Heating Skincare Treatments Market
Heating Skincare Treatments Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Heating Skincare Treatments Market Forecast and Outlook 2026 to 2036
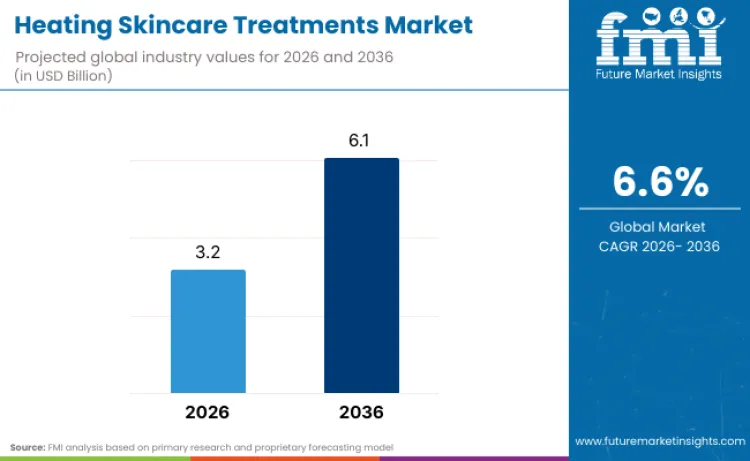
The Heating Skincare Treatments market is forecast to expand from USD 3.2 Billion in 2026 to USD 6.1 Billion by 2036, registering a 6.6% CAGR. Growth is being anchored in scaled beauty players operationalising device-enabled routines that make heat and light delivery repeatable at home, while also building regulatory pathways that de-risk mass rollout. L’Oréal’s CES 2026 announcement positioned infrared and LED-enabled beauty devices as a core innovation frontier, including an LED face mask prototype designed around controlled wavelength delivery and timed sessions, with the company noting that its US launch is subject to the FDA 510(k) premarket notification process.
This decade’s expansion is also being governed by tightening cosmetics compliance obligations that increase the value of large-company systems thinking. In the United States, FDA’s MoCRA implementation has formalised facility registration and product listing expectations for marketed cosmetics, increasing documentation, traceability, and responsible-person accountability across portfolios that are often co-marketed with device routines.
The competitive logic therefore shifts from one-off product novelty to scaled operating capability, where leaders can run multi-year device development cycles, partner with specialist hardware developers, and carry products through clearance and safety frameworks. Barbara Lavernos, Deputy CEO in charge of Research, Innovation and Technology at L’Oréal Groupe, stated: ‘This year, our pioneering advancements in light technologies at CES 2026 once again demonstrate our commitment to pushing the boundaries of new beauty solutions.’
Summary of the Heating Skincare Treatments Market
What is Growth Outlook for Heating Skincare Treatments Market as per Future Market Insights Projection?
Future Market Insights projects the Heating Skincare Treatments market to expand from USD 3.2 Billion in 2026 to USD 6.1 Billion by 2036, reflecting a 6.6% CAGR.
FMI Research Approach: Forecast built by mapping device-enabled skincare adoption to premium routine penetration, at-home usage frequency, and compliance-driven rollout speed across major consumer markets.
How do FMI Analysts Perceive Heating Skincare Treatments Market to Evolve?
FMI expects the category to scale through device-routine ecosystems where brands win by proving safety, controlled energy delivery, and repeatable session design, with regulatory readiness becoming a gating factor for mass launches.
FMI Research Approach: Competitive tracking of beauty-tech roadmaps, clearance requirements where applicable, and cross-market safety compliance constraints for consumer devices.
Which Country is Expected to be the Fastest Growing in Heating Skincare Treatments Market?
China is expected to grow at an 8.7% CAGR from 2026 to 2036, as regulatory supervision of cosmetics and scale retail ecosystems support rapid diffusion of premium at-home routines when compliance and distribution execution are strong.
FMI Research Approach: Country model weighted to premium beauty consumption, compliance scaling under national cosmetics supervision, and platform retail reach for device-led routines.
Which Product Type Leads the Heating Skincare Treatments Market in 2026?
Heating masks lead with a 34.6% share in 2026, reflecting the strongest fit with timed, repeatable home sessions and higher attach rates to anti-aging routines.
FMI Research Approach: Segment share derived from observed routine design patterns in beauty tech launches and the operational simplicity of mask-based session formats.
