these opportunistic territories. [5]
North America Wearable Healthcare Devices Analysis
North America remains the highest value market, characterized by high per capita healthcare spending and a regulatory environment that encourages software-as-a-medical-device innovation. The region is seeing a shift from general wellness to prescribed digital therapeutics.
- Canada: Wearable uptake in Canada is anticipated to climb at 4.7% CAGR over the forecast period as Abbott's diabetes care sales grew 19% organically, helping suppliers align positioning with local buying triggers. [2]
- USA: The USA is likely to see wearable revenues increase at 2.7% CAGR during 2026 to 2036, given the CDC finding that over half of workers favor sensors for occupational safety and the resulting push for capacity and workflow upgrades. [11]
FMI’s report includes a comprehensive assessment of the North American landscape, including Mexico's growing manufacturing base. The analysis points to opportunities in Mexico as a nearshoring hub for medical device production. Buyers in the region are increasingly prioritizing wearable glucometers and cardiac monitors that come with pre-approved reimbursement codes, a critical factor for market penetration. [2]
Competitive Aligners for Market Players
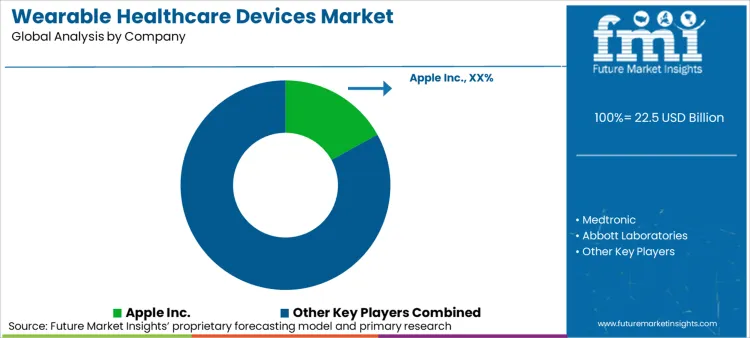
According to FMI's estimates, the competitive landscape is consolidating around players who can offer end-to-end disease management ecosystems rather than standalone hardware. Medtronic's strategic move to spin off its diabetes business into "MiniMed" allows for a more agile pursuit of the automated insulin delivery market, decoupling it from the slower-moving cardiac rhythm segments. [12] This structural separation enables dedicated capital allocation toward software interoperability, which is becoming the primary differentiator.
Market leaders are aggressively pursuing partnerships to close technology gaps, as seen in the collaboration between GE HealthCare and Biofourmis. This partnership extends patient monitoring from the hospital bed to the home, effectively creating a virtual ward that leverages consumer-friendly wearables for clinical-grade data collection. [7] Such alliances are essential for traditional medtech firms to access the agile, user-centric design capabilities of digital health startups without building them from scratch.
Regulatory clearance serves as the ultimate competitive moat. Apple's achievement of De Novo classification for its sleep apnea notification feature signals that consumer tech companies are successfully navigating FDA pathways previously reserved for specialized medical device manufacturers. [3] This development forces traditional players to accelerate their own innovation cycles or risk losing the preventive care market to generalist tech giants who already own the patient's wrist.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In December 2025, Medtronic launched the MiniMed 780G system integrated with Abbott's Instinct sensor in the USA market with a 0.7% HbA1c reduction claim to unify automated insulin delivery with high-fidelity sensing. [1]
- In October 2025, Abbott commissioned an expansion of its AVEIR pacemaker commercial footprint, doubling the number of trained physicians to 2x levels to accelerate adoption of leadless pacing technology in cardiac care. [2]
Key Players in Wearable Healthcare Devices Market
- Apple Inc.
- Medtronic
- Abbott Laboratories
- GE HealthCare Technologies
- Koninklijke Philips N.V.
- Fitbit (Google)
- OMRON Corporation
- Samsung Electronics
- Movano Health
- Corsano Health B.V.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 22.57 billion (2026) to USD 50.99 billion (2036), at a CAGR of 8.5% |
| Market Definition | Wearable healthcare devices utilize sensors to monitor vitals, track fitness, and deliver therapies, bridging consumer wellness and clinical care. |
| Product Segmentation | Trackers, Smartwatches, Patches, Smart Clothing |
| Device Segmentation | Diagnostic & Monitoring, Therapeutic |
| Application Coverage | General Health & Fitness, Remote Patient Monitoring, Home Healthcare |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | India, China, South Korea, Spain, Canada, France, USA, and 40+ others |
| Key Companies Profiled | Apple Inc., Medtronic, Abbott, GE HealthCare, Philips, Fitbit, OMRON, Samsung, Movano, Corsano |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up sensor shipment analysis validated by corporate revenue breakdowns and regulatory clearance tracking |
Wearable Healthcare Devices Market Analysis by Segments
Product:
- Trackers
- Smartwatches
- Patches
- Smart Clothing
Device:
- Diagnostic & Monitoring Devices
- Vital Sign Monitoring Devices
- Glucose Monitoring Devices
- Sleep Monitoring Devices
- Fetal Monitoring & Obstetric Devices
- Neuromonitoring Devices
- Therapeutic Devices
- Pain Management Devices
- Insulin Pumps
- Rehabilitation Devices
- Respiratory Therapy Devices
Grade:
- Consumer-Grade Wearable Healthcare Devices
- Clinical-Grade Wearable Healthcare Devices
Application:
- General Health and Fitness
- Remote Patient Monitoring
- Home Healthcare
Distribution Channel:
- Pharmacies
- Online Channel
- Hypermarkets
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] Medtronic. (2025). FDA Clears MiniMed 780G System to Enable Integration with the Instinct Sensor and Approves Use in Type 2 Diabetes. Corporate Press Release.