Which Application Accounts for the Largest Share in 2026?
Anti aging leads with a 37.9% share in 2026, reinforced by device-enabled routines designed around consistent sessions and visible-results narratives tied to controlled energy delivery.
FMI Research Approach: Application sizing linked to premium skincare spend concentration and brand investment intensity in longevity-positioned routines.
Heating Skincare Treatments Market Key Takeaways
| Items | Values |
|---|---|
| Market Size (2026) | USD 3.2 Billion |
| Forecast Value (2036) | USD 6.1 Billion |
| CAGR (2026 to 2036) | 6.6% |
What is Powering Adoption of Heating Skincare Treatments?
Adoption is being driven by large brands turning heating and light delivery into standardised routines that are easier to repeat at home and easier to govern at scale. L’Oréal’s CES 2026 disclosure framed infrared and LED systems as engineered platforms, with defined session timing and specified wavelengths for skincare delivery, signalling sustained investment in device-enabled regimens rather than short-cycle product launches. Regulatory tightening amplifies this shift by advantaging players with compliance infrastructure. In the United States, FDA’s MoCRA-driven facility registration and product listing expectations raise documentation and accountability requirements across cosmetics portfolios that are increasingly marketed alongside device routines. This combination pushes the category toward operators that can industrialise safety, claims discipline, and multi-market rollout without fragmenting portfolios across incompatible regulatory pathways.
How is the Heating Skincare Treatments Market Segmented?
The market for Heating Skincare Treatments is segmented into Product & Technology Mix (heating masks, heating creams and ointments, heating patches, heating serums, and powered or self-heating systems using chemical reaction, infrared-based heating, or electric heating elements), Application Pathways (anti-aging routines, acne treatment support, muscle relaxation and skin therapy, and deep cleansing and detox), and End User / Setting (home care users, dermatology clinics, beauty salons and spas, and others). This segmentation reflects how heating delivery is engineered and validated for safety, how outcomes are positioned within cosmetic skincare routines, and how adoption is concentrated around repeatable at-home sessions alongside credibility-building professional environments.
Why do Heating Masks Set the Commercial Tempo in Product Type Mix?
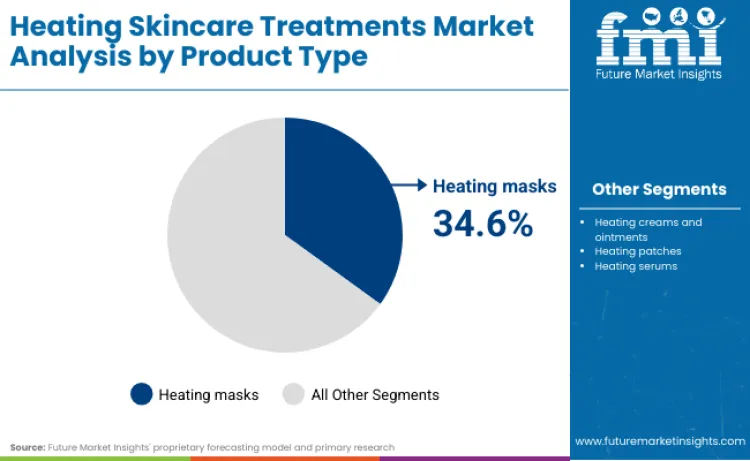
Heating masks lead with a 34.6% share in 2026 because they fit the operating model that scaled beauty-tech players are building: timed sessions, controlled delivery, and routine integration without requiring professional throughput. L’Oréal’s CES 2026 disclosure described an LED face mask concept built around defined wavelengths and a 10-minute session structure, illustrating how mask formats are engineered as repeatable consumer rituals rather than occasional treatments. This structure makes masks easier to standardise across regions because the user workflow is predictable, training load is low, and repurchase economics can be designed around consumables, skincare pairing, or routine bundles. The mask format also reduces variability in application compared with multi-step device handling, which matters as brands scale across diverse retail environments and customer capability ranges. In parallel, where clearance pathways apply, mask-led systems allow companies to document controlled delivery and user instructions more cleanly than open-ended heating tools, supporting launch discipline in regulated markets.
Why does Self Heating Chemical Reaction Outpace Powered Hardware in 2026?
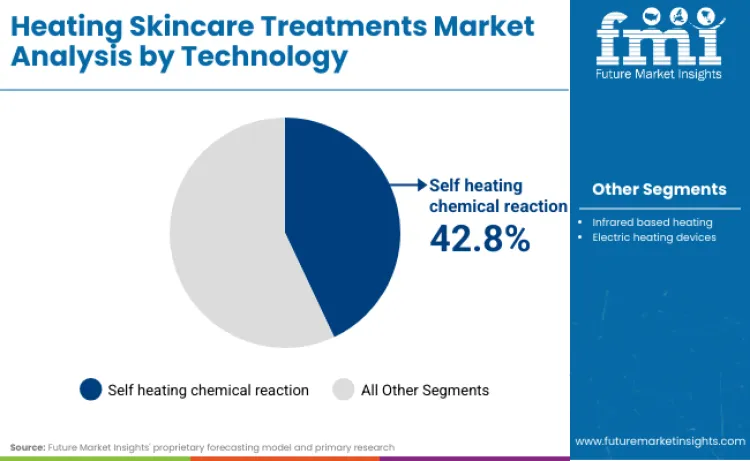
Self heating chemical reaction leads with a 42.8% share in 2026 because it bypasses a major friction point for powered devices: electrical safety certification and market access gating. When products move into mains-powered or electronically controlled heating, manufacturers face appliance safety expectations that vary by jurisdiction, such as BIS-linked safety regimes in India for household electrical appliances and related quality control enforcement timelines. Chemical self-heating formats avoid much of this cross-market electrical compliance burden, enabling faster multi-country rollout through conventional beauty distribution without redesigning hardware architectures for each regulatory environment. This matters in a category that relies on consumer trial and repeat usage frequency, where time-to-shelf and breadth of retail reach shape the adoption curve more than marginal performance differences. The result is a 2026 technology mix that favours formats with lower certification drag and fewer supply chain chokepoints, while powered devices scale more selectively where brands can justify compliance cost through premium pricing and ecosystem lock-in.
What are the Key Trends and Restraints Shaping the Heating Skincare Treatments Market?
A core trend is the industrialisation of device-enabled skincare routines by global beauty leaders, with clearer engineering parameters and regulatory pathways where required. L’Oréal’s CES 2026 announcement described infrared and LED systems with specified wavelengths and session timing, and explicitly linked a planned US launch path to the FDA 510(k) framework, signalling that major players are treating clearance and validation as part of the commercial rollout plan rather than an afterthought. This trend increases the role of partnerships with specialist developers and shifts competition toward operational capability, including the ability to manage multi-year development cycles and compliance documentation across markets.
A central restraint is compliance fragmentation across electrical safety regimes, which slows cross-border scaling for powered heating devices and raises redesign and certification costs. India’s Safety of Household, Commercial and Similar Electrical Appliances (Quality Control) Order, 2025 establishes a mandatory framework for covered household appliances and sets enforcement timelines that manufacturers must align to, creating a practical gating factor for device-led skincare formats that rely on electrical heating. In the United States, MoCRA-linked facility registration and product listing expectations increase compliance workload for associated cosmetics portfolios, raising the operating burden for smaller brands attempting to scale device-plus-topical systems without large compliance teams.
How Will Heating Skincare Treatments Market Expansion Unfold Across Key Global Regions?
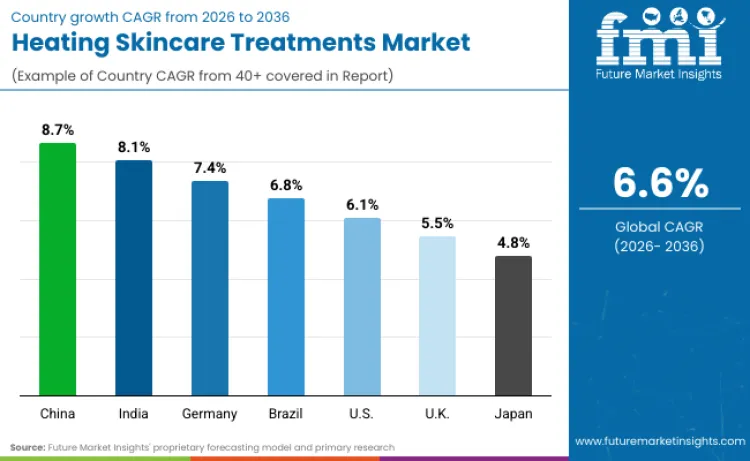
Future Market Insights identifies China (CAGR 8.7%), India (8.1%), Germany (7.4%), Brazil (6.8%), the United States (6.1%), the United Kingdom (5.5%), and Japan (4.8%) as the core demand drivers shaping global expansion of the Heating Skincare Treatments market through 2036. China leads growth as premium beauty consumption converges with strong cosmetics supervision, allowing compliant device-enabled routines to diffuse rapidly through large-scale retail and digital platforms. India’s growth is shaped by rising premium skincare adoption alongside stricter electrical safety enforcement, which filters demand toward certified, brand-led heating formats.