- [2] Abbott. (2025). Abbott delivers strong Q3 results; reaffirms 2025 outlook. Corporate Press Release.
- [3] Apple Inc. (2024). FDA grants De Novo classification to Sleep Apnea Notification Feature. FDA Database.
- [4] GE HealthCare. (2024). GE HealthCare to spotlight industry-leading AI-enabled portfolio at HIMSS 2024. Corporate Press Release.
- [5] World Health Organization. (2024). The rise of telehealth in the European region. WHO Regional Office for Europe.
- [6] Apple Inc. (2024). Q3 FY2024 Consolidated Financial Statements. Corporate Investor Relations.
- [7] GE HealthCare. (2024). New generation patient monitoring: improving efficiency and patient flow. Clinical View.
- [8] Press Information Bureau. (2025). AI-enabled tools in National TB Elimination Programme result in 27% decline in adverse outcomes. Government of India.
- [9] OECD. (2025). Health at a Glance 2025: Korea. OECD Publishing.
- [10] Ministerio de Sanidad. (2025). Operaciones estadísticas oficiales y salud digital. Gobierno de España.
- [11] CDC. (2024). Wearable sensors to track OSH-related risk factors: Survey results. Centers for Disease Control and Prevention.
- [12] Medtronic. (2025). Medtronic announces intent to separate Diabetes business. Corporate Press Release.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market sizing and 10-year forecasts for wearable devices across consumer and clinical grades.
- Analysis of the shift from fitness tracking to regulated medical monitoring.
- Impact of FDA and CE regulatory changes on product interoperability.
- Growth opportunities in remote patient monitoring and decentralized clinical trials.
- Detailed country-level assessments for high-growth Asian and mature Western markets.
- Competitive benchmarking of tech giants vs. traditional medtech incumbents.
- Supply chain trends including the localization of sensor manufacturing.
Frequently Asked Questions
How large is the demand for Wearable Healthcare Devices in the global market in 2026?
Demand for Wearable Healthcare Devices in the global market is estimated to be valued at USD 22.57 billion in 2026.
What will be the market size of Wearable Healthcare Devices in the global market by 2036?
The market size for Wearable Healthcare Devices is projected to reach USD 50.99 billion by 2036.
What is the expected demand growth for Wearable Healthcare Devices in the global market between 2026 and 2036?
Demand for Wearable Healthcare Devices in the global market is expected to grow at a CAGR of 8.5% between 2026 and 2036.
Which Grade is poised to lead global sales by 2026?
Consumer-Grade devices are expected to be the dominant segment, capturing approximately 62.0% of global market share in 2026 due to widespread adoption of smartwatches.
How significant is the role of General Health and Fitness in driving Wearable Healthcare Devices adoption in 2026?
General Health and Fitness represents a critical segment, projected to hold a substantial 43.6% share of the total market in 2026 as users prioritize preventative wellness.
What is driving demand in India?
Demand in India is driven by government-backed AI screening programs and local manufacturing incentives that lower device costs.
What compliance standards or regulations are referenced for India?
The National TB Elimination Programme's AI protocols are referenced as a key regulatory and operational benchmark.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 9.7% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Europe is a priority due to high reimbursement rates for digital health and widespread availability of telemedicine services.
What type of demand dominates in Europe?
Demand for connected devices integrated with national telemedicine infrastructures dominates regional consumption.
What is China's growth outlook in this report?
China is projected to expand at a CAGR of 8.6% during 2026 to 2036.
Does the report cover South Korea in its regional analysis?
Yes, South Korea is included within the Asia Pacific region under the scope of analysis.
What are the sources referred to for analyzing South Korea?
OECD health statistics and data on physical inactivity rates are cited as primary reference sources.
What is the main demand theme linked to South Korea in its region coverage?
South Korea's demand is associated with aging demographics and high digital literacy driving adoption of remote monitoring.
Does the report cover Canada in its regional analysis?
Yes, Canada is included within the North America region under the regional coverage framework.
What is the main Canada related demand theme in its region coverage?
Growth in Canada is linked to strong organic sales in diabetes care and remote monitoring technologies.
Which product formats are strategically important for North America supply chains?
Interoperable devices like insulin pumps and sensor patches are prioritized due to reimbursement requirements.
What are Wearable Healthcare Devices and what are they mainly used for?
They are body-worn technologies used to monitor physiological data for fitness tracking, disease management, and therapeutic delivery.
What does Wearable Healthcare Devices mean in this report?
It refers to the global market for connected health sensors, smartwatches, and medical patches used in home and clinical settings.
What is included in the scope of this Wearable Healthcare Devices report?
Scope covers devices by product, grade, and application, including trackers, patches, and remote monitoring systems.
What is excluded from the scope of this report?
Implantable devices, non-connected medical equipment, and standalone fitness apps without hardware are excluded.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the Wearable Healthcare Devices forecast?
Forecast is developed using bottom-up shipment analysis validated through corporate revenue reports and regulatory filings.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Grade
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Grade , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Grade , 2026 to 2036
- Consumer
- Clinical
- Consumer
- Y to o to Y Growth Trend Analysis By Grade , 2021 to 2025
- Absolute $ Opportunity Analysis By Grade , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- General Health and Fitness
- Remote Patient Monitoring
- Home Healthcare
- General Health and Fitness
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Online
- Pharmacies
- Hypermarkets
- Online
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Grade
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Grade
- By Application
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Grade
- By Application
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Apple Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic
- Abbott Laboratories
- GE HealthCare Technologies
- Koninklijke Philips N.V.
- Fitbit (Google)
- Apple Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Grade , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Grade
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Grade
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Distribution Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Grade
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Grade
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Grade
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Grade
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Grade
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Grade , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Grade , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Grade
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