The United States benefits from established beauty-tech innovation pipelines, supported by structured regulatory pathways for cosmetics and devices that favour scaled operators with compliance infrastructure. Germany’s demand reflects EU-grade product safety discipline, reinforcing consumer trust in validated heating skincare systems. Brazil’s expansion is driven by health-regulator oversight that consolidates demand around compliant portfolios paired with mass retail reach. The UK shows moderate growth as mandatory product notification systems stabilise portfolio execution, while Japan’s slower trajectory reflects mature consumption and stringent electrical safety requirements that favour incumbents with local compliance depth. FMI emphasizes that across regions, adoption is governed less by novelty and more by the ability to align device validation, safety governance, and repeatable at-home routines within disciplined regulatory environments.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.7% |
| India | 8.1% |
| Germany | 7.4% |
| Brazil | 6.8% |
| USA | 6.1% |
| UK | 5.5% |
| Japan | 4.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How is China’s cosmetics supervision model accelerating compliant premium routines from 2026 to 2036?
China will grow at an 8.7% CAGR from 2026 to 2036 because the market combines premium beauty consumption with a regulatory framework that pushes brands toward formalised product governance, which advantaging scaled operators that can execute compliant rollouts quickly. The National Medical Products Administration outlines national responsibility for supervision and administration of cosmetics, signalling a high-governance environment where portfolio discipline, documentation, and compliant claims matter for market access and sustained distribution. As heating skincare routines scale, this governance model favours brands that can align device-led regimens with compliant topical portfolios and deliver consistent consumer instructions across platforms. The result is faster diffusion for players that treat compliance as a commercial capability, using disciplined product registration, safety substantiation, and controlled retail execution to scale device-associated regimens across major urban demand centres.
Why does India’s electrical safety enforcement shape category scaling between 2026 and 2036?
India will expand at an 8.1% CAGR across 2026 to 2036 because the category’s growth is filtered through national product safety governance for household electrical appliances, which raises barriers for powered heating devices and rewards brands that can certify and supply reliably. The Government of India’s Quality Control Order for household, commercial and similar electrical appliances establishes scope and compliance expectations tied to safety standards, creating a formal gate for device-led skincare formats that require electrical heating. This pushes market momentum toward compliant portfolios and distribution partners that can manage certification, labelling, and enforcement readiness, while also supporting growth for simpler at-home formats that avoid heavy electrical certification complexity. Over the decade, companies that align product engineering with mandatory safety regimes and build stable after-sales support are positioned to scale faster than brands that treat India as a straightforward import extension market.
How does Germany’s EU-grade product compliance discipline strengthen premium device routines through 2036?
Germany will rise at a 7.4% CAGR from 2026 to 2036 because adoption is reinforced by high compliance expectations for both cosmetics and consumer devices, which structurally advantages established operators with quality systems and documentation depth. As device-enabled skincare expands, Germany’s market behaviour tends to reward products that can demonstrate safety conformity and clear user instruction, reducing reputational risk for retailers and professional channels. This dynamic aligns with the broader EU operating environment where product safety and compliance are treated as baseline requirements for market access, making compliance capability a differentiator rather than a cost line. For heating skincare treatments, that increases the relative attractiveness of brands that can run validated product development cycles, manage recalls and complaints transparently, and provide consistent technical documentation across retail and professional settings.
Why does Brazil’s health-regulator posture support compliant scaling for skincare portfolios paired with routines?
Brazil will grow at a 6.8% CAGR through 2036 because regulatory oversight and product regularisation requirements increase the advantage of companies that can maintain compliant labelling, packaging, and product control across portfolios marketed into mass channels. ANVISA’s regulatory scope for personal hygiene products, cosmetics and fragrances highlights the role of health-regulator governance in the category’s operating environment, which becomes more important when brands pair topical products with device routines that raise consumer safety expectations. This environment tends to filter out inconsistent operators and supports scaled brands that can manage product stewardship, claims discipline, and channel execution across large retail footprints. As a result, Brazil’s growth trajectory favours firms that can industrialise compliance while sustaining premium positioning and routine-led consumer education.
What is structurally lifting US adoption despite rising compliance load from 2026 to 2036?
The United States will expand at a 6.1% CAGR from 2026 to 2036 because device-enabled skincare is scaling alongside tighter cosmetics governance that shifts advantage toward large operators with compliance infrastructure. FDA’s MoCRA framework sets expectations for cosmetic facility registration and product listing, increasing accountability and documentation across topical portfolios that are often marketed alongside device routines. In parallel, where device pathways apply, FDA’s 510(k) framework defines the requirement for demonstrating substantial equivalence for devices that need premarket notification, which influences launch sequencing and validation investment. These mechanisms increase barriers for smaller brands but reinforce category credibility for scaled players, supporting steady adoption as consumers accept device routines backed by regulated operating discipline.
How does UK product notification enforcement shape disciplined portfolio scaling through 2036?
The United Kingdom will grow at a 5.5% CAGR from 2026 to 2036 because cosmetics governance includes mandatory notification requirements that push brands toward formal portfolio control, improving trust and retail readiness for routine-led categories. The UK’s OPSS notification portal states that every cosmetic product available to consumers in Great Britain must be notified, with penalties for non-compliance, reinforcing a strict compliance baseline for topical products marketed alongside heating routines. This governance pressure favours operators that can maintain accurate product files, responsible-person structures, and consistent labelling and claims discipline, which becomes more important as device-led routines increase scrutiny. Over time, the UK market therefore rewards companies that treat compliance as part of the commercial operating model, enabling reliable scaling across premium retail and direct-to-consumer channels.
Why does Japan remain a market where global scale does not automatically convert into category control by 2036?
Japan will grow at a 4.8% CAGR from 2026 to 2036 because electrical safety compliance and mature consumer expectations favour incumbents with domestic execution depth, limiting easy wins for global players. Japan’s electrical product safety regime under the PSE framework defines mandatory safety and EMI requirements for designated electrical products, creating a practical gate for powered heating devices and increasing the importance of local compliance handling and after-sales quality. In a mature market, this reinforces the position of domestic operators and well-established multinationals with Japan-specific compliance and distribution infrastructure. As a result, global leadership in beauty can fail to translate into heating skincare category leadership in Japan if companies cannot localise compliance, channel strategy, and consumer trust-building at the same intensity as domestic incumbents.
How is the Competitive Landscape Evolving in the Heating Skincare Treatments Market?
Competition is concentrated among global beauty and consumer health companies that can fund device R&D, manage safety compliance, and scale routine-led regimens across regions. This scope includes heating masks, heating patches, heating creams and serums positioned around warming action, and powered or chemical self-heating systems used for cosmetic skincare outcomes. This scope excludes medical-grade thermal therapy devices marketed for clinical indications, hospital heat therapy equipment, and purely pharmaceutical topical treatments without a heating delivery mechanism. Regional leadership differs. In North America, global leadership is reinforced by regulatory readiness under MoCRA-linked cosmetics obligations and device pathways where applicable, favouring operators with compliance scale. In Europe, compliance discipline raises the advantage of quality systems and validated product documentation.
Recent Developments
- In January 2026, L’Oréal announced Light Straight + Multi-styler and an LED Face Mask, both Innovation Award honorees, showcasing infrared and light-based technologies that enhance hair and skin performance and reinforce L’Oréal’s science-led leadership in Beauty Tech.
- In May 2025, DPIIT extended the Safety Electrical Appliances QCO implementation to March 19, 2026, introducing MSME relaxations, legacy stock provisions, and aligning quality enforcement with India’s ‘Made in India’ manufacturing and compliance framework.
Key Players
- L’Oréal
- Procter & Gamble
- Johnson & Johnson
- Beiersdorf
- Unilever
- Shiseido
- Kao Corporation
- Amorepacific
- Estée Lauder Companies
- Panasonic Corporation
Market Definition
Heating Skincare Treatments are consumer and professional skincare solutions that deliver a warming effect to support cosmetic skincare routines, including chemical self-heating masks, warming patches, heating creams, ointments, and serums, and powered systems using infrared or controlled heating elements when positioned for cosmetic outcomes. The market is defined by routine integration, repeat session design, and safety governance requirements that shape product development and market access. Growth is primarily driven by scaled operators that can fund multi-year device development and manage regulated operating obligations, including cosmetics accountability and, where applicable, device premarket pathways. L’Oréal’s CES 2026 disclosure of LED and near-infrared skincare concepts illustrates how market leadership is linked to engineered delivery and regulatory readiness.
Market Inclusion
Included products comprise heating masks, self-heating sheet masks, warming patches for cosmetic skincare routines, heating creams and ointments marketed for warming skincare benefits, heating serums, and powered cosmetic skincare systems using infrared or controlled warming delivery where sold into consumer or aesthetic channels. Included end users cover home care users, dermatology clinics, and beauty salons and spas when the products are positioned for cosmetic skincare outcomes rather than medical treatment. Included market activity covers product launches, compliance disclosures, partnerships with device developers, and governance-led rollout actions such as FDA documentation, UK notification readiness, and electrical safety conformity where relevant.
Market Exclusion
Excluded products comprise clinical thermal therapy devices used for medical indications, hospital-grade heating equipment, rehabilitation heat therapy systems, and regulated medical devices marketed primarily for disease treatment rather than cosmetic skincare outcomes. Excluded are purely topical pharmaceutical products without a heating delivery mechanism, as well as general household heating appliances not designed or marketed for skincare. The exclusion boundary is important because regulatory pathways differ materially. For example, FDA’s 510(k) framework applies to medical devices requiring substantial equivalence demonstration, which is distinct from cosmetics governance obligations under MoCRA for marketed cosmetic products.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type Segments | Heating Masks; Heating Creams & Ointments; Heating Patches; Heating Serums |
| Technology Segments | Self-Heating Chemical Reaction; Infrared-Based Heating; Electric Heating Devices |
| Application Categories | Anti-Aging; Acne Treatment; Muscle Relaxation & Skin Therapy; Deep Cleansing & Detox |
| End User Groups | Home Care Users; Dermatology Clinics; Beauty Salons & Spas |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia, Middle East & Africa |
| Key Countries | China, India, Germany, United States, Brazil, United Kingdom, Japan |
| Key Companies Profiled | L’Oréal; Procter & Gamble; Johnson & Johnson; Beiersdorf; Unilever; Shiseido; Kao Corporation; Amorepacific; Estée Lauder Companies; Panasonic Corporation |
| Additional Attributes | Dollar sales measured across heating skincare formats by product type, technology platform, application focus and end-user adoption; routine-led at-home versus professional usage dynamics; regulatory and safety compliance impact across cosmetics governance and consumer device frameworks (USA MoCRA accountability, FDA 510(k) pathways where applicable, UK product notification systems, electrical safety regimes in India and Japan); innovation roadmap tracking for beauty-tech platforms; channel execution shifts for device-enabled regimens; competitive positioning based on R&D scale and regulatory readiness |
Heating Skincare Treatments Market by Key Segments
By Product Type:
- Heating masks
- Heating creams and ointments
- Heating patches
- Heating serums
By Technology:
- Self heating chemical reaction
- Infrared based heating
- Electric heating devices
By Application:
- Anti aging
- Acne treatment
- Muscle relaxation and skin therapy
- Deep cleansing and detox
By End User:
- Home care users
- Dermatology clinics
- Beauty salons and spas
Bibliography
- Bureau of Indian Standards. (2025). Indian standard for safety of household and similar electrical appliances: General requirements (IS 302-1: 2024). Government of India.
- Office for Product Safety and Standards. (2024). Submit a cosmetic product notification: Guidance for businesses placing cosmetic products on the GB market. UK Government.
- Department for Promotion of Industry and Internal Trade. (2025, May 19). Safety of household, commercial and similar electrical appliances (quality control) order, 2025. Ministry of Commerce and Industry, Government of India.
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Integrated Reports
- Peer-reviewed Medical and Clinical Journals
- Corporate Websites and Product Technical Literature
- Investor Presentations and Earnings Communications
- Regulatory Filings and Consumer Device Safety Standards
- Clinical Guidelines and Public Health Publications
- Trade Journals and Beauty Tech Industry Magazines
- Conference Proceedings and Consumer Electronics Materials
- Government Safety Notifications and Surveillance Data
- Press Releases and Verified Media Coverage
- Specialist Beauty Tech Newsletters
- Sector Databases and Consumer Device Repositories
- FMI Internal Knowledge Base
- Subscription-based Consumer Tech Databases
- Retail and E-commerce Channel Data Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Validation Approach
- Why Primary Evidence is Used
- Field Techniques
- Beauty Tech Product Developer Interviews
- Retail and E-commerce Channel Checks
- Salon and Clinic Protocol Inputs
- Device Safety and Instruction Review Workshops
- Regulatory and Compliance Consultations
- Stakeholder Universe Engaged
- C-suite Executives
- R&D and Engineering Leaders
- Regulatory and Compliance Officers
- Quality and Safety Assurance Heads
- Retail Category Managers and Distributors
- Dermatology Clinics and Aesthetic Service Providers
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Beauty Tech Adoption and At-home Session Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Technology, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By End User, 2026 to 2036
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Oceania
- Middle East and Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- China
- India
- Germany
- Brazil
- USA
- UK
- Japan
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Technology
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
- li>
Frequently Asked Questions
What is the Heating Skincare Treatments Market size in 2026 and 2036, and what is the CAGR?
The heating skincare treatments market is projected to reach USD 3.2 billion in 2026 and USD 6.1 billion by 2036, growing at a 6.6% CAGR.
Which product format leads adoption in the Heating Skincare Treatments Market in 2026?
Heating masks dominate with a 34.6% share due to their timed-session structure and easy integration into repeatable at-home skincare routines.
Why does self-heating chemical technology outperform powered devices in 2026?
Self-heating chemical reactions lead with a 42.8% share because they bypass complex electrical safety certifications that slow cross-market device scaling.
Which application segment captures the largest share of heating skincare demand in 2026?
Anti-aging treatments lead with a 37.9% share as brands design premium device-enabled routines around consistent session-based results.
What regulatory factor most reshapes operational requirements for skincare brands in the United States?
MoCRA-linked FDA cosmetic facility registration and product listing obligations significantly increase documentation and compliance accountability.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Integrated Reports
- Peer-reviewed Medical and Clinical Journals
- Corporate Websites and Product Technical Literature
- Investor Presentations and Earnings Communications
- Regulatory Filings and Consumer Device Safety Standards
- Clinical Guidelines and Public Health Publications
- Trade Journals and Beauty Tech Industry Magazines
- Conference Proceedings and Consumer Electronics Materials
- Government Safety Notifications and Surveillance Data
- Press Releases and Verified Media Coverage
- Specialist Beauty Tech Newsletters
- Sector Databases and Consumer Device Repositories
- FMI Internal Knowledge Base
- Subscription-based Consumer Tech Databases
- Retail and E-commerce Channel Data Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Validation Approach
- Why Primary Evidence is Used
- Field Techniques
- Beauty Tech Product Developer Interviews
- Retail and E-commerce Channel Checks
- Salon and Clinic Protocol Inputs
- Device Safety and Instruction Review Workshops
- Regulatory and Compliance Consultations
- Stakeholder Universe Engaged
- C-suite Executives
- R&D and Engineering Leaders
- Regulatory and Compliance Officers
- Quality and Safety Assurance Heads
- Retail Category Managers and Distributors
- Dermatology Clinics and Aesthetic Service Providers
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Beauty Tech Adoption and At-home Session Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Product Type, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Technology, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application, 2026 to 2036
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By End User, 2026 to 2036
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Oceania
- Middle East and Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- China
- India
- Germany
- Brazil
- USA
- UK
- Japan
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Technology
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Heating Skincare Treatments Market Key Takeaways
- Global Heating Skincare Treatments Market Size, 2021 to 2036
- Heating Skincare Treatments Market Size by Product Type
- Heating Skincare Treatments Market Size by Technology
- Heating Skincare Treatments Market Size by Application
- Heating Skincare Treatments Market Size by End User
- Heating Skincare Treatments Market Size by Region
- Heating Skincare Treatments Market CAGR by Country
- Scope of Report
List of Figures
- Heating Skincare Treatments Market Growth Trajectory, 2026 to 2036
- Governance-led Device-enabled Routine Adoption Framework
- Product Type Share Analysis
- Technology Mix Analysis
- Application Mix Analysis
- End User Share Distribution
- Regional Market Attractiveness Analysis
- Competitive Landscape Overview

